Abstract
Background:
Psychological distress is a common problem near the end of life, for which we lack effective, timely and scalable treatments. No previous study has assessed whether microdose psilocybin can improve symptoms in this population.
Aim:
To determine whether microdose psilocybin is safe, feasible and potentially efficacious in a palliative setting.
Design:
Open label, single-arm clinical trial of a 3-week oral psilocybin intervention, starting with 1 mg daily in week 1, increased to 2 mg in week 2 and 3 mg in week 3. ClinicalTrials.gov NCT04754061.
Setting/participants:
Two-center study in Ottawa, Canada of adults with advanced, incurable illness and an estimated prognosis of 1–12 months, experiencing severe psychological distress.
Results:
We enrolled 20 participants (59% of those screened) between January 2024 and April 2025, of which 17 began and 13/17 (76%) completed the intervention. Participants were 40–84 years old, 53% female, and 82% had cancer. There were no serious adverse events reported, and nine mild or moderate adverse events. Four participants withdrew due to disease progression or poor response. Of the 13 remaining participants, nine (69%) reported a meaningful global improvement (Patient Global Impression of Change ⩾ 5); 8 (62%) reported >50% improvement in Hamilton Depression Rating Scale scores, 7 (54%) reported >50% improvement in Hospital Anxiety and Depression Scale scores and 9 (72%) reported a meaningful improvement in Demoralization Scale II scores.
Conclusions:
Microdose psilocybin is a safe, feasible and potentially efficacious treatment for psychological distress in people with advanced illness.
Keywords
Psychological distress is a common problem near the end of life, for which we lack effective, timely and scalable treatments.
Psychedelic-assisted psychotherapy, which involves the use of large doses (macrodoses) of psychedelic medication, has shown promising results, but there are barriers to widespread use of this approach in the palliative setting.
Microdose psilocybin is not subject to many of the barriers seen with macrodosing, but no previous study has assessed whether microdose psilocybin can improve symptoms in this population.
We found that microdose psilocybin was safe, feasible and potentially efficacious for improving psychological distress in a small, uncontrolled study in a palliative setting.
Our findings are promising but this intervention must be tested in a large, placebo-controlled study powered for efficacy.
Introduction
Approximately 35%—50% of people with advanced illness experience severe psychological distress (including depression and anxiety, or existential distress) as they approach the end of life.1–3 This distress is associated with reduced medication adherence, and poor quality of life, 4 and is the most common reason people request Medical Assistance in Dying (MAiD). 5
Psilocybin is a serotonergic hallucinogen that can produce durable improvements in psychological distress in people with serious illnesses when given in high doses along as part of psychedelic-assisted psychotherapy.6,7 However, macrodose psychedelic-assisted psychotherapy involves many sessions and requires a commitment (and an operational delay) of several weeks, which may not be feasible for many people in a palliative context.
Psychedelic microdosing involves taking sub-psychedelic doses of a serotonergic hallucinogen on a regular basis. 8 Large surveys and anecdotal reports suggest that microdosing psilocybin or LSD can produce meaningful and sustained improvements in psychological symptoms without any important side-effects. 9 However, there are no published clinical studies of microdose psilocybin in people with psychological symptoms. To address this gap, we conducted a study to assess the safety, feasibility, and preliminary efficacy of microdose psilocybin for severe psychological distress in people with advanced and incurable disease in the palliative context.
Methods
Design
Open-label, single-arm clinical trial.
Setting
Two academic hospitals in Ottawa, Canada (Appendix)
Participants
Adults with advanced, incurable illness who were under the care of a specialist palliative care provider, judged to have a prognosis of 1–12 months, and experiencing severe psychological distress. Psychological distress was defined as a score of ⩾7 on either the Depression, Anxiety or Well-being dimension of the Edmonton Symptom Assessment System, confirmed by a Hamilton Depression Rating Scale (HDRS) score of >13 or a Demoralization Scale-II (DS-II) score of >10. Exclusion criteria are listed in the Appendix.
Sampling and recruitment
Potential participants were identified and referred for screening by palliative care providers affiliated with the participating sites (see Appendix), who were sent regular reminders about the study. After a screening call with the principal investigator and a member of the research team, participants who met inclusion criteria, did not meet exclusion criteria, and who agreed to the conditions of participating in the study (see Appendix) were offered enrollment in the study.
Intervention
Participants received a 3-week psilocybin microdosing intervention, starting with 1 mg oral psilocybin daily for 5 days in week 1, and increasing to 2 mg oral psilocybin in week 2, and 3 mg oral psilocybin in week 3 as tolerated. After the 4-week follow-up assessment, participants could request enrollment into an open-label extension of the study for up to 1 year (to be reported separately). Purified psilocybin was supplied by Psilo Scientific Ltd., a wholly owned subsidiary of Filament Health Corp, Dealer’s License #6-1289, located in Burnaby, British Columbia, Canada.
Outcomes
The primary outcomes were:
Feasibility
a.
b.
c.
Safety
a. We recorded the proportion of participants who experienced an adverse event, including the dose the participant was receiving when they experienced the adverse event. Based on previous literature, adverse events that were specifically monitored for safety are:
i. Elevated blood pressure (BP) and heart rate (HR): BP and HR were measured 1 h post-medication administration and once in the evening each day during the intervention. Elevated blood pressure was defined as a systolic blood pressure of >180 mmHg with symptoms or >200 mmHg (regardless of whether symptoms are present). Elevated heart rate was defined as >130 bpm.
b. Mood: participants were provided with a diary and asked to record their mood and behavior each day during the intervention and at 1 day, 2-weeks, and 4-weeks of follow-up.
c. Delirium incidence: measured by the Confusion Assessment Method (CAM) status in inpatient settings or the Family Confusion Assessment Method (FAM-CAM) status in outpatient and community settings
d. Serotonin syndrome: any participant who reported a combination of tachycardia (elevated heart rate) and cognitive changes (delirium, confusion), or a tremor, were referred immediately to the treating palliative care physician for an in-person assessment of possible serotonin syndrome.
The secondary outcomes were related to efficacy, including:
(a) Psychological distress, as measured by the Hospital Anxiety and Depression Scale (HADS)10,11 and the 17-item Hamilton Depression Rating Scale (HDRS). 12 For each tool we considered a 50% reduction in score from baseline as a clinically meaningful improvement.
(b) Subjective global improvement, evaluated using the Patient Global Impression of Change (PGIC) score 13 : we considered a score of 5 (moderately better – slight but noticeable change), 6 (better – definite improvement that has made a real and worthwhile difference), or 7 (a great deal better – considerable improvement that has made all the difference) as a clinically meaningful improvement.
(c) Psychological wellbeing, evaluated using the Edmonton Symptom Assessment System-revised (ESAS-r) Depression, Anxiety, and Well-being item scores 14 : we considered a 2-point reduction in item score from baseline, or an absolute score value of 4 or less as a clinically meaningful improvement.
(d) Existential distress, evaluated using the Demoralization scale II, 15 a 16-item questionnaire that measures loss of meaning and purpose, distress, and coping abilities.
(e) Wish to die, evaluated using the Categories of Attitudes Toward Death Occurrence (CADO). 16
Analysis
Analysis was descriptive, including counts and means for the patient demographics, and proportions for the safety and efficacy outcomes. The descriptive analysis also focused on the dose at which participants reported adverse events or improvements in outcomes.
Ethics
The study was approved by the Ottawa Health Sciences Network Research Ethics Board (OHSN-REB) via Clinical Trials Ontario (Project ID #3980). A No Objection Letter and an Exemption to use a Controlled Substance for Clinical Studies (i.e. exemption license) was obtained from Health Canada to store, distribute, and administer microdose psilocybin for the study. A Data Safety Monitoring Board reviewed the outcomes after three and eight participants were enrolled.
Changes to the trial
Initially, the protocol was a 4-week intervention. In the first week, participants took 1 mg oral psilocybin on Monday and on Thursday, and only started 1 mg daily in the second week. We removed this initial week after the first eight enrolled participants because we found no effect or adverse events, and participants were frustrated by the delay in reaching the 2 mg or 3 mg doses where most experienced benefit. We also removed the collection of the Death Anxiety Scale, as participants found it upsetting to answer the questions.
Results
Out of 34 referred and screened, we enrolled 20 participants over 15 months (58.8%; 95% CI: 0.42–0.74) between January 2024 and April 2025 (1.3 per month; Figure 1). Three withdrew prior to starting the intervention, due either to clinical factors or new inability to adhere to driving restrictions. The demographics, clinical characteristics and baseline psychological symptom scores of the remaining 17 are provided in Table 1. Participants were aged between 40 and 84 years and 53% female, with mostly cancer (82%) as the underlying illness. Participants all reported either mild (n = 12) or moderate (n = 5) symptoms of depression, with eight having moderate symptoms of anxiety.
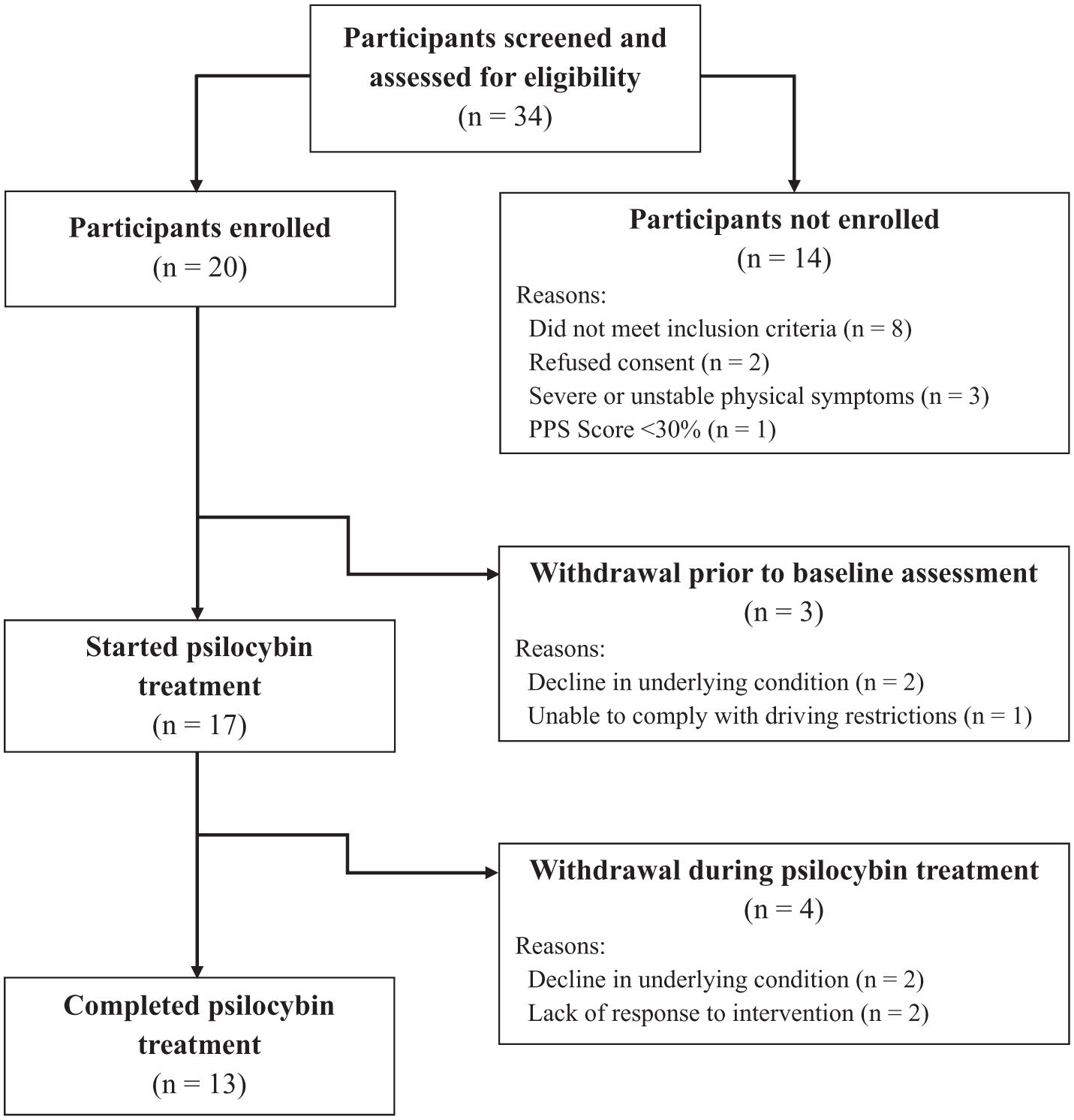
Flow diagram for study participants.
Participant demographics and baseline symptom scores.

Of the 17 participants who started the intervention, 13 (76%; 95% CI: 0.53–0.90) completed all 3 weeks of treatment, while four withdrew due to progression of their underlying disease (n = 2) or lack of response (n = 2). Four participants paused (but later resumed) the intervention due to complications or treatments related to their underlying illness. Two experienced adverse events while taking the 2 mg dose; their dose was lowered to 1 mg and completed 3 weeks of treatment with no further adverse events. Of the 13 who completed treatment, four withdrew during follow-up due to death (n = 1), progression of disease (n = 1) or assessment burden (n = 2).
Of the 13 participants who completed the intervention (Table 2), nine (69%) reported a PGIC score of 5 or greater (corresponding to moderately better, better, or a great deal better) either on the final day of treatment or the first day of follow-up (Table 2), with one additional participant reporting a score of 5 at only the 2-week follow up. In eight of these nine who experienced benefit, the participant experienced their greatest response at the 3 mg dose. One participant (who weighed only 37 kg) experienced a good response at 1 mg, but experienced lightheadedness when we increased the dose to 2 mg; they completed the study using a dose of 1 mg daily. Depression scores improved by 50% in 38% (HDRS) and 62% (HADS-Depression) of participants. HADS-Anxiety scores improved by 50% in 54%. Demoralization scores improved by 2 points in 72% of participants. Mean scores for all measures improved post intervention, with moderate effect size for demoralization (0.64) and large effect size estimates for the other measures (Cohen’s d 0.89–2.15; Table 3).
Changes in symptom scores from baseline.
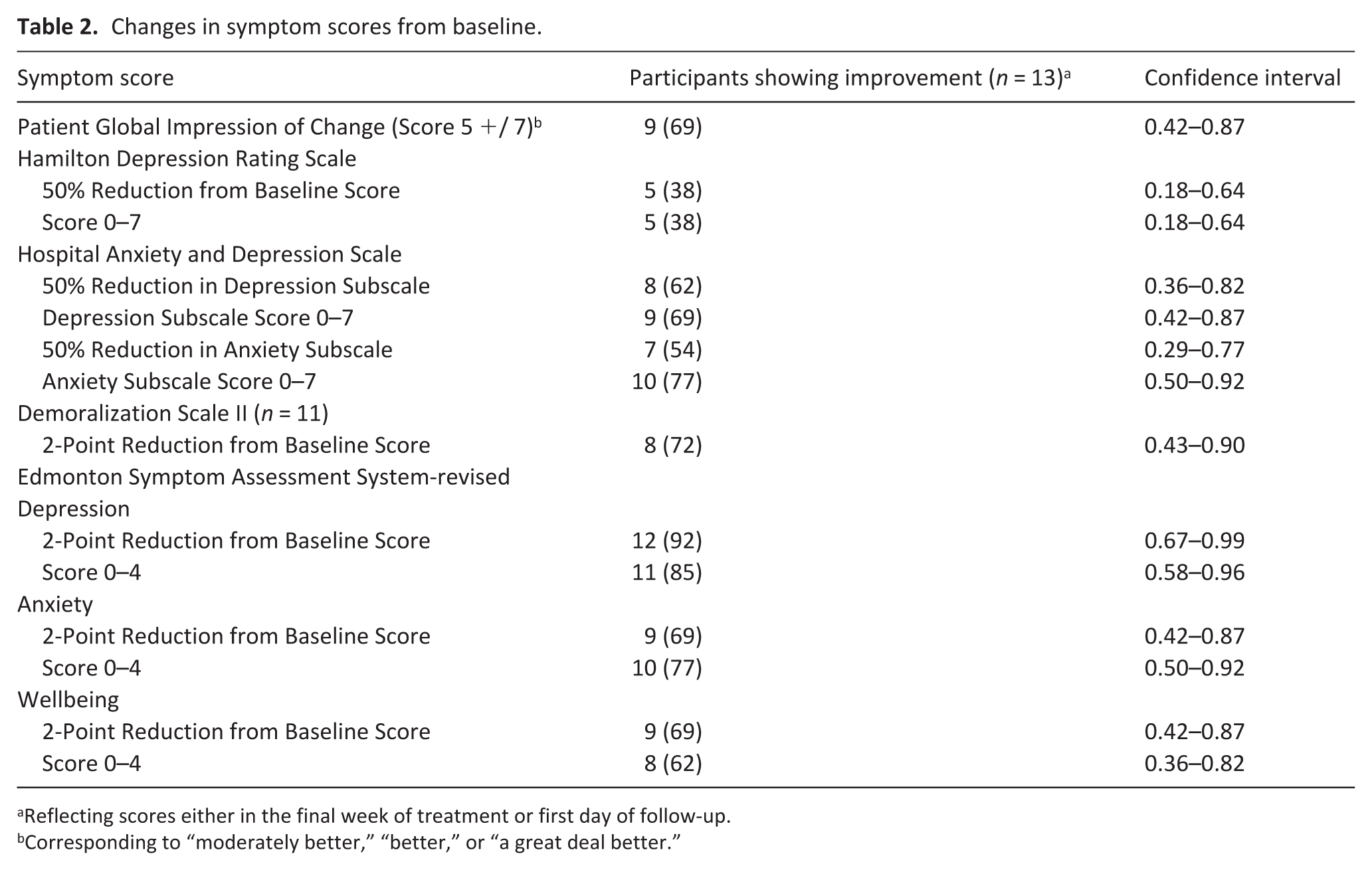
Reflecting scores either in the final week of treatment or first day of follow-up.
Corresponding to “moderately better,” “better,” or “a great deal better.”
Effect sizes for changes in symptom scores from baseline.
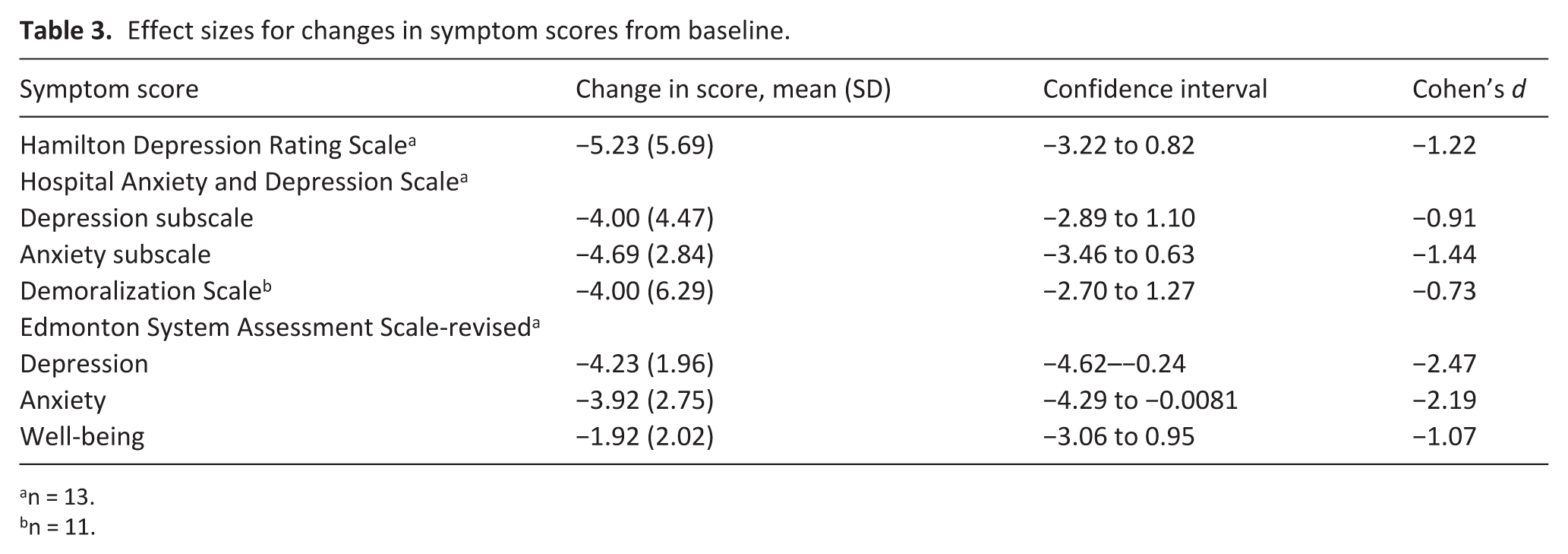
n = 13.
n = 11.
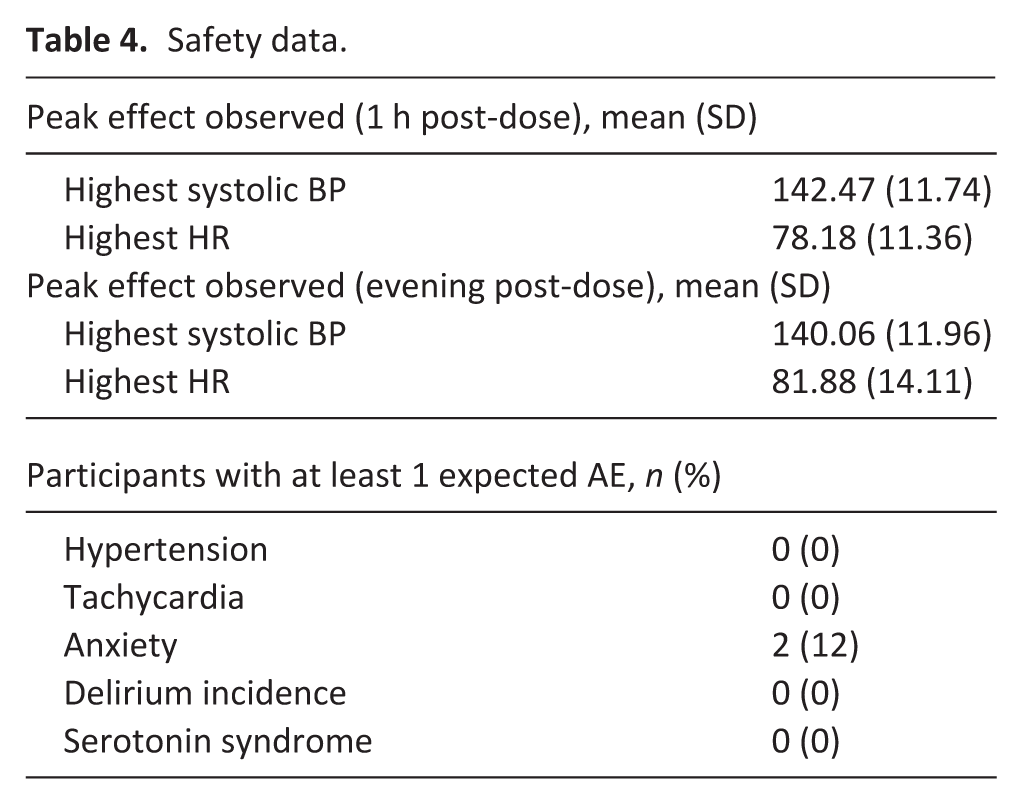
There were no serious adverse events. Vital sign measurements remained within acceptable limits for all participants (Table 4), 18 adverse events were reported, of which 14 were graded as mild (e.g. lightheadedness) and 4 as moderate (e.g. anxiety) in severity; nine were assessed as possibly or probably related to the study medication. Of the two participants who withdrew for lack of response, one was admitted to a community hospice for palliative sedation for their persistent mood symptoms after failing to respond to 3 mg, while the other withdrew after failing to respond to 1 mg and received MAiD. In both cases, these plans had been discussed with their clinical team prior to enrollment, but had been put on hold by the participant in order to try psilocybin as a last resort.
Safety data.
Of the eight participants who reported a PGIC ⩾ 5 and survived until the 4-week follow-up, six (75%) requested enrollment into the open-label extension of the study, while the other two had persistent benefit and did not require further treatment.
In terms of subgroups, three participants had non-cancer illness. All three completed the protocol; two reported a PGIC score of 5+ and enrolled in the open-label extension. Of the four participants who had a PPS < 50, one did not complete the protocol, two had a PGIC score <5, and one had a PGIC score 5+ and enrolled in the open-label extension. Of the six participants who were taking oral antidepressants, one did not complete the protocol, one had a PGIC score <5, and four had a PGIC score 5+ (three enrolled in the open-label extension).
Discussion
Main findings of the study
In this open-label study, we found that microdose psilocybin was safe and feasible for use in a palliative context. We also found that people taking open-label microdose psilocybin experienced meaningful improvements in psychological symptoms compared to baseline. To our knowledge, this is the first registered trial of microdose psychedelic medication used for symptom management.
What this study adds
Palliative care providers have effective treatment options for physical suffering, but often struggle to address psychological distress. A recent meta-analysis of 38 Randomized Controlled Trials (RCTs) of palliative care interventions found no significant effects on anxiety, depression, or psychological distress. 17 Evidence of efficacy of antidepressants and anxiolytics is limited18,19 and many patients avoid these medications due to adverse effects, delays in treatment effect, or cost. Psychotherapeutic interventions (e.g. cognitive behavioral therapy, dignity therapy) have shown modest results with poor durability. 3
A meta-analysis by Metaxa and Clarke 6 found that macrodose psychedelic-assisted psychotherapy is effective for treating symptoms of depression, particularly when the depressive symptoms are driven by an underlying medical illness. Schipper et al. 20 performed a meta-analysis of six studies of macrodose psychedelic assisted psychotherapy in people with life-threatening conditions; they concluded that this intervention is safe and may be effective in treating symptoms of depression, anxiety and existential distress. Smaller studies have suggested that macrodose psychedelics may also be effective for treating other psychiatric illnesses or substance use disorders. 8 Microdose psilocybin and LSD have not been well studied outside of anecdotal reports and surveys, but they have been widely reported to improve mood, cognition or relationships with others. 8
Microdose psilocybin offers several potential advantages over macrodose psilocybin in a clinical setting. Our approach to microdosing did not include psychotherapy or supervision, which simplifies the logistics and shortens the time to experience symptomatic benefits. Microdoses do not appear to produce the nausea or cardiovascular side-effects often seen with macrodoses, which may improve tolerability for people with advanced diseases. Microdosing studies would also not be as susceptible to “functional unblinding,”20,21 where participants can easily determine their group assignment.
Strengths and limitations
Strengths of our study include the novel intervention and broad inclusion criteria, which improve the generalizability of our results. We also used a subjective patient-centered measure of efficacy; the psychological distress experienced by patients in a palliative setting is often not purely depression or anxiety, and many patients are less familiar with terms like “existential distress,” “demoralization,” or adjustment disorder. Limitations include the potential for a placebo or expectation effect due to the uncontrolled, open-label design, which we plan to address in a future double-blind, placebo-controlled study powered for efficacy. In addition, while we considered 3 mg to be a microdose and none of our participants described a psychedelic experience, Griffiths et al. 22 expressed concern that this dose had the potential to induce psychedelic effects. Our inclusion of people with more advanced illness also meant a higher dropout rate, as patients deteriorated more quickly than anticipated. We did not employ any formal psychotherapeutic techniques as used in PAP, but this could be incorporated into future studies either as a standard of care or a factorial design. Finally, the small sample size, which is common in psychedelic studies, precluded any definitive subgroup analyses.
In conclusion, microdose psilocybin appears to be a safe, feasible and potentially efficacious treatment for psychological distress in people with advanced illness in the palliative setting. This represents a promising and highly-scalable option for an indication that is in urgent need of effective and scalable treatments.
Footnotes
Appendix
Authors’ note
The supplier of the investigational product (Filament, Inc.) was not responsible for the study concept, design, data collection or interpretation, and were paid by the research budget for the investigational product.
Author contributions
JD, JL, GL and PL designed the study. JL, KA and GP enrolled participants and were involved in preparation of regulatory documents relevant to this study. JD, JL, KA, NP, GL and PL were involved in analysis and interpretation of the data. JD drafted the manuscript, and all authors were involved in critical review and revision of the manuscript. All authors approved the final version of the manuscript submitted.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by The Ottawa Hospital Academic Medical Organization (TOHAMO) and the Bruyere Health Academic Medical Organization (BHAMO). The funders had no role in the design, conduct, or publication of the trial.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data collected and analyzed during this study will be available from the corresponding author on reasonable request.
