Abstract
Background:
Psychological and existential suffering affects many people with advanced illness, and current therapeutic options have limited effectiveness. Repetitive transcranial magnetic stimulation (rTMS) is a safe and effective therapy for refractory depression, but no previous study has used rTMS to treat psychological or existential distress in the palliative setting.
Aim:
To determine whether a 5-day course of “accelerated” rTMS is feasible and can improve psychological and/or existential distress in a palliative care setting.
Design:
Open-label, single arm, feasibility, and preliminary efficacy study of intermittent theta-burst stimulation to the left dorsolateral prefrontal cortex, 600 pulses/session, 8 sessions/day (once per hour) for 5 days. The outcomes were the rates of recruitment, completion of intervention, and follow-up (Feasibility); and the proportion of participants achieving 50% improvement on the Hamilton Depression Rating Scale (HDRS) or Hospital Anxiety and Depression Scale (HADS) 2 weeks post-treatment (Preliminary Efficacy).
Setting/participants:
Adults admitted to our academic Palliative Care Unit with advanced illness, life expectancy >1 month and psychological distress.
Results:
Due to COVID-19 pandemic-related interruptions, a total of nine participants were enrolled between August 2021 and April 2023. Two withdrew before starting rTMS, one stopped due to clinical deterioration unrelated to rTMS, and six completed the rTMS treatment. Five of six participants had a >50% improvement in HDRS, HADS-Anxiety, or both between baseline and the 2 week follow up; the sixth died prior to the 2-week follow-up. In this small sample, mean depression scores decreased from baseline to 2 weeks post-treatment (HDRS 18 vs 7, p = 0.03). Side effects of rTMS included transient mild scalp discomfort.
Conclusions:
Accelerated rTMS improved symptoms of depression, anxiety, or both in this small feasibility and preliminary efficacy study. A larger, sham-controlled study is warranted to determine whether rTMS could be an effective, acceptable, and scalable treatment in the palliative setting.
Trial registration:
NCT04257227
Keywords
Psychological and existential suffering is common, highly burdensome, and difficult to treat in people approaching the end of life.
Repetitive Transcranial Magnetic Stimulation (rTMS) is a safe and effective therapy for refractory depression, but no previous study has used rTMS to treat psychological or existential distress in the palliative care setting.
An accelerated 5-day rTMS treatment caused rapid and meaningful improvements in depression, non-significant decreases in anxiety and demoralization, and no difference in quality of life among patients admitted to a palliative care unit in this small, open-label study.
Future studies, including a sham-controlled study, are warranted to determine whether rTMS could be an effective, acceptable, and scalable treatment for one of the most challenging forms of suffering faced by people with advanced illness.
Severe psychological and existential suffering affects up to 35%–50% of people with advanced cancer as they approach end-of-life,1,2 impairing their quality of life and medication adherence, worsening their “total pain” experience, and increasing their use of acute care services. 3 Therapeutic options for psychological and existential suffering in the palliative setting are limited. Specialized palliative care consultation provides little or no benefit for mood symptoms. 4 Pharmacological treatments have little evidence of effectiveness,5,6 and the delayed treatment effect and risk of adverse reactions are a deterrent to patients and clinicians alike. Psychotherapeutic interventions require a substantial time commitment, and there is little evidence for effectiveness beyond the immediate post-treatment period. 7 There is a need for brief, scalable, and rapidly effective means of relieving psychological and existential distress in people nearing the end of life.
Repetitive Transcranial Magnetic Stimulation (rTMS) is a safe and non-invasive brain stimulation technique approved for treating major depression and anxiety, 8 and under study for other neurological and psychiatric disorders. Using an induction coil placed against the scalp, rTMS delivers focused magnetic pulses to brain regions responsible for cognition and emotion regulation.9,10 Previously, the broad use of rTMS was limited by the availability of rTMS devices and the duration of treatment (4–6 weeks). However, a 5-day “accelerated” rTMS protocol rapidly improved symptoms in people with treatment-refractory depression.11–13 No previous study has used rTMS to treat psychological or existential distress in the palliative setting, so we conducted this phase 2 feasibility and preliminary efficacy study.
Methods
Design
Open-label, single arm study.
Setting
Single center, 31-bed inpatient academic Palliative Care Unit with a regional referral base in Ottawa, Ontario, Canada.
Participants and recruitment
Eligible individuals included those with advanced illness who were under the care of a palliative care provider, judged to have >1 month life expectancy, and experiencing psychological or existential distress. Potential participants were identified by a score of 4 or greater 14 on the Depression, Anxiety, or Well-being subscale of the Edmonton Symptom Assessment System. Psychological or existential distress was confirmed by a Hamilton Depression Rating Scale (HDRS) 15 score of >13 16 or a Demoralization Scale-II (DS-II) score of >10. 17 Exclusion criteria included a diagnosed seizure disorder, documented brain lesions, contraindications to rTMS (e.g. intracranial metallic clips or plates), or an inability to remain still while sitting up (45°) for the duration of therapy. See Appendix for details.
Intervention
A trained technician administered rTMS to the participant’s left dorsolateral prefrontal cortex,18,19 located using the BeamF3 technique. 20 Each treatment session delivered 600 pulses of intermittent theta-burst stimulation at a target intensity of 120% of resting motor threshold. 21 Participants received up to eight sessions daily (approximately 1/h) for 5 days consecutively or in a 7-day window (if a weekend was involved). See Appendix for details.
Outcomes
The primary outcome for feasibility was the enrolment rate. The primary outcome for preliminary efficacy was the proportion achieving 50% improvement on the HDRS or HADS 22 at 2 weeks post-treatment (or last observation prior to death if death occurred <2 weeks) compared to baseline. Secondary outcomes included changes in existential distress, evaluated using the Demoralization (DS-II) scale, 17 and quality of life, evaluated using the World Health Organization Quality of Life Scale, brief version (WHOQOL-BREf). 23 Outcomes were measured daily during treatment, and then at 2, 4, and 8 weeks post-treatment.
Sample size and analysis
Prior to the COVID-19 pandemic, we had aimed to enroll 15 participants; one per month, or 20% of admitted patients who would normally be eligible (estimated at 30% of the ~500 annual admissions to our unit), and we pre-specified a goal completion rate of 85%. Data were analyzed descriptively for response compared to baseline. Changes in symptom scores were analyzed using a Wilcoxon pairs test.
Ethics
All participants provided written consent to participate. The study was approved by the local Research Ethics Board (Bruyère REB #M16-19-035 and OHSN-REB #20200017-01H).
Results
Nine people met inclusion criteria and consented to participate in the study between June 2021 and April 2023. Study enrolment was repeatedly interrupted and admissions were frequently affected by the COVID-19 pandemic, precluding any realistic estimation of enrolment rate. Two chose not to proceed with therapy after consent, and one patient withdrew on day 2 after a sudden clinical deterioration unrelated to rTMS. Six participants (86%) started and completed the therapy (mean age 64, four males (66%)). Five had metastatic cancer (three colorectal, one lung, and one cervical) as an underlying diagnosis, while one had pulmonary fibrosis. Five had a history of treatment for depression, and three had a history of treatment for anxiety. Two were taking escitalopram, and one was taking regular clonazepam at the time of enrolment. One participant completed 38 out of 40 sessions, and one died prior to the 2-week follow-up assessment. Side effects of rTMS included mild scalp discomfort, headache, or tingling sensation, all of which resolved spontaneously.
Five of the six participants who completed the treatment had a >50% improvement on the HDRS, HADS-Anxiety, or both between baseline and the 2-week follow up. The remaining participant died before the 2-week follow-up. Four of five participants with DS-II data demonstrated at least a 2-point improvement, which is considered meaningful. 17 Mean depression scores decreased from baseline to 2 weeks post-treatment (HDRS 18 vs 7, p = 0.03; Figure 1). There were non-significant decreases (Table 1) in mean anxiety (HADS-Anxiety 14.7 vs 8.7, p = 0.06) and demoralization (n = 5, DS-II 25 vs 17.4, p = 0.13), and no difference in quality of life (n = 5, WHOQOL-BREf 2.6 vs 2.6).
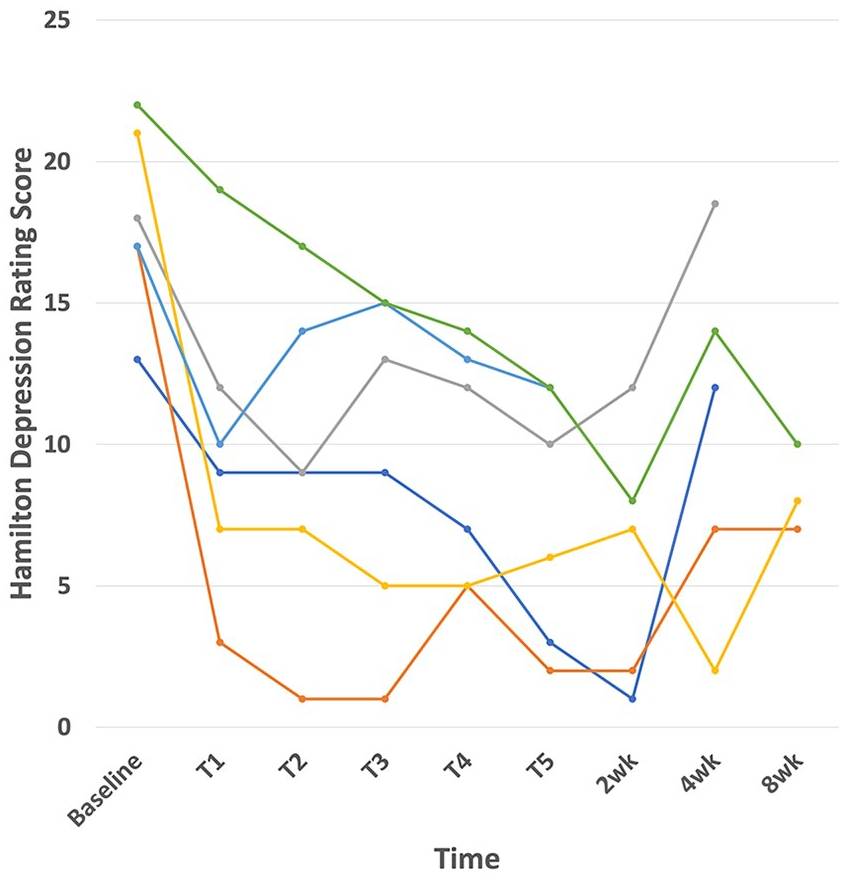
Depression scores among participants.
Symptom scores.
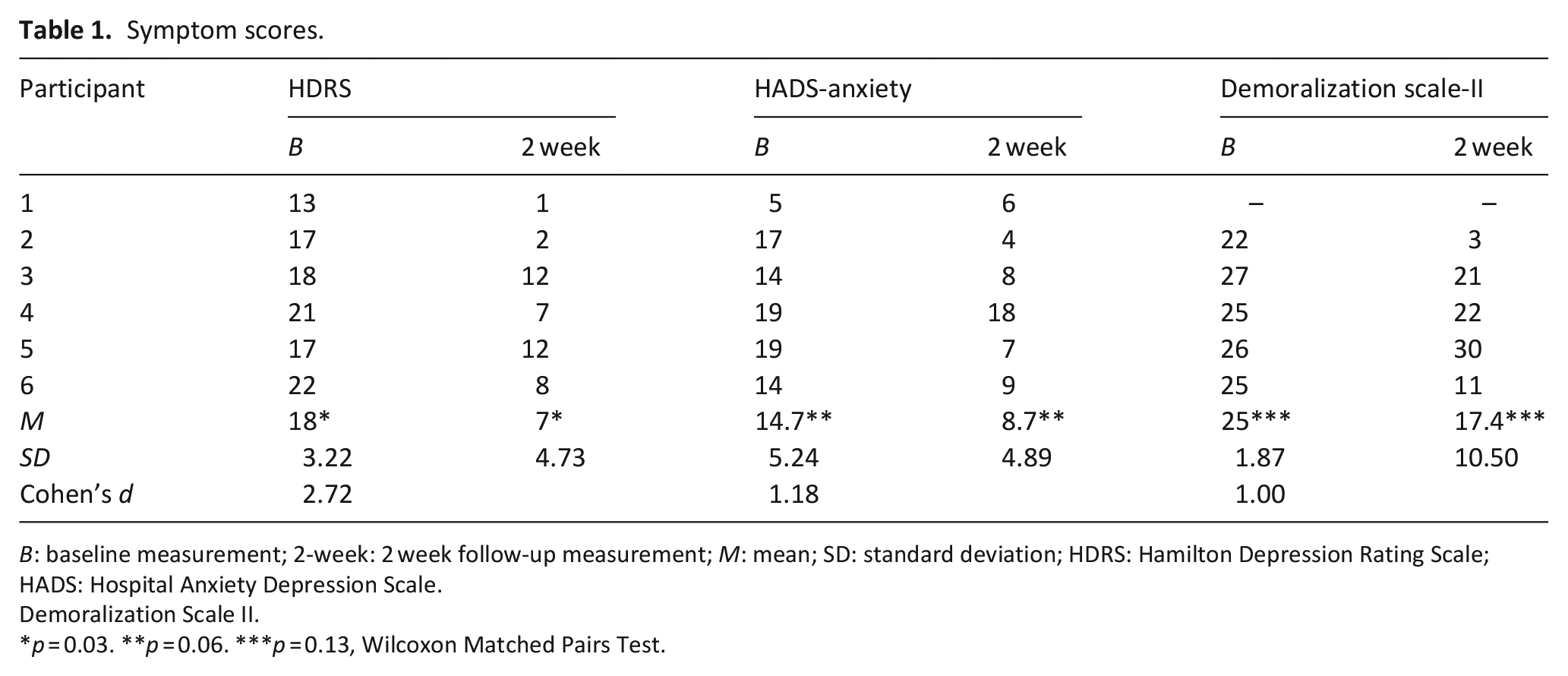
B: baseline measurement; 2-week: 2 week follow-up measurement; M: mean; SD: standard deviation; HDRS: Hamilton Depression Rating Scale; HADS: Hospital Anxiety Depression Scale.
Demoralization Scale II.
p = 0.03. **p = 0.06. ***p = 0.13, Wilcoxon Matched Pairs Test.
Discussion
Main findings of the study
In this single center, open label study in people with advanced illness and moderate to severe psychological distress, accelerated rTMS produced remission of depression, anxiety, or both in five of six participants. Enrolment was heavily affected by the COVID-19 pandemic, making a poor enrolment rate difficult to interpret, but the completion rate met our prespecified goal. The symptomatic improvements seen in our study, with minimal side effects and high tolerability, suggest that rTMS may be effective for relieving one of the most challenging forms of suffering experienced in the palliative setting.
What this study adds?
Although >100 sham-controlled trials of rTMS have demonstrated efficacy for treating refractory depression,24,25 this study is the first report of rTMS for psychological distress in a palliative setting. Historical barriers to rTMS in the palliative setting (equipment cost, long treatment courses, and delayed effects) may be less relevant now. Accelerated rTMS protocols offer both a shorter duration of therapy and a rapid time-to-effect, while newer rTMS devices offer portability and could remove the need for a trained in-person technician or for the patient to come to a healthcare facility. rTMS is generally well-tolerated, and although accelerated rTMS can produce headaches and fatigue, none of our participants stopped their treatment because of side-effects. Given the limited effectiveness and duration of existing palliative and psychotherapeutic interventions for psychological and existential distress,4–7 rTMS may represent a much-needed treatment that is effective, rapid, acceptable and scalable. Transcranial Direct Current Stimulation (tDCS) is another noninvasive neuromodulatory therapy that uses simpler equipment and does not require admission, although the treatment protocols are typically several weeks long. 26 Although a meta-analysis of tDCS showed a moderate effect size in depression, 27 a recent negative trial suggests that more studies are needed to identify individualized treatment approaches, and populations most likely to benefit. 26
Strengths and limitations of this study
Strengths include the use of prospective design and proven rTMS protocols, and inclusion criteria that reflect a broad range of psychological suffering. Limitations include the potential for a placebo or expectation effect, which could be evaluated in a future sham-controlled trial. Our enrolment was poor, and while this was probably due to the COVID-19 pandemic, we cannot exclude the possibility that many terminally ill people would be unwilling to commit to even five days of inpatient treatment. We chose to finish the study and proceed to a definitive sham-controlled study given that our results suggested both preliminary efficacy and a high completion rate. Finally, we used an rTMS protocol developed for refractory depression; better-optimized anatomical targets or dose regimens might yield better results than what we observed, or achieve the same results with a shorter treatment course. Our limited results did not suggest that a shorter or longer duration of treatment would be appropriate, but this could be investigated in future studies.
In conclusion, accelerated rTMS could be a much-needed treatment option for one of the most challenging forms of suffering faced by people with advanced illness. A future multi-site sham-controlled study is planned.
Footnotes
Appendix
Author contributions
All listed authors made substantial contribution to the concept or design of the work; or acquisition, analysis or interpretation of data; drafted the article or revised it critically for important intellectual content; approved the version to be published; and have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Data management and sharing
On publication, the data for this study will be made available publicly.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JD and JD have a financial interest in Ampa, Inc., a company that manufactures rTMS devices. The other authors have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by an Innovation Grant from the Canadian Cancer Society and the Lotte and John Hecht Memorial Foundation. The funders had no role in the design or conduct of the trial, the analysis/interpretation of the results, or the drafting/revision of the manuscript.
Research ethics and consent
All participants provided written consent to participate. The study was approved by the local Research Ethics Boards (Bruyère REB #M16-19-035 and OHSN-REB #20200017-01H).
