Abstract
Background:
Malignant gastric outlet obstruction is a frequent complication in advanced gastric, pancreatic and duodenal cancers, and can cause nausea, vomiting, pain, and malnutrition. No individual guideline comprehensively addresses the palliative management of malignant gastric-outlet-obstruction symptoms.
Aim:
To develop evidence-based recommendations for non-surgical palliative management of malignant gastric-outlet-obstruction, incorporating pharmacological, decompressive, endoscopic and nutritional strategies.
Design:
Practice review using scoping methodology to evaluate the strength of evidence supporting guideline-recommended interventions. Data sources: We identified relevant national and international guidelines via web searches and the TRIP database; guideline reference lists were hand-searched for relevant primary studies; a supplementary structured MEDLINE search (2024) captured emerging research.
Results:
A multidisciplinary team approach, with early evaluation of prognosis and patient preference, should guide intervention choice. Pharmacological therapies (opioids, antiemetics, antisecretory agents) are frequently used for symptom control, but evidence of their efficacy in malignant gastric-outlet-obstruction is limited. Duodenal stenting remains first-line for endoscopic palliation, while endoscopic gastroenterostomy has emerging evidence supporting its effectiveness and should be considered. Nasogastric or venting gastrostomy decompression is advised for acute obstruction when endoscopy is not feasible, but prolonged NG tube use should be avoided. Early nutritional assessment is recommended, although the optimal modality and duration of nutritional supplementation have yet to be determined.
Conclusions:
Patient-centred, multidisciplinary care is essential for malignant gastric-outlet-obstruction palliation. Further research is needed to establish optimal drug combinations. Duodenal stenting remains first-line, but guidelines should incorporate newer minimally-invasive interventions such as endoscopic gastroenterostomy. Standardised quality-of-life measures are required for developing integrated care pathways.
Malignant gastric outlet obstruction frequently arises in advanced malignancies and significantly impacts quality of life.
There is no guideline that addresses all aspects of malignant gastric outlet obstruction management.
This review summarises evidence for non-surgical palliative management of malignant gastric outlet obstruction
We identified strong evidence supporting emerging minimally invasive techniques, however current guidelines have yet to be updated.
Approaches to pharmacological management vary, and there is limited evidence supporting existing recommendations.
Malignant gastric outlet obstruction management should be patient-centred, led by a multidisciplinary team, and tailored to individual prognosis and preferences.
Commonly recommended drugs have limited specific evidence to malignant gastric outlet obstruction, highlighting the need for cautious, individualised use considering efficacy and side-effects.
Duodenal stenting remains first-line intervention, but guidelines should incorporate emerging interventions like endoscopic ultrasound-guided gastroenterostomy, which is supported by growing evidence.
Further research focused on symptom management and quality-of-life outcomes is needed to establish an optimal, integrated care pathway for malignant gastric outlet obstruction
Background
Malignant gastric-outlet-obstruction commonly complicates advanced gastric, pancreatic and duodenal cancers, accounting for 50%–80% of adult gastric outlet obstructions and affecting up to 15%–20% of patients with late-stage pancreatic cancer. 1 It arises when tumour infiltration or external compression mechanically blocks the gastric outlet, leading to nausea, vomiting, abdominal pain and inability to eat – symptoms that severely diminish quality of life and are associated with a median survival measured in months. 2 Optimising palliative management is essential both to alleviate these challenging symptoms and to improve quality of life in the context of advanced disease.
Effective palliation of malignant gastric-outlet-obstruction requires interventions that directly target the physical blockage. Surgical gastrojejunostomy offers durable relief, 3 but carries high morbidity and is unsuitable for many palliative patients. 4 Minimally invasive endoscopic interventions now predominate offering effective symptom relief with lower procedural risk. Duodenal stenting with self-expanding metal stents achieves technical success rates exceeding 90%, allowing most patients to resume a mechanical soft diet. 5 More recently, endoscopic ultrasound-guided gastroenterostomy using lumen-apposing metal stents has emerged as a promising alternative, potentially providing better outcomes. 6
Pharmacological support – opioids, anti-emetics, antisecretory agents – are necessary for patients waiting, or unsuitable for intervention, yet evidence for these drugs in malignant gastric-outlet-obstruction is limited and optimal regimens are undetermined. 7 Nutritional and decompressive strategies also vary.
Management must be individualised to tumour stage, prognosis, functional status and local expertise, yet no guideline addresses the full spectrum of palliative management for malignant gastric-outlet-obstruction. This review therefore aims to synthesise existing guidance to generate evidence-based recommendations for analgesic, anti-emetic and antisecretory therapy; appraise the effectiveness of duodenal stenting, endoscopic ultrasound-guided gastroenterostomy and decompressive measures; and assess how current guidance integrates pharmacological, endoscopic and nutritional strategies within a multidisciplinary framework.
Methods
We reviewed clinical guidelines and supporting primary evidence using scoping methodology 8 and appraised included evidence to provide an evidence-based summary of recommendations. The review followed the methodological framework proposed by Arksey and O’Malley 9 for scoping reviews, and reporting was informed by the PRISMA extension for Scoping Reviews (PRISMA-ScR) checklist (Supplemental Material 1). 10
Eligibility criteria
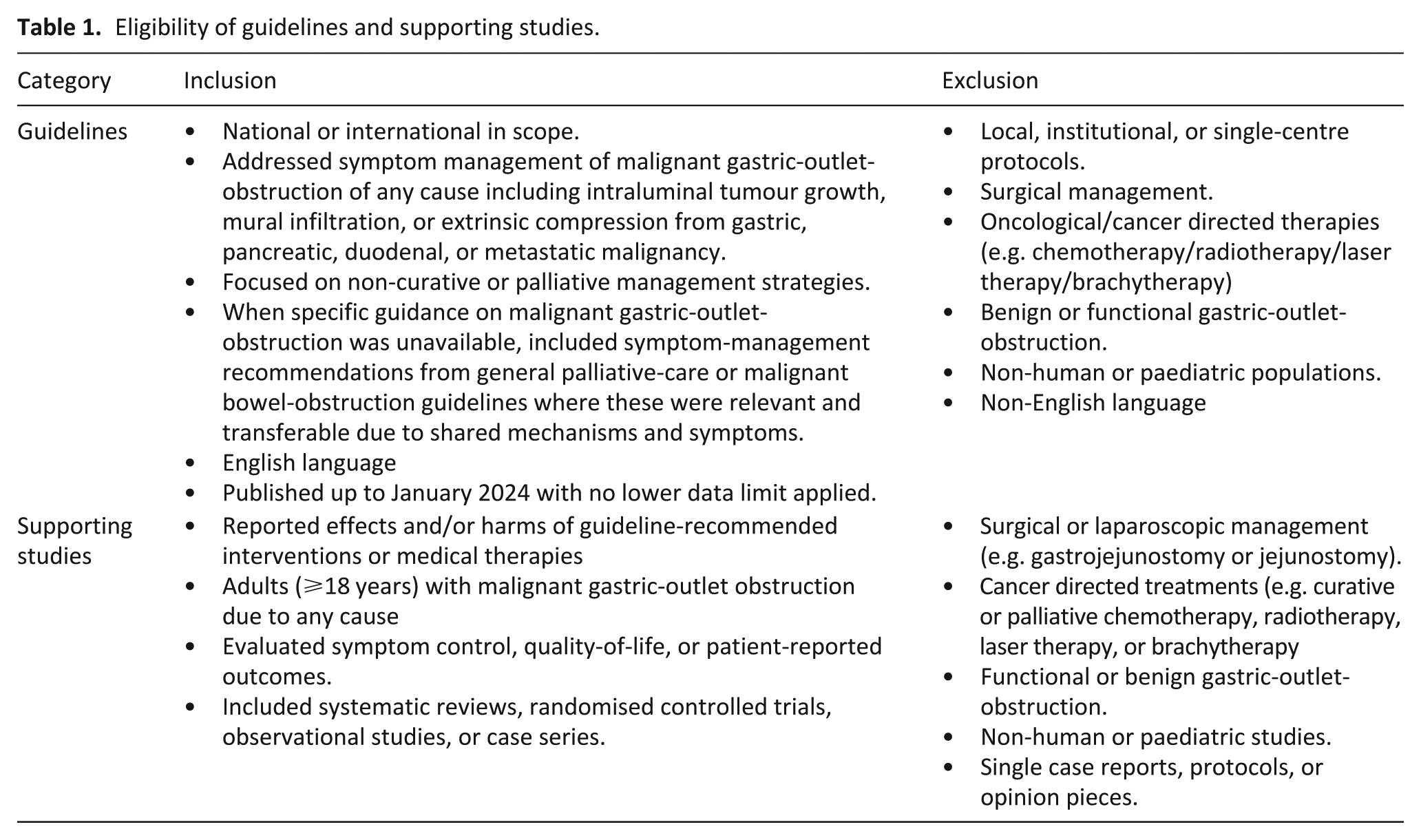
We included guidelines and evidence relating to any non-surgical symptomatic management of malignant gastric-outlet-obstruction, irrespective of aetiology. Cases secondary to intraluminal tumour growth, mural infiltration or extrinsic compression (e.g. from pancreatic, gastric, duodenal, or metastatic disease) were all eligible. Owing to the limited number of specific guidelines to malignant gastric-outlet-obstruction, recommendations from malignant bowel-obstruction guidelines were also considered when symptom-management principles were deemed transferable, given the shared pathophysiology and clinical features between these conditions. Whilst we recognise oncological/cancer directed therapies may have a role in management, we considered recommendations about specific cancer directed therapies to be beyond the scope of this review.
The inclusion and exclusion criteria are summarised in Table 1.
Eligibility of guidelines and supporting studies.
Search strategy and data extraction
Guideline search
National and international guidelines addressing various aspects of malignant gastric-outlet-obstruction management were identified by searching the PubMed, TRIP database and web-searching using terms including ‘luminal obstruction’ OR ‘gastric outlet obstruction’ OR ‘obstruction of the lumen’. We extracted information on relevant interventions or symptom-management approaches from each included guideline to populate the recommendations table.
Additional primary evidence search
We hand-searched reference lists from all included clinical guidelines to identify and evaluate evidence supporting relevant recommendations within the guideline. To capture emerging research, and ensure recent and relevant studies not yet reflected in existing guidelines were included, we conducted a structured MEDLINE search using predefined terms (Jan 2024, Supplemental Material 2). During searching, we paid attention to relevant gaps or limitations in the cited evidence base.
Analysis and strength of evidence
We identified relevant recommendations to management of malignant gastric-outlet-obstruction from included guidelines and evidence, and each treatment recommendation was allocated to a practice recommendation category of ‘Do’, ‘Do not’ or ‘Don’t know’ based on the direction and strength of the supporting evidence. 11 We determined the strength of evidence for commonly recommended treatments and strategies. A ‘strong’ recommendation was made if a large and consistent body of evidence existed, such as a systematic review specific to the malignant gastric-outlet-obstruction cohort. Comparatively, solid empiric evidence from one or more papers or a guideline advocacy justified a ‘moderate’ recommendation, with a ‘tentative’ recommendation made if limited empiric evidence existed (Table 2).
Category of practice recommendation and strength of evidence.

Results
We identified 19 relevant guidelines from 10 organisations in the US, the UK, Europe and Worldwide. Table 3 summarises each guideline along with its recommendations, while Table 4 details the strength of those recommendations. The additional primary evidence supporting our practical recommendations can be found in Supplemental Material 3.
Summary of guidelines and recommendations.

Pink shading denotes areas addressed within each guideline.
Both the ASGE and NICE in the table above each comprise several relevant sub-guidelines, as follows:
ASGE (American Society for Gastrointestinal Endoscopy) Guidelines:
a. Role of Endoscopy in Premalignant & Malignant Gastric Conditions (2015)
b. Role of endoscopy in the management of benign and malignant gastroduodenal obstruction (2021)
c. Endoscopy in Solid Pancreatic Neoplasia (2016)
d. Role of Endoscopy in Gastroduodenal Obstruction and Gastroparesis (2011)
e. Adverse Events of Upper GI Endoscopy (2012)
f. Appropriate Use of GI Endoscopy (2012)
g. Adverse Events Associated with EGD and EGD-Related Techniques (2022)
NICE (National Institute for Health and Care Excellence) Guidelines
a. NG83: Oesophago-Gastric Cancer: Assessment and Management (2018, updated 2023)
b. CG140: Palliative Care for Adults: Strong Opioids for Pain Relief (2012)
c. NG31: Care of Dying Adults in the Last Days of Life (2015)
d. Clinical Knowledge Summaries (CKS)
Note. Please refer to the online version of the article to view this table in colour.
Practice recommendations and strength of evidence.
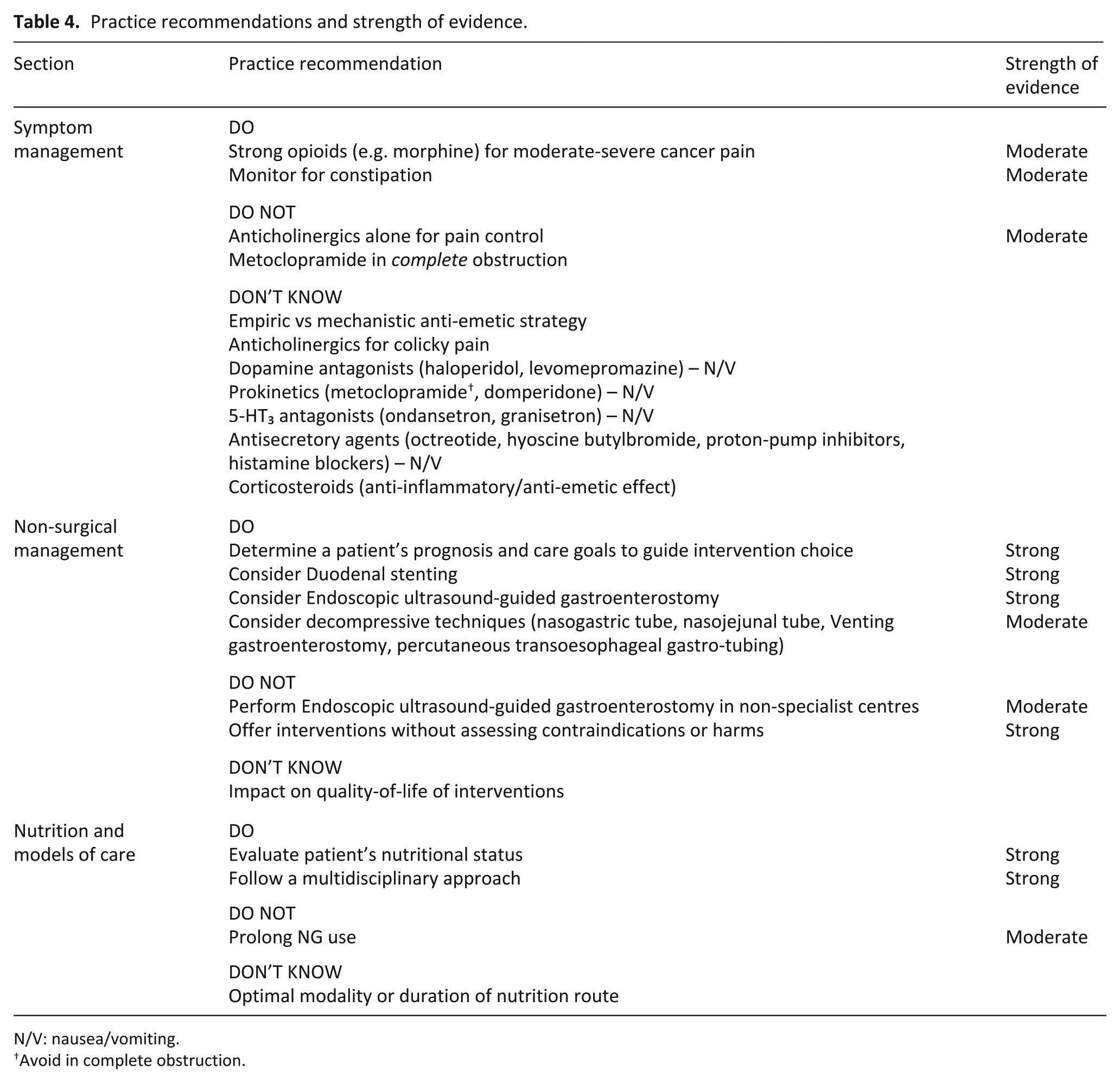
N/V: nausea/vomiting.
Avoid in complete obstruction.
Symptom management
DO strong opioids (e.g. morphine) for moderate-severe cancer pain and monitor for constipation
Pain is a common symptom in advanced cancers 12 but no trials address pain management in malignant gastric-outlet-obstruction. Current guidance is extrapolated from general palliative care guidelines, advocating individualised treatment that considers pain characteristics, patient response and the optimal drug delivery – particularly given that patients with malignant gastric-outlet-obstruction may be unable to take medications orally. In general, strong opioids – most commonly morphine – are endorsed by NICE CG140, the BNF and PCF as first-line treatment for moderate to severe cancer-related pain, with careful titration and monitoring for constipation and reduced gut motility.13,14 Typical starting regimens include morphine sulphate 5–10 mg subcutaneously every 4 h or continuous subcutaneous infusion (CSCI) 20–30 mg/24 h with breakthrough doses. Alternative agents include oxycodone and hydromorphone. Escalation without managing side-effects may worsen obstruction-related symptoms. 15
DON’T KNOW anticholinergics for colicky pain
For persistent colicky pain associated with bowel obstruction, MASCC and PCF advise to consider anticholinergic medication. Some studies have shown that hyoscine butylbromide (20 mg SC QDS or 60–120 mg/24 h CSCI) may provide beneficial antispasmodic effects when used in combination with opioids, but data is of low quality. 16 The BNF/PCF advise glycopyrronium (0.2–0.6 mg SC QDS or 0.6–2.4 mg/24 h CSCI) as an alternative, particularly when a patient’s cardiac profile precludes higher doses of anticholinergics. However, support for these agents are largely based on expert consensus and studies in bowel obstruction rather than high-quality trial evidence specific to the malignant gastric-outlet-obstruction population.
DO NOT anticholinergics alone for pain control
Avoid using these agents as monotherapy for pain control; their role is to complement opioid analgesia rather than replace it. 16
DON’T KNOW Empiric versus mechanistic anti-emetic strategy
Around 80% of malignant gastric-outlet-obstruction patients suffer from nausea and vomiting due to blockage at the distal stomach, pylorus or proximal duodenum, leading to retention of food and secretions, gastric distension, and an emetic reflex. 17 Anti-emetic therapy in advanced cancer can be approached either mechanistically – selecting agents based on the specific receptor pathways implicated in the emetic process – or empirically by trialling different drugs, though no studies have directly compared these methods.18,19 Different antiemetics vary in receptor affinities, side-effects and interactions; although current guidelines favour an aetiology-based approach to tailor treatment, limited data indicate either strategy offers similar benefit. 20 Importantly, the authors agreed that there is insufficient evidence to draw firm conclusions about the efficacy of drugs commonly recommended for managing malignant gastric-outlet-obstruction symptoms.
DON’T KNOW dopamine antagonists (haloperidol, levomepromazine) – N/V
Haloperidol and levomepromazine control chemical or metabolic nausea without impairing gut motility – making them safe when prokinetics are contraindicated. 18 However, their antiemetic effect is limited to central mechanisms and doesn’t address the mechanical obstruction that triggers the emetic reflex in malignant gastric-outlet-obstruction. Based on the NICE CKS, MASCC and BNF/PCF guidelines, haloperidol (0.5–3 mg/24 h via CSCI) is recommended in patients with persistent nausea, with evidence from small palliative trials. 21 Additionally, BNF/PCF endorse levomepromazine (6.25–25 mg/24 h via CSCI) as a broad-spectrum antiemetic in refractory cases, but caution is advised due to potential sedation and hypotension. Data is too limited to draw firm conclusions about the efficacy of these drugs in patients with malignant gastric-outlet-obstruction.
DON’T KNOW prokinetics (metoclopramide†, domperidone) – N/V
Metoclopramide and domperidone enhance gastric emptying making them effective for nausea and vomiting due to stasis, but they’re contraindicated in mechanical obstruction where increased contractions may worsen symptoms. 22 MASCC and NICE NG31 guidelines recommend that in cases of partial obstruction, metoclopramide (30–60 mg/24 h via CSCI or 10 mg SC TDS) or domperidone (10–20 mg PO TDS) could be used to improve GI transit. Whilst small case studies have indicated some benefits in bowel obstruction, 23 robust conclusions cannot be drawn for patients with malignant gastric-outlet-obstruction. Moreover, in clinical practice, it is often challenging to differentiate between partial and complete obstruction, a difficulty that complicates treatment decisions in palliative care. 24 Furthermore, in individuals for whom anticholinergics are being used for antisecretory effects or pain control, metoclopramide’s opposite action may worsen colicky pain. 25
DO NOT metoclopramide in complete obstruction
Avoid metoclopramide in complete obstructions because of the increased risk of bowel perforation. Although this risk has not been demonstrated in trials, it is widely accepted based on the known mechanism of action of metoclopramide.
DON’T KNOW 5-HT₃ antagonists (ondansetron, granisetron) – N/V
Ondansetron and granisetron block central and peripheral emetic pathways, but their constipating, anti-motility effects limit use in obstruction-related vomiting. 20 A small phase II trial in patients with bowel obstruction suggested that granisetron, when combined with other antiemetics, may offer benefit, but the evidence is too limited to draw firm conclusions with regards to gastric-outlet-obstruction. 26 These drugs warrant cautious use in the setting of gastric-outlet-obstruction as further impairment of peristalsis can worsen stasis. 5
DON’T KNOW antisecretory agents (octreotide, hyoscine butylbromide, proton-pump inhibitors, histamine blockers) – N/V
The pathophysiology of malignant gastric-outlet-obstruction involves the accumulation of gastric secretions that contribute to the emetic reflex – thus, an antisecretory approach is logical. MASCC and NICE NG31/PCF guidelines recommend octreotide (0.25–0.5 mg/24 h via CSCI) – a somatostatin analogue – as the first-line agent for reducing gastrointestinal secretions in patients with large-volume vomiting. This recommendation is primarily based on small, low-quality RCTs in malignant bowel obstruction.27,28 Although hyoscine butylbromide is used for its antispasmodic and antisecretory properties, a small trial suggests its antiemetic effect may be inferior to octreotide, making it more appropriate for use as an adjunct rather than first-line. 29 Small RCTs have shown some benefit of the role of PPI and histamine antagonists to reduce intestinal secretions in malignant bowel obstruction, supporting their inclusion in ASCO guidelines; however, it remains unclear whether these findings can be extrapolated to the malignant gastric-outlet-obstruction population. 30
DON’T KNOW corticosteroids (anti-inflammatory/anti-emetic effect)
ASCO guidelines 31 recommends considering steroids for their anti-inflammatory, indirect analgesic and central antiemetic effect. There is limited high-quality evidence for the use of corticosteroids (e.g. dexamethasone 4–8 mg PO/IV/SC daily) to control nausea and vomiting in malignant gastric-outlet-obstruction but their use is supported by small trials in cancer-related obstruction and is recommended on an individual basis. 32
Non-surgical management
DO determine patient’s prognosis and care goals to guide intervention choice
Evidence and guidelines support two minimally invasive palliative techniques – duodenal stenting and endoscopic ultrasound-guided gastroenterostomy – for malignant gastric-outlet-obstruction in patients with limited life expectancy. Interventions should align with patient prognosis. This review targets those with an estimated life expectancy of ⩽3 months – recognising clinician estimates can be inaccurate. 13 Objective markers (declining performance status, metastatic burden, hypoalbuminemia) and validated prognostic models may refine predictions. 33 Following prognostic evaluation, we advise clinicians to engage in discussions that clarify patient wishes, weigh the risks/benefits of each intervention, and set realistic expectations for symptom control and quality of life. 13
DON’T KNOW impact on quality-of-life of interventions
Although many studies report high technical and clinical success, 34 few assess quality of life and inconsistent quality of life measures hinder comparison. Standardising quality of life endpoints in future trials will clarify these the value of these interventions for palliative care.
DO consider endoscopic duodenal stenting
Endoscopic duodenal stenting using self-expanding metal stents is a minimally invasive procedure in which a flexible, metal-mesh tube is inserted via an endoscope into the blocked duodenum to keep it open, allowing food to pass through. This technique was first described in the early 1990 s as a palliative treatment for malignant gastric-outlet-obstruction and offers rapid symptomatic relief – with cessation of vomiting and a return to oral diet within days.35,36 Multiple Cochrane reviews and guidelines (NICE NG83, ESMO, ASGE) endorse stenting as first-line palliation for unresectable disease, citing a low serious-complication rate (1%–3%).37,38 However, stent dysfunction (20–30 % of patients) from tumour ingrowth, overgrowth or migration often necessitates re-intervention and limits durability.39,40 Consequently, stenting should not be offered to patients with an anticipated longer survival (e.g. >3–4 months) who might benefit from a more durable bypass, such as surgical gastrojejunostomy or emerging alternatives like endoscopic ultrasound-guided gastroenterostomy. Furthermore, stenting should be avoided in cases of multi-level obstruction or extensive malignant infiltration, as these factors are associated with a higher risk of stent failure.
DO consider endoscopic ultrasound-guided gastroenterostomy
Introduced in 2015, endoscopic ultrasound-guided uses a lumen-apposing metal stent to create an internal bypass from the stomach to the jejunum, combining the durability of surgical gastrojejunostomy with the low morbidity of endoscopy. 41 In expert centres, technical and clinical success rates range from 85% to 95%, with patients resuming liquids within 1–2 days and advancing diets rapidly. 42 A growing body of evidence – including a meta-analysis of 12 studies involving 285 patients reported 92% technical success, 90% clinical success, low rates of symptom recurrence/unplanned reintervention (9%) and adverse events (12%) 43 – suggests that Endoscopic ultrasound-guided gastroenterostomy may be superior to traditional duodenal stenting. Meta-analyses show it has lower odds of recurrent obstruction than duodenal stenting, and an RCT found shorter hospital stays (4 vs 6 days; p < 0.05) with endoscopic gastroenterostomy, suggesting faster recovery – though more high-quality trials are needed. 44
Despite its promising evidence, endoscopic gastroenterostomy has not yet been acknowledged in many guidelines including NICE, highlighting a need for updates to keep pace with evolving technology. Comparative data suggest endoscopic ultrasound-guided gastroenterostomy may offer superior symptom relief and fewer reinterventions than duodenal stenting, but its slightly lower technical success mandates performance by expert endoscopists in high-volume centres (ESGE 2022). 45 Long-term efficacy of this procedure versus duodenal stenting is still under evaluation, with current recommendations drawn from retrospective and network meta-analyses.46,47
DO NOT perform endoscopic ultrasound-guided gastroenterostomy in non-specialist centres
Endoscopic ultrasound-guided gastroenterostomy is contraindicated in patients with diffuse peritoneal metastases or massive ascites, as these conditions may compromise the creation of a stable anastomosis (ESGE 2022).
Additionally, this procedure may require general anaesthesia, whereas duodenal stenting is usually performed under moderate sedation with local anaesthetic support; this higher potential anaesthetic burden can limit suitability in those with comorbidities or frailty. 42
DO NOT offer interventions without assessing contraindications or harms
Recent meta-analyses and reviews indicate that stent placement for malignant gastric outlet obstruction is generally safe, though clinicians should remain aware of potential risks – including perforation (1.2%), stent migration (4%), infection (0.5%), bleeding, stent occlusion (12.6%) and the need for re-intervention.48–50
DO consider decompressive techniques (NG, NJ, venting gastrostomy, percutaneous transoesophageal gastro-tubing)
Effective decompression is important in malignant gastric-outlet-obstruction to control vomiting, relieve distension and improve comfort. For acute decompression, a nasogastric tube is recommended as a temporary measure to rapidly evacuate gastric contents 51 by NICE NG31, MASCC and ASGE guidelines. Some patients may be suitable for a venting gastrostomy (PEG) for continuous drainage – an option endorsed by the PCF, NICE NG31 and ASGE guidelines –though evidence is limited to observational data. 13
Emerging techniques such as percutaneous transoesophageal gastro-tubing (PTEG), which involves creating a decompressive channel through the oesophagus into the stomach, have shown promising early results in a small randomised trial comparing NG tube decompression, 52 although further high-quality studies targeting the malignant gastric-outlet-obstruction cohort are needed before its adoption.
DO NOT prolong nasogastric tube use
Prolonged nasogastric tube use should be avoided due to risks of discomfort, aspiration and mucosal injury 53 and nasogastric decompression in contraindicated in cases of multi-level obstruction or extensive malignant infiltration.
Nutritional management and models of care
DO evaluate patient’s nutritional status
Chronic vomiting and reduced oral intake in malignant gastric-outlet-obstruction lead to malnutrition, making early nutritional intervention important for maintaining functional status and quality of life. Guidelines (NICE NG83; ESMO; MASCC; ESPEN) advocate early nutrition screening and tailored support – from dietary changes and supplements to enteral feeding (NG/PEG) or parenteral nutrition. Enteral feeding is preferred over parenteral, with duration of support adjusted for prognosis and treatment risks.
DON’T KNOW optimal modality or duration of nutritional support
There is little evidence as to specific approaches to nutrition, in particular there is little evidence to support effectiveness of parenteral nutrition in cancer end of life care. Recommendations are largely extrapolated from studies in broader oncological populations rather than from RCTs focused solely on malignant gastric-outlet-obstruction. 54 Guidance from ESPEN 55 and palliative-care frameworks emphasises the importance of early nutritional assessment to guide decision-making, balancing the potential benefits and burdens of nutritional interventions within the individual’s overall illness context. Assessment may incorporate biochemical markers such as serum albumin and validated tools including the Patient-Generated Subjective Global Assessment (PG-SGA) and the Global Leadership Initiative on Malnutrition (GLIM) criteria, 56 which help identify malnutrition and inform supportive care planning. Further rigorous studies are needed to define the role and impact of nutritional interventions in this specific patient group.
DO follow a multidisciplinary team approach
All guidelines (NICE NG83, ESMO, MASCC, and ASGE) strongly recommended that complex cases of malignant gastric-outlet-obstruction should be managed by an experienced multidisciplinary team. Involving oncologists, gastroenterologists, surgeons and palliative care specialists leads to a more holistic and individualised treatment plan that improves symptom control, reduces hospital admissions, and enhances quality of life. Evidence indicates that early integration of palliative care can not only reduce symptom burden but may also improve survival. 57 Comprehensive assessment – including evaluation of vomiting characteristics, pain, nutritional status and comorbidities – is essential to guide clinical decision-making.
Limitations
Whilst we are confident we have identified relevant guidelines, the scoping review of primary data was not as comprehensive as a full systematic review. Therefore it is possible that some primary evidence is not included. However much of the primary evidence is already cited in relevant guidelines. Furthermore, evidence underpinning pharmacological recommendations is largely based on extrapolation from bowel obstruction and advanced cancer guidance, as there is little data specific to the malignant gastric-outlet-obstruction population. Finally, we have not summarised evidence on cancer directed therapies such as chemotherapy and radiotherapy which may be relevant to certain patients in this cohort.
Discussion
Malignant gastric outlet obstruction often arises in advanced cancers and requires an integrated, symptom focused approach to care. Consensus supports a multidisciplinary approach, bringing together gastroenterology, oncology, interventional, nutrition and palliative-care teams. Early evaluation of performance status, prognosis, comorbidities and patient wishes is essential to align interventions with patient goals and expected benefit.
Robust randomised trial data support endoscopic interventions as safe and effective for rapid symptom relief, with endoscopic duodenal stenting remaining the first-line option in many guidelines. Emerging techniques, such as Endoscopic ultrasound-guided gastroenterostomy, show promise for improved stent patency, reduced need for reintervention and potentially lower recovery time. By author consensus, we therefore recommend that endoscopic ultrasound-guided gastroenterostomy be incorporated into future guideline revisions.
Pharmacological therapies remain central for symptom control, although evidence is largely extrapolated from malignant bowel obstruction and general palliative-care literature. Agents such as opioids, dopamine antagonists, prokinetics, anticholinergics, antisecretory agents and corticosteroids are used empirically to relieve pain, nausea, and secretions, but comparative data is limited. Further research is needed to determine optimal drug dosing and combinations specific to malignant gastric-outlet-obstruction, and treatment should be tailored to individual tolerance and response. Oncology input is also important, as some patients with malignant gastric-outlet-obstruction may be suitable for palliative chemotherapy or radiotherapy to help reduce tumour burden. 58 While not explored in this review, we acknowledge its role as part of overall multidisciplinary management.
When invasive interventions are unsuitable, gastric decompression through a nasogastric tube or venting gastrostomy remain an important palliative option. Alongside symptom relief, early attention to nutrition is crucial, as poor nutritional status limits function and tolerance of further interventions and treatment. However, optimal nutritional strategy is uncertain, with limited evidence for parenteral nutrition; care should be guided by patient goals and quality-of-life considerations.
Overall, guidance for malignant gastric-outlet-obstruction remains fragmented and it is unclear whether current recommendations enhance quality-of-life in patients with limited life expectancy. Inconsistent evaluation of quality-of-life outcomes reduces understanding of patient benefit, highlighting the need for greater focus on patient-reported outcomes in future research. Rigorous pharmacotherapy trials and prompt guideline updates that incorporate emerging endoscopic techniques are essential to define an optimal, integrated care pathway and improve outcomes for this complex patient group.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163251403430 – Supplemental material for Palliative management of malignant gastric outlet obstruction: A practice review
Supplemental material, sj-docx-1-pmj-10.1177_02692163251403430 for Palliative management of malignant gastric outlet obstruction: A practice review by Tanvi Acharya, Anna Spathis, Edmund Godfrey, Gareth Corbett, Isla Kuhn and Simon Noah Etkind in Palliative Medicine
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

