Abstract
Background:
There is growing demand for home-based palliative care because of patient preference, and increased number of deaths. Optimal models for community-based palliative and end-of-life care are unknown.
Aim:
To identify, synthesise and describe review-level evidence to better understand models of palliative and end-of-life care for adults living at home, and examine components of these models and their association with outcomes.
Design:
Systematic umbrella review, using key concepts established a priori from Firth et al. and Brereton et al.’’s model descriptions. Quality assessment used AMSTAR-2 or equivalent.
Data sources:
MEDLINE, EMBASE, CINAHL, Cochrane Database, Epistemonikos (inception – 2024), supplemented by CareSearch, PROSPERO and citation searches.
Results:
From 6683 initial papers, n = 66 reviews were included. Seven models of care were identified; by setting (in-home, outpatient); type of professionals (specialist, integrated, non-specialist); or mode (telehealth, education/training). Components included: holistic person-centred assessment, skilled professionals, access to medicines/care/equipment, patient/family support, advance care planning, integration of services, virtual/remote technology and education. We categorised outcomes into: (i) patient outcomes, (ii) family/informal caregiver outcomes, (iii) professional outcomes and iv) service utilisation/cost outcomes. The ‘in-home palliative care’ model was most researched with good evidence of positive benefit. Specialist and integrated models of care were next most researched, with evidence of improved patient and service utilisation outcomes. Cost-effectiveness evidence was lacking.
Conclusion:
This meta-level evidence supports provision of in-home palliative care, with most review level evidence showing positive effect on patient outcomes. There was also evidence to support specialist palliative care and integration of primary palliative care with specialist support.
Keywords
Care at home for people approaching the last months or year of life has become increasingly important in recent years, due to the increase in deaths, multimorbidity and preference of the majority for care at home.
Individual reviews of the evidence on palliative and end of life care at home have been undertaken, with some evidence of benefit.
This paper reports the overall evidence, which largely supports in-home palliative care, especially if delivered via specialist palliative care models or integrated palliative care models (where integration refers to coordination between specialist and non-specialist services).
It also provides evidence of benefit for education and training, both for informal family carers and for professionals.
Detailed narrative synthesis links models of care, with their components and sub-components and related outcomes.
There is clear evidence supporting provision of in-home palliative care, with common components related to addressing (and delivering positive impact on) patients’ symptoms, psychological distress and functional status.
Introduction
There is growing demand for home-based palliative care because of patient and family preference, growing demand (increased number of deaths) and policy imperative towards community-based care. This means care at home for people approaching the last months or year of life has become increasingly important.1,2 Unplanned and urgent home and community-based care is increasingly also needed and used by those with palliative and end of life care needs.3,4 Enabling individuals to be cared for and die at home has been identified as a top priority for patients and their family carers, as well as for policy makers.5–7
Palliative care provided at home is delivered by a range of different professionals. This includes those who may provide care day-to-day as part of their wider roles (such as general practitioners/family physicians, community/district nurses, pharmacists, or allied healthcare professionals) and those who specialise in palliative and end of life care (such as specialist palliative care doctors, nurses, allied health professionals, and social workers). 8 An essential aspect of enabling patients to stay at home is the care provided around the clock (including care ‘out of hours’; i.e. between 6 pm and 8 am, weekends and public holidays), which accounts for over two-thirds of a patient’s week. 9 Good quality out-of-hours care has also been identified as an important component of cost-effective care. 10 As care provided inside and outside of normal working hours comprises many different service types (including expertise and delivery), there is limited understanding of which service models and components of care work best and for whom, and under what circumstances.8,10,11 Evidence is necessary to inform the best type of care needed outside of normal working hours to avoid crises and support people to stay in their preferred place of care, 5 and how community care can be a realistic alternative to hospital-based care.3,12 From a service planning perspective, enabling people to be cared for and die at home in line with their preference is also likely to reduce (proportionately very expensive) hospital admission costs.3,12–14
Existing reviews of reviews have focused on (i) identifying the range of models of palliative care across different care settings (including care provided at home),10,15 (ii) identifying components of in-home end-of-life care programmes and examining their effectiveness (focusing on the last months or days of life), 11 (iii) quantifying the impact of home palliative care services on whether people will die in a home setting 13 or (iv) are focussed solely on out-of-hours services. 16 However, an over-arching review and synthesis of the evidence on palliative and end-of-life care delivered to adults living at home (including models, components of care and associated outcomes) has not been undertaken.
Research aim
This review therefore aimed to identify, synthesise and describe review-level evidence on models of palliative and end-of-life care for adults living at home, and examine the components of these models and their association with outcomes.
Review Question: From existing evidence, can we characterise and better understand models, components and related outcomes for adults living at home with palliative care needs?
Methods
Study design
A systematic umbrella review of reviews – useful for combining findings from a large volume of reviews into a single synthesis of evidence 17 – and registered the review protocol on the international prospective register of systematic reviews (PROSPERO Registration Number: CRD42022362156). Reporting adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 statement 18 and the Prognostic factor Review Outcomes in Systematic reviews (PRIOR) checklist 19 (see Supplemental Material 1). We also adopt the original seven critical items established by Shea et al. 20 The main concepts considered as part of this review are defined in Box 1.
Definitions and considerations around key concepts for this review.

Eligibility criteria
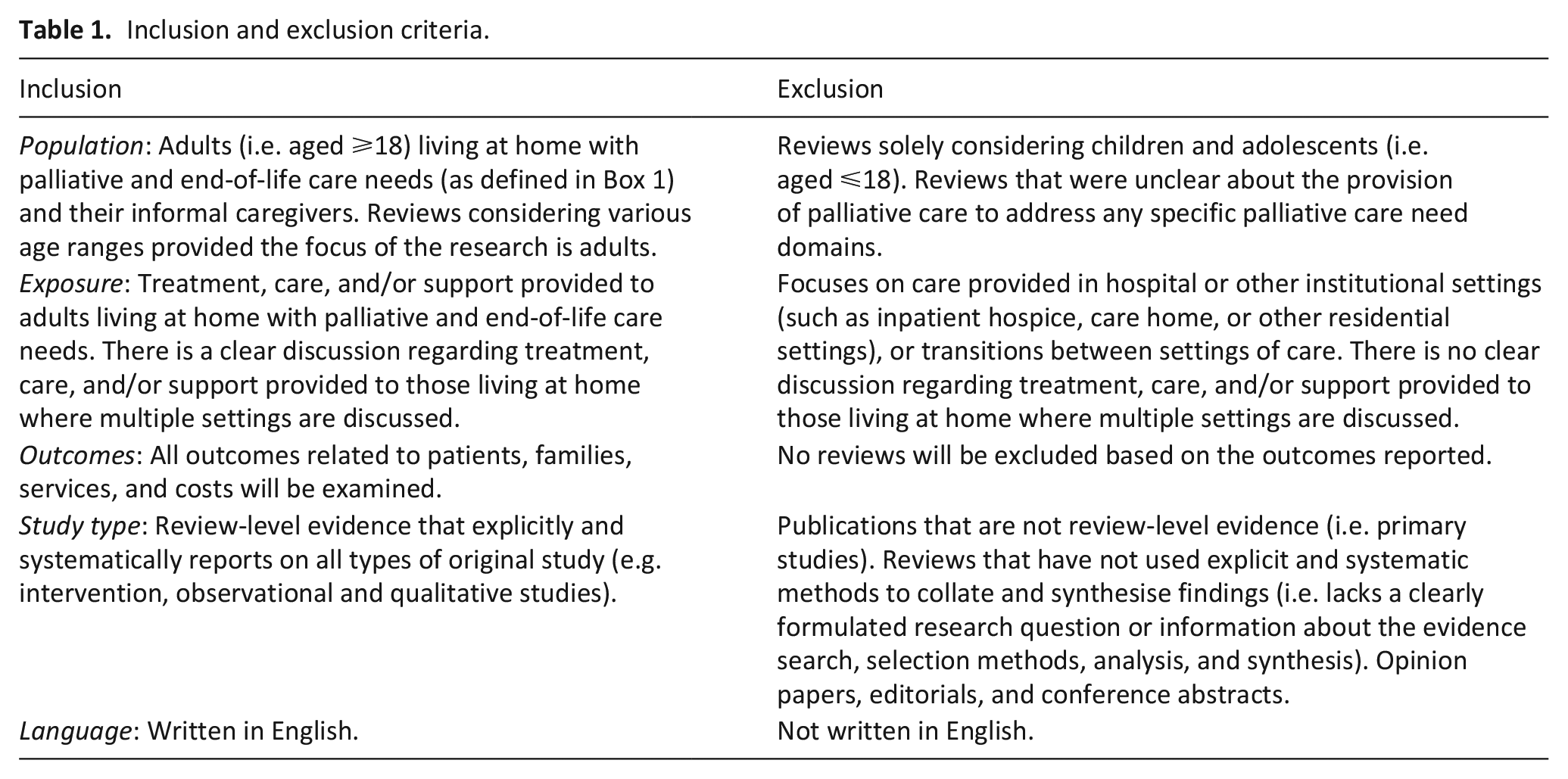
We included reviews which reported evidence on home or community-based palliative and end-of-life care. Detailed inclusion and exclusion criteria are shown in Table 1.
Inclusion and exclusion criteria.
Search strategy
The search strategy was developed with an information specialist (SG) drawing on relevant reviews to refine search terms. The search strategy was refined using ‘sentinel’ reviews (reviews we expected to find through searching). Hence, scoping searches were used to refine our approach; for example, initial searches revealed that although most of the gold standard papers were identified, some reviews that report on out-of-hours palliative and end-of-life care within a primary care context were not identified. Therefore, final searches used database appropriate subject headings terms and keywords relating to palliative care, primary care, home-based or community setting, and out-of-hours (see Supplemental Material 2 for all search strategies for databases used), combined with study design filters for reviews.
MEDLINE, EMBASE (via OVID), CINAHL (via EBSCOHost), Cochrane Databases of Systematic Reviews (via Cochrane library) and Epistemonikos (via www.epistemonikos.org) were originally searched from inception to September 2021, with updates in December 2022, and September 2023. We also searched the CareSearch Project systematic review collections which consolidates online palliative care knowledge 31 and the following systematic review collection subjects were reviewed and cross-checked for missing reviews: site of care, models of service delivery, place of death, general practitioners and occupational therapists. In addition, PROSPERO was used as a source to check for any additional reviews. The date of the last search for all sources was August 1, 2024. Forward and backward citation searching of included reviews using Citation Chaser was used to identify any additional reviews that potentially meet the inclusion criteria. Returned records from the search were imported into EndNote 21 32 for deduplication, and then transferred to another reference management software; Rayyan. 33 This software allowed for remote working and collaboration within a distributed team.
Review selection
The titles and abstracts of retrieved results were independently screened by at least two review authors (SP, RP, CO, AM and AS) against the inclusion criteria. The full texts of potentially eligible reviews were then retrieved and independently assessed for eligibility by two review authors (SP, RP, CO, AM and AS). Where there was any disagreement between the two review authors over the eligibility, this was resolved through discussion with a third review author (FM). Upon confirmation of eligibility of the reviews, primary studies included in the eligible reviews were assessed for overlap using the Corrected Covered Area method by Pieper et al. 34 This is an established method effective in reducing overestimation of the strength of evidence in systematic reviews.
Data extraction
The development of the data extraction template was guided by the definitions provided by Firth et al.27,28 and Brereton et al., 10 and considering the descriptive picture of practice that was likely to be provided by the authors of reviews (see Box 1). The development of the data extraction form was an iterative process that considered the models and components of care described and reported within each review, including: target population of the review (i.e. care recipients), setting of care, health and social care professionals involved in delivering care (including presence of multiple disciplines), the intervention and objectives of care (including mode of delivery, and timing and duration), outcomes reported in the review, and the clinical effectiveness and cost-effectiveness of the interventions (i.e. models of care and components). To identify and manage primary study overlap at the level of data extraction following the method by Pieper et al., 34 primary studies of included reviews were imported into Zotero, 35 and assessed for duplicates using EndNote. 32 To identify unique studies, their occurrence in each review was mapped as ‘1’ if present and ‘0’ if absent. These and extraction of data from the reviews were managed using Microsoft Excel (2019). 36 Five review authors (SP, RP, CO, AM and AS) independently completed data extraction from the included reviews, including the models of care, components and outcomes. 10% were double extracted and compared at the start of extraction to help standardise extraction. Any discrepancies identified were resolved through discussion.
Quality assessment
The quality of included reviews was assessed independently by at least two review authors (TC, AM, JY and CO) depending on the methodology of the included review using one of the assessment tools:
For reviews which were systematic (i.e. systematic reviews, rapid systematic reviews, umbrella reviews conducted systematically, and integrative reviews conducted systematically), we used A Measurement Tool to Assess Systematic Reviews (AMSTAR-2). 20 This is a 16-component critical appraisal tool for systematic reviews that include randomised and non-randomised studies of healthcare interventions that generates a descriptive judgement of a review’s quality by evaluating critical and non-critical domains, has been recommended for its precision, critical domains, risk of bias assessment and wide acceptability.37–39 We classified the quality appraisal results from AMSTAR-2 as critically low in quality, low in quality, moderate or high in quality, depending on the severity of the flaws. Critically low quality indicates multiple flaws with or without weaknesses; low quality indicates one flaw with or without non-critical weaknesses; moderate indicates more than one non-critical weakness but no flaw; and high quality indicates no or one non-critical weakness without flaw. Pieper et al. demonstrated that assessments of which domains are considered critical can vary, which can impact on the overall evaluation of a review’s quality – using AMSTAR-2. 40
For narrative reviews that followed a systematic approach, we used the Scale for the Assessment of Narrative Review Articles (SANRA). 41 SANRA, a brief critical appraisal tool for the assessment of non-systematic articles has good specificity for narrative reviews, internal consistency and inter-rater reliability for multiple reviewers. 41 The six items that form the revised SANRA scale are rated in integers from 0 (low standard) to 2 (high standard), with 1 as an intermediate score. The maximal sum score is 12. 41
For scoping reviews that followed a systematic approach, we assessed them based on their adherence or not, to the Preferred Reporting Items for Systematic Reviews and Meta-analyses Extension for Scoping Reviews (PRISMA-ScR) checklist 42 or the Levac et al. methodology for scoping reviews. 43
No reviews were excluded from the synthesis due to poor quality. However, the quality assessments were used to inform critical reflection on the strengths and limitations regarding the robustness of the synthesis.
Data synthesis
The synthesis aimed to explain the models and components of care identified, whether any components identified could be related to evidence of effectiveness (i.e. any significant positive effects on any of the outcomes measured) or cost-effectiveness.
Due to expected heterogeneity in the included reviews, we conducted a narrative synthesis following the guidance outlined by Popay et al.. 44 The initial development of the data extraction form involved identifying models, components and outcomes within and across the reviews. Hence, we developed a preliminary synthesis to summarise and organise findings. Where reported, the definitions and descriptions of the reported models and components of care were summarised. The evidence reported in each review was tabulated using textual descriptions to provide a descriptive summary of the elements earlier described and explore relationships. The relationships between models and components, then models and outcomes were explored and summarised (e.g. chart/matrix) by quantifying the reviews that explored the identified model, component or outcome, and then developing an accompanying narrative synthesis. Applying exploratory subgroup analysis, we aimed to understand the differences in the components and outcomes and group these as subgroups.
Results
Sixty-six reviews were eligible for inclusion. Full details are shown in a PRISMA flow diagram 18 (Figure 1). See Supplemental Material 3 for list of excluded studies.
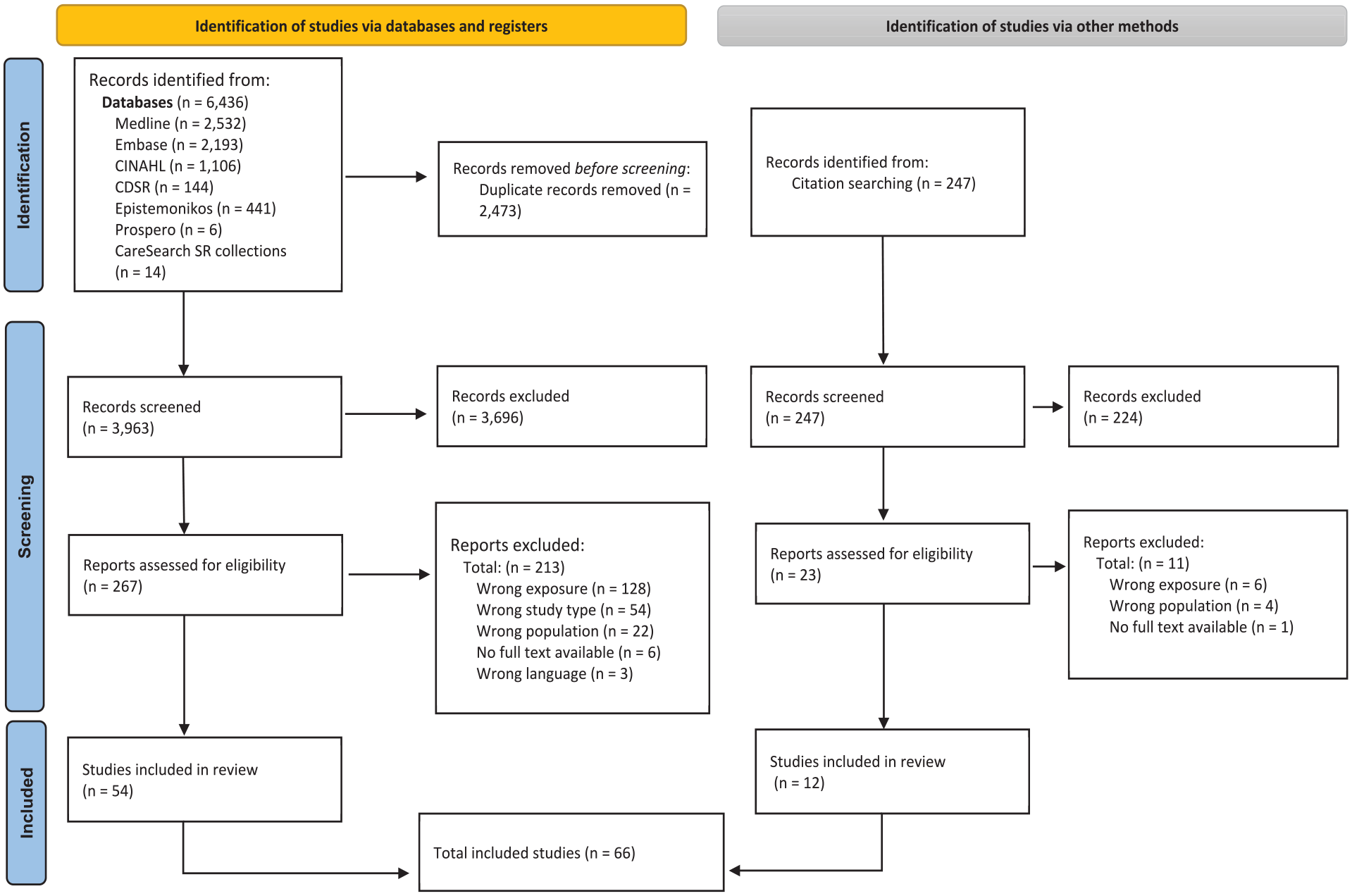
PRISMA flowchart . 18
Description of included reviews
The included reviews (n = 66) had varying publication dates with the majority published from 2019 onwards (n = 47).16,27,45–89 Fourteen reviews were published between 2013 and 201810,11,13,15,90–99 two between 2007 and 2012100,101 and three prior to 2007.102–104
The reviews were led by teams in Europe (n = 25),10,13,16,46,47,50,53,55,56,58,59,61,65,75–78,83,85,86,95,100,101,103,104 North America (n = 18),11,45,51,54,57,60,63,68,72,74,87,89,92–94,96,99,102 Australia (n = 7),15,52,69,71,90,91,98 Asia (n = 2),49,67 South America (n = 2)80,82 and Africa (n = 1). 81 Reviews were also published from more than one continent (n = 11).27,48,62,64,66,70,73,79,84,88,97
The study design of the included reviews was categorised for consistency into reviews which we judged were formal systematic reviews (n = 51),10,11,13,15,16,45,47–52,54–58,60–69,71,73,76,79–87,89,91–93,95,97–99,101–104 narrative reviews which adopted a systematic approach (n = 6),27,59,70,90,96,100 and scoping reviews which adopted a systematic approach (n = 9).46,53,72,74,75,77,78,88,94 The sources of data in the included reviews included experimental, quasi-experimental, feasibility studies, economic evaluations, cohort and observational studies, case studies, qualitative studies and mixed methods studies.
Both cancer and non-cancer patients requiring palliative and end-of-life care were considered in the included reviews. Advanced cancer and incurable/metastatic cancer patients were included,11,16,27,45,46,49,52,54,57,59,61,62,65–67,69,71,72,74,76,77,80,85,86,88,90,93–100,102–104 while the non-cancer populations with palliative needs encompassed those with neurodegenerative diseases or advanced heart failure, dementia, or chronic respiratory conditions.10,13,15,47,48,50,51,53,55,56,58,60,63,64,68,70,73,75,78,79,81–84,87,89,91,92,101 Comparators were the ‘usual care’ group. Further details of these reviews can be found in Supplemental Material 4: Characteristics of Included Reviews.
Detection of primary study overlap
From the included reviews, we identified n = 1604 primary studies. Upon deduplication, n = 410 were excluded and n = 1196 unique primary studies was used to determine the CCA. 34 We calculated the CCA 34 as:

where Nr = 1604 (total number of primary study occurrences across all included reviews)
N = 1196 (unique number of primary studies)
r = 66 (number of included reviews)
Pieper et al. 34 reports a CCA of 0.52% as slight overlap between primary studies included in reviews. This implies a minimal risk of double-counting and overestimation of evidence from these included reviews. Further details can be found in Supplemental Material 5: Detection of Primary Study Overlap in Included Reviews.
Results of quality appraisal
Fifty-two of the included reviews conducted a quality appraisal of their included studies using different tools while 14 did not conduct any quality appraisal. Using AMSTAR-2, 20 we appraised 52 reviews as follows: Critically low quality (n = 28)10,11,15,45,48,51,54,55,60–64,67,68,76,82,86,89,92,93,95,98,99,101–104 low quality (n = 14)16,47,50,57,58,65,69,71,73,80,83,87,91,97 moderate quality (n = 5),49,52,56,79,81 and high quality (n = 4).13,66,84,85 One scoping review was reported according to PRISMA guidelines, thus assessed with AMSTAR-2. 94 Using SANRA, 41 we appraised six narrative reviews as 9/12 (n = 2),96,100 11/12 (n = 1), 90 and 12/12 (n = 3)27,59,70 (higher scores represent better quality; categorisation e.g. to low/medium/high categories is not recommended). Six scoping reviews adhered to the PRISMA-ScR 42 checklist,46,53,72,77,78,88 while one did not adhere to PRISMA-ScR or any equivalent, 74 and one utilised Levac et al. 43 (which is a design approach and not a guideline), (which is methodology for scoping reviews instead of PRISMA-ScR checklist. 75 We did not exclude any review based on review quality. Further details can be found in Supplemental Material 4.
Models of care
We identified seven models of care from the included reviews, including ‘education and training’ as a ‘model of care’ for clarity, while recognising education and training is only indirectly (rather than directly) related to care. We categorised these models according to: setting of care; professional group delivering the care; mode of delivery of care; and education and training to support delivery of care (See Table 2). A number of reviews reported on multiple models of care.
Identified models of care.
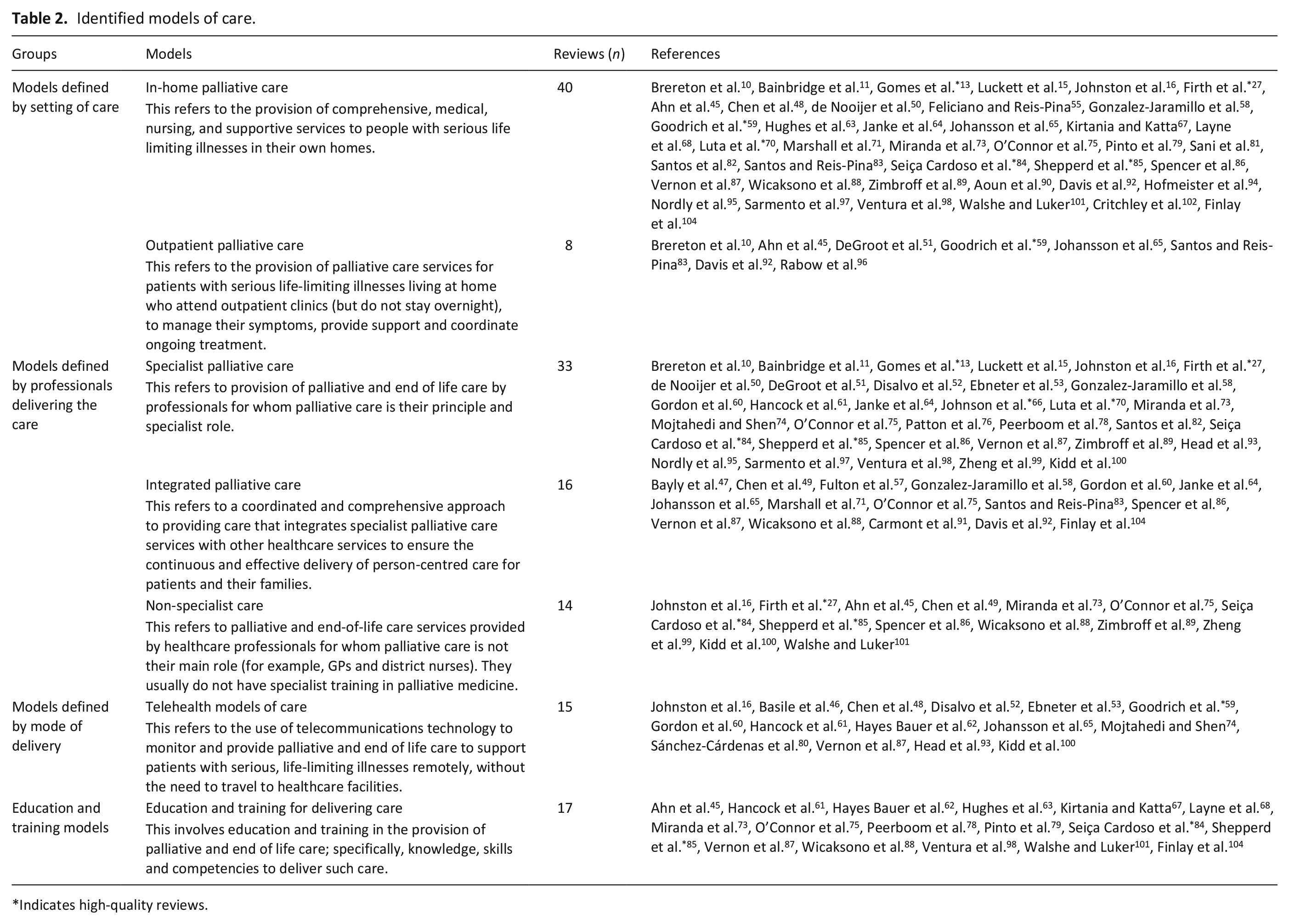
Indicates high-quality reviews.
The In-home model had the most evidence (n = 40 sources) while the outpatient model had the least (n = 8 sources).
Components and subcomponents of care
Within the seven models of care, we identified components and subcomponents. These components and subcomponents were derived from 59 of included reviews (seven reviews did not describe components or subcomponents). The main components (often common across the seven models) were: (a) holistic and person-centred assessment (n = 35 reviews), (b) skilled professional care (n = 40 reviews), (c) access to medicines, care and equipment (n = 16 reviews), (d) support for patients and their families (n = 43 reviews), (e) advance care planning (n = 27 reviews), (f) integration of services (n = 31 reviews), (g) virtual and remote technology (n = 19 included reviews) and (h) educational interventions (n = 32 reviews; see Table 3).
Numbers of included reviews reporting on the components and subcomponents of care, grouped by the seven different models of care we identified.
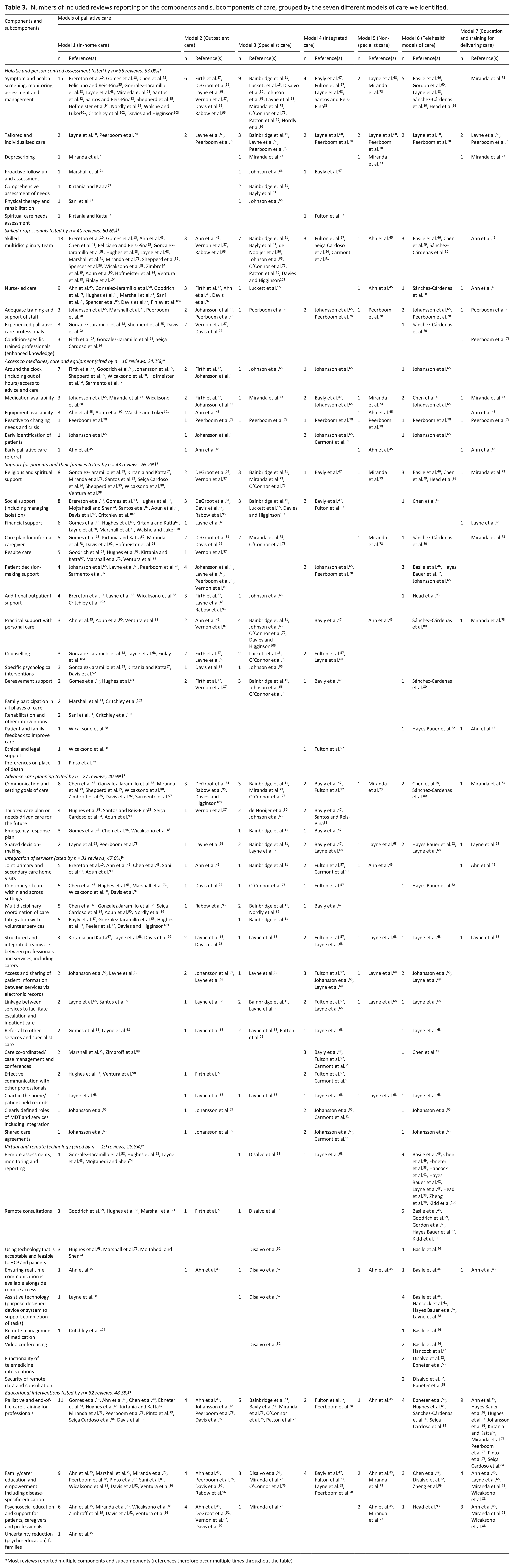
Most reviews reported multiple components and subcomponents (references therefore occur multiple times throughout the table).
Sixty-four subcomponents of care were identified and grouped under the appropriate component above. The most frequently reported subcomponents were ‘skilled multidisciplinary team’ (n = 33 reviews), under the component ‘skilled professionals’, and ‘symptom and health screening, monitoring, assessment and management’ (n = 31 reviews), under the component ‘holistic and person-centred assessment’. See Figure 2 for details of the components, grouped by the identified models of care.
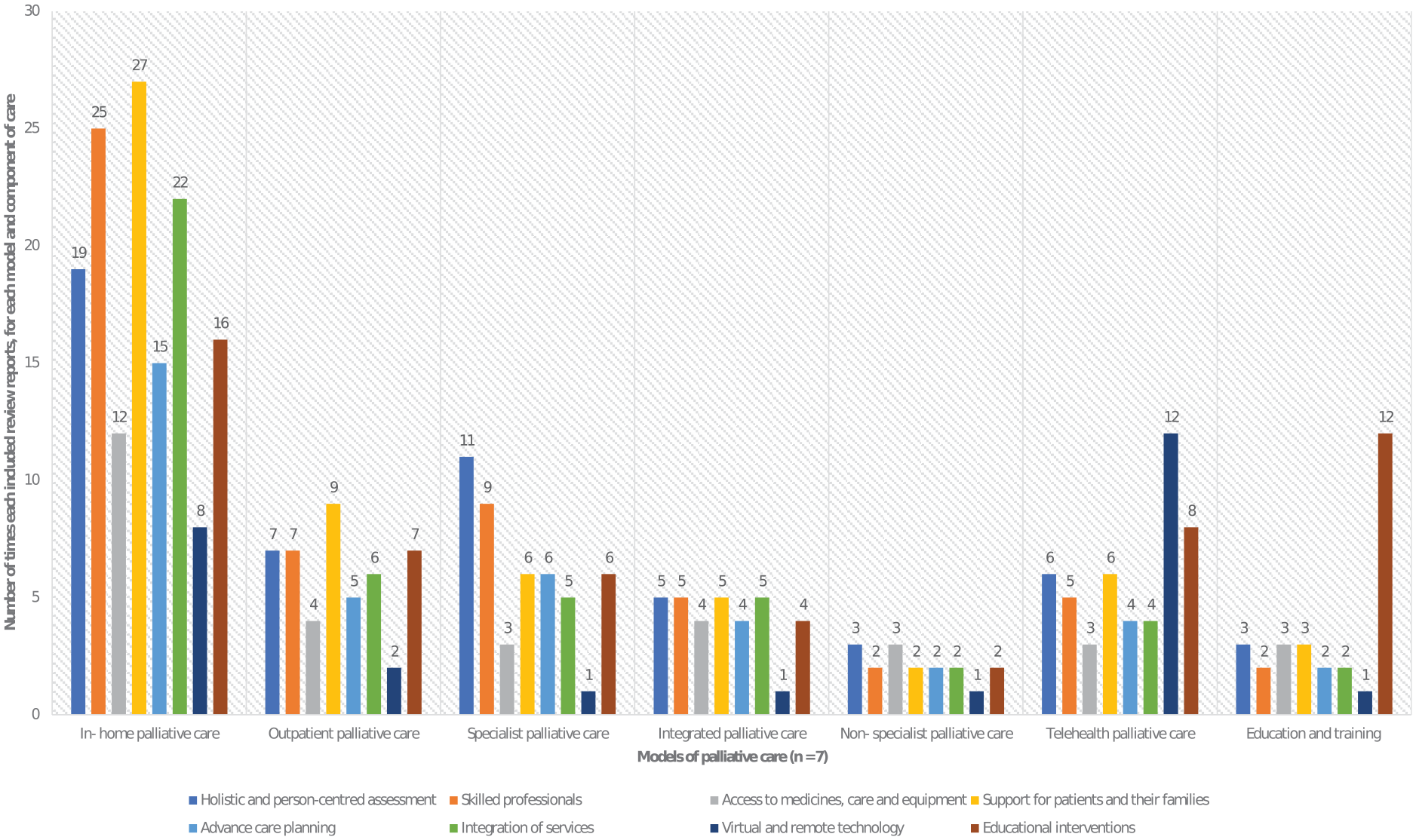
Identified components of care within the included reviews and their frequency across the identified models of palliative care (Included reviews which report on more than one model and/or component are counted more than once).
Outcomes of care
Twenty-four outcomes of care (see Table 4) were identified from all included reviews (these were reported by n = 55 reviews); we categorised these into four groups:
a) patient outcomes (reported by 46 reviews across all models of care): symptoms (general), behavioural symptoms, psychological distress, functional status, quality of life, advance care planning, survival, satisfaction with care, coping, adherence and autonomy and patient unmet needs;
b) family or informal caregiver outcomes (reported by 21 reviews across all models of care): include caregiver behavioural symptoms, caregiver psychological distress, caregiver grief, caregiver quality of life, caregiver burden, caregiver self-efficacy/competence, caregiver satisfaction with care, and caregiver unmet needs;
c) professional outcomes (reported by 10 reviews across all models of care): professional competence, and professional’s satisfaction with care;
d) service utilisation and costs (reported by 34 reviews across all models of care): place of death, hospitalisation, emergency department visits, and overall or specific healthcare costs.
Summary of reported evidence from the included reviews, grouped by model of care.

Indicates high-quality reviews.
The majority of the patient outcomes were reported in relation to the in-home (26 reviews), and specialist (21 reviews) models of care. The family or informal caregiver outcomes were mostly reported in the in-home (10 reviews) and education and training (eight reviews) models of care (where education or training was provided to families). Professional outcomes were mostly reported in the education and training (five reviews) model (where education and training were provided to professionals). Service utilisation and cost outcomes were mostly reported in relation to the in-home (25 reviews) model of care. Across all included reviews and models of care, the most common outcomes (reported by ⩾ 20 reviews and ordered by most common first) were: symptoms (general); satisfaction with care; overall or specific healthcare costs; quality of life; place of death; and hospitalisation.
Positive benefits were most commonly reported in relation to patient outcomes for in-home and specialist models of care; relatively less evidence was reported for the non-specialist model of care. For family or informal caregiver and professional outcomes, these were most commonly reported in relation to the education and training model of care, where education or training was delivered to these groups. Evidence of changes in service utilisation or costs were reported for the in-home and specialist models of care, but less often that for patient outcomes. See Figure 3 and Table 4 for further details of the direction and volume of evidence.
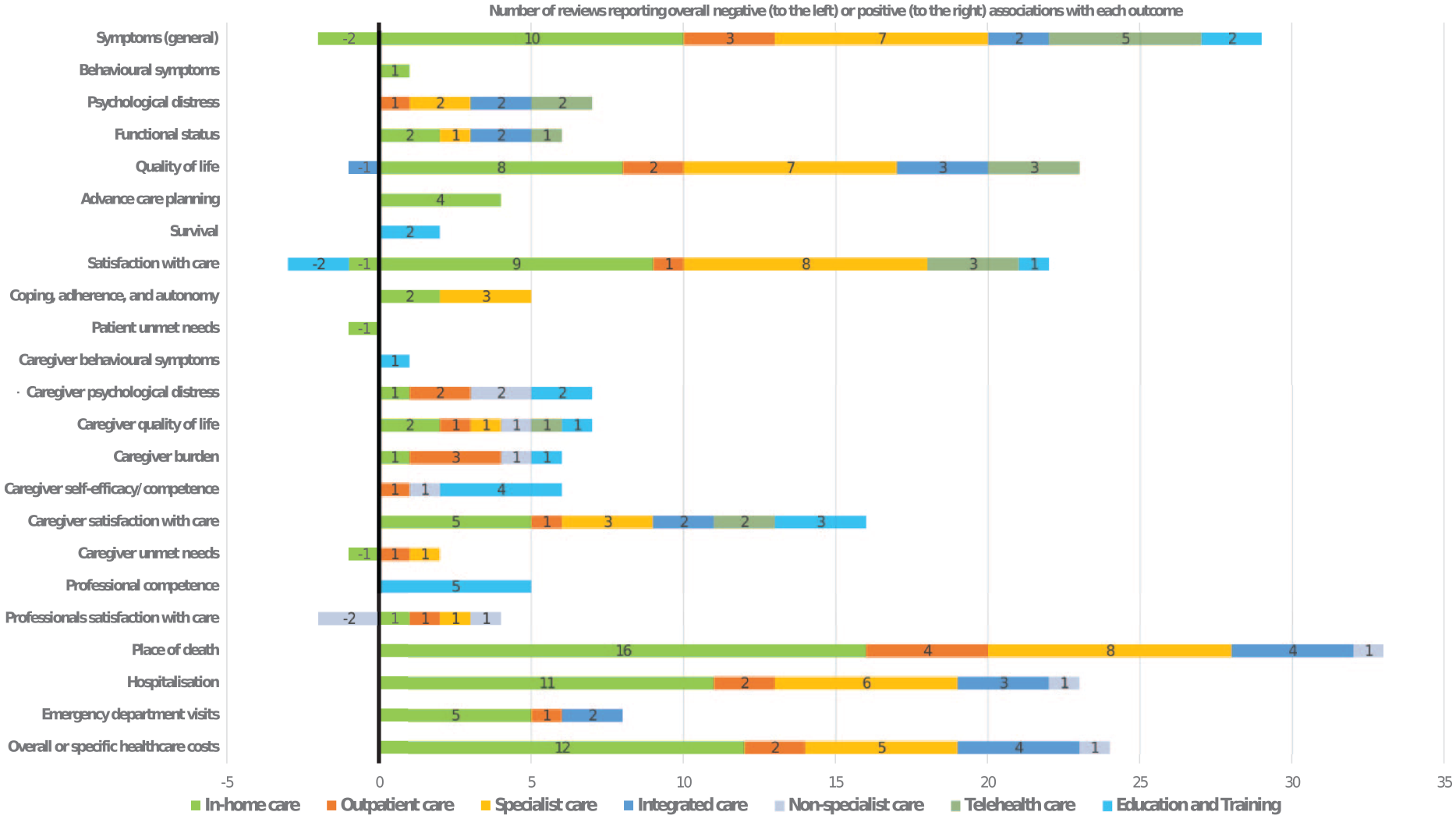
Illustration of the identified benefits and associations between some of the models of palliative care and outcomes from the included reviews (Included reviews which report on more than one outcome are counted more than once).
All the identified models of care had some reported evidence of a positive effect, mostly in relation to patient outcomes and service utilisation and costs. Figure 3 provides an overview of the numbers of reviews reporting each outcome, by model of care, and the reported direction of effect. Caution should be exercised in interpreting Figure 3, given that inconsistent findings are not captured in the figure, there may be some ‘amplification’ where individual studies are reported by more than one review and the quality appraisals of reviews are not incorporated. However, it does demonstrate where most research has been conducted, and gives a headline indication of the direction of effects identified.
In general, we found evidence on service utilisation, costs and cost-effectiveness very limited. Several studies examined the overall healthcare costs of various interventions in the different models of care using outcomes such as overall costs, specific hospital costs, cost effectiveness, hospital utilisation and impact of place of death on costs. For example, an increase in the likelihood of dying at home was shown for the in-home model of care which included access to home nursing compared to alternative models13,48,50,55,58,63,70,71,73,85,87,102 and this did not compromise quality of life. 10 Home-based specialist palliative care from specialist nurses increased home deaths without compromising symptoms.15,70 Comprehensive and robust evidence on the cost implications of telephone advice lines for telehealth models of care related to the benefits is ‘very limited’, but there is some limited indication that costs can be reduced through out-of-hours telephone advice lines 65 and videoconference-based palliative care. 72 Janke et al., 64 in a non-cancer population where most were home-based, suggests that palliative care is ‘cost-saving or at least cost-neutral’. Spencer et al. 86 in their review conducted from the perspective of the healthcare system, showed that hospital-based palliative care costs are higher than hospice or home-based palliative care (however, patient and informal caregiver costs were not included).
Discussion
Main findings of the review
In this extensive and complex systematic review of review-level evidence about palliative and end-of-life care for people at home, we have been able to draw together a large volume of evidence and present an overarching picture of the evidence for models of palliative and end-of-life care for those at home.
First, we provide insights into the existing models and components of in-home palliative care. We identified seven models, distinguished by setting (where care is delivered; either at home or through outpatient care), who is delivering the care (type of professionals; either specialist, integrated with specialist or non-specialist palliative care), and mode of delivery (telehealth or education/training). Not all models are equivalent: some only represent part of care for example, outpatients, telehealth, education/training. It is clear that two models – the outpatient model, and the model delivered by non-specialists in palliative care – have been much less researched.
Second, we synthesise the overall evidence, and show that this largely supports in-home palliative care, especially if delivered via specialist palliative care models or integrated palliative care models (integration is between specialist and non-specialist services). Careful interpretation of our findings (see Table 4) shows surprising consistency in the evidence; consistency between included reviews (as evidenced by the same direction of effect for a number of reviews for the same outcomes) and consistency in the outcomes showing benefit (for instance, the positive effect of several models on patients’ symptoms, psychological distress, and functional status, in contrast to the more mixed evidence across models on patients’ quality of life, where it is perhaps harder to show impact). This consistency is also apparent across the higher-quality reviews. The in-home and specialist models of care have most evidence for reducing patients’ symptoms, overall or specific healthcare costs, attaining patient, family and professional satisfaction with care, and reducing hospitalisation. This is supported by a recent meta-analysis and meta-regression on specialist palliative care. 66 There is less evidence for non-specialist palliative care, but there may also be more variation in this model, accounting for less consistency in what evidence there is (see Table 4). In addition, the education and training model extensively supports family or informal caregivers in delivery of care, with evidence suggesting training, increases their competence, quality of life, and reduces psychological distress; an important finding given that 1 in 10 carers report unwillingness to care again. 105
The in-home care model sometimes showed inconsistent results for service utilisation and cost (see Table 4), such as emergency department visits, hospitalisation and overall healthcare costs. One review indicated that care at home may be approximately 30% more costly in the last 24 months of illness than hospice or conventional care. 102 and it is likely that this reflects different cost horizon and perspectives. Despite this, achieving preferred place of care and death, improved overall healthcare costs, symptom relief, quality of life and wellbeing, alongside satisfaction, by the identified models of care, especially the in-home, and specialist models of care, is in line with recent findings,55,59,62–64,66,71,78,79,84 and may reflect affordability, and meeting preferences of patients and family to care for and die in a place of comfort – home.
Considering the benefits that was observed for patients,16,49,52,58,62,63,65,87,93,100 and caregiver52,60,65 outcomes, supporting professionals in effective service delivery and provision of care services to support individuals to be cared for – in their own homes – through telehealth care is promising. However, there is limited evidence to support effectiveness of specific services such as telephone advice lines, and out-of-hours telephone lines. 65 There has been little study of the effects of telephone support and telehealth in general on symptom management, functional status and advance care planning. 49 This requires more investigation as it may support wellbeing while reducing overall costs.
Strengths and limitations of the review
We have endeavoured to present an overview of a large volume of evidence, and at the same time provide sufficient detail to identify the sources of evidence for any one model of care and related outcomes. We used two previous meta-level reviews10,27,28 on models, components and outcomes of palliative care to inform our analysis. We used an established method 34 to manage overestimation of our reported evidence. We used narrative synthesis to address the heterogeneity that results from including multiple types of reviews without a uniform model of care or standard outcomes. There is a challenging trade-off between synthesis and detail. Strengths of this review include: registration of, and adherence to a study protocol including reporting changes; detailed search strategy developed by specialist; dual reviewer screening; estimation of overlap of primary studies in included reviews; quality appraisal; and a focus on more recent evidence given most included reviews were less than 10 years old. Limitations include: restriction to English-language searches; no exclusion of low-quality review articles (although we report high quality reviews in Table 4); potential publication bias (with positive findings more likely to be published). Our application of narrative synthesis exposed our analysis to inherent subjectivity that can affect the reproducibility and validity of our findings. However, we applied a critical reflexive approach in all stages of the research, were transparent in our reporting, and adopted systematic techniques and PRISMA guidance to control for this bias. We found it was not possible to explore directly, the associations between components of care and outcomes as earlier planned. It is worthy of note that most of the included reviews were from high-income countries and findings may not be generalisable to low or middle-income countries.
What this review adds and implications for practice, policy and research
This review of reviews provides an overview of a large volume of evidence with sufficient detail to understand current evidence on any one model of care, components and related outcomes. The evidence most strongly supports the provision of in-home palliative care, specialist palliative care and integrated services, with positive effects largely reported on patient outcomes for these models of care.
Future experimental and observational studies are required to assess the cost effectiveness of the identified models, components and outcomes of care in different economic regions and healthcare systems. System wide research is particularly required. Also, research is needed to standardise outcomes of care and explore multidisciplinary and integrated approaches to provision of holistic care.
Conclusion
This meta-level evidence most strongly supports the provision of in-home palliative care, as this is where there is most review level evidence showing positive effect on patient outcomes. There was also evidence to support specialist palliative care and integrated teams (integration of primary palliative care with specialist support), and education and training for both informal family carers and professionals.
Differences between protocol and review
We made minor revisions to the title and objectives from the protocol to streamline the focus of this review, along with refining the inclusion and exclusion criteria, outcomes and search strategy to improve findings. We made several minor changes to the style and organisation of the text to improve the clarity and conciseness.
Also, the extracted information and approach to quality appraisal were updated to include additional elements aimed at improving the quality of the review.
We mapped the models to outcomes, not components as previously planned but endeavoured to reflect on how the models and components relate to the outcomes.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163251362567 – Supplemental material for Models, components and outcomes of palliative and end-of-life care provided to adults living at home: A systematic umbrella review of reviews
Supplemental material, sj-docx-1-pmj-10.1177_02692163251362567 for Models, components and outcomes of palliative and end-of-life care provided to adults living at home: A systematic umbrella review of reviews by Sophie Pask, Chukwuebuka Okwuosa, Ahmed Mohamed, Rebecca Price, Jennifer Young, Thomas Curtis, Stuart Henderson, Ishbel Winter-Luke, Anisha Sunny, Rachel L Chambers, Sarah Greenley, Therese Johansson, Anna E Bone, Stephen Barclay, Irene J Higginson, Katherine E Sleeman and Fliss EM Murtagh in Palliative Medicine
Supplemental Material
sj-xlsx-2-pmj-10.1177_02692163251362567 – Supplemental material for Models, components and outcomes of palliative and end-of-life care provided to adults living at home: A systematic umbrella review of reviews
Supplemental material, sj-xlsx-2-pmj-10.1177_02692163251362567 for Models, components and outcomes of palliative and end-of-life care provided to adults living at home: A systematic umbrella review of reviews by Sophie Pask, Chukwuebuka Okwuosa, Ahmed Mohamed, Rebecca Price, Jennifer Young, Thomas Curtis, Stuart Henderson, Ishbel Winter-Luke, Anisha Sunny, Rachel L Chambers, Sarah Greenley, Therese Johansson, Anna E Bone, Stephen Barclay, Irene J Higginson, Katherine E Sleeman and Fliss EM Murtagh in Palliative Medicine
Footnotes
Author’s contributions
SP, FEMM, KES, SB and IJH were responsible for conceptualisation and review design, with critical input from AM, RLC, SG, TJ and AEB. SG and SP developed and refined the search strategy. SP, CO, AM, RP, JY, TC, SH, IW, AS, RLC and SG were responsible for review identification, screening, inclusion and extraction of data. SP, CO, RP, JY, TC, SH, IW, AM, TJ, AEB, SB, IJH, KES and FEMM were involved in the analysis and interpretation of results. SP, CO, RP and FEMM were involved in preparing the draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted as part of the Better End of Life Programme, which is funded by Marie Curie (grant MCSON-20-102) and awarded to KES, IJH, FEMM and SB. The research was carried out in collaboration between King’s College London, Hull York Medical School, University of Hull and University of Cambridge. The funder was not involved in the study design, data collection and analysis or interpretation of results. This study was also supported by the UK National Institute for Health and Care Research Applied Research Collaboration for Yorkshire and Humber [NIHR200166]. KES is the Laing Galazka Chair in palliative care at King’s College London, funded by an endowment from Cicely Saunders International and the Kirby Laing Foundation. IJH is an NIHR Senior Investigator Emeritus. FEMM is a National Institute for Health Research (NIHR) Senior Investigator. IJH and SB are supported by the NIHR Applied Research Collaboration (ARC) South London (SL) and NIHR ARC East of England, respectively. SG’s time was funded by Yorkshire Cancer Research under the TRANSFORM programme (award reference HEND405SPT). The views expressed in the report are those of the authors and not necessarily those of the UK NIHR, or the UK Department of Health and Social Care.
Supplemental material
Supplemental material for this article is available online.
Research ethics and patient consent
This study did not require ethics approval as it was a review of already published reviews.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
