Abstract
Background:
Despite global support, there remain gaps in the integration of early palliative care into cancer care. The methods of implementation whereby evidence of benefits of palliative care is translated into practice deserve attention.
Aim:
To identify implementation frameworks utilised in integrated palliative care in hospital-based oncology services and to describe the associated enablers and barriers to service integration.
Design:
Systematic review with a narrative synthesis including qualitative, mixed methods, pre-post and quasi experimental designs following the guidance by the Centre for Reviews and Dissemination (PROSPERO registration CRD42021252092).
Data sources:
Six databases searched in 2021: EMBASE, EMCARE, APA PsycINFO, CINAHL, Cochrane Library and Ovid MEDLINE searched in 2023. Included were qualitative or quantitative studies, in English language, involving adults >18 years, and implementing hospital-based palliative care into cancer care. Critical appraisal tools were used to assess the quality and rigour.
Results:
Seven of the 16 studies explicitly cited the use of frameworks including those based on RE-AIM, Medical Research Council evaluation of complex interventions and WHO constructs of health service evaluation. Enablers included an existing supportive culture, clear introduction to the programme across services, adequate funding, human resources and identification of advocates. Barriers included a lack of communication with the patients, caregivers, physicians and palliative care team about programme goals, stigma around the term ‘palliative’, a lack of robust training, or awareness of guidelines and undefined staff roles.
Conclusions:
Implementation science frameworks provide a method to underpin programme development and evaluation as palliative care is integrated within the oncology setting.
Keywords
The hospital based palliative care services have been ascertained as enabling access to early or timely palliative care. Yet integration of palliative care into existing cancer services remains limited with patients continuing to be referred to palliative care at a late stage, if at all.
Implementation science offers an approach to underpin the development of integrated palliative care services.
Just seven studies utilised implementation frameworks to guide service delivery which included RE-AIM, SELFIE model, Medical Research Council framework, and the quality improvement approach of Define, Measure, Analyze and Improve.
The main facilitators for palliative care integration included the value of co-design with ‘end users’ of the intervention and the processes of implementation; staff education and training including around processes, communication, and introducing referrals; dedicated communication across the multidisciplinary teams in the hospital. Barriers included lack of time and rapid staff turnover, maintaining fidelity and consistency of the intervention across individual patients and different sites, and a lack of relevant health policy regulations and resources to support palliative care services.
A deeper understanding of the enablers and barriers to the success or failure of the previous palliative care implementation efforts and the opportunities provided by a systematic approach as offered by implementation frameworks will benefit the effective establishment and sustainability of future integrated palliative care programs.
The findings from this systematic review may help stakeholders including policy makers and service providers in allocating precious resources towards planning and systematically implementing integrated palliative care programs in hospital-based cancer services.
Introduction
Palliative care is increasingly recognised 1 as a core component of oncology care.2,3 Significant evidence from randomised clinical trials has validated the proven benefits of early palliative care for people with advanced cancer including better symptom management, 4 increased satisfaction with care, and reduced psychological suffering.5–7 This has led to a strong global consensus supporting the integration of early palliative care in routine cancer practice.2,8
Implementation science is the study of methods and approaches that facilitate the uptake of research into clinical practice. 9 It includes the identification and response to barriers that block the uptake of proven interventions into care, thereby closing the information gap.10,11 There is limited literature to guide the implementation of timely palliative care in cancer care services.10,12 and timely referral to hospital based palliative care services remains challenging. 13 Many clinicians, who oversee the introduction of programs in clinical settings, are unfamiliar with the current implementation science evidence meaning that ‘real-world changes’ do not result. 12
There is a need for clinical researchers to consider the acceptability, fidelity, and viability of the intervention that is to be implemented, particularly during the initial phases of its development, when its appraisal and utility may be assessed with iterative checks over time. 14 There is also an opportunity for clinical researchers to take a ‘systems’ approach as available in implementation science to the translation of research findings around early palliative care. This systematic review and narrative synthesis aimed to identify the existing utilisation of implementation science frameworks, to describe the barriers and enablers encountered, and strategies adopted to implement integrated palliative care into hospital-based oncology services.
Design
A systematic review was conducted with a narrative synthesis approach. Due to the diversity of included study designs (qualitative, mixed methods, pre-post and quasi experimental designs, this systematic review utilised a narrative synthesis approach to presenting the data as guided by Popay et al. 15 and followed the guidelines by the Centre for Reviews and Dissemination. 16 This systematic review was registered on the International Prospective Register of Systematic reviews PROSPERO CRD42021252092. Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed for reporting.
Review aim/questions
The aim of this systematic review was to identify the existing utilisation of implementation science frameworks, and to describe the barriers and enablers encountered and strategies adopted to implement integrated palliative care into hospital-based oncology services.
Inclusion/exclusion criteria
Inclusion: Studies conducted in English language, on humans 18 years and older, reporting on the process of implementing hospital-based palliative care and oncology integration, including qualitative, quantitative, or mixed methods studies (including studies involving interviews, focus groups, surveys and cross-sectional questionnaires) as well as formal randomised controlled trials or implementation studies published in the year 2010 onwards. 17 The target clinical group included adult patients with advanced cancer including those with distant metastases, or locally advanced cancer that is life limiting. 2
Exclusion: Publications classified as book reviews or sections, case reports, comments, conference abstracts, posters or oral presentations, conference reviews, dissertations, editorials, lectures, letters or review papers were excluded.
Information sources/data sources
Publications were identified through systematic searches of six bibliographic databases, conducted on 12 March 2021: Ovid MEDLINE(R) ALL 1946 to March 10, 2021; Embase 1974 to 2021 March 10 (Ovid); Ovid Emcare 1995 to 2021 Week 08; APA PsycInfo 1806 to March Week 1 2021 (Ovid); CINAHL (EBSCOhost) and Cochrane Library (Wiley). The reference lists of included papers were also searched. An updated additional search was conducted in Medline Ovid from 2021 to 2023 (current) for any pertinent papers published recently. In addition, a hand search was conducted in Google Scholar.
Search strategy
Search strategies were developed by a medical librarian in consultation with a topic expert. Potential search terms were identified through text mining in PubMed PubReminer 18 using the query: (‘palliative care’[Mesh:NoExp] OR palliative[tiab]) AND (neoplasms[Majr] OR cancer*[tiab]) AND hospital*[tiab] AND ((implement*[tiab] OR integrat*[tiab]) OR ‘implementation science’[Mesh:NoExp]). Forty publications identified from those results were further analysed using Yale Text Analyser. 19 Search terms retrieved through text mining were extensively tested for usefulness and relevance in Ovid Medline to develop the final search strategy.
Final search strategies combined the general concepts of Palliative Care AND Cancer AND hospital AND Implementation using a combination of subject headings and text words. Searches were limited to English language publications, but no date limits were applied. An initial search was developed for Ovid Medline (Supplemental File 1) and then adapted for other databases adjusting subject headings and syntax as appropriate (Supplemental Appendix 1). Search syntax used in the Ovid databases was adapted for CINAHL (EBSCOhost) and Cochrane (Wiley) using the Polyglot Search Translator. 20
Study selection process
Search results were exported to EndNote bibliographic management software 21 and duplicates were removed by one author. In accordance with inclusion and exclusion criteria (Table 1), records were screened on publication type by the same author, and book reviews; books and book sections; case reports; comments; conference abstracts; conference reviews; dissertations; lectures and letters were excluded.
Inclusion/exclusion criteria.

A decision was made to include only those studies published after 2010 based on the development of the field of integrated palliative care following the publication of a landmark palliative care trial (Temel et al. 17 ).
All remaining records were loaded into Covidence 22 systematic review software for screening on title and abstract by a second author and included records were independently screened a second time on title and abstract by another author within Covidence. These two authors made the decision to include only those studies published after 2010 based on the development of the field of integrated palliative care following the publication of Temel et al., 17 a landmark study which heralded integrated palliative care using the model of service delivery. This study was helpful in shaping the discourse in literature and the service development that followed.
The remaining records were exported to EndNote and assessed for eligibility on full text by another two independent reviewers. Disagreements and uncertainties around screening outcomes were resolved by an iterative inter-reviewer discussion between the two authors until a consensus was reached.
The following data were independently collected by two authors from included articles in tabulated formats: author(s), date of publication, participant group, study design. Data were also collected for key findings around descriptions of the implementation process including references to implementation frameworks and models and any barriers and enablers to the implementation. The critical quality appraisal for risk-of-bias was conducted by two authors.
Strategy for data synthesis
A narrative synthesis approach was used to summarise the data. The defining characteristic of narrative synthesis as described by Popay et al. 15 is that it adopts a textual approach to the process of synthesis to ‘tell the story’ of the findings from the included studies. 15 The purpose of the narrative synthesis was to organise findings from the included studies in order to provide an assessment of the strength of the evidence for drawing conclusions about the facilitators and/or barriers to implementation identified in the synthesis. The steps towards data synthesis involved two researchers reading the included papers multiple times to facilitate thematic analysis, ascertaining the main, repeating and/or pertinent findings or themes (based on the review question) and/or concepts emerging from the various studies. 15 Thematic analysis provided the researchers with a means of organising and summarising the main ideas and conclusions across the diverse research studies. Patterns were identified in the sections including methods, results and discussion by reviewing the full-text papers and potential themes were extracted in a table and discussed among the two researchers in order to ensure the context of themes, which were later refined under categories. Any inter researcher disagreement about the themes was resolved through consensus and a third senior researcher’s advice. Regular meetings were arranged among all researchers throughout the process to progress discussion and finalisation of findings. The study results were then transformed and summarised into a common rubric. This rubric in Table 4 included; identified implementation models and framework, and results/themes of implementation approaches but not the outcomes of the intervention itself.
Results
Following removal of duplicates and excluding publication types, 4642 records were screened on title and abstract. An additional update to the search was conducted via Medline Ovid 2021 to 2023 (current) and a further 181 records were screened on title and abstract, leading to a total of 60 records that were subject to full text review. Of these, 16 studies met the inclusion criteria (see Figure 1 for PRISMA flow chart).
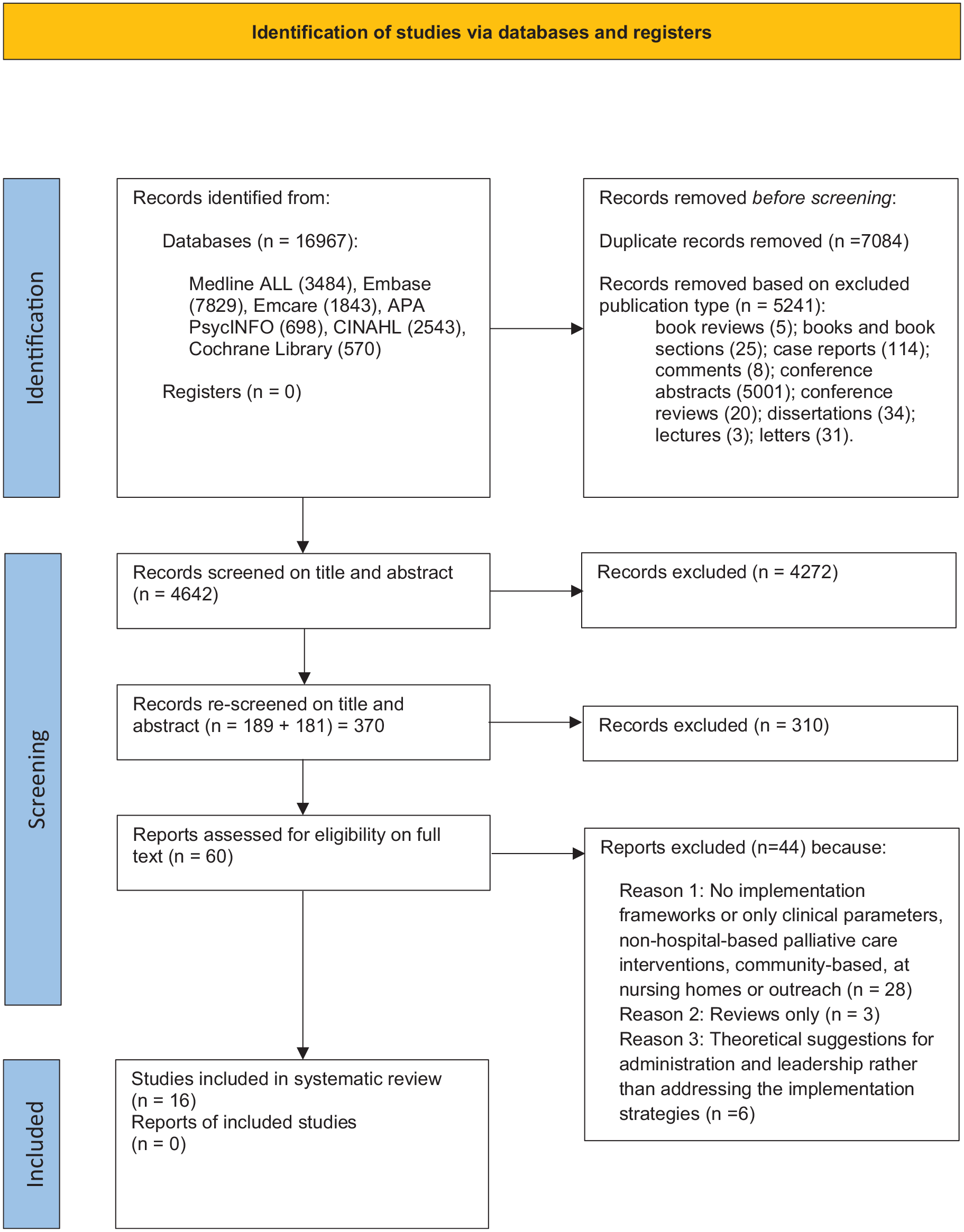
PRISMA 2020 flow diagram.
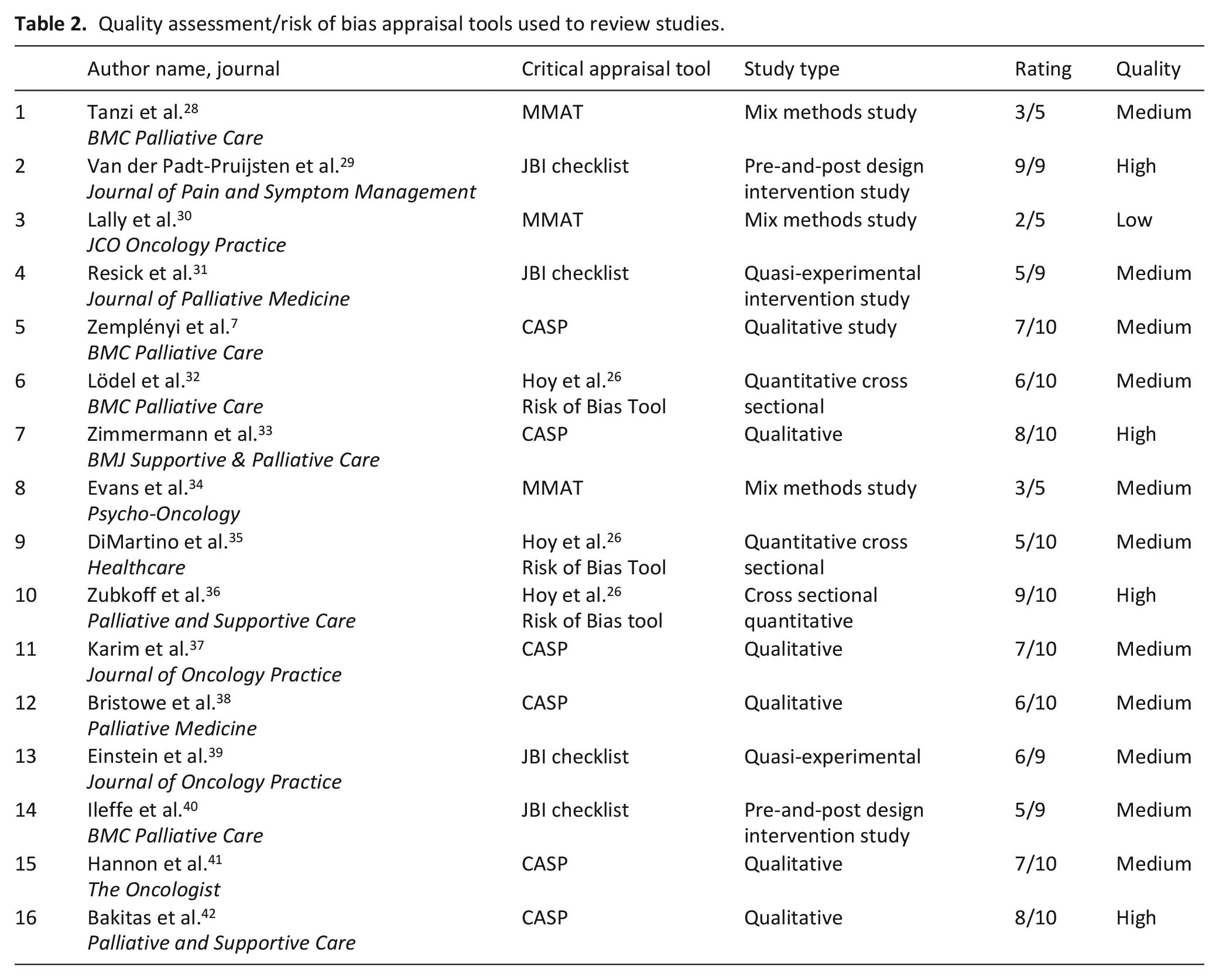
Quality assessment/risk-of-bias appraisal tools used to review studies
Study appraisal, also called validity assessment or quality assessment and critical appraisal pertains to a process of assessing the methodological quality of individual studies, by utilising the risk of bias validated tools. 15 This is pertinent as it may affect both the results of the individual studies and ultimately the conclusions reached from the body of studies.
The following validated tools were used to assess quality, rigour and risk of bias; Critical Appraisal Skills Program (CASP) tool 23 was used for critically appraising the qualitative studies, cohort studies and experimental/intervention studies, Mixed Method Assessment Tool (MMAT) 24 for mixed methods studies and Joanna Briggs Institute (JBI) 25 tool for pre-and-post (quasi-experimental) studies. Another risk of bias tool 26 was used for the quantitative cross-sectional studies to assess 10 characteristics of the study. Quality assessment was based on the implementation data provided, rather than the overall study data. Qualitative studies were given a ranking of 1 to 10, cohort studies were given a ranking of 1 to 12 and experimental studies were given a ranking of 0 to 11 based on how many of the CASP questions could be answered positively.23,27 The Mixed Method Assessment Tool was used with a ranking of 1 to 5 based on how many of the five Mixed Method Assessment Tool questions could be answered positively 24 (see Table 2).
Quality assessment/risk of bias appraisal tools used to review studies.
Study characteristics
The 16 included studies (Table 3) largely originated in high-income countries including the United States (US),30,31,36,35,39,42 Canada,33,34,37,41 United Kingdom (UK) 38 and Europe including Hungary, 7 Germany 32 and Netherlands 29 and Italy 28 One study was conducted across multiple European countries 40 (Table 3).
Project and sociodemographic summaries.
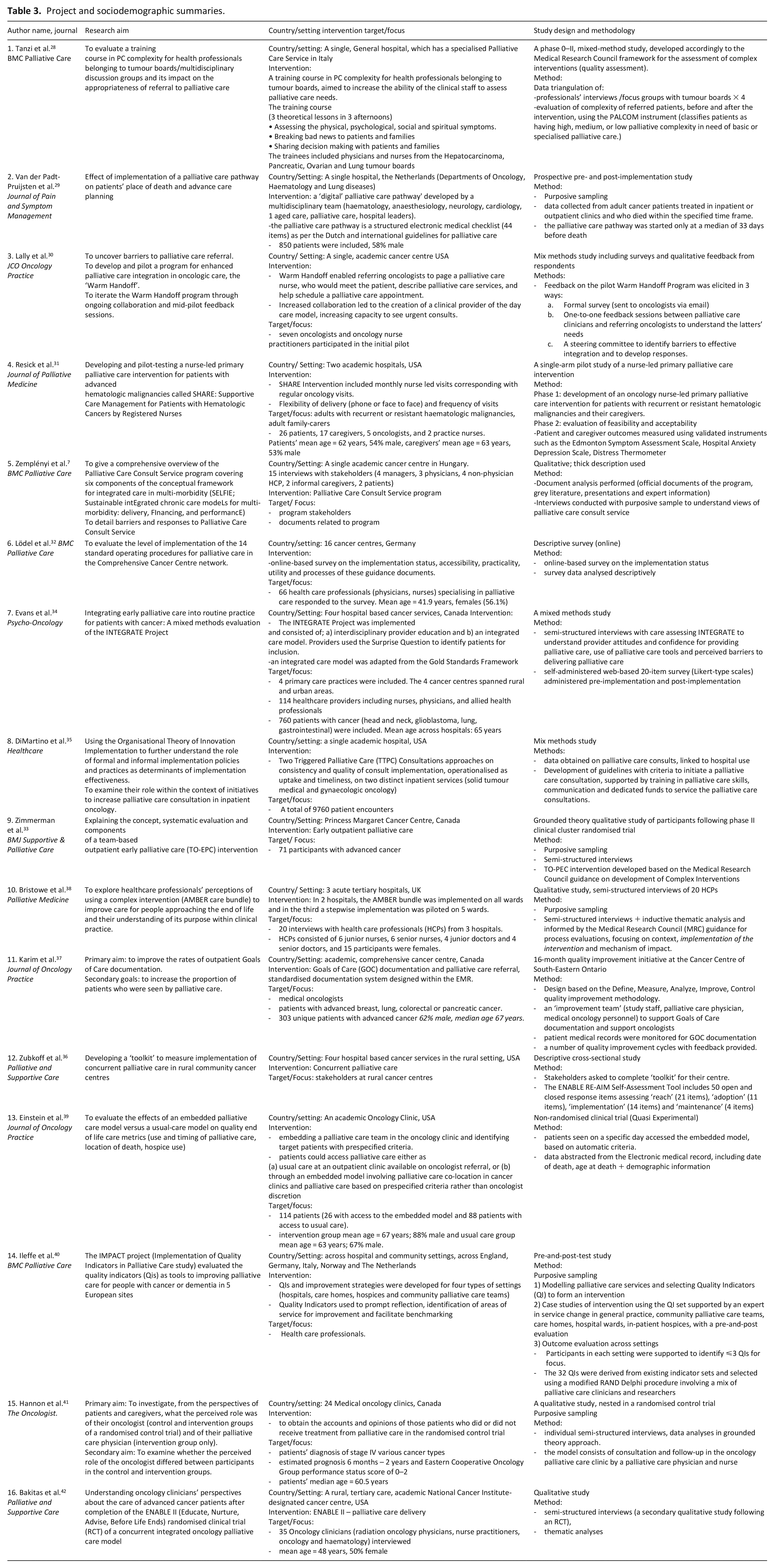
Studies designs included qualitative studies (n = 6 out of 16), descriptive/cross-sectional (n = 3), mixed-methods (n = 3), pre-and-post implementation designs (n = 2) and clinical/quasi-experimental studies (n = 2) studies.
Study participants included individuals (health professionals, patients) and the participating urban and rural oncology services centres (Table 3).
Types of implementation frameworks
Seven studies satisfied all our criteria and utilised a validated implementation framework7,28,33,36–38,42 (Table 4). Of these, two studies included the implementation of a palliative care intervention (ENABLE RE-AIM Self-Assessment Tool for reach, effectiveness, adoption, implementation and maintenance)36,43 across four United States rural cancer services in hospitals. 36 One study employed a quality improvement framework based on domains of Define, Measure, Analyze, and Improve to implement an intervention aiming to increase documentation of goals of care and secondarily, palliative care referral. 37 Three studies utilised the Medical Research Council (MRC) guidance for process evaluation, focusing on context, application of the intervention, mechanism of impact 38 and evaluation phases when establishing hospital based palliative care services28,33 (Table 4). A study in Hungary used the SELFIE implementation model (Sustainable integrated chronic care models for multi-morbidity: delivering, financing, and performance) to guide their review of a palliative care consultation service programme 7 (Table 4). This framework organises elements of integrated care into the six World Health Organisation components of health systems (leadership and governance, service delivery, information and research, technologies and medical products, financing and workforce). 7
Implementation models, frameworks and emerging themes/results.
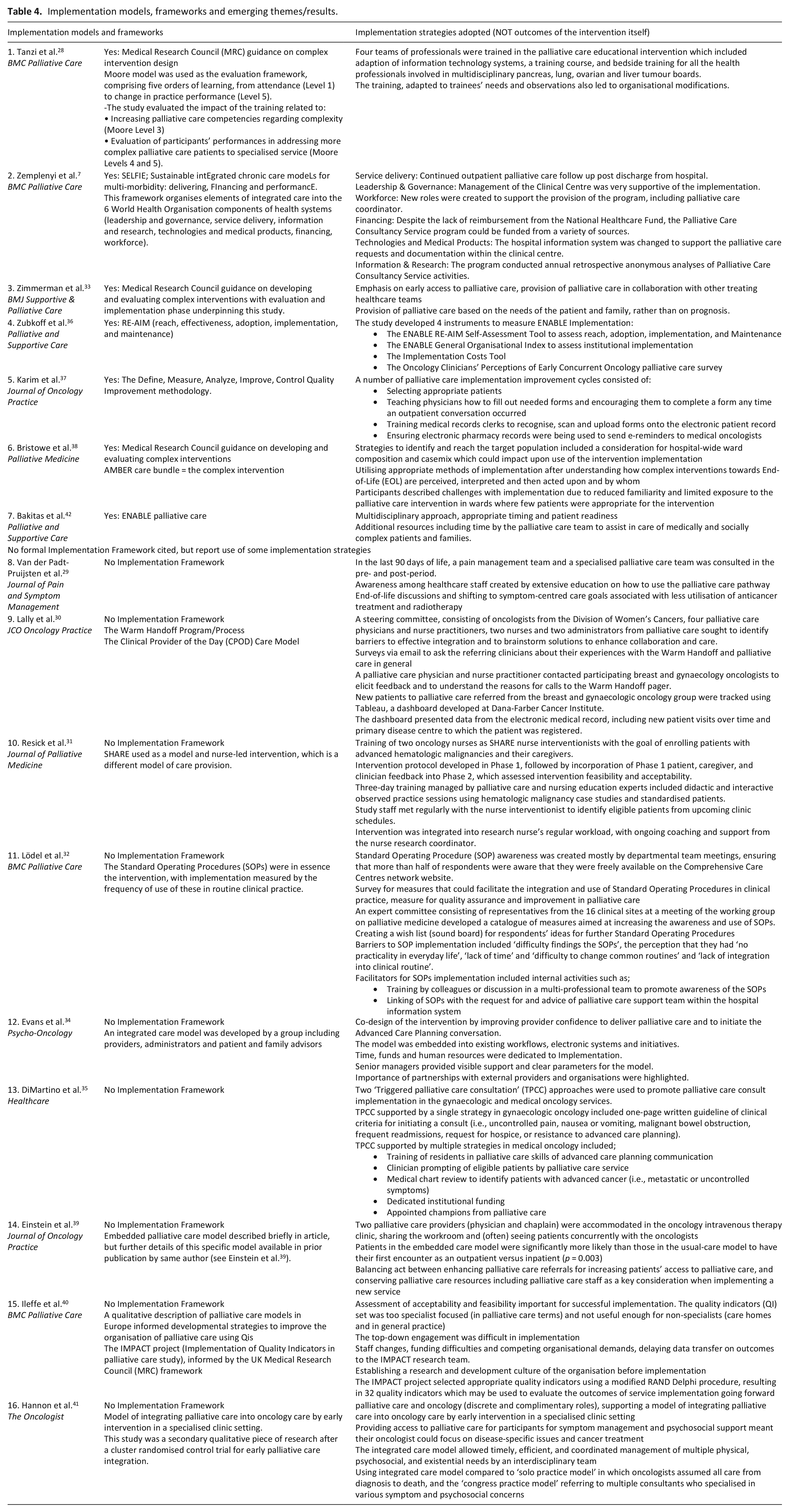
A further nine studies included components of implementation science to report upon outcomes of interventions for integration of palliative care in oncology settings but did not cite a specific implementation framework. These studies were establishing different integrated palliative care models, for example, a digital ‘palliative care pathway’ developed by the multidisciplinary team, 29 and a nurse-led primary palliative care implementation programme for patients with advanced hematologic malignancies 31 (Table 4). Strategies used and reported by these nine studies which aligned with implementation domains included: strategies to identify and reach the target population29,39; relationship building and engagement 30 ; co-design of the intervention 34 ; training and education 32 ; attention to workplace culture and practices34,39 and assessment of acceptability and feasibility.30,31,40
Synthesis of enablers and barriers reported in palliative care implementation studies in cancer services
The details of the implementation frameworks for the included studies along with the study descriptions, and the nature of the evaluations undertaken are provided in Table 4. The key findings of enablers and barriers from our included studies with more detailed explanation have been grouped based on: a) staff; b) patients; c) system and d) implementation guidelines.
Staff enablers and barriers
The availability of dedicated palliative care specialist staff was key to the success of programs, 36 including the ability to respond to urgent consults and the availability of clinicians to discuss challenging cases. 30 A designated ‘clinical provider of the day’ programme allowed for a noteworthy increase in the capacity to see urgent palliative care consults with an average of 19 patients seen per month. 30
There was a generalised perception amongst oncologists that the roles of palliative care clinicians were as ‘co-managers’ and ‘consultants’ 42 with the need for clarity emphasised around the roles and responsibilities of clinicians. 34 Cancer-directed treatments were perceived to be the main domain of the oncologist, while pain and symptom control, and care for the patient and their carer were identified as the primary role of palliative care. 41 A nurse led supportive care clinic with clearly defined roles was set up for patients with haematological cancer which was widely supported by service oncologists, 75% of whom agreed it improved the quality of patient care. 31
Integrating early palliative care into routine care for patients with advanced cancer led to an improvement in oncologists’ confidence to deliver palliative care 34 and to initiate the Advanced Care Planning conversation with patients.28,42 Nevertheless, some physicians expressed doubts about starting discussions with patients as a result of uncertainty regarding prognosis and unknown consequences for the physician−patient relationship. 29 This prognostic uncertainty was particularly cited as a barrier in the setting of haematological cancers. 31
Patient enablers and barriers
Patients and caregivers were largely satisfied (84%) with the palliative care intervention 31 and described it as providing: swift and personalised symptom management; holistic support for patients and caregivers 41 ; guidance in decision-making and preparation for the future28,37 and assisting the caregivers with understanding their family member’s illness and cancer ‘journey’.31,42 However, potential barriers included the uncertain course of hematologic malignancies and low levels of patient interest in palliative care research (89%) with few patients completing surveys reporting feedback about palliative care. 31 The stigma surrounding the name ‘palliative’ was reported as a barrier to early palliative care referral that needed to be explicitly addressed within the intervention programme. 33
System enablers and barriers
The integrated palliative care model was most effective when it was implanted into existing workflows and supported by electronic systems. 34 Conversely, challenges to implementation included a lack of clinician readiness at the clinical site was observed as an implementation challenge 36 and practices that involved non-palliative care clinicians being tasked with additional clinical responsibilities such as completing symptom assessment tools. 40 Others noted barriers included lack of acceptability and feasibility, with work tools that were considered too specialist focused 40 or not practical, difficult to locate, time consuming and poorly integrated into routine workflows and routines. 32
Administrative difficulties included securing optimum space, staff, and sufficient time to launch the implementation programme. 36 Authors of early palliative care intervention studies described potential cultural or professional conflicts for implementing a complex intervention in clinical settings under the care of multiple specialities. 38
The integrated palliative care model was most successful if the senior managers and heads of tumour streams provided support, 28 and time, funds, education and human resources were dedicated to the implementation programme.31,34,38 An important provision is that a balance must be struck between broadening palliative care referral criteria for increasing patient access to palliative care, 28 and taxing limited available palliative care resources including workforce shortages and space constraints. 39 A lack of a standardised Institutional Review Board approach including their hesitancy to approve palliative care research and quality audit activities was encountered by the researchers when undertaking evaluation of outcomes. 36
The rapid turnover of staff in the hospital/clinical settings was reported as having a detrimental impact upon implementing a new service intervention, resulting in delays and funding difficulties with ongoing requirements for staff training,31,40 disrupted data collection, clinical relationships and lines of communication.31,40
Within the organisation, the role of a multidisciplinary team 28 was recognised to be pertinent in the delivery of palliative care interventions.38,41 Considering those studies involving education interventions aimed at increasing clinician ability to recognise palliative care complexity,28,42 the role of a multidisciplinary team was regarded positively and facilitated earlier access to the palliative care team.
Others noted positive organisational change available in early palliative care through the AMBER study which provided a tool to facilitate change to the way care was delivered and also facilitated change to the culture of care delivery. 38 Potential facilitators for palliative care implementation included internal activities such as intercollegiate palliative care training or multidisciplinary team discussions to promote awareness of tools to assist palliative care provision.28,32
The value of clinical advocates and champions was highlighted as supporting a successful palliative care programme implementation. 34 To meet the patient and clinician needs, a strong collaboration between palliative care and oncology teams was necessary. 30
The oncologists viewed the role of integrated palliative care as facilitating a comprehensive approach to treating the patient with an advanced cancer, and palliative care consultants were seen as sharing the load.28,42 The importance of ‘timing’ and ‘time’ was emphasised for the palliative care conversations and oncologists involved palliative care clinicians early on in order to establish good communication enabling end-of-life treatment planning for a patient with advanced cancer. 42 However, many haematologists felt the unpredictable nature of hematologic malignancies made it difficult to identify the appropriate time to introduce palliative care consultations. 31 Hence, there were a few missed opportunities where palliative care could not be introduced. 42
Physicians conveyed doubts about starting discussions with patients around palliative care as a result of prognostic uncertainty and concerns about destroying patients’ hopes and negatively affecting the physician−patient relationship. 29 Training in having such discussions was identified as an important aspect of doctor-patient communication.28,34
Implementation guidelines
The maintaining of fidelity and consistency of the intervention across individual patients and different sites required ongoing training and education of staff.28–30,34,38,39 A number of authors cited the lack of relevant health policy regulations and financial schemes to support palliative care services. They highlighted the need for distinction on an institutional level between feasibility and ongoing sustainability, which required a more systematic approach.7,28,33,36,41 Furthermore, the divergent levels of development across services required flexibility in the methods for service engagement and the setting of benchmarks to assess success.32,35,40
In order to demonstrate the application of implementation frameworks to this field, the enablers and barriers for palliative care implementation identified through the narrative synthesis are presented in Table 5, and grouped into categories using the example of the RE-AIM Frame-work (Reach, Effectiveness, Adoption, Implementation and Maintenance). 44
Narrative synthesis of enablers and barriers for palliative care implementation under RE-AIM Framework.

Discussion
This systematic review supports the need to translate the strong evidence-base supporting integrated palliative care into oncology practice by examining the rigour with which implementation science principles have been employed.
This review indicates that many researchers have hinted at components of implementation frameworks without explicitly using them when establishing integrated palliative care in their hospital settings. This finding is consistent with observations made in the implementation science literature about the paucity of explicitly naming frameworks.45,46 Those studies that clearly utilise implementation frameworks to formally address the issues related to system, staff, patients and intervention have largely been published in recent years, suggesting this is an emerging area in the field. 12
The few studies in this review which have utilised implementation frameworks to guide service delivery have variously used RE-AIM (Reach, Effectiveness, Adoption, Implementation and Maintenance) framework,36,42 SELFIE model (Sustainable integrated chronic care models for multi-morbidity: delivering, financing and performance), 7 MRC (Medical Research Council) framework to guide complex interventions,28,33,38, and the quality improvement approach of Define, Measure, Analyze and Improve. 37 The increase of studies in the recent past which have utilised implementation frameworks may suggest that it is an emerging area in the field of palliative care integration.
This review utilises multiple, validated tools for included studies to assess the quality, rigour and risk of bias as part of quality appraisal, with medium scoring allocated to most studies.23–26 The quality appraisal ranking was high for four studies, medium for 11 studies, and low for one study.
While the evidence-base underpinning early integration of palliative care is now mature, it appears that the evidence guiding implementation into practice is less developed. The effective utility of the implementation frameworks for systematically implementing palliative care in hospital clinical settings has been shown in this systematic review with narrative synthesis as well as by previous studies conducted in other settings (such as community settings and aged care facilities) and with other populations such as non-oncology (chronic disease) cohorts and chronic asthma sufferers.47,48
We posit the use of an implementation framework such as RE-AIM should be considered going forward to systematically present the barriers and enablers in palliative care implementation programs across hospitals in the oncology units. Having a systematic approach to the implementation of a new service or a new aspect of a service will make explicit not only of those areas which have facilitated the success of the project, but also enable a clearer understanding of reasons underpinning failure to make a change. This in turn may help stakeholders including policy makers and service providers in allocating precious resources towards planning and systematically implementing integrated palliative care programs in the hospital-based cancer services.
The systematic approach offered by implementation science frameworks is also key to ensuring the sustainability of those successfully established programs. Importantly, having a common language to report these experiences of service implementation such as those constructs of RE-AIM framework will allow the learning to be available to other services considering establishing integrated palliative care programs.
There are few limitations to this review. While care was taken in screening studies, it is possible that some studies were missed, as implementation methodologies were not consistently reported. Only the studies written in English language were selected, and these were conducted in high-income countries, so the implementation strategies presented may not be applicable in the low-to-middle-income countries. Some qualitative studies did not provide adequate details about the interview methodologies, or justification for observing rigour and reflexivity and so were omitted, as was the considerable grey literature and unpublished work in this area. However, the strengths of this review include a robust and systematic screening process, and using multiple, validated risk-of-bias tools for quality appraisal of various study designs, to have a holistic view of the palliative care implementation strategies.
The type of Implementation framework that a researcher may want to select could be affected due to the diverse interventions (nurse led, education training, collaborative) and settings for the studies. However, using a formal implementation framework prompts comprehensive consideration of the different areas of influence, of activity and evaluation, ensuring a systematic approach to the programme development is available.
Conclusion
There is very limited utilisation of implementation frameworks to integrate early palliative care into hospital oncology services. Yet the multidimensional, system-wide approaches within implementation science frameworks offer a structure to guide and evaluate programme development. Key elements of implementation to consider when establishing palliative care service integration in cancer centres include a focus on co-design of the intervention and processes of implementation, staff education and training including around processes, communication and introducing referrals, dedicated resources, recognition of champions and more effective communication across the multidisciplinary teams in the hospital. The opportunities offered by implementation science to guide the effective integration of palliative care into cancer care suggest that the formal use of implementation frameworks should be adopted in future service development initiatives.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231186177 – Supplemental material for An evidence-base for the implementation of hospital-based palliative care programs in routine cancer practice: A systematic review
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231186177 for An evidence-base for the implementation of hospital-based palliative care programs in routine cancer practice: A systematic review by Farwa Rizvi, Helen Elizabeth Wilding, Nicole M Rankin, Roslyn Le Gautier, Lorna Gurren, Vijaya Sundararajan, Kylee Bellingham, Joyce Chua, Gregory B Crawford, Anna K Nowak, Brian Le, Geoff Mitchell, Sue-Anne McLachlan, Tanara Vieira Sousa, Robyn Hudson, Maarten IJzerman, Anna Collins and Jennifer Philip in Palliative Medicine
Supplemental Material
sj-pdf-2-pmj-10.1177_02692163231186177 – Supplemental material for An evidence-base for the implementation of hospital-based palliative care programs in routine cancer practice: A systematic review
Supplemental material, sj-pdf-2-pmj-10.1177_02692163231186177 for An evidence-base for the implementation of hospital-based palliative care programs in routine cancer practice: A systematic review by Farwa Rizvi, Helen Elizabeth Wilding, Nicole M Rankin, Roslyn Le Gautier, Lorna Gurren, Vijaya Sundararajan, Kylee Bellingham, Joyce Chua, Gregory B Crawford, Anna K Nowak, Brian Le, Geoff Mitchell, Sue-Anne McLachlan, Tanara Vieira Sousa, Robyn Hudson, Maarten IJzerman, Anna Collins and Jennifer Philip in Palliative Medicine
Footnotes
Data management and sharing
Additional materials are provided (e.g. Supplemental Files of databases search strategies).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding is the Medical Research Future Fund (MRFF) grant: This research work was supported by the NHMRC: APP1174028 – MRFF KAOH
Research ethics and patient consent
Ethics approval not required for this systematic review; however, the included papers in this review received the relevant Ethics Committee or Institutional Review Board provided (or waived) approval and participants provided informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
