Abstract
Background:
Pain is a prevalent symptom in patients with advanced cancer. Recognition of prognostic factors associated with pain intensity, could help provide better assessment, leading to better pain management.
Aim:
identifying prognostic factors which could guide improvements on cancer pain classification.
Design:
a prospective observational study on chronic cancer pain, exploring the association between average mean pain intensity during a 28 days study follow-up and patients’ clinical and pain-related characteristics, including pain syndromes. To evaluate these associations, a mixed model was built.
Setting/participants:
Patients attending a Palliative Care and Pain Outpatient Clinic from May 2015 to June 2019 were screened. Patients with moderate to severe cancer pain who were already receiving or needed treatment with third step WHO ladder opioids were enrolled in the study. Data from 342 patients with at least one follow-up visit were analyzed.
Results:
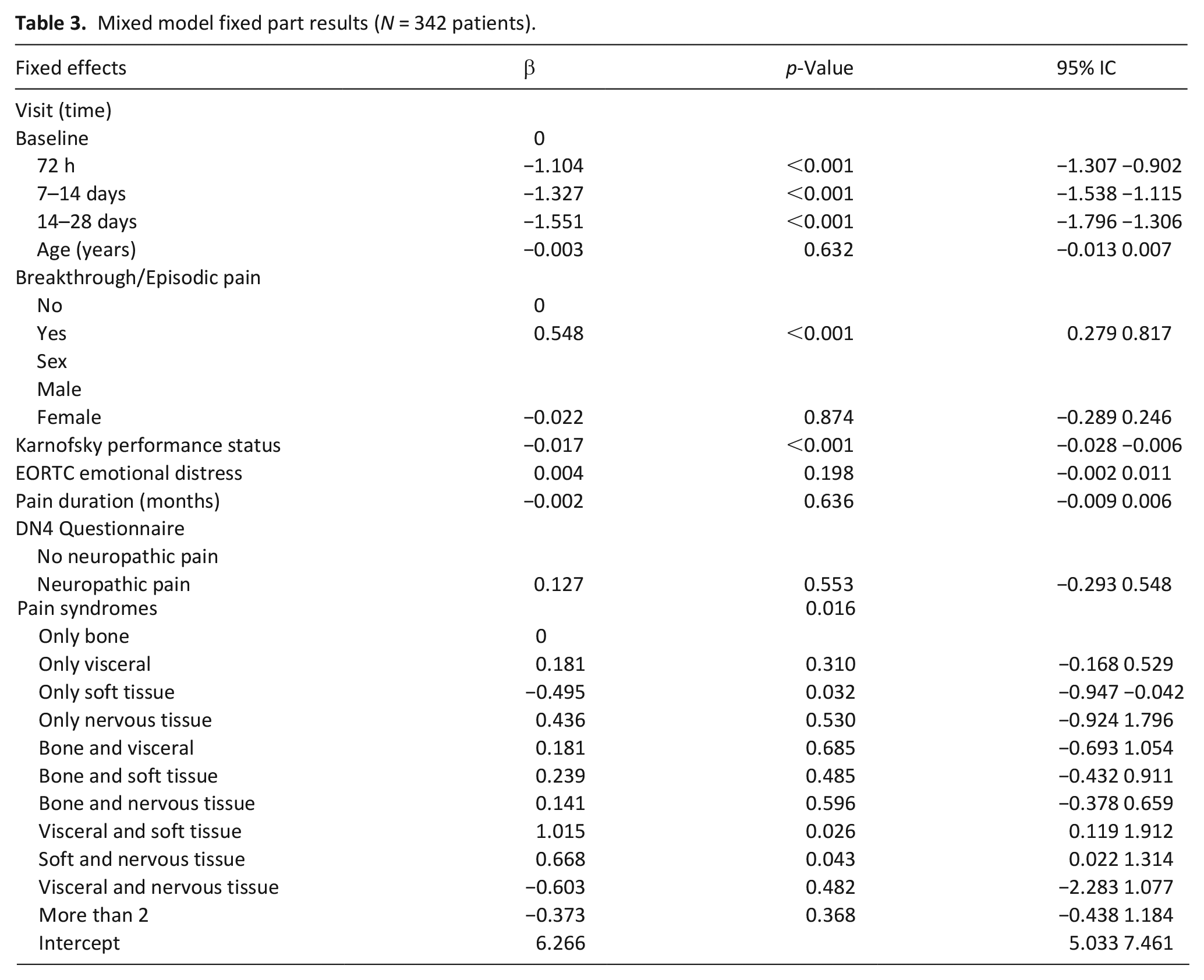
Pain intensity decreased significantly for all patients during time (p < 0.001). Age, sex, emotional distress, pain duration and neuropathic pain presence evaluated by the Douleur Neuropathique 4 Questions (DN4) questionnaire were not significantly associated to pain intensity. Breakthrough/episodic pain was associated with higher pain intensity during follow-up (p < 0.001). The diagnosis of pain syndrome was overall significantly associated with mean pain intensity during follow-up (p = 0.016). Particularly, the concurrent presence of visceral and soft (p = 0.026) or soft and nervous tissue pain (p = 0.043) were significantly related to worse outcome, whereas pain due to only soft tissue damage with better outcome (p = 0.032).
Conclusions:
The recognition of specific pain syndromes may help to better classify cancer pain.
Undertreatment or lack of satisfactory pain relief remains common in people with cancer pain.
Better pain characterization is helpful to better classify cancer pain and is associated with treatment outcomes.
The role of the presence of specific pain syndromes, characterized by different clinical presentations and tissue involvement, has not been previously evaluated for this purpose.
This study analyzes the association between pain intensity during opioid therapy and patients’ and pain characteristics.
We identify baseline prognostic factors which could guide improvements on cancer pain classification.
We show that specific pain syndromes are associated with pain intensity during pain management with opioids and confirm the relevance of breakthrough/episodic pain in conditioning pain relief.
The recognition of distinct cancer pain syndromes could lead to better cancer pain classification and treatment in clinical practice.
Training programs on appropriate pain assessment and multicenter clinical trials confirming the results of the present article are needed to confirm the clinical usefulness of this approach.
Introduction
Pain is estimated to affect around 65% of people with advanced cancer, significantly impacting their quality of life. 1 The World Health Organization (WHO) program against cancer pain and the three-step analgesic ladder have been crucial in building a global internationally accepted management strategy,2,3 which has since then been followed by many guidelines.4–6 However, under-treatment or lack of satisfactory analgesic response is still common and an appropriate assessment remains key to identify difficult pain conditions. 7
Cancer-related pain, according to the ICD-11 classification, 8 is a wide definition including both pain directly due to the tumor progression or metastases, and pain caused by surgery, chemotherapy, target biological agents, immunotherapies or radiation consequences. Pain due to cancer and treatment-related pains need furthermore to be distinguished as entities characterized by different causes, mechanisms and clinical presentations, with different implications in care strategies.
Several studies have described domains or variables that can be useful in classifying cancer pain and identifying prognostic factors for analgesia. These include pain characteristics, such as: pain intensity, breakthrough pain and neuropathic pain presence and patients’ characteristics such as: drug-related behaviors, psychological and cognitive function.9–13 These characteristics and their combination have been used in some classification systems, such as the Edmonton Classification System for Cancer Pain, but only neuropathic pain, breakthrough pain, psychological distress and baseline pain intensity resulted to have a clinical impact on pain outcomes.9–13
An additional diagnostic approach, recognizes cancer pain syndromes identifying the actual cancer lesions causing pain as consequences of the underlying disease or its treatment.14,15 These syndromes have been summarized in a checklist based on the anatomical site of tumor invasion causing the pain or with the lesion caused by anticancer treatment.16–18 Using this simplified pain syndrome list, a prospective international study 16 demonstrated that the majority of patients with pain directly due to cancer present with one or more pains caused by different types of tissues involvements. This syndromes checklist has never been specifically tested as to its potential impact on clinical outcomes. In this article we explore the association of pain syndromes with pain intensity during opioid medication-based therapy, together with other patients’ and pain characteristics already used in available classification systems.10,13
Methods
Aim of this study was to assess if a pain diagnosis based on a syndrome checklist together with other pain characteristics is associated with the analgesic outcome of pain due to cancer.
Study design and setting
A prospective longitudinal observational study (MOLO 13) on interaction between clinical and genetic factors and opioid analgesia in patients with cancer pain is being conducted at the Palliative Care and Pain Outpatient Clinic of the National Cancer Institute of Milan. Here we present a secondary analysis of preliminary data aimed at identifying clinical factors from the baseline visit (the day of enrollment in the study) and their association to pain intensity during the study follow up period of 28 days. The pharmacological treatment of enrolled patients was based on existing clinical guidelines2–5 and on opioid titration according to common clinical practice and was not subject to any modifications due to enrollment, therefore, no pre established dose limitations were planned or applied.
Study population and recruitment
Patients with cancer pain seen at our outpatient clinic from May 2015 to June 2019 were prospectively screened. Patients were eligible if they were older than 18 years, had diagnostic evidence of locally advanced or metastatic solid tumor with a life expectancy of 1 month or longer, had moderate to severe cancer pain (mean pain intensity in the last 24 h > = 4 on a 0–10 numerical rating scale (NRS)). Only pain directly caused by cancer was considered in this study and patients could be included if they were already receiving or needed treatment with WHO ladder step III opioids (morphine, oxycodone, fentanyl, hydromorphone, or buprenorphine).
Criteria of exclusion were the presence of psychiatric diseases or pathologies leading to impaired state of consciousness and cognitive capabilities, antalgic radiotherapy in the last 2 weeks or planned during the study, documented presence of moderate to severe renal failure (plasma creatinine >1.5 mg/mL with a creatinine clearance <60 mL/min) and pain due to anticancer therapies.
Data collection and clinical assessments
After giving written informed consent a standardized assessment was performed for all enrolled patients both at study enrollment (baseline) and follow-up period. Patients’ follow-up consisted of five subsequent visits, respectively 3, 7, 14, 21, and 28 days after baseline evaluation. In the event that the patient could not reach the clinic in person, the follow up assessment could be conducted by phone. Questionnaires were completed on paper during in-person visits or by the physicians, based on the patients’ answers, when the visits were conducted by phone. Schedule of enrollment and assessments is explained below and summarized in Figure 1.
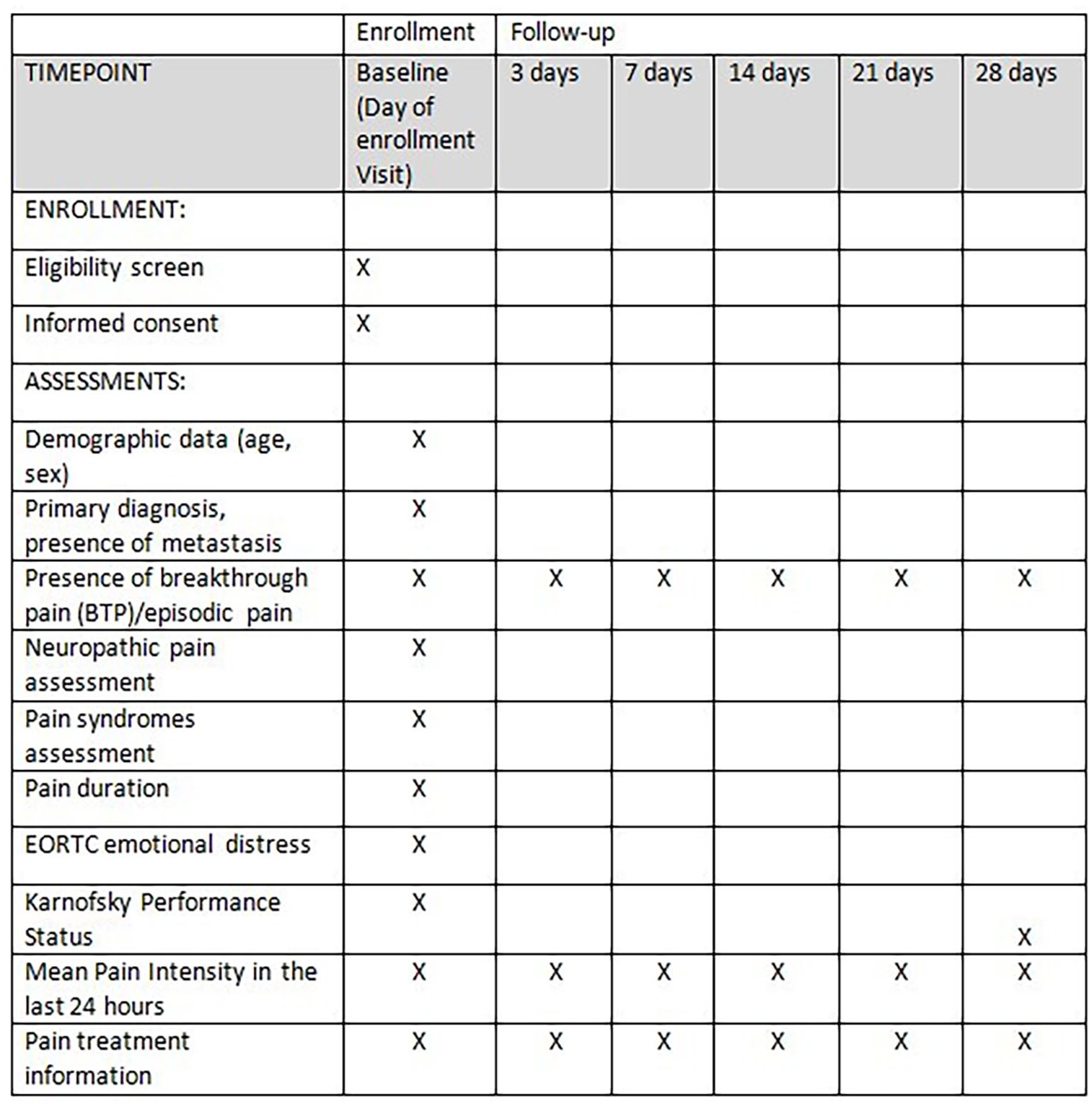
Schedule of enrollment and assessments.
Study enrollment visit
Basic demographic and clinical data were collected by a clinical researcher, including primary cancer diagnosis, presence of metastases and anticancer treatments. Pain treatment pre-study was recorded, including opioids and adjuvants with their respective dosages; opioid dosages were converted into milligram oral morphine equivalent daily dose (MMED) 19
Clinician collected pain data
A palliative care physician, recorded the following pain related data: pain duration; presence of breakthrough pain (BTP)/episodic pain; pain treatment and pain syndromes. In this study the presence of any transient pain exacerbations was defined as presence of BTP/episodic pain. 20 Pain duration referred to how long the patient was experiencing pain at the moment of enrollment, and was measured in months. Pain syndrome were identified based on cancer disease localization, pain clinical history, physical examination and available diagnostic tests demonstrating tissues’ involvement. The physician had to choose one or more pain syndromes using a codified list.16–18 For the purpose of this study, pain syndromes are grouped in one of four general etiologies using the classification of specific syndromes provided in the list16,23: pain due to bone, visceral, soft or nervous tissue involvement by cancer. The pain syndrome was therefore used to assign patients to one or more of the previous four grouping categories, resulting in 16 possible combinations depending on the number of tissues involved.
Patient reported pain measurements data
Pain intensity was assessed with the Italian Brief Pain Inventory–short Form questionnaire. 21 Mean pain intensity in the last 24 h using 0–10 numerical rating scales on day of enrollment and at follow up visit were used as outcomes of the present study.
Functional status and psychological distress assessment
Performance status was rated using the Karnofsky Performance Status Scale (KPS). Emotional distress was evaluated using the four questions (Q21-Q24) regarding emotional functioning domain of the EORTC QLQ-C30 questionnaire. 22 All four items in the EORTC QLQ-C30 module were scored on a Likert scale from “not at all” to “very much” and their average was linearly transformed to 0–100 scores, in accordance with the scoring instructions given by the EORTC Quality of Life Study Group; a higher score represents a higher (“better”) level of functioning.
Neuropathic pain assessment
the Douleur Neuropathique 4 Questions (DN4) was used as a screening questionnaire for neuropathic pain.23,24 The DN4 has good specificity and sensitivity in screening for the presence of neuropathic pain in chronic non-malignant pain. 25 It contains both interview questions and an objective examination. The interview consists of seven verbal pain descriptors (burning, painful cold, electric shocks, tingling, pins and needles, numbness, and itching), while the objective part contains three items assessing for sensory abnormalities: pinprick, tactile hypoesthesia, and pain to light touch. A score of 1 is given to each of the 10 items when positive. The final score ranges from 0 to 10 and a score of 4 or greater is the cut off value indicative of neuropathic pain. 25
Follow-up visits
Pain assessment included mean pain intensity in the last 24 h and worst pain intensity in the last 24 h. Any therapy or pain treatment variation was recorded. Reasons for ending prematurely the study were registered, including missing follow-up, abandonment of opioid therapy, or death.
Statistical analysis
Frequencies and percentages were used to describe categorical variables while means and standard deviations (SD) were used for continuous ones. In total, 13 categories of syndrome combinations were observed and included in the analysis. Given the longitudinal nature of the data and missing follow-up data for a few patients, a mixed model for repeated measures (MMRM) 26 was chosen to model the longitudinal mean pain intensity in the last 24 h measures as a function of pre-specified patient and pain-specific characteristics based on clinical experience and documented previous findings (sex, age, KPS, presence of BTP/episodic pain at enrollment, EORTC emotional distress score, pain duration and DN4), and pain syndrome classification. These variables were inserted in the fixed part of the model as continuous or as binary, except for nominal factor pain syndromes. Considering evidences of no substantial differences in efficacy among WHO third step opioids for cancer pain management,27,28 we decided not to include them in the model.
In the random part of the model, time was included as categorical variable with four categories: baseline; 2nd follow-up; 3rd and 4th visit; 5th and 6th. This selection was based on a preliminary estimation of coefficients of each time point, and those with close estimated coefficient were grouped in the same categories. Lastly, the reference category used for pain syndromes was “bone pain only,” based mainly on the numerosity of this category.
Mixed effect models are especially useful for identifying the role of individual differences in responses, while incorporating information from different measures at both individual or group levels, enhancing associations of the underlying components influencing response. The MMRM model did not specify any random effects on patient level, but instead modeled the correlation within the repeated measures over time taking into account that the residual errors are correlated. An unstructured correlation matrix of the repeated measures was selected among others 29 by Akaike Information Criterion (AIC) and Bayesian Information Criterion. The MMRM estimation method used was restricted maximum likelihood estimation. The model diagnostics were the verification of the linearity and independence of all variables and the normal distribution of the residuals. The model p values shown in the results are Wald test. The significance level was set at p < 0.05.
All data analysis was performed using STATA IC 16. 30
Ethical issues and study approvals
All participants gave written informed consent before enrollment. The study protocol and supporting documentation was approved by INT Research Ethics Committee (INT 153/13).
Results
From May 2015 to June 2019 3400 patients were screened at the outpatients clinic and 45% of them (1544) had pain. Of these, 27% did not require the use of WHO Step III opioids, 24% had pain due to cancer treatment or other causes unrelated to the oncological disease, 18% were undergoing antalgic radiotherapy, 4% had either a poor prognosis or follow-up was not possible, 2% had cognitive impairment and were therefore considered not eligible for the study. Only around 3% of the remaining eligible patients refused to participate, and a total of 350 patients (97% of potentially eligible) were enrolled in the study 8 patients (2.3%) had no follow-up visits and were excluded from the analysis (see Figure 2). Seventy-eight patients did not conclude the study but had at least one follow-up visit. The mean follow-up duration for these patients was 13.2 days (±6.4). Reasons for early dropping out were: loss to follow-up (31 patients, 8.8% of all 350 enrolled patients), death (17 patients), lack of compliance to detailed pain assessments (13 patients), transfer to other healthcare centers (13 patients), and other reasons (4 patients).
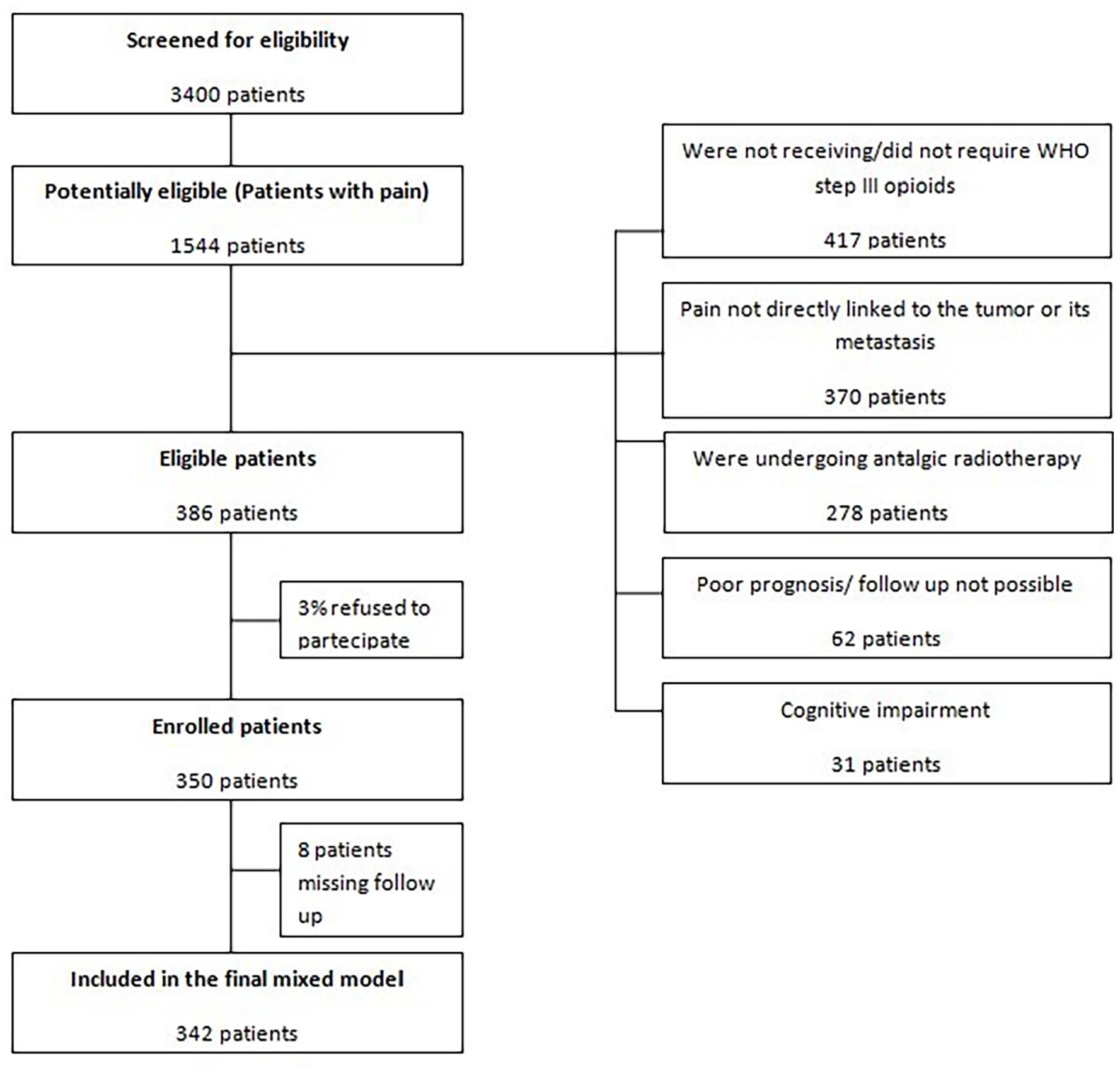
Flow diagram of screening and eligibility.
Table 1 reports baseline demographic and clinical characteristics of the 342 analyzed patients. Mean follow up time was 24.6 (±6.8) days. Mean age was 63.5 years old. The most frequent diagnosis were breast (19%) and lung cancer (15.8%), with 93% of patients having metastatic disease.
Demographic and clinical characteristics of patients at baseline (n = 342).
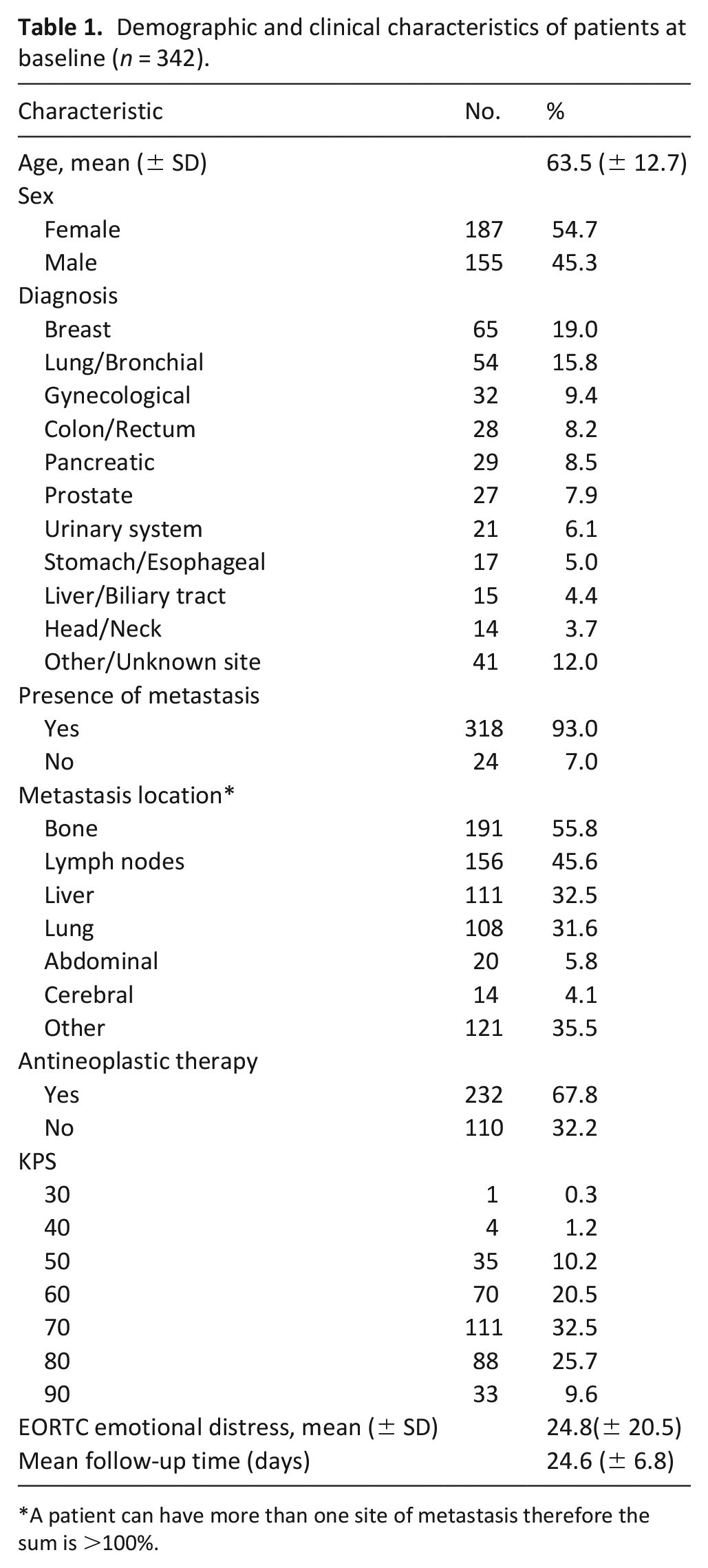
A patient can have more than one site of metastasis therefore the sum is >100%.
Pain and analgesic treatment characteristics data are reported in Table 2. The average pain duration at baseline was 12 months (± 17.4). Average of mean pain intensity in the last 24 h and worst pain intensity in the last 24 h scores were 5.4 (± 1.4) and 6.9 (± 1.8) respectively. The most common pain syndromes were bone pain (33.6%), visceral pain (26.9%) and pain due to soft tissue damage (10.8%). Around 28% of the analyzed patients presented with more than one tissue involvement and over 60% had BTP/episodic pain at enrollment. About 20% of patients had a positive DN4 result, indicating a possible neuropathic pain component. Eighty six percent of patients were already receiving WHO step III opioids before enrollment, 11% weak opioids and only 3% were not receiving any opioids. At baseline, 51.5% of patients were prescribed fentanyl, 43% oxycodone, 3.2% morphine and 2.3% buprenorphine. An opioid-switch during follow-up was done for only 12.6% of patients. Average Morphine Milligram Equivalents for baseline and during follow-up were 114.6 (± 118.2) and 144.8(± 136.9) respectively. 83% of patients were prescribed additional adjuvant analgesic drugs, mostly corticosteroids (45%) and anticonvulsants (31%).
Pain and treatment characteristics (n = 342).
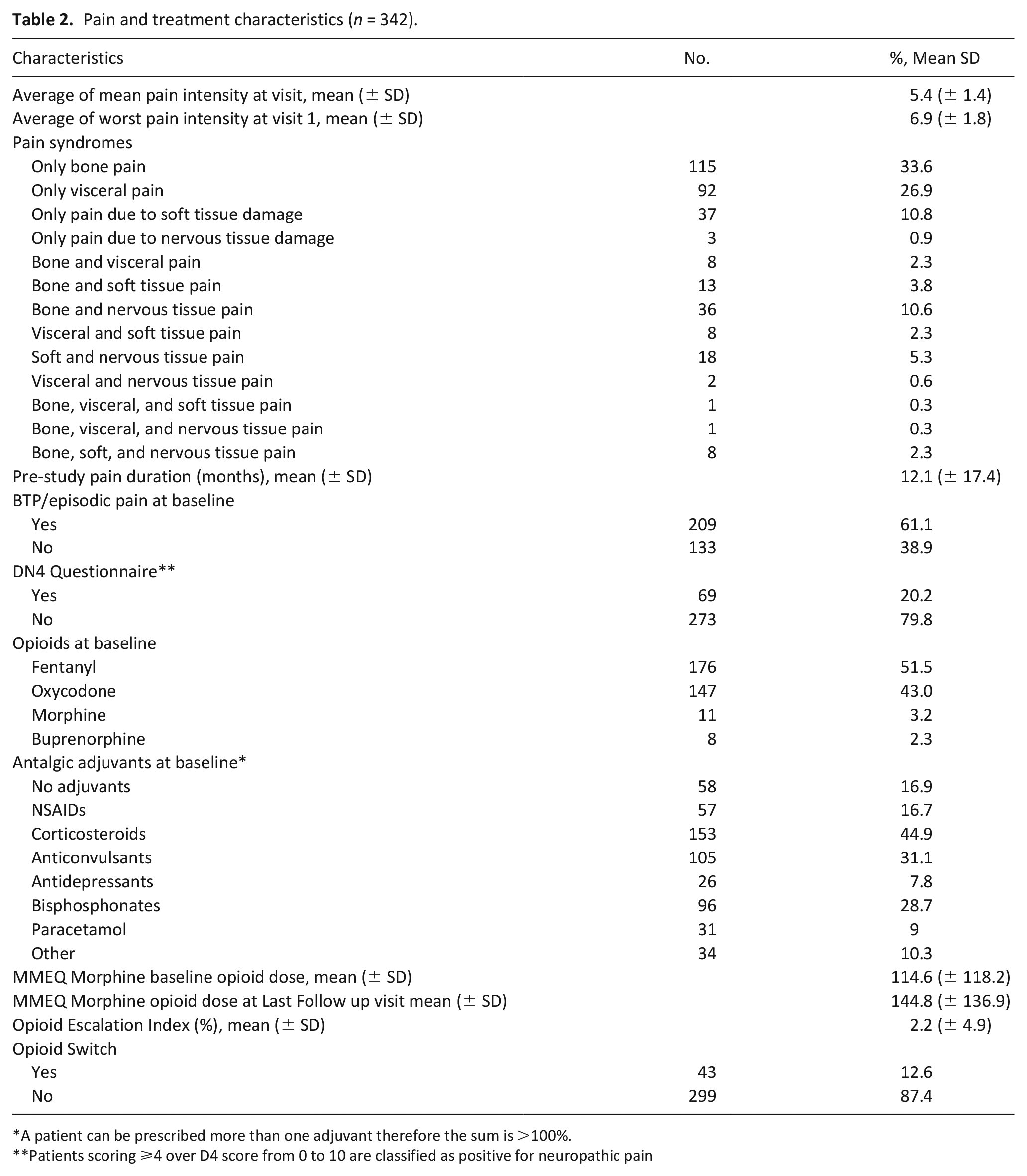
A patient can be prescribed more than one adjuvant therefore the sum is >100%.
Patients scoring ⩾4 over D4 score from 0 to 10 are classified as positive for neuropathic pain
Patients were classified based on the combination pain syndromes diagnosed by the treating physician. Figure 3 reports the mean pain intensity in the last 24 h score average during follow-up by pain syndrome combination groups, each represented by a different color.
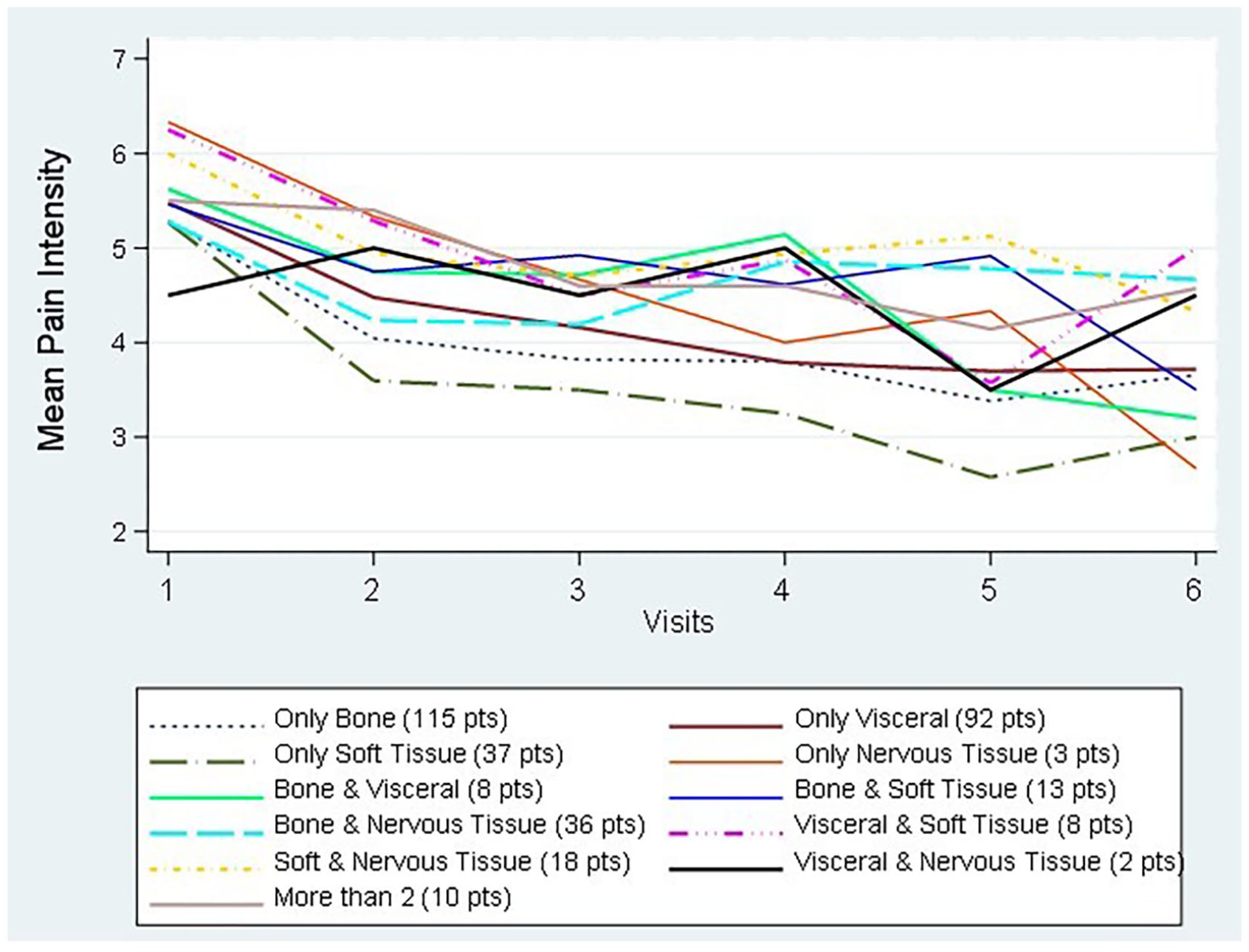
Average of mean pain intensity during follow-up for the different pain syndrome combinations.
Results from the fixed part of the multivariable MMRM (Table 3) showed several variables to be significantly associated to pain intensity during follow-up. Pain intensity decreased significantly during time with estimated average decreases from baseline of 1.1, 1.3, and 1.5 for the three time points identified. Age, sex, emotional distress, DN4 classification of neuropathic pain and pain duration were not associated to pain intensity. Patients with BTP/episodic pain had 0.55 pain intensity score higher than those without (p ⩽ 0.001). Overall, the presence of different pain syndromes was significantly associated to mean pain intensity during follow-up (p = 0.016). In particular, compared to bone tissue syndromes, the concurrent presence of visceral and soft tissue (β = 1.015, p = 0.026) or soft and nervous tissue (β = 0.67, p = 0.043) were significantly correlated to higher mean pain intensity in the last 24 h. The opposite was true for pain due to only soft tissue damage (β = −0.49, p = 0.032).
Mixed model fixed part results (N = 342 patients).
Of the patients with concomitant soft tissue and visceral pain, most were affected by pararectal–pelvic tissue infiltration resulting in pain associated with tenesmus, and retroperitoneal and abdominal pain due to distension or infiltration. The group of patients affected by both nervous and soft tissue damage, were mainly presenting pain due to infiltration of muscles and fasciae of the chest, abdominal wall or limbs with peripheral nerves damage.
Discussion
Main findings
This study reports on pain and patient characteristics associated with analgesic response. In particular, for the first time, we show that specific pain syndromes are associated with pain intensity during opioid based pain management.
The pain syndrome assessment resulted in being overall significantly associated with pain intensity in the statistical model. Using the group of patients with only bone pain as comparator, patients with only soft tissue pain and only visceral lesions have lower pain intensity levels, whereas, those with the association of soft and nervous tissue pains have higher pain intensities during follow-up (Figure 2). Similar findings were reported in another study, which also identified soft tissue pain as associated to shorter time for achieving pain control 31 but the authors do not provide information on how the classification was performed and whether patients had one or more types of pains.
In a small group of patients (eight patients), the concurrent presence of visceral and soft tissue induced pain was significantly correlated to worse pain control. Most of these patients (six patients) had pararectal–pelvic tissue infiltration and pain associated with tenesmus, a complex type of pain, for which little information is available in terms of both pathophysiology and appropriate management. 32
Neuropathic pain has been often associated with greater analgesic requirements, poorer outcomes, and greater disability,33,34 but its recognition as a component of pain due to cancer is far from homogeneous and standardized.35,36 In this study, DN4 classification on presence of neuropathic pain did not result in a significant effect in the multivariate model. In a previous study on this population, we showed that clinicians diagnosed neuropathic pain in some cases that did not reach the cut-off on the DN4 scores when a nervous tissue lesion was the cause of pain. 23 Furthermore, only in three patients (1%) we found that the pain cause was attributed to a neurological lesion only. More commonly nervous tissue lesions are associated with bone or soft tissue invasion, presenting with different outcomes. We hypothesize that depending on the tissues involved, a component of nervous tissue damage could lead to differences in clinical presentations and mechanisms. Still, the number of patients included in the above groups is relatively small, making it difficult to discuss about the generalizability of this result. A better assessment based on clinical and etiological information, considering the IASP criteria, but also the peculiarity of cancer dissemination across different tissues and different local tissue/cancer pain inducing mechanisms, may improve the recognition of neuropathic pains in subsets of patients leading to a better overall classification. 37
The relevance of BTP/episodic pain in conditioning worse pain is confirmed in this analysis. BTP/episodic pain has been associated with higher interference with general activities and poor pain management,38–40 resulting in the need of using additional specific medications.40,41 Our finding further emphasizes the relevance of the recognition of the presence of pain flares and their appropriate management.
Psychological distress is one of the domains reported and used in cancer pain classification systems9,10 which has also been associated with worse response to treatments.42,43 However, we did not find a significant relationship with pain intensity in this study. Average EORTC-QLQ C30 emotional status scoring for the patients under study was approximately 25/100, indicating a rather low level of psychological distress. This could in part explain the lack of significant differences in the analgesic response. Furthermore, it could be that part of the lack of statistically significant differences could be explained by the need of using a more thorough and specific for evaluating psychological distress.
Pain duration is another component that can be relevant in terms of pain intensity evolvement, considering phenomena such as tolerance to commonly prescribed analgesics or central sensitization. Yet, there is currently little information in regards and with one study confirming that patients with prolonged uncontrolled pain are likely to need more complex treatments and have higher pain intensities. 44 We, however, found no significant association between pain duration and pain intensity in the present work. This could be related to the fact that all the patients in this study had chronic pain in a range that did not account for a large variability in terms of pain history.
Demographic characteristics included in the model were age and sex, which were both found to not affect significantly pain intensity during follow-up. Previous reports about these two factors have been contradictory. Past clinical experience has led to the impression that females have an increased risk of experiencing pain with greater pain sensitivity compared to males. 45 Some studies have confirmed sex differences, showing that either females 46 or males 47 were less susceptible to common pain analgesics, while others, similarly to ours, have found no such differences.48,49 A meta-analysis on 13 different studies also demonstrated no significant differences in self-perceived pain between genders in cancer pain patients. 50
Similarly, older age has been associated with more 51 or less52,53 pain. Other studies,54,55 as ours, have found no significant relation between age and pain intensity. Different characteristics of patients enrolled in different studies, including sex and age distributions, can partly explain the high variability among different reports.
Strengths and limitations
This study’s strengths include its prospective longitudinal design with a significant follow-up period and a comprehensive and standardized assessment, with the identification of cancer pain syndromes. The inclusion in the study of only cancer pain directly linked to the tumor provides a unique homogenous picture, often lacking in similar studies.
There are some limitations to acknowledge. This is a single-site study held at a tertiary-level cancer center with its specific characteristics, in terms of sociocultural patterns and clinical characteristics of patients and clinical practice. These could limit the generalizability of the findings. Patients enrolled were all patients with chronic cancer pain but at different time points of their disease and with different pain duration. This could bring some more heterogeneity in the data. Cognitively impaired patients were not included in the present study and probably specific tools and assessment protocols are needed in the future in order to evaluate appropriately pain in these patients. In addition, biological differences and opioid tolerance development could contribute to unknown variability in the clinical outcome. Missing data, mostly due to loss during follow-up, affected 22% of cases although the statistical analysis based on mixed models, such as those used in our analysis are appropriate for handling this problematic.
Conclusions
In this study we have shown that cancer pain syndromes assessment carries a prognostic information regarding pain relief, and it provides a standardized way to classify pain according to presentation and anatomical lesion. The syndromic checklist, considering the different dimensions that characterize pain, cannot be proposed as a single component for an appropriate pain evaluation, but it could be helpful in integrating previous classification systems. 13 Testing in larger cohorts of patients and in clinical intervention trials in different centers the validity of a cancer pain syndrome classification would be necessary to confirm the results presented. Integration with additional components such as metabolomics and genetics of patients could help in enlightening factors affecting pain susceptibility and analgesic response among individuals.
Footnotes
Authorship
Augusto Caraceni, Morena Shkodra, Cinzia Brunelli, Stein Kaasa and Ernesto Zecca were involved in the conception and original study design. Ernesto Zecca, Morena Shkodra, Mariangela Caputo, Paola Bracchi, and Silvia Lo Dico were involved in the data collection process. Cinzia Brunelli, Morena Shkodra, Rosalba Miceli and Gabriele Infante have worked on the analysis and interpretation of the data. Morena Shkodra wrote the first draft and all authors contributed to reviewing and editing the final draft. The corresponding author attests that all listed authors meet authorship criteria and have approved the version to be published.
Data management
The data related to this study is maintained and managed according to organizational guidelines and ethical regulations. In the interest of patient confidentiality and anonymity, this information will not be made publicly available. Requests for further information can be directed to the corresponding author.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.C. has received honoraria from Angelini, Italfarmaco, Shionogi, Kyowa Kirin, Molteni, Pfizer/EliLilly Italia Spa, Mundipharma and Ipsen Spa institutional grant. The sponsors had no role in the interpretation or writing of the study. The other authors declare no conflicts of interest.
Funding
The authors(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research received funding by the PAIN-Net project, part of the EU Research Framework Programme H2020/Marie Skłodowska-Curie Actions, grant agreement number 721841.
