Abstract
Background:
Malignant bowel obstruction, a complication of certain advanced cancers, causes severe symptoms which profoundly affect quality of life. Clinical management remains complex, and outcome assessment is inconsistent.
Aim:
To identify outcomes evaluating palliative treatment for inoperable malignant bowel obstruction, as part of a four-phase study developing a core outcome set.
Design:
The review is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA); PROSPERO (ID: CRD42019150648). Eligible studies included at least one subgroup with obstruction below the ligament of Treitz undergoing palliative treatment for inoperable malignant bowel obstruction. Study quality was not assessed because the review does not evaluate efficacy.
Data sources:
Medline, Embase, the Cochrane Database, CINAHL, PSYCinfo Caresearch, Open Grey and BASE were searched for trials and observational studies in October 2021.
Results:
A total of 4769 studies were screened, 290 full texts retrieved and 80 (13,898 participants) included in a narrative synthesis; 343 outcomes were extracted verbatim and pooled into 90 unique terms across six domains: physiological, nutrition, life impact, resource use, mortality and survival. Prevalent outcomes included adverse events (78% of studies), survival (54%), symptom control (39%) and mortality (31%). Key individual symptoms assessed were vomiting (41% of studies), nausea (34%) and pain (33%); 19% of studies assessed quality of life.
Conclusions:
Assessment focuses on survival, complications and overall symptom control. There is a need for definitions of treatment ‘success’ that are meaningful to patients, a more consistent approach to symptom assessment, and greater consideration of how to measure wellbeing in this population.
Keywords
Malignant bowel obstruction is a complex condition, both in terms of its aetiology and management. For patients nearing the end of their life, multiple options for palliative treatment exist and the best option is not always clear.
Recruitment to control arms of randomised clinical trials is difficult in inoperable malignant bowel obstruction when patients are at the end of life.
Outcome assessment in inoperable malignant bowel obstruction is currently inconsistent.
Adverse events and survival are the most prevalent outcomes measured in studies of inoperable malignant bowel obstruction patients, when symptom relief might be the most appropriate objective.
Definitions of treatment success and methods of measuring key symptoms vary across palliative interventions used to achieve symptom relief.
Few studies measure patients’ quality of life, and those that do struggle to conduct meaningful assessments because of patient deaths in the follow-up period.
There is a need for greater consistency in the way that measures of pain, nausea and vomiting are captured in the assessment of palliative interventions for inoperable malignant bowel obstruction.
Success of treatment should encompass a measure related to patients’ wellbeing.
Quality of life should be measured in a way which is appropriate for palliative settings, and captured in a short window of time in a way that is meaningful and minimises the burden of assessment for patients.
Background
Bowel obstruction is a common complication of advanced cancer1,2 which prevents intestinal transit and digestion. This causes severe pain, nausea, abdominal distension and vomiting 3 and can have profound effects on a person’s quality of life.4,5 An obstruction can be mechanical (caused by the infiltration of a tumour) or functional (caused by a lack of motility), and can present as a singular blockage or multiple blockages caused by diffuse carcinomatosis. The wide range of definitions of the condition contribute to difficulties in establishing its incidence.6–8 It is most prevalent in colorectal cancer, affecting up to 29% of patients, 9 and in ovarian cancer, affecting up to up to 51% of patients. 6
The management of malignant bowel obstruction is complex and controversial. 10 Surgery to remove the blockage is often not an option in advanced disease 3 because symptom relief is often short term, and patients are at risk of complications and an increased length of hospital stay.11,12 Use of nasogastric tube decompression can relieve the symptoms, but is often uncomfortable for patients.13,14 For inoperable malignant bowel obstruction, palliative intervention options include placing an expandable stent or a venting gastrostomy for decompression, or a more conservative approach using medication to reduce intestinal secretions, nausea, vomiting and pain.
There is currently little consensus over how to evaluate the outcome of treatments for inoperable malignant bowel obstruction.2,10 A mix of procedural and non-procedural interventions are used to relieve the obstruction and/or its symptoms. Often, symptoms are addressed simultaneously, using drugs such as somatostatin analogues to reduce intestinal secretions, antiemetics to control nausea and vomiting and analgesics for pain relief, but experience in pharmacological interventions is limited and sometimes theoretical when it comes to sequencing and combining medications.12,15 There is also a lack of agreement on clinically relevant outcomes and timepoints for measuring symptom control. 15
The development and use of a standardised set of outcomes across clinical research studies of inoperable malignant bowel obstruction would improve the consistency of outcome reporting, allow comparisons between clinical trials and inform clinical decision-making. The aim of this review was to identify the range and suitability of outcomes currently used to evaluate palliative treatments for inoperable malignant bowel obstruction, including procedural interventions for intestinal decompression (stenting or venting gastrostomy), non-procedural pharmacological interventions and the administration of parenteral nutrition. It comprises Phase I of a four-phase study developing a core outcome set (COS) for the assessment of inoperable malignant bowel obstruction in research and clinical practice. 16
Methods
The protocol for this review was registered with the International Prospective Register of Systematic Reviews (PROSPERO ID: CRD42019150648). The review follows methodology recommended by the Core Outcome Measures in Effectiveness Trials (COMET) initiative, 17 and is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 18
Search strategy
The following databases were searched in October 2021 using strategies developed through discussion with an information retrieval specialist: the Cumulative Index to Nursing and Allied Health Literature (CINAHL), Cochrane CENTRAL, Embase, MEDLINE and psycINFO. Additional searches were conducted through Caresearch, OpenGrey and BASE. The search was limited to studies of adults, with no date or language restrictions. Search strategies are available via the PROSPERO registry. Reference lists of systematic reviews of studies of palliative interventions for inoperable malignant bowel obstruction were hand searched for relevant primary studies not captured by the database search.
Study eligibility and selection
Given the complex aetiologies of malignant bowel obstruction and the difficulties in comparing studies that this presents, our definition of the condition for the purposes of this review is restricted to the obstruction of the intestines distal to the ligament of Treitz as a result of a cancerous tumour.8,12 In order to capture as wide a range as possible of outcomes, the review included RCTs, quasi-RCTs, single arm trials and observational studies reporting outcomes on clearly defined palliative groups or subgroups of patients with advanced, unresectable cancer undergoing pharmacological (‘medical’ or ‘conservative’) treatment, endoscopic or temporary decompression procedures (stents or venting gastrostomy) or parenteral nutrition to treat patients with malignant bowel obstruction without concurrent chemotherapy. Eligible studies included at least one subgroup undergoing a non-surgical intervention with palliative intent, in any study setting, with no restrictions on the period of follow-up.
We excluded studies in which patients with gastric outlet obstruction (above the ligament of Treitz) made up all or the majority of the palliative sample, and studies solely focused on obstructions of benign aetiology (adhesions or radiation enteropathy). We also excluded studies without a clearly defined palliative inoperable malignant bowel obstruction group, studies of interventions including chemotherapy unless a non-chemotherapy group was assessed separately, and studies evaluating the technical success of endoscopic procedures without assessing patient-relevant outcomes. Studies were also excluded if the abstract cited clinical success as the sole outcome with no accompanying patient-relevant definition of ‘success’. Qualitative studies were excluded; a systematic review of qualitative studies was undertaken separately (PROSPERO ID: CRD42020176393). 19
Papers were collated using Endnote X7 (Thompson Reuters, New York, USA) and duplicates removed. All abstracts were screened independently by AB and GO against eligibility criteria using Abstrackr (Centre for Evidence Synthesis in Health, Brown School of Public Health, Providence, Rhode Island, USA). Full texts were also screened independently by AB and GO. Disagreements over inclusion were resolved by discussion (AB, GO, JB). Abstracts citing outcomes of completed trials meeting the eligibility criteria were included where a published full text was not available. For papers in languages other than English, full texts of methods, results (including tables) and discussion sections of each study were translated using GoogleTranslate and edited for clarity; this produced a level of translation adequate to meet the data extraction requirements of a review of outcome terminology, and enabled the inclusion of a broader range of papers.
Data extraction
Data on study designs, aims, settings, sample sizes, comparison groups/interventions and cancer types were extracted from full text articles by AB and GO using a data extraction form in Microsoft Excel® piloted before extraction commenced. We anticipated that the heterogeneity of study designs and outcomes would not allow the synthesis of statistical data, and measures of effect were not extracted. The aim of the review was to conduct a descriptive synthesis of outcome reporting. 20
Indexing of outcomes and domain categorisation
Outcomes, the frequency of their occurrence, outcome definitions, timepoints and patient-relevant statements in descriptive text were extracted verbatim using NVivo 12 (QSR International, Burlington, MA, USA) to retain contextual information, categorised by intervention type. ‘Outcome’ was defined as any term used in included papers to specify measurement of a clinical endpoint or physiological event, in any domain. Where a primary outcome was not specified, this was inferred as the first outcome reported in study results. Details of patient-reported outcome measures (PROMs) used in included studies were also collected.
Two lists of verbatim outcomes were produced: Outcomes List 1 included all stand-alone clinical and physiological endpoints (Supplementary File 2), Outcomes List 2 included individual items extracted from patient-reported outcome measures (Supplementary File 3), as recommended by the Core Outcome Measures in Effectiveness Trials (COMET) initiative. 17 All outcomes were categorised under the following COMET domains 21 : physiological/clinical (including gastrointestinal and nutrition outcomes), life impact, resource use, death (including mortality and survival) and adverse events. Synonymous outcomes in each list were pooled and combined into standardised terms, and this process was reviewed by members of the study Steering Group (EB, JB, DC, MJ, FM, GO, SN, KS).
Assessment of bias
The objective of the review was to extract, analyse and pool outcome terms (verbatim) and to count the frequency of their use to indicate which outcome measures are most prevalent in the assessment malignant bowel obstruction. Inclusion criteria focused on gathering as broad a range of outcome measures as possible. The review did not assess the methodological quality of studies as it did not aim to draw any conclusions related to the efficacy of treatments, or to evaluate the research design of included studies.
Results
Search results
Search results are summarised in Figure 1. Of the 80 papers included in the review, 12 reported RCTs (two papers reporting different outcomes for the same RCT), 3 quasi-RCTs, 8 single-arm trials and 57 observational studies, with a total of 13,898 participants. For one single-arm trial not yet published, outcomes were extracted from trial results on ClinicalTrials.gov and a published abstract. 22 Study characteristics are summarised in Supplementary File 1. The distribution of included studies by year and intervention type is shown in Figure 2.
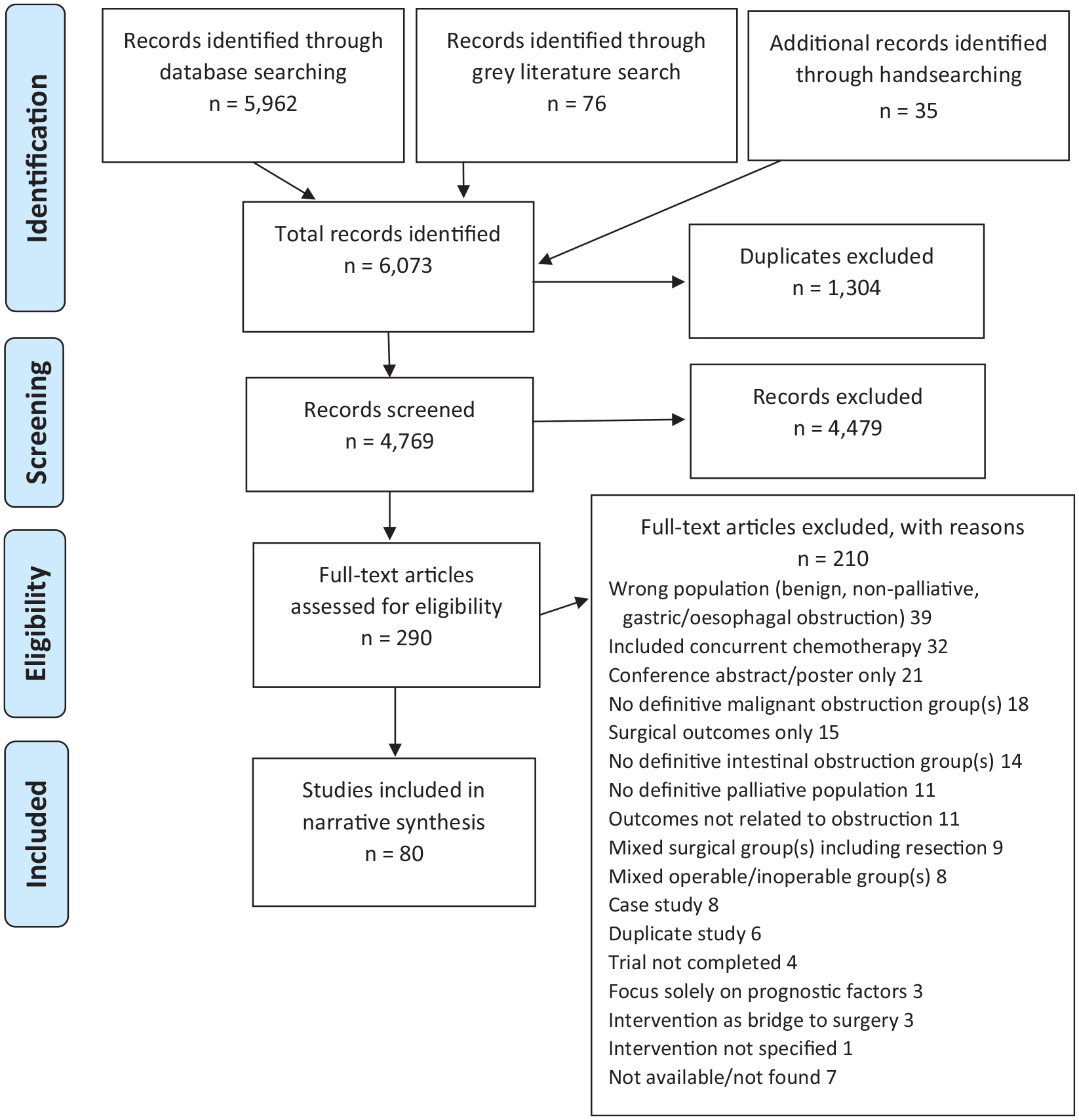
PRISMA diagram of studies investigating palliative treatment of inoperable malignant bowel obstruction.
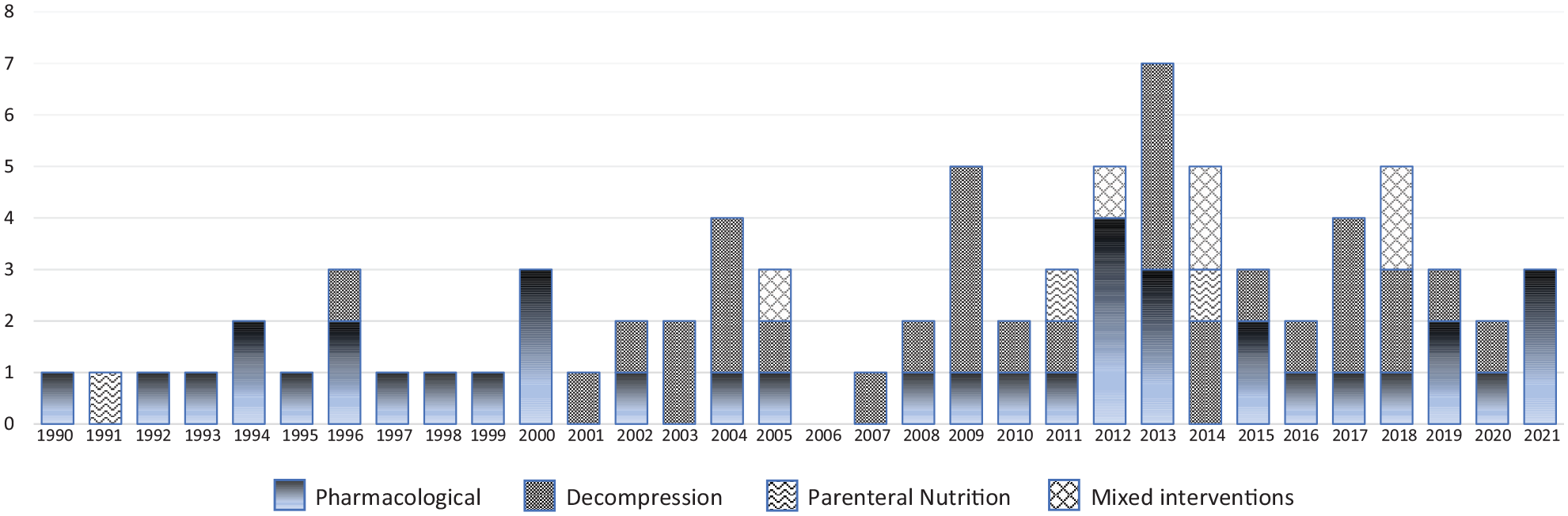
Distribution of included studies by year and intervention type.
Characteristics of included studies
Participants and interventions
The 23 papers reporting on clinical trials included 1311 participants, of which 53% took part in pharmacological trials,22–37 14% in trials of decompressive procedures (stenting/venting gastrostomy),38–42 4% in a trial of a traditional Chinese remedy (Da-Cheng-Qi) 43 and 3% in a trial of parenteral nutrition. 44 The number of participants enrolled on the 11 randomised controlled trials ranged between 17 34 and 106.23,29 One early RCT compared dexamethasone to a placebo, 24 eight RCTs compared treatment with somatostatin analogues to standard pharmacological treatment,23,26–28,30–32,34 one compared percutaneous transoesophagal gastrostomy (PTEG) to decompression using a nasogastric tube, 39 and one compared stenting with surgery (resection or stoma). 42
The 57 observational studies45–101 reported on the treatment of 12,587 patients, 92% of which were patients with malignant bowel obstruction. Of these palliative patients, 27% underwent surgery for their obstruction (initially or after temporary stenting) and 73% were inoperable. Surgery was conducted with the primary intention of relieving symptoms (adhesiolysis, bypass, colostomy, enterostomy, laparotomy, ileostomy, open gastrostomy, resection); outcomes for subgroups of operable patients were not extracted. Of the inoperable patients, 74% underwent pharmacological treatment and 3% unspecified pharmacological or decompressive treatment, 11% underwent stenting, 11% gastrostomy and 1% parenteral nutrition as a primary intervention. The grouping of samples without distinguishing between operable/inoperable or non-palliative/palliative patients was a common reason for exclusion during abstract screening. None of the included observational studies focused exclusively on palliative surgery. Two quasi-RCTs included subgroups undergoing palliative surgery, both comparing defunctioning colostomy with stenting.40,41
Settings
The majority of studies took place in hospital settings (88%), 14% of these in specialist cancer centres, 5% reporting the inclusion of patients in palliative care units, 5% included hospice patients and 10% included patients being cared for in their home. Studies took place in Europe (34%), North America (28%), Asia (26%) and remaining studies in Russia and the Middle East. Italy was the source of 48% of the European studies, exploring a mix of palliative interventions and outcomes related to home care.
Cancer types
The primary cancers of study participants are shown in Table 1. The majority of the studies (63%) recruited mixed samples including people with a range of advanced cancers, based on their need for symptom palliation. Nine studies focused exclusively on patients with colorectal cancers,45,57,60,73,74,77,89,92,93 nine studies focused on patients with gynaecological cancers.32,37,61,69,79,80,86,87,94
Primary cancers of patients recruited to the 80 studies included in the review.
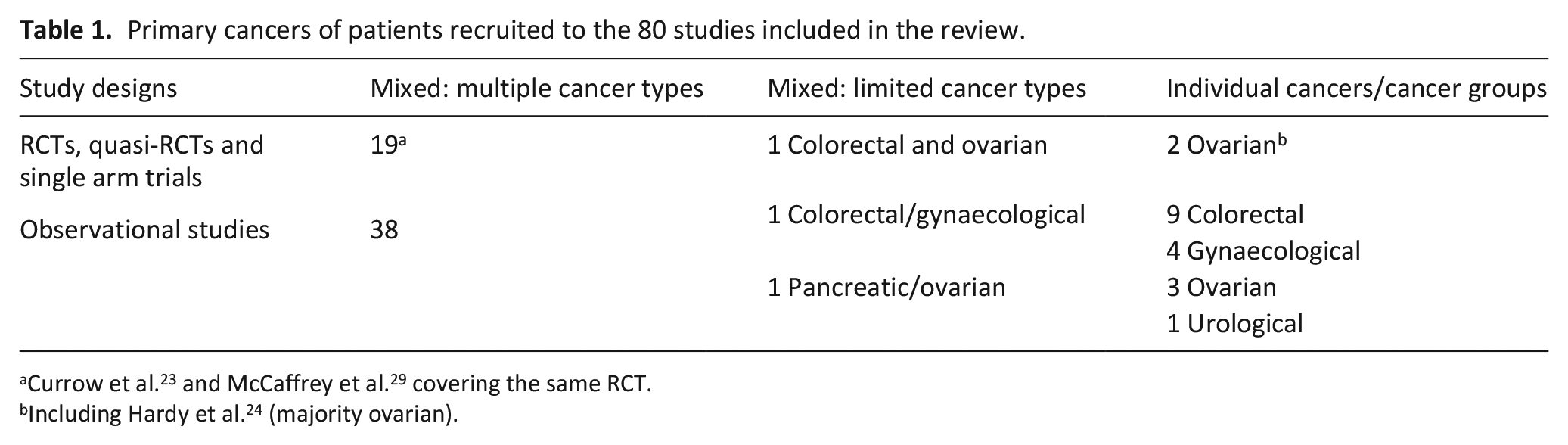
Including Hardy et al. 24 (majority ovarian).
Identification of outcomes
A total of 343 individual terms reflecting individual and composite outcome measures were extracted verbatim from the 80 studies and categorised under COMET domains. 21 Synonymous outcomes were pooled into 90 standardised terms (see Supplementary File 2). The distribution of these outcome measures across COMET core areas and domains is shown in Table 2. A summary of the frequency of outcomes, listed by intervention, is supplied in Supplementary File 4. The majority of outcome measures were related to gastrointestinal symptoms and nutritional intake, reflecting the symptoms of malignant bowel obstruction. Composite quality of life measures were poorly represented. The number of outcomes under the ‘life impact’ domain reflect the reporting of discharge settings or place of death, which were categorised under ‘personal circumstances’, defined by the COMET taxonomy as relevant to the patient’s environment or place of care.
Distribution of the 90 standardised individual outcome terms across COMET domains.

Composite outcome measures
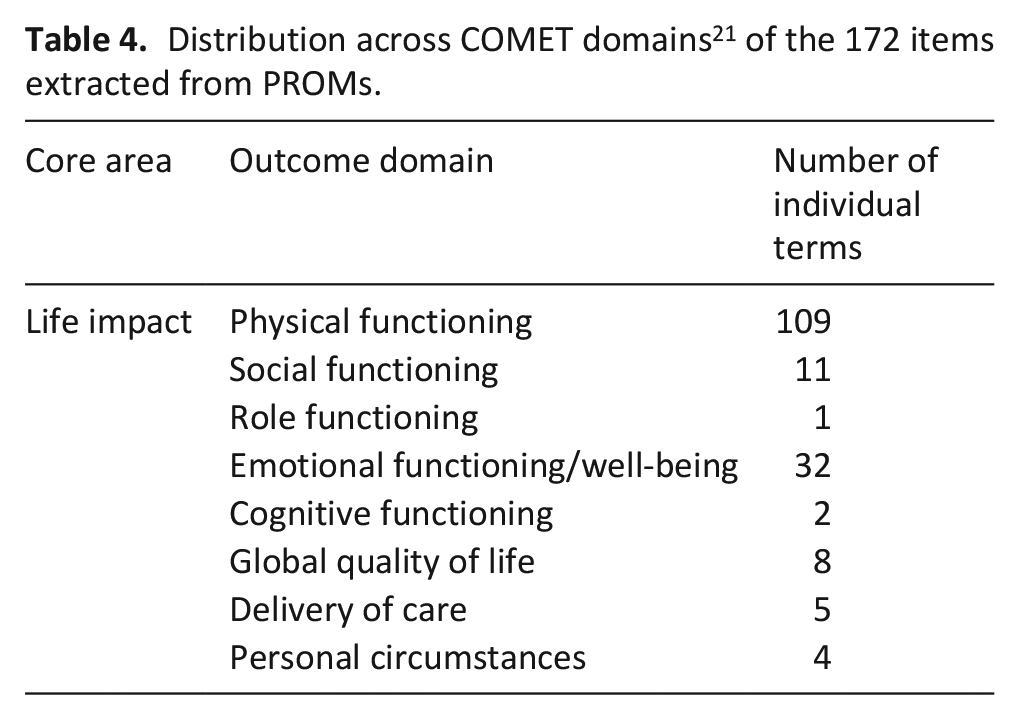
A total of 21 patient-reported outcome measures (PROMs) were used across all the studies included in the review, 14 of which used validated measurement tools and 8 of which created customised scales. Multiple-item validated scales used to assess physical symptoms and health related quality of life are shown in Table 3. Measurement tool items were separated out into individual items as recommended by COMET 17 ; the distribution of 172 items across domains is shown in Table 4. Synonymous outcomes were recategorised under COMET domains 21 and pooled into 50 standardised terms (see Supplementary File 3). Table 4 shows a predominance of items assessing physical functioning over items assessing emotional and social functioning.
Multiple item assessment scales used in studies.

Distribution across COMET domains 21 of the 172 items extracted from PROMs.
Evaluation of outcome definitions
Of the 343 individual outcome terms, 67 were accompanied by a definition: 38 of these definitions were related to measures of overall treatment success or efficacy, 22 to measures of overall symptom control and 6 to other individual outcomes (complications, lumen patency, readmission, remission rate, resolution of bowel obstruction, hospital-free days). Definitions of success varied according to intervention type. Procedural or technical success in stenting and venting gastrostomy studies was distinguished from clinical success. Definitions of clinical success for decompression included the resolution of obstruction, the relief of symptoms and/or the return of normal bowel function accompanied by toleration of oral intake. Symptom control was reported as ‘response’ in 21% of studies reporting pharmacological interventions. In studies of pharmacological interventions, ‘success’ and ‘response’ were defined as the reduction of symptoms, the most prevalent associated measure being a reduction in vomiting.
Approaches considered as ‘conservative treatment’ or ‘medical management’ were variously defined as the insertion of a nasogastric tube and administration of fluids, 32 pharmacological treatment23–25,30,31 or tube decompression with fluids and pharmacological management,36,51 or undefined. 100 A rationale offered for including endoscopic procedures under conservative or medical management was that they do not require general anaesthesia or involve the same recovery time as surgery. 51
Outcome measures
The frequency of occurrence of outcome measures in the 80 included papers is supplied in Supplementary File 4, which lists outcomes by intervention type under COMET taxonomy categories. 21 Figure 3 shows outcomes ranked by frequency.
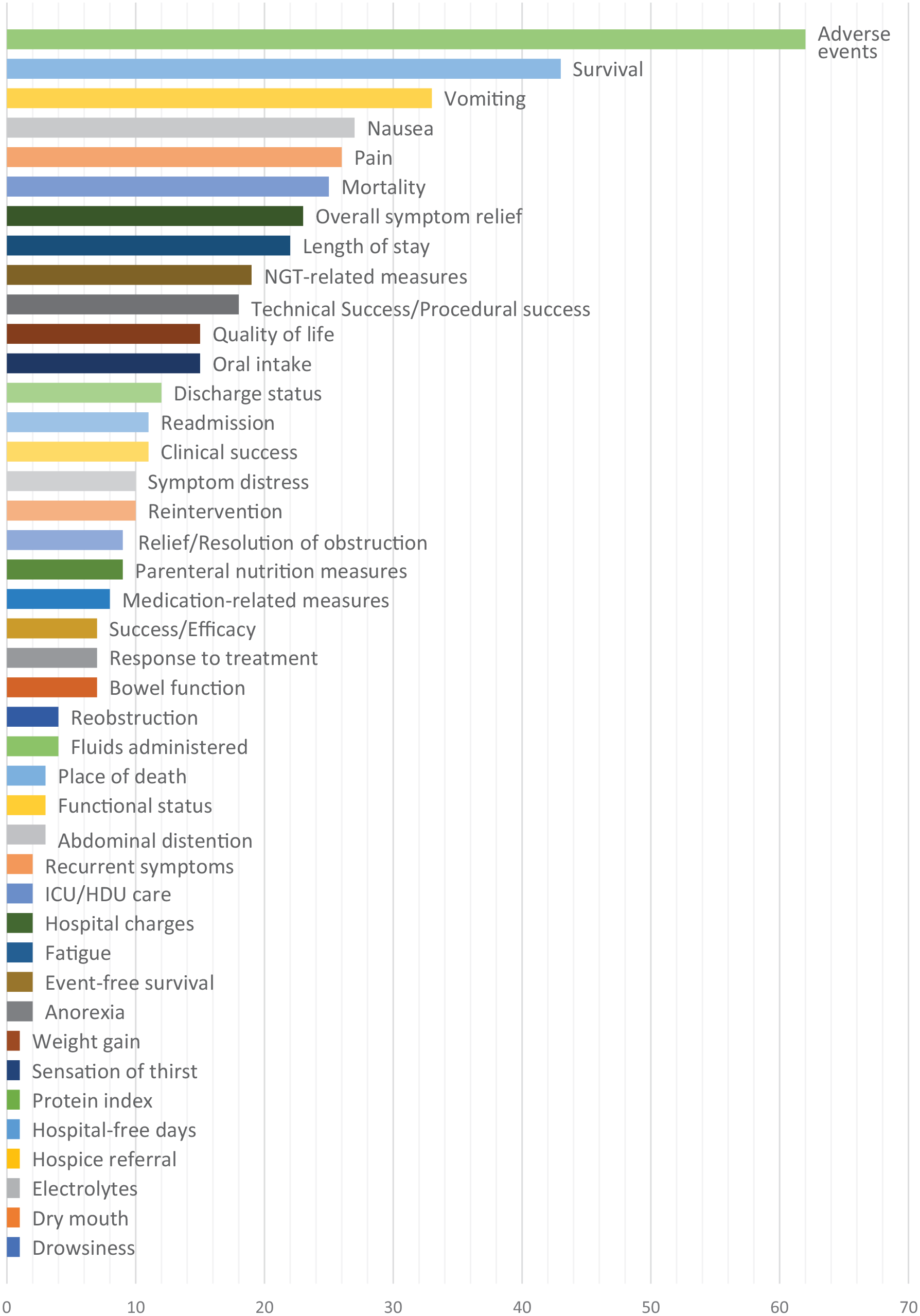
Outcomes used to assess inoperable malignant bowel obstruction in the 80 studies, ranked by frequency of occurrence.
Physiological and clinical
The range of approaches taken to the measurement of key symptoms are shown in detail in Supplementary File 4. In the gastrointestinal subdomain, the most prevalent outcome was overall symptom control or ‘response to treatment’, measured in 39% of studies. The most prevalent symptoms measured individually or as part of a composite symptom control measure included vomiting (41% of studies), nausea (34%) and abdominal pain (33%). Outcomes related to the use of a nasogastric tube to relieve vomiting were included in 24% studies (15 related to pharmacological interventions). Removal of nasogastric tube/changes in nasogastric tube secretion volume were reported as proxy measures for the control of vomiting (evaluated daily), and requirement for a nasogastric tube indicated a failure to adequately control symptoms.
The most prevalent nutritional measure was oral intake (19% of studies), reported as an indicator of symptomatic improvement and most often assessed in three stages: ability to tolerate fluids only, fluids and soft foods or fluids and solid foods. Measures related to parenteral nutrition were included in 13% of studies33,44,45,48,54,69,82,100; only three papers focused on parenteral nutrition met inclusion criteria for the review,44,48,56 all of which discussed controversies around its administration and patient and caregiver concerns about death by starvation when parenteral nutrition is withdrawn.
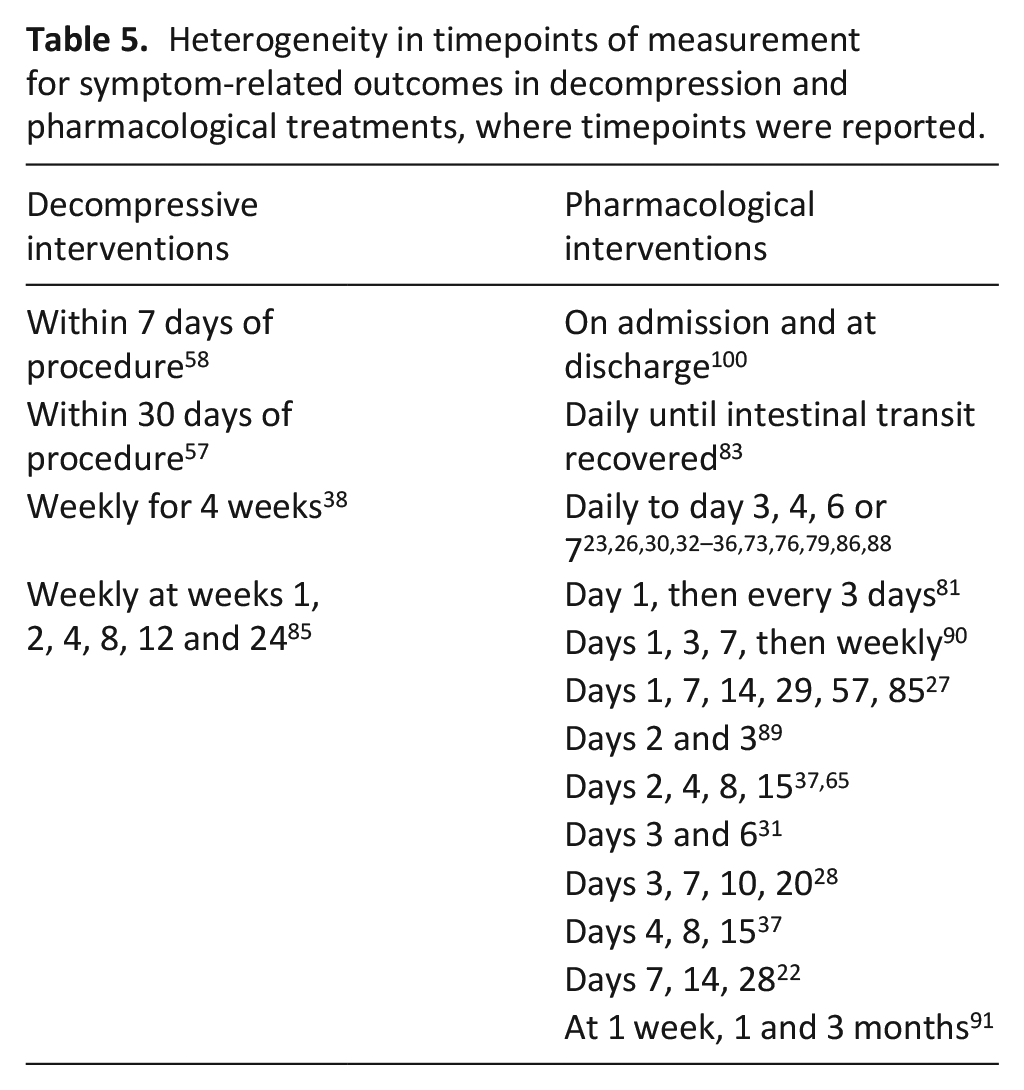
PROMs including measures of physical symptoms were used by 24% of pharmacological studies.22,23,27–29,33,35,65,81,100 Pharmacological studies assessed a more diverse range of symptom measures than papers exploring decompression procedures (see Supplementary File 4), only four of which made use of PROMs assessing global quality of life.39,42,85,101 The majority of pharmacological studies measured key symptoms daily, while studies of decompression conducted weekly assessments. The degree of heterogeneity in timepoints of measurement for symptom-related outcomes is shown in Table 5.
Heterogeneity in timepoints of measurement for symptom-related outcomes in decompression and pharmacological treatments, where timepoints were reported.
Life impact
Life impact is a core area in the COMET taxonomy, 21 and includes domains related to physical, psychological, social, emotional and cognitive functioning, global health related quality of life and personal circumstances. Assessment of quality of life was attempted in 19% of studies. Validated summary measures used to assess quality of life are listed in Table 3, and were used in four studies.23,29,42,49,85 Two RCTs23,29,42, two observational studies49,85 chose quality of life as a primary outcome, but did not recruit or retain enough patients at follow up to enable successful analyses.
Shima et al. 35 used a customised summary quality of life measure. Implicit composite global quality of life outcomes 17 were assessed in three studies: ‘30 good days’, defined as days out of hospital subsequent to the date of consultation 49 ; well-being, defined on a VAS from ‘I don’t feel well at all’ to ‘I feel very well’ 28 ; and quality of life recorded using a VAS 102 showing faces with graded expressions from unwell/unhappy to well/happy. 65 Quality of life was assessed at treatment cessation, 29 daily in the week following intervention23,35,65 or weekly/monthly22,27,42,85,91 (with follow-up to 24 weeks in studies evaluating decompressive procedures). Treatment-related preferences or goals of care expressed by study participants rather than clinicians were referred to in the descriptive text of 15% of studies.34,48,50–52,59,66,68,69,72,85,86
Given that one of the aims of palliative care is to support patients achieve their preferred place of care, place of death (which does not appear elsewhere in the COMET taxonomy) was categorised under the life impact domain. This outcome appeared in 4% of studies53,59,69 evaluating percutaneous decompression tube/gastrostomy procedures, two of these citing the procedure as facilitating home death.53,69
Resource use and adverse events
Studies of procedural interventions reported a higher proportion of resource related outcomes than studies evaluating pharmacological interventions or parenteral nutrition (see Supplementary File 4). Length of hospital stay was the most frequently assessed (28% of studies), the other most prevalent outcomes being discharge status (15%) readmission (14%) and reintervention (13%), reflecting the likelihood of re-obstruction and the recurrence of symptoms or the occurrence of complications.
The most frequently measured outcomes were adverse events (78% of studies); terminology to describe adverse events included ‘adverse effects’, ‘side effects’, ‘toxicity’ or ‘complications’. All papers reporting on decompression procedures reported details of complications (pain or bleeding; stent migration, perforation or tumour overgrowth; tube occlusion or infection at tube insertion site). Early complications were defined as events occurring within 30 days 57 of a decompression procedure. Overall, 66% of the pharmacological studies recorded details of adverse effects. The National Cancer Institute Common Terminology Criteria for Adverse Events (CT-CAE v3) was used to record toxicity in 4% of studies.23,37,65 About 16% of studies did not report adverse events, including three RCTs exploring pharmacological interventions.30,32,34
Studies focussing on nutrition recorded complications such as bone pain, catheter dislodgement, febrile episodes, hyperkalemia, infection, metabolic complications, pancreatitis and sepsis,48,56,66 with the exception of one quasi-RCT 44 which closed early because of poor patient accrual, reporting concerns from patients and families about starvation in the case of allocation to the control arm.
Mortality and survival
Mortality was a more prevalent outcome in evaluations of procedural interventions (56% of decompression studies) than pharmacological studies (8%), with timepoints of follow-up ranging up to 8 months. Survival was assessed in 54% of studies (63% of decompression studies and 42% of pharmacological studies), with variable follow up timepoints (days or months).
Discussion
From the 80 studies included in this review, 343 individual outcomes were extracted and pooled into 90 standardised terms. Items from 21 PROMs were separated out into 175 individual items and pooled into 50 standardised terms. All unique standardised terms were then categorised into six domains: physiological, nutrition, life impact, resource use, mortality and survival. The highest number of outcomes were categorised under the physiological domain, representing the gastrointestinal symptoms of inoperable malignant bowel obstruction. Other key domains represented in the studies include quality of life, nutrition, the need for further intervention, adverse events, mortality and survival. Assessment of survival and adverse events is comprehensive, but there is wide variation in the level of detail reported for adverse events, with some studies describing intervention-related events and others recording concomitant major events related to comorbidities.
The outcomes summarised in this review have been used to assess a patient population with advanced cancer, many of whom are approaching the end of life. For this population, survival is not always the most important outcome from the perspective of patients or clinicians – the aim of palliation is ‘a good outcome under. . .unfavourable circumstances’. 98 Previous reviews of treatment for malignant bowel obstruction exploring surgery, 103 surgery and medical management104,105 and parenteral nutrition 106 point out that the clinical resolution of bowel obstruction is not an adequate proxy measure for symptom relief or quality of life. These outcomes apply across all interventions (procedural and non-procedural, and parenteral nutrition), and this review indicates a need to be more precise in our definitions of ‘treatment success’ with this population.
Trials of treatments for inoperable malignant bowel obstruction are difficult to conduct – recruitment raises ethical concerns in a population suffering from distressing symptoms towards the end of life, and there is an understandable reticence among patients and caregivers to agree to randomisation.44,85 Difficulties also arise where symptom control is the primary outcome and control arms include patients with a poorer prognosis. Currow et al. 23 was the only study to address the issue of pre-consent, where patients who might be expected to develop an obstruction give their permission for inclusion in a trial before it commences.
The review demonstrates a level of consensus across studies on the central importance of pain, nausea and vomiting in bowel obstruction and how these key symptoms should be measured – the majority of studies assess the severity of pain and nausea, and the severity and frequency (number of daily episodes) of vomiting. Assessing the absence of key symptoms is not sufficient in this population, 19 and placing nausea on a continuum with vomiting may not allow the assessment of the balance between these symptoms for individual patients. The details of this can, however, be difficult to tease out where patient-reported outcome measures fail to focus either on symptoms specific to bowel obstruction or symptoms appropriate to the end of life. The review also indicates that currently, a focus on physical symptom assessment overshadows the measurement of psychological, social and spiritual outcomes in inoperable malignant bowel obstruction.
There is a need for further consideration of which patient-reported outcomes measures might best suit this particular population. Quality of life assessment includes an individual’s perceptions in the context of their personal values and beliefs, and considers symptom improvement alongside physical deterioration, reflecting the core values of the WHO definition of palliative care. 107 This can only be meaningful when patients have good communication with health care professionals and reasonable expectations of treatment. Evidence of the improvement of quality of life is important to determining the utility of palliative treatment. 104 It can be limited in relation to inoperable malignant bowel obstruction because of difficulties in conducting meaningful assessment,27,42,49 for example because of short windows of time available for measurement. The use of visual analogue scales to assess wellbeing27,65 does not consider the challenges this may present to patients who may have difficulty with vision and the interpretation of emotions at the end of life. 108 Self-report is often feasible, however,109,110 and COMET suggest that the scope and nuance of quality of life measurement, in general terms, is often inadequate. 21
In the studies included in this review, evaluating the assessment of quality of life relies on global measures. In the light of our qualitative review, 19 this fails to capture some of the associations between quality of life and outcomes listed under other COMET categories that are evident from studies of patient experience. For example, the resumption of oral intake, when measured to evidence the mechanical resolution of obstruction, does not reflect the psychological effects on the patient. The inability to eat is often experienced as a deep loss on a social and emotional level, 19 and the degree and duration of its restoration are likely to have deep implications for quality of life. Further, issues to do with patient comfort are rarely discussed in any depth in the discussion sections of study reports. A minority of papers noted patients’ physical discomfort with nasogastric tubes, and studies of parenteral nutrition explored patient and caregiver concerns about starvation when treatment is withdrawn.
Strengths and limitations of the review
The search was necessarily broad to catch as wide a spectrum of outcomes as possible from palliative approaches to treatment. Searching by the condition (malignant bowel obstruction) was necessary as searching by intervention proved too indiscriminate, retrieving (e.g.) multiple papers focused on the treatment of non-malignant obstruction or evaluating procedural techniques. Studies use a variety of approaches in their titles – some specifying obstruction by cancer type, others by its location, many including benign and malignant obstruction in the same study. Our broad search strategy might have led to the omission of studies which met the eligibility requirements. It is possible that studies where concurrent chemotherapy occurred have been included in the review, if this has not been reported. A strength of the review is that papers reporting on decompression and pharmacological management reached a point of saturation where no new outcomes arose in multiple additional papers. This point was not reached in relation to parenteral nutrition, because of the low number of included studies in this area.
This review cannot demonstrate whether quality of life tools focused on palliative patients may be more suitable for assessing inoperable malignant bowel obstruction than tools focused more generally on symptoms of advanced cancer. For example, the Palliative care Outcome Scale (POS), 111 developed in 1999 as a successor to the Support Team Assessment Schedule (STAS) for use with patients with advanced disease and refined as the Integrated Palliative care Outcome Scale (IPOS) in 2019, 112 did not appear in the included papers, which date from 1990 to 2021. This may reflect the time lag between uptake of new measurement tools in practice and reports of their use in journal publications.
Conclusion
This review demonstrates that outcome measurement in the majority of studies of palliative interventions for inoperable malignant bowel obstruction currently focuses on survival and adverse events, and that routine assessment of patients’ quality of life is scarce. Definitions of treatment success centre around technical aspects of decompressive procedures and the reduction of symptoms by pharmacological interventions, but fail to include measures of wellbeing appropriate to patients at the end of life. A clear distinction needs to be made between studies evaluating the technical success of procedural interventions in resolving obstruction and studies evaluating patient-relevant outcomes related to symptoms and wellbeing. The majority of studies focus on the three key symptoms of pain, nausea and vomiting, assessing them in a variety of ways for their severity, frequency and/or duration; measures placing nausea and vomiting on a continuum may be inappropriate for the assessment of inoperable malignant bowel obstruction patients because they do not distinguish the balance between these two key symptoms. Three recommendations can be made from the results of the review. In assessing inoperable malignant bowel obstruction, we need increased patient relevance in definitions of treatment success to align with the aims of end of life care, a more consistent approach to the nuances of symptom assessment, and greater consideration of how to measure wellbeing in this patient population.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163221122352 – Supplemental material for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review
Supplemental material, sj-pdf-1-pmj-10.1177_02692163221122352 for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review by Alison Bravington, George Obita, Elin Baddeley, Miriam J Johnson, Fliss EM Murtagh, David C Currow, Elaine G Boland, Annmarie Nelson, Kathy Seddon, Alfred Oliver, Simon IR Noble and Jason W Boland in Palliative Medicine
Supplemental Material
sj-pdf-2-pmj-10.1177_02692163221122352 – Supplemental material for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review
Supplemental material, sj-pdf-2-pmj-10.1177_02692163221122352 for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review by Alison Bravington, George Obita, Elin Baddeley, Miriam J Johnson, Fliss EM Murtagh, David C Currow, Elaine G Boland, Annmarie Nelson, Kathy Seddon, Alfred Oliver, Simon IR Noble and Jason W Boland in Palliative Medicine
Supplemental Material
sj-pdf-3-pmj-10.1177_02692163221122352 – Supplemental material for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review
Supplemental material, sj-pdf-3-pmj-10.1177_02692163221122352 for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review by Alison Bravington, George Obita, Elin Baddeley, Miriam J Johnson, Fliss EM Murtagh, David C Currow, Elaine G Boland, Annmarie Nelson, Kathy Seddon, Alfred Oliver, Simon IR Noble and Jason W Boland in Palliative Medicine
Supplemental Material
sj-pdf-4-pmj-10.1177_02692163221122352 – Supplemental material for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review
Supplemental material, sj-pdf-4-pmj-10.1177_02692163221122352 for The range and suitability of outcome measures used in the assessment of palliative treatment for inoperable malignant bowel obstruction: A systematic review by Alison Bravington, George Obita, Elin Baddeley, Miriam J Johnson, Fliss EM Murtagh, David C Currow, Elaine G Boland, Annmarie Nelson, Kathy Seddon, Alfred Oliver, Simon IR Noble and Jason W Boland in Palliative Medicine
Footnotes
Acknowledgements
The research team would like to thank Sarah Greenley for a significant contribution to the development of the search strategy.
Author contributions
JB, SN, MJ, FM, DC, GO, EGB, AN and KS were involved in the conception of the study. All authors contributed to protocol development; JB, SN, MJ, FM and DC made significant contributions to the direction of the review. AB and GO led on data collection and analyses, and AB led on writing, with oversight of data collection, analysis and writing by JB. All authors read and approved the final manuscript.
Data management and sharing
Data analysis files are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Marie Curie Research Grants Scheme, grant number MCRGS-20171220-8020. Fliss EM Murtagh is a National Institute for Health Research (NIHR) Senior Investigator. The views expressed in this article are those of the author(s) and not necessarily those of the NIHR, or the Department of Health and Social Care.
Research ethics and patient consent
The review did not directly involve human participants and did not require ethical approvals. The overarching core outcome set study of which this is a part was reviewed by the Wales Research Ethics Committee 5 (Wales REC) on 10th December 2019 (Ref 19/WA/0340).
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
