Abstract
Background:
The circumstances and care provided at the end of a child’s life have a profound impact on family members. Although assessing experiences and outcomes during this time is challenging, healthcare professionals have a responsibility to ensure high quality of care is provided.
Aim:
To identify available tools which measure the quality of dying, death and end-of-life care for children and young people; describe the content, and data on validity and reliability of existing tools.
Design:
Scoping review was conducted following the Arksey and O’Malley methodological framework.
Data sources:
Four electronic databases (MEDLINE, EMBASE, CINAHL and PsycINFO) and grey literature were searched for studies published in English (January 2000–June 2021). A review of reference lists and citation searching was also undertaken. Tools needed to include a focus on the ‘dying’ phase of illness (defined as the last month of life).
Results:
From 2078 articles, a total of 18 studies, reporting on 11 tools were identified. All tools were completed by primary caregivers or healthcare professionals as ‘proxy’ assessments; all except one was undertaken after death. Question items about quality of life and preparation for death were found in all tools; items relating to cultural aspects of care, grief and financial costs were less common. Only 6/11 had undergone psychometric testing within a paediatric palliative care setting.
Conclusions:
Future research should include ways to adapt, refine and improve existing tools. Assessing their wider application in different clinical and cultural settings and conducting further psychometric assessment represent areas of focus.
The circumstances and care received at the end of a child’s life can have a profound effect on parents and siblings.
Measuring experiences and outcomes during this time is challenging but extremely important to ensure high quality of care is provided.
This is the first scoping review to systematically identify tools assessing the quality of dying, death and end-of-life care for children and young people.
Gaps were identified in the assessment of salient domains relating to cultural aspects of care, economic costs and grief.
Only 6 of the 11 tools had conducted specific psychometric testing within a paediatric palliative care setting.
Rather than developing new tools, future focus should include ways to adapt, refine and improve existing ones.
Further work is needed to determine whether the existing tools are suitable for use in a wider cultural context.
The direct views of the dying child and those of the sibling are not captured by existing measures.
Introduction
Despite marked improvements in health services, medical treatments and public health, over 4600 babies, children and young people aged 0–19 years die each year in high-income countries, such as the United Kingdom (UK). 1 Globally, the Lancet Commission highlighted that 2.5 million children die each year with ‘serious health related suffering’, with the majority of deaths occuring in low and middle income countries. 2 Therefore, a large number of parents and other family members worldwide suffer the consequences of a child bereavement. The effects of the death of a child on parental health and wellbeing are well known.3–6 However, the circumstances and care received at the end of a child’s life can have a profound effect on parents and siblings in terms of their subsequent relationships, roles, friendships, and ability to carry on with their lives. 7 The key elements of a ‘good death’ 8 from the perspective of a dying child, the child’s family and the healthcare providers, include: preserving quality of life; preparation for death; specific aspects of care such as continuity, addressing cultural and spiritual concerns; and considering the impact on survivors. 9
The period of care up to and during the end of a child’s life is extremely important and healthcare professionals have a responsibility to ensure high quality care, including dignity, respect and symptom control, is provided during this time. Defining high quality care at the very end of life is greatly dependent on the preferences and priorities of the patient and their family and their views are central to any efforts to measure quality. Measuring care, outcomes and experiences during end-of-life is challenging but patient reported outcome measures (PROMs) can be used. 10 Although the patient’s perspective on the quality of end-of-life care should be sought whenever possible, this is not easy, especially with children. Children receiving palliative care may be non-verbal, too young or too unwell to complete self-report tools. 11 Debate also exists about who is best placed to complete outcome measures for children and young people. 11 Potential ‘proxy’ assessments can be undertaken by a parent, carer, or professional, but their degree of agreement with child self-report measures is variable. For example, child and parent scores tend to be better correlated for more observable, physical aspects of care and poorer for issues such as emotional problems. 12
When evaluating outcomes, it is often the case that a range of PROMs are available that could be used for a given purpose (i.e. to assess quality of end-of-life care and death). Reviews and evaluation work are therefore necessary for researchers and clinicians to help map what tools are available and their supporting psychometric evidence. In adults, a number of systematic reviews have identified, appraised and assessed tools used with ‘proxies’ that is bereaved family carers after the death to measure quality of end-of-life care.13–16 None have specifically focussed on tools used to assess quality of dying, death and care at the very end of life for children and young people.
Scoping reviews represent a way of mapping broad areas; they provide breadth, as compared to depth, and help identify any research gaps in the literature.
17
Within this scoping review, we aimed to address the following research question:
What existing tools are available to measure the quality of dying, death, and end-of-life care for children and young people?
An additional sub-question was:
What can we determine about the quality of these tools e.g., comprehensiveness of content, assessment for validity and reliability (as demonstrated by their development process and reported psychometric testing)?
Methods
Design
The scoping review was conducted in five stages following the Arksey and O’Malley 17 framework: identifying the research question; identifying relevant studies; study selection; charting the data; and collating, summarizing and reporting the results. Additionally, we incorporated enhancements to this original framework using the Joanne Briggs Institute guidance (https://jbi.global/scoping-review-network/resources). Reporting was informed by the PRISMA extension for Scoping Reviews (PRISMA-Scr). 18
Search strategy
Working in collaboration with a subject librarian (MC), an initial limited search of EMBASE was undertaken to identify relevant target papers. Text words within titles and abstracts and the index terms of these articles were used to generate a full search strategy. The search strategy consisted of four main concepts: ‘quality of death’, ‘tool’, ‘palliative care’ and ‘children and young people’ (Textbox 1). We defined ‘children and young people’ as those less than 25 years of age, to include adolescents as well as younger children. 19 We did not include studies which focussed solely on neonatal deaths (within first 27 days of life) 1 as these tend to relate to perinatal factors, 1 infections and premature birth. 20 For the purposes of this review, the ‘dying period of their illness’ was regarded as the last month of life, reflecting that advanced, incurable illnesses have different disease trajectories. Where a specific time period was not stated, tools which had specific questions about the quality of dying or death were also included.
The National Institute for Health and Care Excellence (NICE) guidance for end-of-life care for children 21 was used to inform the chosen search terms under each search concept. Modifications were made, for example, to ensure the search strategy focussed on the ‘dying period’ rather than the broader remit of palliative care. An electronic literature search was conducted on 15th June 2021 with four electronic databases (MEDLINE, CINAHL, EMBASE and PsycINFO) covering the years from January 2000 to June 2021 (Supplemental file 1). This time period reflects more recent changes within paediatric palliative care (e.g. formation of the Association of Paediatric Palliative Medicine within the UK (https://www.appm.org.uk/)). Specified inclusion and exclusion criteria (Textbox 1) were used to identify studies.
Inclusion and exclusion criteria.

Titles and abstracts were initially screened by teams of two independent reviewers. A full text review of all potentially eligible studies was conducted independently the same teams; any areas of uncertainty were resolved by discussion with the lead author. Review articles were not included, but reference lists were screened to identify any additional papers. A citation search of all selected articles was completed, and reference lists of all included papers were screened for potentially relevant studies. Grey literature was also searched using the search terms
Data extraction
Data was extracted using a specially designed proforma (piloted prior to use) by one member of the research team and verified by the lead author. Data was mapped out, using selected principles developed by the Scientific Advisory Committee of the Medical Outcomes Trust to assess quality-of-life instruments, 22 namely: conceptual model and reported psychometric testing (validity and reliability). The content of each tool was mapped to the seven key dimensions of a ‘good death’ (from research which incorporated the perspectives of the dying child, the child’s family and healthcare providers). 9 These considerations were supplemented by information on the study objective, tool purpose and description, assessment period, setting, population, participants and key study findings.
Collating and summarizing data
Charted data were then tabulated into the following categories, reflecting the predominant use of the tools:
Healthcare professional: tool used solely with healthcare professionals
Cancer: tool used solely within a cancer population
Cancer and non-cancer: tool used in populations with more than one disease groups (both malignant and non-malignant illnesses)
Life-limiting cardiac disease: tool used solely within an advanced cardiac disease population.
Comparisons were made between the extracted results focussing on development and use, content, participants and psychometric testing. This method highlighted dominant areas and allowed gaps to be identified. In keeping with the accepted remit of scoping review guidance, specific quality appraisal (e.g. of the methodology or psychometric properties), was not conducted. Rather, where documented within the manuscripts, these details were directly extracted. Where specific details were missing about tool content, the corresponding author of the relevant study was contacted and invited to provide further information.
Results
Range of studies
The initial search identified 2078 articles across all databases. Removal of duplicates resulted in 1663 papers, 65 of which were retrieved for full text review. A further 49 papers were excluded on reviewing full papers (Figure 1). Two additional articles were identified through reference lists and citation searches. A total of 18 papers23–40 were included in the review, reporting on 11 tools. One study reported on the use of two different tools, 27 whereas all other studies used a single tool.
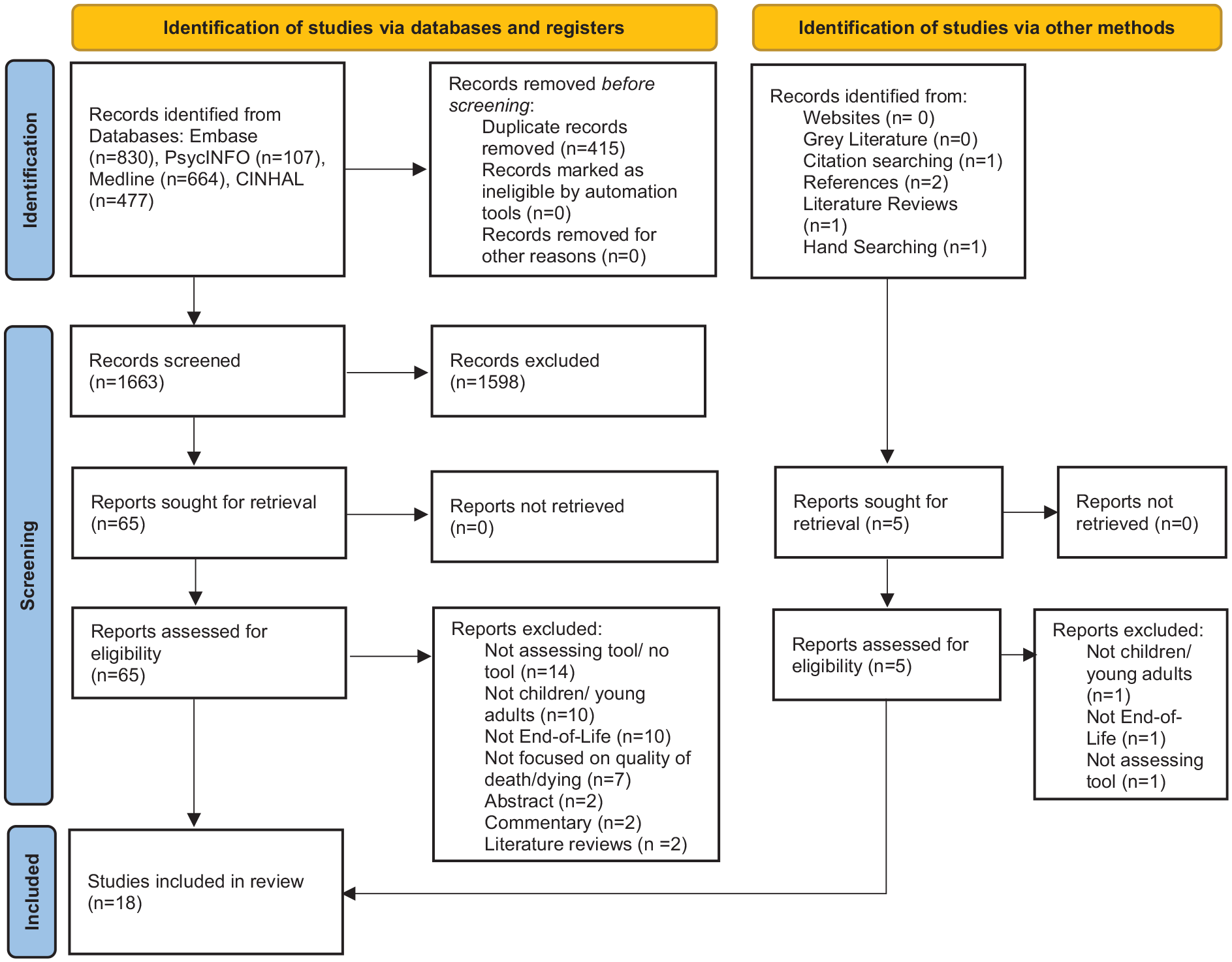
PRISMA flow diagram for the scoping review process.
The 18 studies were conducted in seven countries: USA (
Range of tools
The 11 tools were sub-categorized into the defined groups: sole use by healthcare professional tools (
Studies detailing the development, validation and initial use of healthcare professional tools assessing quality of dying, death and end-of-life care for children/young adults.
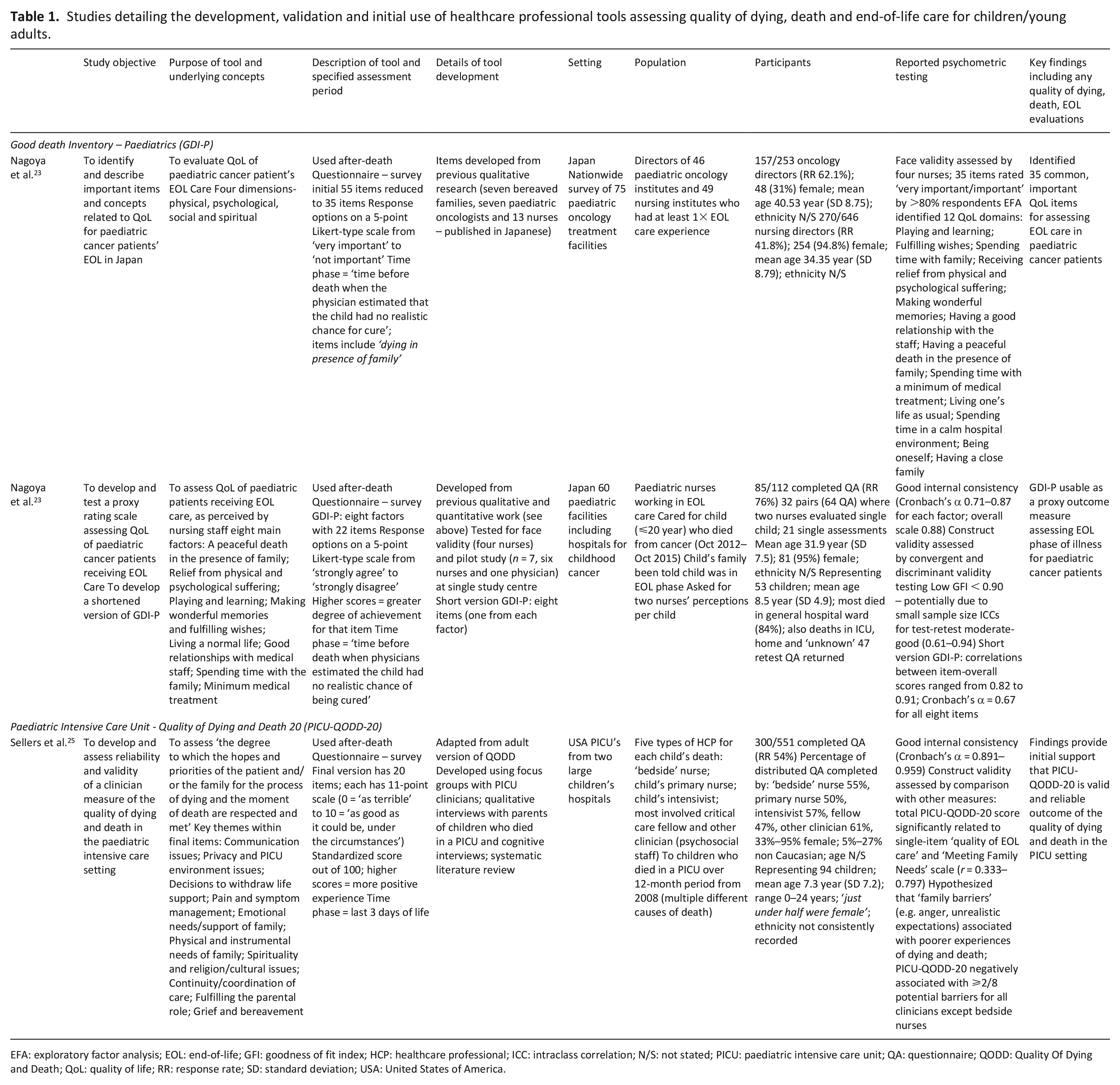
EFA: exploratory factor analysis; EOL: end-of-life; GFI: goodness of fit index; HCP: healthcare professional; ICC: intraclass correlation; N/S: not stated; PICU: paediatric intensive care unit; QA: questionnaire; QODD: Quality Of Dying and Death; QoL: quality of life; RR: response rate; SD: standard deviation; USA: United States of America.
Studies detailing the development, validation and initial use of tools assessing quality of dying, death and end-of-life care for children/young adults within a cancer population.
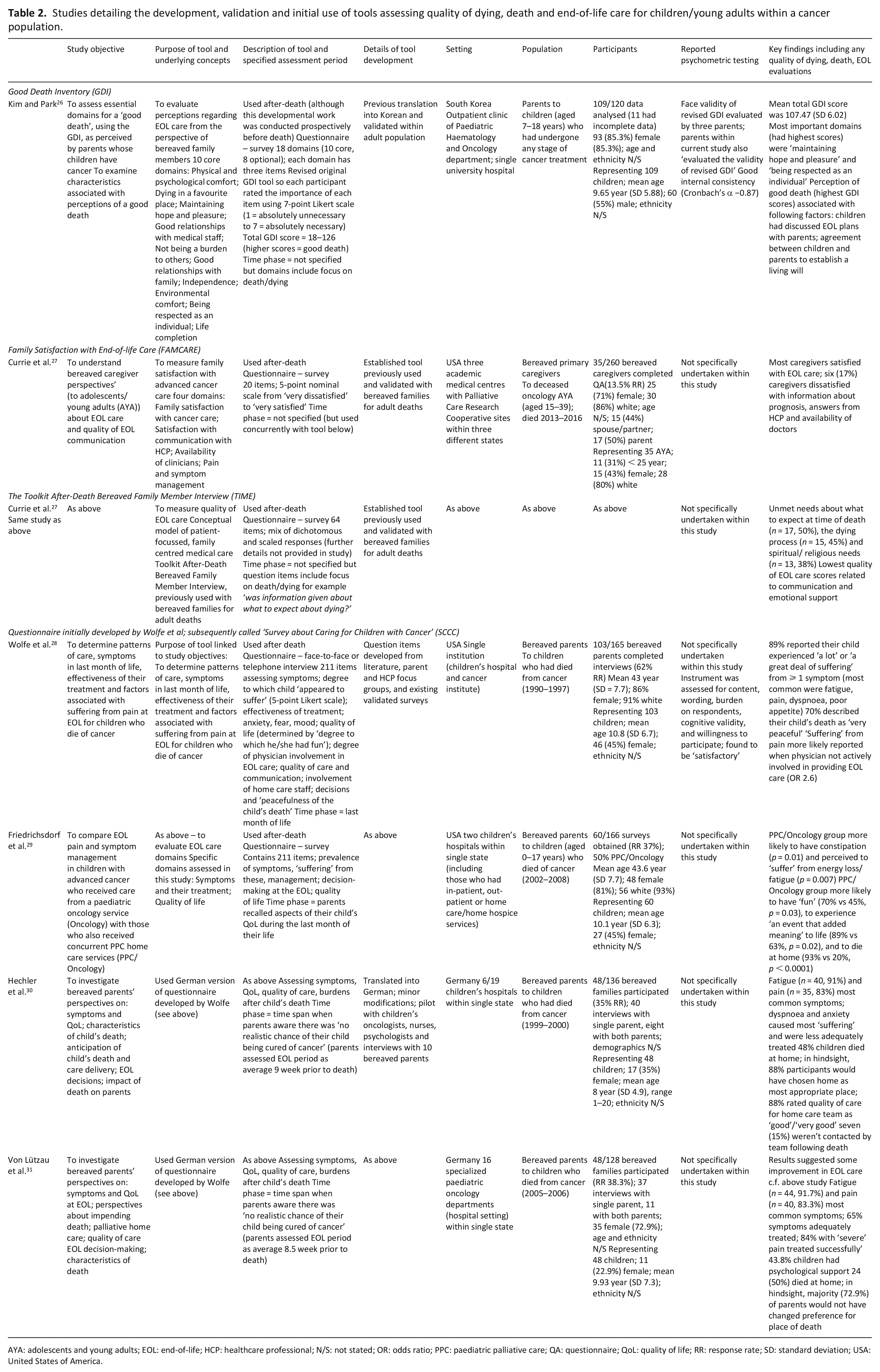
AYA: adolescents and young adults; EOL: end-of-life; HCP: healthcare professional; N/S: not stated; OR: odds ratio; PPC: paediatric palliative care; QA: questionnaire; QoL: quality of life; RR: response rate; SD: standard deviation; USA: United States of America.
Studies detailing the development, validation and use of tools assessing quality of dying, death, and end-of-life care for children/young adults within a mixed cancer and non-cancer population.
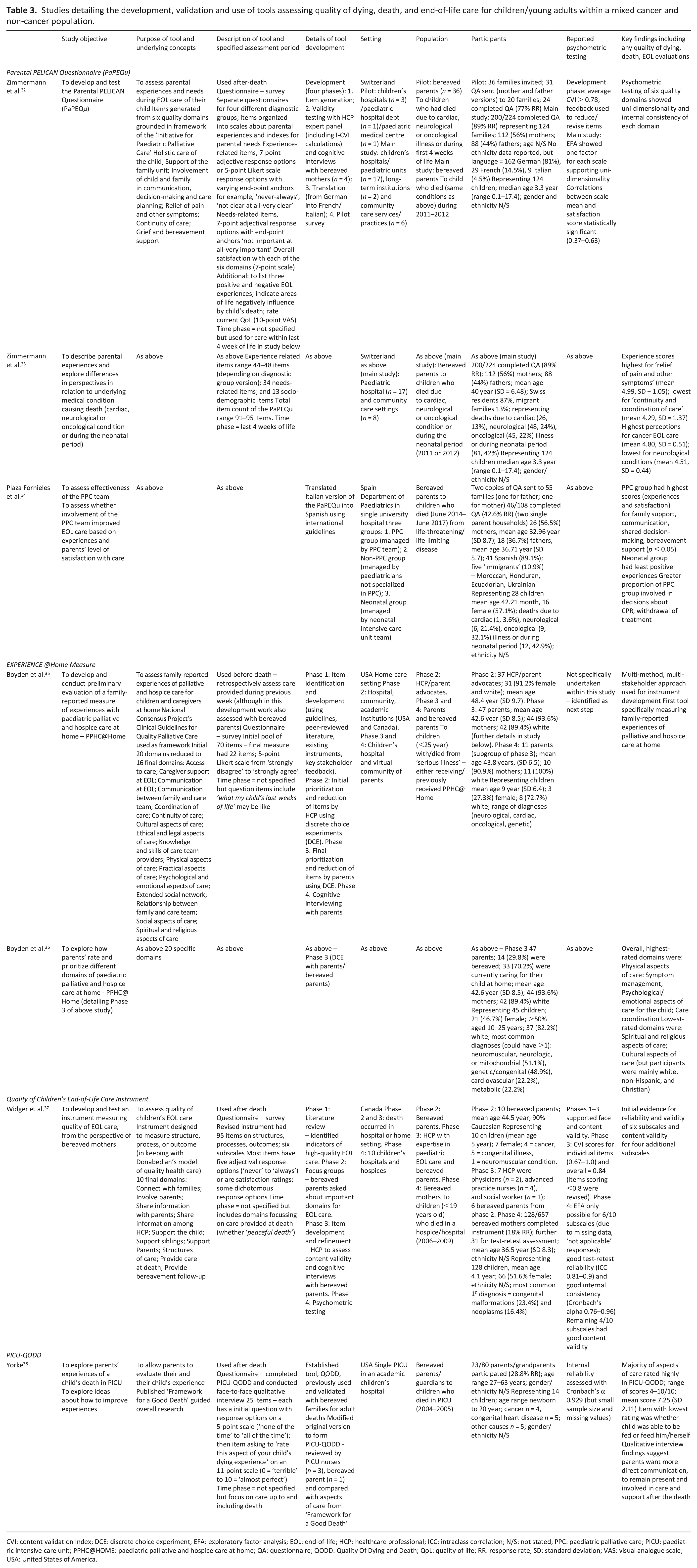
CVI: content validation index; DCE: discrete choice experiment; EFA: exploratory factor analysis; EOL: end-of-life; HCP: healthcare professional; ICC: intraclass correlation; N/S: not stated; PPC: paediatric palliative care; PICU: paediatric intensive care unit; PPHC@HOME: paediatric palliative and hospice care at home; QA: questionnaire; QODD: Quality Of Dying and Death; QoL: quality of life; RR: response rate; SD: standard deviation; VAS: visual analogue scale; USA: United States of America.
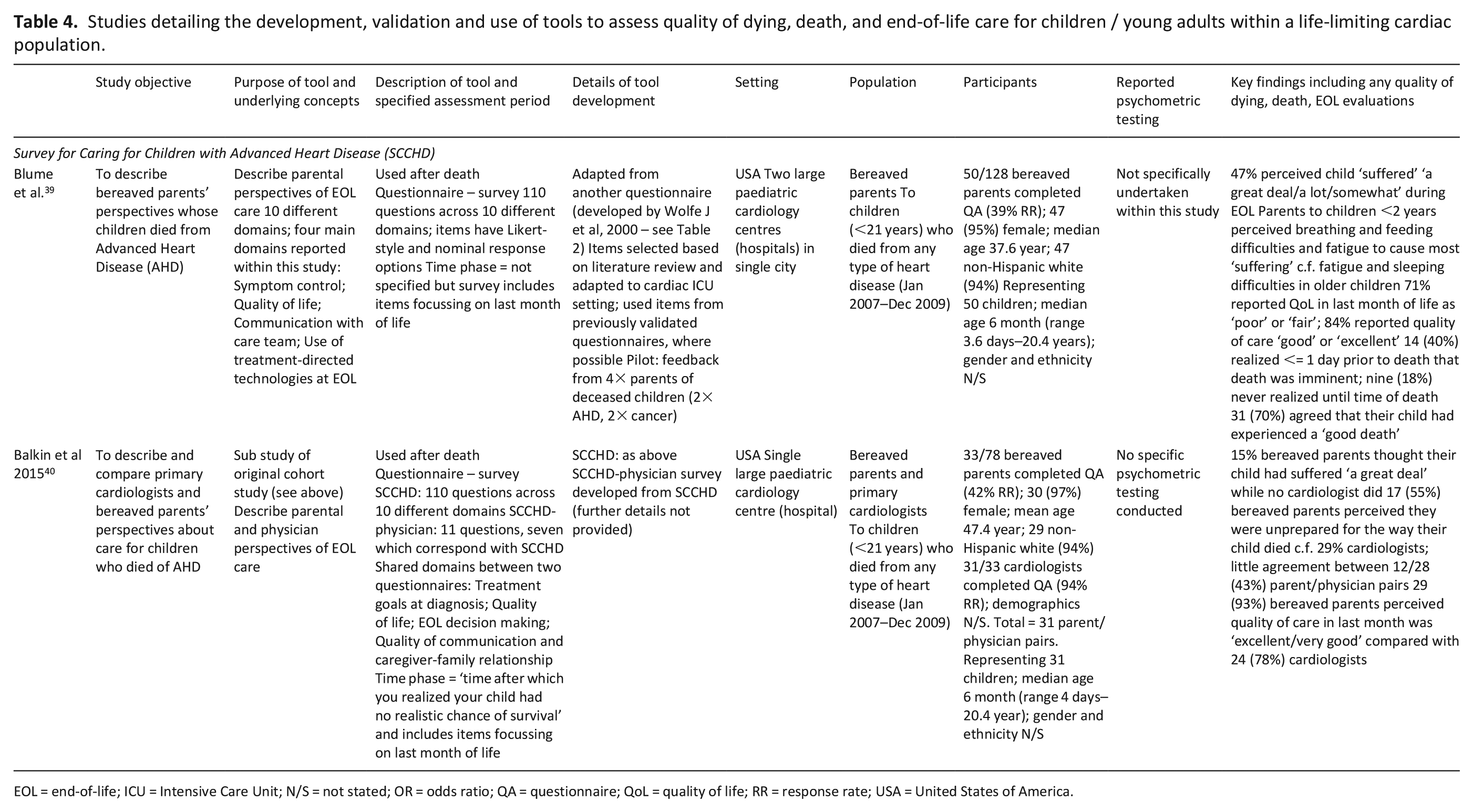
Studies detailing the development, validation and use of tools to assess quality of dying, death, and end-of-life care for children / young adults within a life-limiting cardiac population.
EOL = end-of-life; ICU = Intensive Care Unit; N/S = not stated; OR = odds ratio; QA = questionnaire; QoL = quality of life; RR = response rate; USA = United States of America.
Content of the tools mapped to the ‘good death of a child’ dimensions. 8

Y: yes; N: No; N/S: not stated (detail not provided within study); DNACPR=do not attempt cardio-pulmonary resuscitations; EOL: end-of-life; HCP: healthcare professional; PICU: paediatric intensive care unit.
Only communication items were reported within study; further information about FAMCARE items obtained from http://www.npcrc.org/files/news/famcare_scale.pdf.
Full details of question items used not provided within study and did not receive response from corresponding author.
With the exception of one tool,
35
all the other tools were developed for use after death. The time period in which the child’s death had occurred ranged from within a previous 12-month period
25
up to the previous 7 years.
28
No tool had been developed or used directly with patients (child or young person) during the dying phase of their illness nor specifically with siblings. The definitions of the specified assessment period varied and could include the last 3 days (
All the tools assessed aspects of quality of life (e.g. pain and symptom control) and preparation for death (e.g. communication, decision-making). Items relating to ‘legacy’ (e.g. establishing meaning, importance of ritual/funeral), were assessed within five tools (Table 5). Question items less frequently asked about cultural aspects of care (
Tools used predominately with healthcare professionals
The two tools used with healthcare professionals were the Good Death Inventory – Paediatrics (GDI-P)23,24 and the Paediatric Intensive Care Unit – Quality of Dying and Death 20 (PICU-QODD) 25 (Table 1).
Quality of tool
Both tools underwent a robust process of development and have been tested for validity and reliability.23,24 PICU-QODD-20 has question items mapping across all seven dimensions of a ‘good death’. 25
Clinical implications
Whereas the GDI-P purpose is focussed on nursing perspectives of paediatric cancer deaths across several care settings (including hospital, PICU and home), 23 the PICU-QODD-20 seeks to obtain a variety of healthcare professional perspectives about deaths due to different illnesses but only for those occurring in PICU. 25
Tool used solely within a cancer population
The four tools used solely within a cancer population were: Good Death Inventory (GDI), 26 Family Satisfaction with the End-of-Life Care (FAMCARE), 27 the Toolkit After-Death Bereaved Family Member Interview (subsequently referred to as the ‘Toolkit’) 27 and a questionnaire, initially developed by Wolfe et al., 28 which was later called Survey about Caring for Children with Cancer (SCCC)29–31 (Table 2).
Quality of tool
The SCCC is the most extensive tool (211 items)28–31 with question items spanning across many different aspects of cancer care as well those relating to care at the very end of life. It has undergone a careful process of question item development and selection. FAMCARE and the ‘Toolkit’ 27 are established, validated tools previously used with bereaved families for adult deaths. Only the GDI, 26 however, has undergone initial psychometric testing of validity and reliability specifically within a palliative paediatric population. None of the tools incorporated all aspects of multi-dimensionality in terms of a ‘good death’.
Clinical findings
Findings from the study using the GDI indicated that aspects of advance care planning (e.g. establishing a ‘living will’) were associated with more positive parental perspectives about a ‘good death’. 26 Both FAMCARE and the ‘Toolkit’ were used within the same study, assessing the quality of end-of-life care for adolescents and young people (aged 15–39 years) from the caregiver perspective. 27 The study showed most caregivers were satisfied with care, but there were unmet information and religious/spiritual care needs. 27 SCCC has been used within four studies conducted in two different countries. Within the first study, 92 (89%) bereaved parents reported their child experienced ‘a lot’ or ‘a great deal’ of suffering’, although 70% said the actual death was ‘very peaceful’. 28 A further study found that those receiving home care services were more likely to die at home. 29 An additional two studies, conducted within a single state in Germany, enabled a comparison of quality of end-of-life care over two time periods.30,31 Although symptom reporting was similar, preferences about place of death were more concurrent with actual place of death in the second study. 31
Tool used with both cancer and non-cancer populations
The four tools used within both cancer and non-cancer populations were: the PELICAN questionnaire (PaPEQu) 32,33; the Experience @HOME Measure 35,36; the Quality of Children’s End-of-life Care Instrument 37 and the PICU-QODD 38 (Table 3).
Quality of tool
The first three tools have all undergone a robust process of development 32,35,37; the PICU-QODD was modified from an existing, validated tool used with bereaved families for adult deaths. 38 All tools except the Experience @HOME Measure have reported on their psychometric properties with the PaPEQu being the most extensively reported. 32 Only the PICU-QODD covers all seven dimensions of a ‘good death’. 38
Clinical findings
The Experience @HOME Measure focuses purely on the home care setting. It is the only tool intended to be used before death and retrospectively assesses care provided in the previous week.35,36 The Quality of Children’s End-of-life Care Instrument focuses on the bereaved mothers’ perspective of the quality of end-of-life care. 37 Both the PaPEQU and the PICU-QODD have been used within clinical studies. PaPEQU has been used to assess quality of end-of-life care for children who died from a variety of illnesses (cardiac, neurological or oncological illness or during the first 4 weeks of life). 32 Studies show that bereaved parents’ perceptions about overall care were highest for children dying with cancer, those who had engaged with Paediatric Palliative Care teams, and lowest for children dying with neurological conditions or in the neonatal period.32,33 The PICU-QODD was used alongside a qualitative interview and explored both bereaved parents and grandparents’ views about end-of-life care. The majority of aspects of care within the PICU-QODD were rated highly, whereas the qualitative findings highlighted the need for more direct communication with healthcare professionals. 38
Tool used solely within a life-limiting cardiac population
The one tool used within a life-limiting cardiac population is the Survey for Caring for Children with Advanced Heart Disease (SCCHD)39,40 (Table 4).
Quality of tool
This was developed from the Wolfe et al. 28 questionnaire, 39 although no psychometric testing has been reported.
Clinical findings
A subsequent study used the SCCHD to assess both bereaved parents and cardiologist views reflecting different perspectives about the degree of preparation for death and overall quality of care. 40
Discussion
Main findings
This scoping review identified 11 tools, developed and used across seven countries, which assess the quality of dying, death and end-of-life care for children and young people. The majority of tools have been used after the child’s death with bereaved parents, predominantly mothers, in a hospital setting. In terms of content, all tools asked about quality of life and preparation for death whereas aspects relating to cultural concerns, financial costs, grief and bereavement were more variable. The PICU-QODD-20 and PICU-QODD had the most comprehensive content across the dimensions of a ‘good death’.
Only six tools have undergone some degree of psychometric testing for validity and reliability specifically within a paediatric palliative care population. Those which have reported the most extensive testing for validity and reliability are GDI-P, PICU-QODD-20 and PaPEQu, whereas initial findings were more limited for the GDI, the Quality of Children’s End-of-Life Care Instrument and PICU-QODD. Although the SCCC has not undergone formal psychometric validation, it represents an extensive ‘question bank’ which has been developed and used across two different countries to assess quality of end-of-life care. No tool has addressed the challenges of assessing the views of children or young people themselves or specifically been used to assess the perspective of siblings.
What this study adds
Whilst previous systematic reviews, have focussed on health-related quality of life outcome measures, 11 none have been directed towards identifying tools used to assess quality of care provided at the end of a child’s life. This scoping review allows comparison of tools and helps identify gaps for which future research is needed.
Establishing whether the identified tools are suitable for use in a wider cultural context is required. Existing studies have predominately been undertaken within the USA, which has a specific type of healthcare system, reliant on health coverage and economic resources. No tools have been developed or revised to be used within the UK, Ireland, Canada nor Australia, which are all regarded as having a high level of palliative care integration into mainstream children’s healthcare services. 41 The majority of studies were conducted, at least in part, within hospital settings. This may reflect specific cultures such as that within the UK, where most children and young people’s deaths occur in hospital. 42 International partnerships have previously been recommended to enhance learning and inform tool validation. 43 Hence, there is a need to establish whether existing tools are relevant and meaningful across much more diverse countries and cultures. This is especially pertinent when terms such as ‘grief’ and ‘distress’ can be specific to the English language.44,45
Rather than developing new tools, future focus should be on further improving and validating existing tools. It is also important to consider whether the identified tools have utility within different clinical settings. For example, the content of PICU-QODD-20 covered all seven dimensions of a ‘good death’ and has been assessed for some aspects of validity and reliability. 25 The remit of the tool, however, is within a very specific intensive care environment. It would be important to establish whether this tool could be adapted and have wider application. The SCCHD offered comparative views about care from both the bereaved parents and the cardiologists’ perspective. 40 As there are two different versions of the GDI and the PICU-QODD (one for healthcare professionals; one for bereaved parents),24–26,38 these tools also offer that possibility. Establishing whether tools such as these could be adapted to incorporate the views of siblings would also be of value. The Experience @HOME Measure is the only tool used before death. 35 Hence, exploring the possibility of the dying child’s ability to participate in completion would be a further area of exploration.
Only one study combined the use of a tool with an individual qualitative interview. 38 The opportunity for bereaved relatives to be able to ‘tell their story’, to share narrative accounts, is recognized to have potential therapeutic benefit. 46 Hence, it would seem important for existing tools to include free-text space to enable opportunities for sharing experiences not captured within the specific question domains. Additionally, it has been recognized that there is strength in combining both qualitative and quantitative approaches for paediatric palliative care research 47 – evaluation of quality of dying, death and end-of-life care would be an area where both rigorously developed outcomes and qualitative approaches would enrich the detail of reported experiences.
Strengths and limitations of the study
The search strategy conducted followed a robust, systematic methodology and included grey literature, reverse citation searching and screening of reference lists. We were not able to contact every individual author to enquire about additional work/unpublished studies, hence some relevant studies may have been overlooked. Additionally, our main focus was on the identification and development of available tools so subsequent studies focussing only on their use, may have been omitted. In keeping with the aims of a scoping review, we did not undertake a formal assessment of study quality nor psychometric properties. As the reporting of these details within each study was not always consistent, there may be some ambiguity when directly comparing different tools. Additionally, we did not consider all the principles which can be used to assess quality-of-life instruments for example respondent and administrative burden. The choice of our dimensions for a ‘good death’ came from a study which, although involved multiple stakeholders, was focussed on children dying from cancer. 9 Experiences about what constitutes a ‘good death’, however, is complex and multi-faceted, potentially varying for different types of life-limiting illnesses.48,49
Conclusion
This review has identified 11 available tools for assessing quality of dying, death and end-of-life care in paediatrics, yet there is variability in terms of instrument content and evidenced quality (i.e. degree of assessment of validity and reliability). Improvement of existing tools should involve the inclusion of additional items representing salient domains of a ‘good death’ and further psychometric testing to ensure more valid, reliable and comprehensive assessment. International partnerships are key to determining suitability for wider use, informing tool validation and application across different countries and cultures. Despite the recognized challenges, sensitive and timely ways to identify data about the last weeks of life, can help facilitate learning about experiences, leading to further improvements in quality of care both before and after the death.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163221105599 – Supplemental material for Measuring quality of dying, death and end-of-life care for children and young people: A scoping review of available tools
Supplemental material, sj-docx-1-pmj-10.1177_02692163221105599 for Measuring quality of dying, death and end-of-life care for children and young people: A scoping review of available tools by Catriona R Mayland, Katy A Sunderland, Matthew Cooper, Paul Taylor, Philip A Powell, Lucy Zeigler, Vicki Cox, Constance Gilman, Nicola Turner, Kate Flemming and Lorna K Fraser in Palliative Medicine
Footnotes
Acknowledgements
We wish to acknowledge the wider membership of the White Rose collaboration for their input and discussions into this work.
Author contributions
CRM conceived the initial idea and designed the study along with MC, PT, PAP, LZ, KF and LKF. MC undertook the searches. CRM, KAS, PT, PAP, LZ, VC, CG and NT conducted initial screening and full manuscript reviews. CRM and KAS analysed and interpreted the data. CRM and KAS drafted the initial manuscript. All authors have reviewed the article critically for clarity and intellectual content, provided revisions and have approved this version for submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding for this study was provided by the White Rose collaborative. This funder did not have a role in the study design, collection, analysis or interpretation of the data nor in the writing of the manuscript. Dr Catriona Mayland is funded by Yorkshire Cancer Research.
Research ethics and consent
As this study represents a scoping literature review, not formal ethics approval was required.
ORCID iDs
Data availability statement
Further information about the search strategies are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
