Abstract
Background:
Anaemia is a common sequela of advanced disease and is associated with significant symptom burden. No specific guidance exists for the investigation and management of anaemia in palliative care patients.
Aim:
We aim to offer a pragmatic overview of the approaches to investigate and manage anaemia in advanced disease, based on guidelines and evidence in disease specific patient groups, including cancer, heart failure and chronic kidney disease.
Design:
Scoping review methodology was used to determine the strength of evidence supporting the investigation and management of anaemia in patients with advanced disease.
Data sources:
A search for guidelines was performed in 2020. National or international guidelines were examined if they described the investigation or management of anaemia in adult patients with health conditions seen by palliative care services written within the last 5 years in the English language. Searches of MEDLINE, the Cochrane library and WHO guidance were made in 2019 to identify key publications that provided additional primary data.
Results:
Evidence supports patient-centred investigation of anaemia, results of which should guide targeted intervention. Blanket use of blood transfusion should be avoided, with evidence supporting a more restrictive approach to transfusion. Routine use of oral iron and erythropoetin stimulating agents (ESAs) are not recommended. Insufficient evidence exists to determine the effectiveness of IV iron in this patient group.
Conclusion:
We advocate early consideration and investigation of anaemia, guided by symptom burden and patient preferences. Correction of reversible causes should be the mainstay of treatment, with a restrictive approach to blood transfusion. Research is required to evaluate the efficacy of IV iron in these patients.
The majority of palliative patients are anaemic, with a variety of underlying causes.
No guideline exists for the overarching investigation and management of anaemia in palliative care patients, although certain disease states have their own tailored guidelines.
Differences in the approach to investigation and management is common in practice.
We make recommendations for practice which include investigating the cause of anaemia, consideration of targeted treatments, discussion of risks and benefits of treatments with patients and reducing blood loss.
Anaemia should not be considered a diagnosis in itself, and should be acknowledged, investigated and management tailored in patients with advanced disease.
The blanket use of red blood cell transfusion should be avoided, with evidence supporting a more restrictive approach to transfusion.
Oral iron and ESAs should not be routinely used in practice.
Further research is required to determine the efficacy of intravenous iron in palliative care patients.
Introduction
Anaemia is a common sequela of advanced disease and can cause symptoms including fatigue and breathlessness.
A study of palliative care patients found that 77% of men and 68.2% of women were anaemic. 1 In terms of severity and cause of anaemia a cohort of 1797 patients with advanced cancer referred to palliative care services showed 38% had moderate to severe anaemia (defined as haemoglobin between 80 and 110 g/dL and below 80 g/dL, respectively). Functional iron deficiency, a component of anaemia of chronic disease was present in 39%–43% of all patients. 2 Anaemia is also common in non-malignant disease. Prevalence in chronic obstructive pulmonary disease can be as high as 33%, with anaemia of chronic disease likely the most common cause.3,4
Despite its prevalence in palliative care patients, no specific guidelines for the management of anaemia in this patient group exist. Some treatments for anaemia, such as red cell transfusion, carry their own risks,5–7 particularly in frail patients with advanced disease. A UK audit of red cell transfusions in hospices found that haematinics (serum B12, folate and iron levels) were not checked in 70% of anaemic patients, highlighting a lack of investigation in this setting. 8 Furthermore, treatments such as B12, folate and iron were rarely used. 8 Although evidence in a specific palliative cohort is lacking, the management of anaemia in actively treated oncology patients demonstrated improvements in symptoms and improved quality of life. 1
Detailed guidance for the management of anaemia exists for several disease specific patient groups, which overlap with palliative care. We aim to offer palliative care professionals practical guidance on the management of anaemia in advanced disease, based on guidelines and evidence in disease specific patient groups, such as cancer, heart failure and chronic kidney disease.
Methods
A scoping methodology was employed for this practice review. Scoping reviews can report on the types of evidence that address and inform practice in the field and the way the research has been conducted. 9 As it had already been established that there no specific guidelines for palliative care the authors agreed by consensus a list of conditions which would include patients under the care of palliative care services. This included:
Cancer
Chronic kidney disease (CKD)
Heart failure
Guidelines were identified by searching databases and grey literature (Table 1). We included guidelines written within the last 5 years in the English language (Table 2). WHO guidance, the trial registry and Cochrane database were searched using the term ‘anaemia’ (May 2019). An additional search of MEDLINE was made for further key publications that provided additional primary data to add to the evidence base for the interventions considered (search date: 31 May 2019 – see Appendix 1 for search strategy). Studies relating to patients with cancer who were receiving chemotherapy were excluded.
Medline search for guidelines 20/1/21.

Guidelines examined: Shading indicated whether the intervention is included.
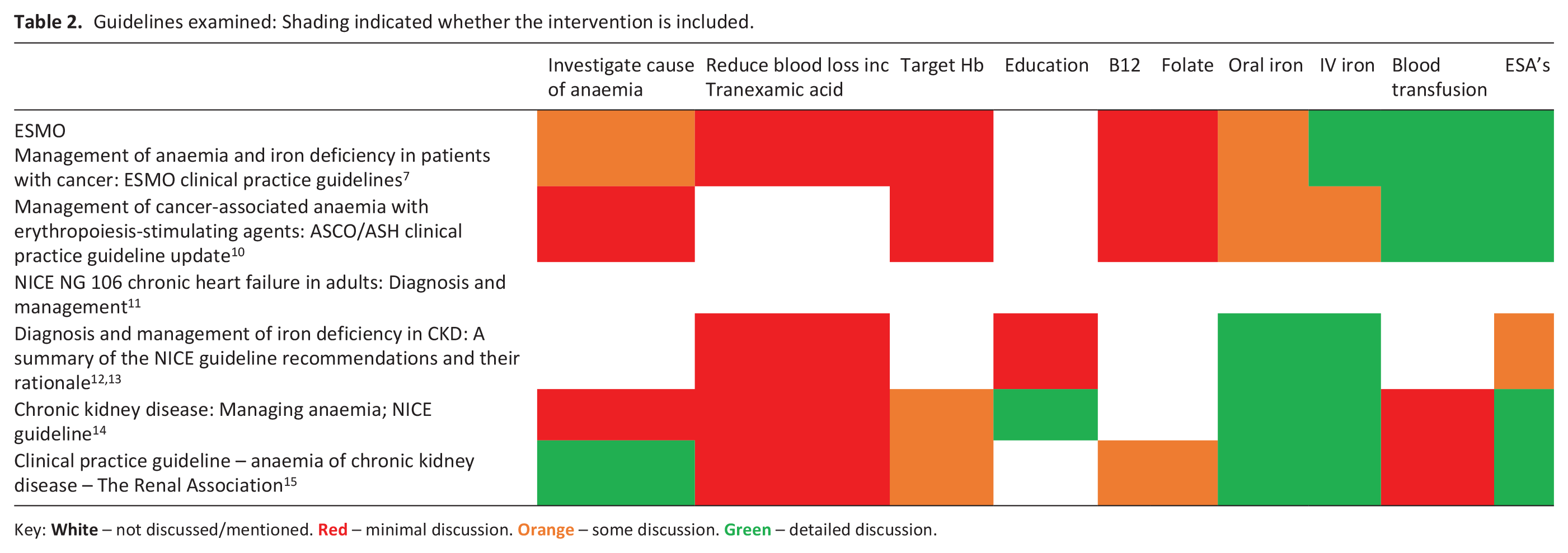
Key:  – not discussed/mentioned.
– not discussed/mentioned.  – minimal discussion.
– minimal discussion.  – some discussion.
– some discussion.  – detailed discussion.
– detailed discussion.
In addition, evidence for specific treatments used to treat anaemia in palliative care has been included. Red cell transfusions have been researched in palliative care patients, but evidence is more limited for other treatments in this cohort.
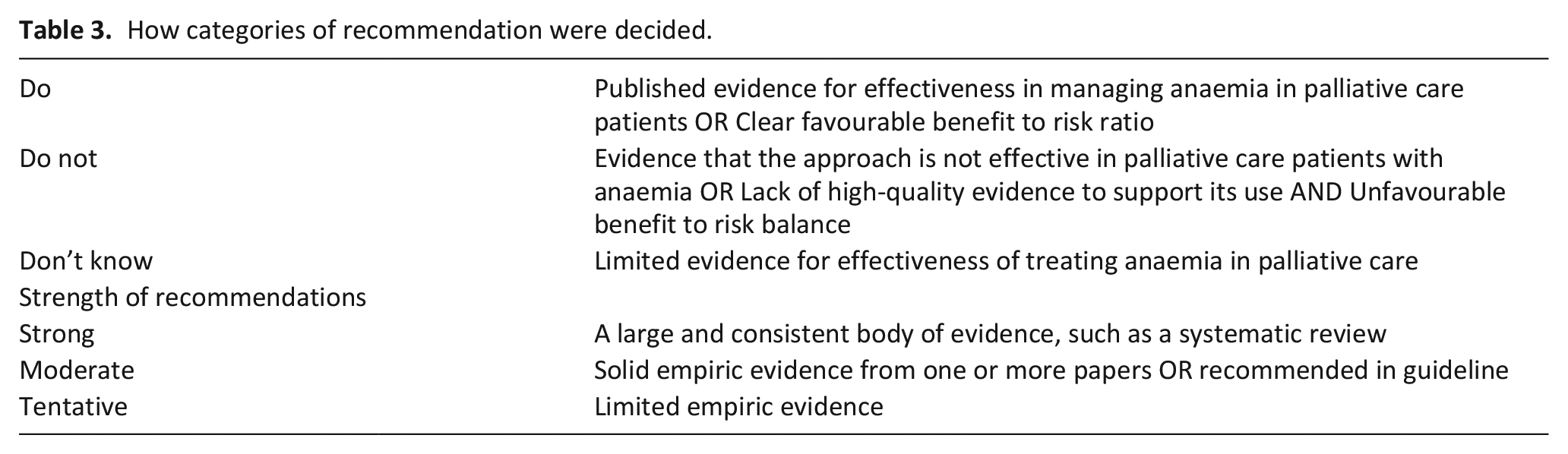
Methods were compliant with the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) Checklist. We determined the strength of evidence for commonly recommended treatments and strategies. Each treatment was then allocated to a practice recommendation category of ‘Do’, ‘Do not’ or ‘Don’t know’ based on the direction and strength of the supporting evidence (Table 3). A ‘strong’ recommendation was made if a large and consistent body of evidence existed, such as a systematic review. Comparatively, solid empiric evidence from one or more papers or a guideline advocacy justified a ‘moderate’ recommendation, with a ‘tentative’ recommendation made if limited empiric evidence existed. Please see Table 4 for summary of recommendations.
How categories of recommendation were decided.
Summary of recommendations.

Results
Do
Investigate the cause of anaemia (moderate)
Anaemia is the end point in a process, not a diagnosis. The majority of patients referred to palliative care services are elderly. 2 In the general elderly population anaemia is caused by nutritional deficiencies, chronic inflammation/CKD, or other causes in approximately one third each. 13 It is important to establish the cause of a patient’s anaemia in order to consider appropriate treatments. Renal guidance outlines baseline blood parameters to check.13–15 Equally, NICE recommends a haemoglobin threshold and symptoms that warrant investigation, as well as the acknowledgement of excluding blood loss. 14 Oncology guidelines advocate investigating for cause of anaemia. 10
Consider targeted treatments based on results for example, correct haematinics (strong)
This is a heterogeneous group of patients but who are likely to share some commonalities, such as an underlying inflammatory process. Review of haematinics and iron studies should form part of the assessment and a targeted treatment plan instigated. While the ESMO guidance states B12 and folate deficiencies are rare causes of anaemia in cancer patients, an audit of blood transfusion practice in UK hospices (96% cancer diagnosis) found that of the patients who had a B12 level checked, 12% may have benefitted from B12 injections, of the patients who had a folate level checked, 41% may have benefitted from folic acid. 8 NICE guidance recommends and outlines the management of folate and B12 deficiency 16 ; longitudinal studies support the effectiveness of replacement. 17 This recommendation is made based on the evidence and favourable risk benefit ratio.
Reduce bleeding where possible (tentative)
While not all the guidance we reviewed specifically discusses reducing blood loss it is part of a logical approach that this should be done. This would include investigating any ongoing bleeding, consider endoscopic cautery and reducing the amount of blood taken during blood sample investigations. Oncology guidelines discuss patient blood management, includes minimising blood loss and bleeding. 18 A review of a patient’s medications to consider stopping any that could exacerbate bleeding, such as NSAIDs, 10 and potentially a trial of tranexamic acid can be considered.
Discuss the risks and benefits of treatments to allow an informed decision to be made with the patient (strong)
Palliative care advocates a holistic, person centred approach. 19 This is only possible through involvement of a patient (or their family) with decisions. Education is included in NICE guidance in CKD 14 including practical information about how anaemia of CKD is managed and knowledge about symptoms, and causes of anaemia. The guidelines specifically discuss education around adaptation to chronic disease, including managing patient expectations. An audit of blood transfusion practice in UK hospices found that only 18% of patients had lasting benefit from a blood transfusion, 8 information that should be discussed with patients when taking informed consent.
Do Not
Consider red blood cell transfusion unless haemoglobin is below 70 g/dL (or 80 g/dL in presence of acute coronary syndrome) (strong)
Red blood cell transfusions are commonly used in palliative care to treat anaemia or its symptoms, despite little evidence of benefit to patients and risk of harms. A systematic review of 13 studies of blood transfusion in palliative care found that any symptom benefit is short lived. 20 Equally, a Cochrane review of 12 observational studies found limited benefits, and that approximately one-third of patients died within 2 weeks of transfusion. 21 Nice published red blood cell transfusion guidelines for adults and recommends the use of a restrictive approach: transfusing one unit when haemoglobin falls below 70 g/L to a target concentration of 70–90 g/L. 22 The trigger threshold is raised to 80 g/L in those who are actively bleeding, have acute coronary syndrome or chronic anaemia (as the reason for their transfusion). Recent guidance from the American Association of Blood Banks (AABBs) 23 also recommends a restrictive threshold but concluded that there is insufficient evidence in some patient groups to guide trigger thresholds. The European Society for Medical Oncology (ESMO) 18 advocates a threshold of 70–80 g/L and recommends that transfusions are only used in anaemic patients with severe symptoms in need of rapid haemoglobin improvement. A review found that restrictive transfusion strategies reduced the risk of receiving a blood transfusion by 43% with no impact on clinical outcomes, 30-day mortality or adverse events. Importantly, the authors felt that there were insufficient data to draw firm conclusions about the safety of transfusion policies in certain subgroups including acute coronary syndrome, bone marrow failure and blood cancers. 24
Transfuse more than one unit of blood without reassessing (strong)
A national audit of blood transfusion practice in UK hospices 8 found that 84% of patients were given more than one unit of blood. Transfusion-associated circulatory overload (TACO) is cardiogenic pulmonary oedema caused by the infusion of blood products, with a mortality of 6%–10%. 25 TACO is more common with rapid or large volume infusions. Patients treated in hospices commonly have risk factors for TACO which include hypo-albuminaemia, low body weight, being physiologically compromised by cardiac, respiratory or renal insufficiency, as well as older age.5–7 TACO is likely underdiagnosed; one study across 157 UK hospitals showed that 4.3% of inpatients aged over 60 years developed increasing respiratory distress after transfusion. 26 Despite this, only one-third of patients diagnosed with TACO were reported to the SHOT Haemovigilance Group. 26 Patients should receive a weight-related transfusion, which may be less than one unit if the patient weighs less than 70 kg.
Routinely use Erythropoetin Stimulating Agents (ESAs) (strong)
ESAs are used in chemotherapy induced anaemia. The palliative population is likely to have a different cause of their anaemia. With the exception of selected patients with myelodysplastic syndromes, ESAs should not be offered to most patients with non- chemotherapy associated anaemia. 18
In those with heart failure ESAs successfully correct haemoglobin levels, but without significant improvement in clinical outcome. ESAs increase rates of thromboembolic events and ischemic stroke, therefore their use for anaemia in heart failure is not recommended. In those with chronic kidney disease there may be specific situations in which ESA’s may be indicated. 27 For example, NICE recommend ESAs with concurrent iron supplementation. 14
Routinely use oral iron (moderate)
Oral iron is poorly tolerated and is not well absorbed when systemic inflammatory processes are present (indicated by raised C-reactive protein (CRP) marker). These processes raise serum hepcidin and inhibit gastrointestinal iron uptake. In many advanced diseases, raised CRP is a common finding and would make oral iron supplementation ineffective. In a study of palliative care patients CRP was raised in 84% of participants in which it was recorded, highlighting the prevalence of inflammation in this population. 2
A RCT comparing oral iron with placebo in heart failure noted increased iron stores but no significant effect on exercise capacity. 28 A significant relationship between higher baseline hepcidin levels and lack of iron repletion was noted, supporting the view that oral iron is unlikely to be helpful in those with active inflammation.
Due to the effects of inflammation on oral iron absorption, the difference between absolute and functional iron deficiency should be considered. In absolute iron deficiency, total body iron stores are reduced, therefore demand cannot be met. Comparatively, in functional iron deficiency, body iron stores are adequate but alterations in iron homeostasis limit the supply of iron for erythropoiesis. This is most frequently observed in chronic inflammatory states and/or infection, from which pro-inflammatory cytokines, especially interleukin-6 and tumour necrosis factor, induce hepcidin production, a peptide produced by the liver. Hepcidin inhibits intestinal iron absorption and limits the recycling of iron from red blood cell by degrading the macrophage transmembrane protein ferroportin. The processes underlying functional iron deficiency are considered a component of the pathophysiology of the anaemia of chronic disease. The distinction between absolute, functional or a mixed deficiency is challenging, considering a history of inflammation and chronic disease alongside appropriate laboratory investigation. 29 Although not in a palliative population, a trial of oral iron replacement in management of sole absolute iron deficiency has shown benefit and is advocated in guidelines. 29 Translation to a palliative population remains unevidenced but, considering the prevalence of inflammation, the routine use of oral iron should be avoided. 2 A more detailed discussion about functional iron deficiency or the anaemia of chronic disease is not possible in this review.
Don’t know
The effectiveness of IV iron in this patient group
Trials of IV iron in palliative care patient groups are rare. Studies in heart failure indicate that IV iron may be effective in improving symptoms (functional class, exercise capacity and QOL) but further trials are needed. Oncology guidelines 18 advocate treatment of absolute iron deficiency with IV iron, as well as those with functional iron deficiency being considered for ESA treatment. Advantages include a reduction in the number of red blood cell transfusions required and an improved response to ESAs. However, the long-term safety in oncology has not yet been fully established.
Recent British Society of Gastroenterology guidelines for the management of iron deficiency anaemia in adults 29 advocate the consideration of IV iron when oral iron is contraindicated, ineffective or not tolerated. Therefore, there may be scope for the wider use of IV iron in patients with either absolute or functional iron deficiency, in whom inflammation would render oral administration ineffective or following a failed or poorly tolerated trial of oral iron in absolute iron deficiency. IV iron is significantly more expensive than oral iron and more invasive to administer, factors which need to be taken into account. Considering its complex pathophysiology, research is required to define the role, if any, of IV iron replacement in many patient groups with functional iron deficiency.
Management of anaemia in patients with myelodysplastic syndrome (MDS) receiving palliative care
Only a minority of patients with MDS can be treated with curative intent (i.e. allogenic haematopoietic cell transplantation). Therefore, a combined approach that includes both disease-modifying agents and palliative care is needed early in the course of illness. The supportive management of anaemia in MDS may include ESAs and occasionally granulocyte colony stimulating factor (G-CSF), but this is often limited to MDS patient with lower risk factors. Even in this subgroup only the minority patients (approximately 1/3) show improvement in erythropoiesis, and with no overall survival benefit. 30 RBC transfusions are generally reserved for patients with symptomatic anaemia with a haemoglobin of <80–100 g/L (8–10 g/dL). 18
Limitations
Limitations include the method of using a scoping review technique which is not as rigorous as a systematic review. Therefore there may be additional guidelines or studies not included.
Conclusion
Anaemia is common in those with advanced disease and a large proportion of this may be due to functional iron deficiency. We recommend early consideration and investigation of anaemia, guided by symptom burden and patient preferences. Correction of reversible causes should be the mainstay of treatment, with a restrictive approach to red blood cell transfusion considered only in circumstances supported by international guidelines. Routine use of oral iron and ESAs are not recommended. Research is required to evaluate the efficacy of IV iron in palliative care patients.
Footnotes
Appendix 1
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
