Abstract
Background:
Use of implantable cardioverter defibrillators is increasingly common. As patients approach the end of life, it is appropriate to deactivate the shock function.
Aim:
To assess the prevalence of implantable cardioverter defibrillator reprogramming to deactivate the shock function at the end of life and the prevalence of advance directives among this population.
Design:
Following a previously established protocol available in PROSPERO, we performed a narrative synthesis of our findings and used the logit transformation method to perform our quantitative synthesis.
Data sources:
We searched seven bibliographic databases (Embase, Cochrane Central register of controlled Trials, Medline-Ovid, Web-of-Science, Scopus, PsychInfo, and CINAHL) and additional sources until April 2019.
Results:
Of the references we identified, 14 were included. We found a pooled prevalence of implantable cardioverter defibrillator reprogramming at the end of life of 28% (95% confidence interval, 22%–36%) with higher reprogramming rates after the recommendations for managing the device at the end of life were published. Among patients with advance directives, the pooled prevalence of advance directives that explicitly mentioned the device was 1% (95% confidence interval, 1%–3%).
Conclusions:
The prevalence of implantable cardioverter defibrillator reprogramming and advance directives that explicitly mentioned the device was very low. Study data suggested reprogramming decisions were made very late, after the patient experienced multiple shocks. Patient suffering could be ameliorated if physicians and other healthcare professionals adhere to clinical guidelines for the good management of the device at the end of life and include deactivating the shock function in the discussion that leads to the advance directive.
Keywords
Although implantable cardioverter defibrillator is a successful therapy, all patients eventually progress to the end-of-life phase.
In patients with a progressive and marked decline, the shock function of the implantable cardioverter defibrillator no longer prolongs life and may instead cause them and their families unnecessary distress.
Expert consensus statements and several guidelines recommend discussing deactivating the implantable cardioverter defibrillator shock function with patients who are nearing the end of their lives.
In these meta-analyses, including data from six different developed countries, we found a low device-reprogramming rate and a very low rate of advance directives explicitly mentioning the device.
This study shows an improvement in the rates of implantable cardioverter defibrillator reprogramming at the end of life, after the publication of a consensus statement as the ones from the Heart Rhythm Society and the European Heart Rhythm Association.
We highlight the importance of more physicians and other health professionals adhering to clinical guidelines in discussing with the patient their preferences regarding the management of the implantable cardioverter defibrillator at the end of life and in documenting them.
Healthcare directors could facilitate the compliance with the guidelines by organizing training for physicians to initiate discussions on device reprogramming, as well as by organizing informative talks for patients and their families to inform them about end-of-life device management options and encourage them to take the initiative to discuss this with their treating physicians.
Introduction
Both the incidence and prevalence of heart failure are growing as the population ages and risk factors increase. 1 Implantable cardioverter defibrillator has become the standard of care for both primary and secondary prevention of sudden cardiac death in selected patients.2,3 With the expansion of the indications for its use, the number of patients with prolonged survival increased.4–6
Although implantable cardioverter defibrillator is a successful therapy, all patients eventually progress to the end-of-life phase. If the health of a patient with an irreversible condition is deteriorating, the device’s shock function no longer prolongs life and may instead cause them and their families unnecessary distress. 7 Patients who have a recognizable end-of-life phase, with marked progressive decline, may need to reappraise their treatment goals. A discussion should be initiated with these patients so that they and their health providers can decide on the treatments that best meet the patient’s goals of alleviating symptoms and preventing suffering. The discussion should cover the reprogramming of the implantable cardioverter defibrillator to deactivate the shock function, as mentioned in the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA) recommendations for managing implantable cardioverter defibrillator at the end of life, published in 2010.8,9 Note that the term implantable cardioverter defibrillator “deactivation” is a misnomer since besides shocks, the device has other functions such as pacemaker capacity and antitachycardia pacing should be kept active and helps controlling symptoms. 10 Therefore, the term implantable cardioverter defibrillation reprogramming will be used thorough the manuscript.
Discussing implantable cardioverter defibrillator reprogramming during a patient’s end-of-life phase is a Class I recommendation in the 2017 AHA/ACC/HRS guideline for Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death. 2 The result of these discussions is often indicated in a written document called an advance directive, which specifies the procedures and actions required or allowable when a patient is incapacitated or no longer able to communicate. Although implantable cardioverter defibrillator management should be discussed and explicitly mentioned in the advance directive, we do not yet know if advance directives change end-of-life outcomes for patients with the device.
We thus conducted a comprehensive and systematic appraisal of the literature on managing implantable cardioverter defibrillator at the end of life, with the goals of (1) assessing the prevalence of implantable cardioverter defibrillator reprogramming to deactivate its shock function at the end of a patient’s life, (2) determining the prevalence of advance directives among patients with implantable cardioverter defibrillator, and (3) evaluating the role advance directive plays in implantable cardioverter defibrillator reprogramming at the end of a patient’s life. Finally, we set out to critically appraise limitations and gaps in the literature.
Methods
Literature search
Our systematic review followed the Joanna Briggs Institute Guide for conducting a systematic review of incidence and prevalence and was reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline;11,12 the protocol was registered in PROSPERO (code CRD42019131219).
We searched for formal, peer-reviewed scientific literature on studies published before April 20, 2019 (date of last search), in seven electronic databases (Embase, Cochrane Central register of controlled Trials, Medline (Ovid) Web of Science, Scopus, PsychInfo, and CINAHL). We built our search construct for each database in consultation with an experienced medical information specialist. We combined terms related to the exposure (implantable defibrillators, intracardiac defibrillator, implantable cardioverter defibrillator) and the outcomes (deactivation, withholding treatment, turnoff, advance directive). We did not apply language or date restrictions.
To identify informal sources and gray literature, we searched Google Scholar and screened the first 200 hits. To identify more sources, we inspected the reference lists of studies that qualified for a full-text evaluation (backward searching). We performed a forward search by searching these titles in Google Scholar with the “cited by” function. We continued this procedure until it returned no new results.
Study selection and inclusion criteria
A pair of persons screened all titles and abstracts and then conducted full-text reviews to evaluate potentially relevant articles. Reviewers resolved disagreements through discussion. In the case of no consensus between the reviewers, a third reviewer was available to solve any disagreement.
We included prospective and retrospective observational studies (cross-sectional, case-control, or cohort) and randomized and nonrandomized interventional studies of adults (⩾ 18 years old), if the studies assessed the prevalence of implantable cardioverter defibrillator reprogramming at the end of life or assessed prevalence of advance directives among patients with the device.
Data extraction
We used a predesigned data collection form to extract data on study design, characteristics of the study population, and sample size. We extracted each outcome assessed, and the corresponding measure of associations (e.g. prevalence of implantable cardioverter defibrillator reprogramming, prevalence of advance directives, prevalence of shocks at the end of life, and place of death).
Assessing the risk of bias
Two reviewers independently rated the quality of studies, based on the Joanna Briggs Institute Critical Appraisal tools used for the JBI Systematic Reviews Checklist for Prevalence Studies. 13
Data synthesis
We performed a narrative synthesis of findings of included studies and a quantitative synthesis using random-effects models to minimize the effect of between-study heterogeneity. We used STATA release 15 (Stata Corp, College Station, Texas) for all statistical analyses. We used the logit transformation method to combine proportions, using the command
Results
Relevant studies
After deduplication, we identified 2422 potentially relevant citations. We screened titles and abstracts and selected the full texts of 34 articles to evaluate in detail. After full-text assessment, we excluded 20 papers (see Figure 1). We included 14 articles in the systematic review, 11 of which were included in the meta-analysis.
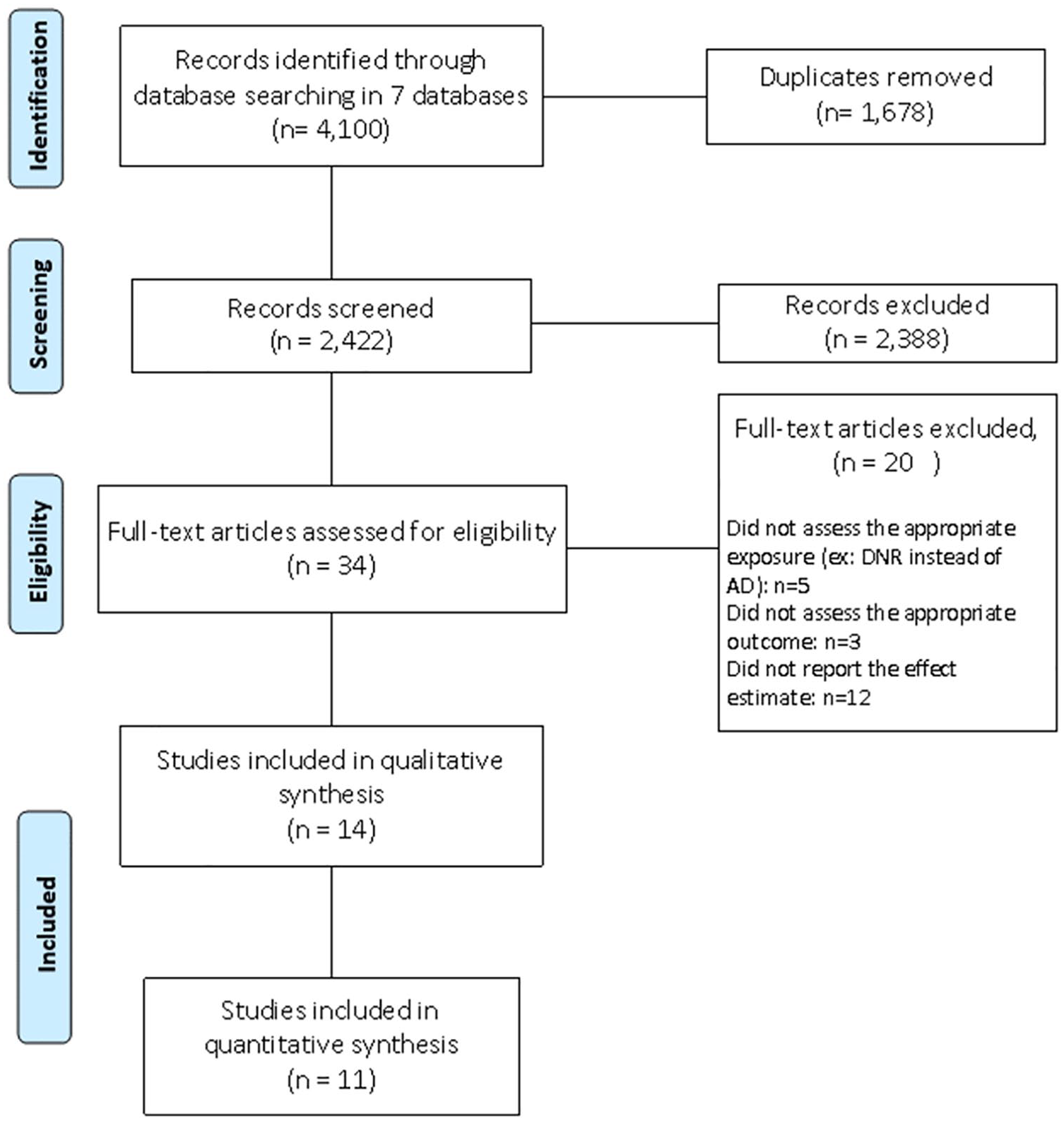
PRISMA 2009 flow diagram.
General characteristics of the included studies
Detailed characteristics of the 14 included studies are summarized in Table 1, eight of which were retrospective cohorts, two were prospective cohorts, two had a cross-sectional design, one was a case-control study, and one was a noncontrolled intervention study. Participants totaled 2745 (range 26–701), 21% of whom were women. Eight studies included participants from the United States, two from Sweden, one from Canada, one from Ireland, one from England, and one from the Netherlands. Two studies included only patients who died in a hospital setting; the others did not distinguish between patients by place of death.
Demographic characteristics and results of the studies included in the systematic review.
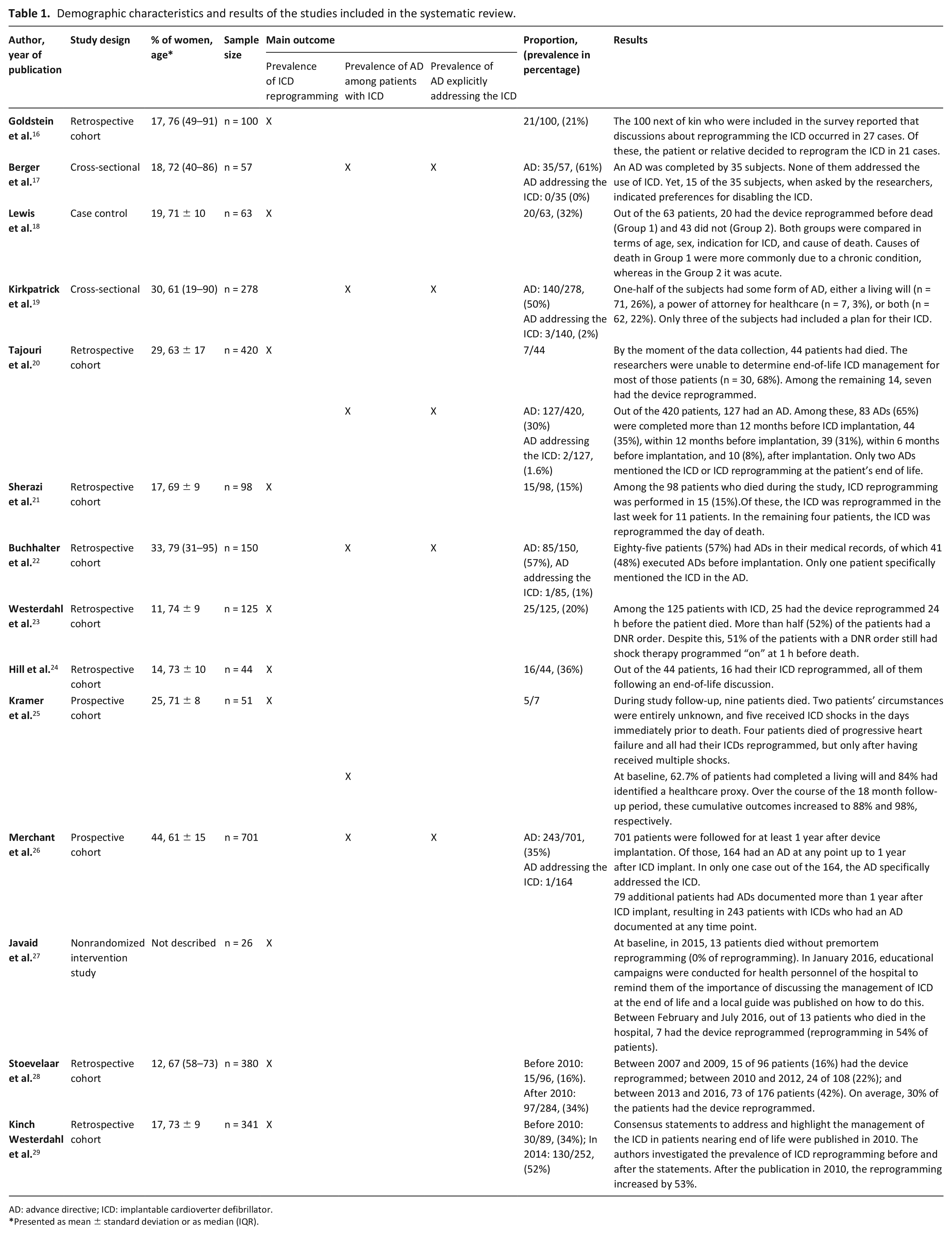
AD: advance directive; ICD: implantable cardioverter defibrillator.
Presented as mean ± standard deviation or as median (IQR).
Of the 14 included studies, two assessed both prevalence of implantable cardioverter defibrillator reprogramming and prevalence of advance directives, eight assessed only prevalence of implantable cardioverter defibrillator reprogramming, and four assessed only prevalence of advance directives.
Supplemental Table 1 shows the risk-of-bias assessment for each study.
Main outcomes
Prevalence of implantable cardioverter defibrillator reprogramming (shock function deactivation)
Ten studies reported prevalence of implantable cardioverter defibrillator reprogramming at the end of life; we included seven in the quantitative synthesis of the results (Figure 2). We explain why we excluded three studies in Supplemental Table 2.
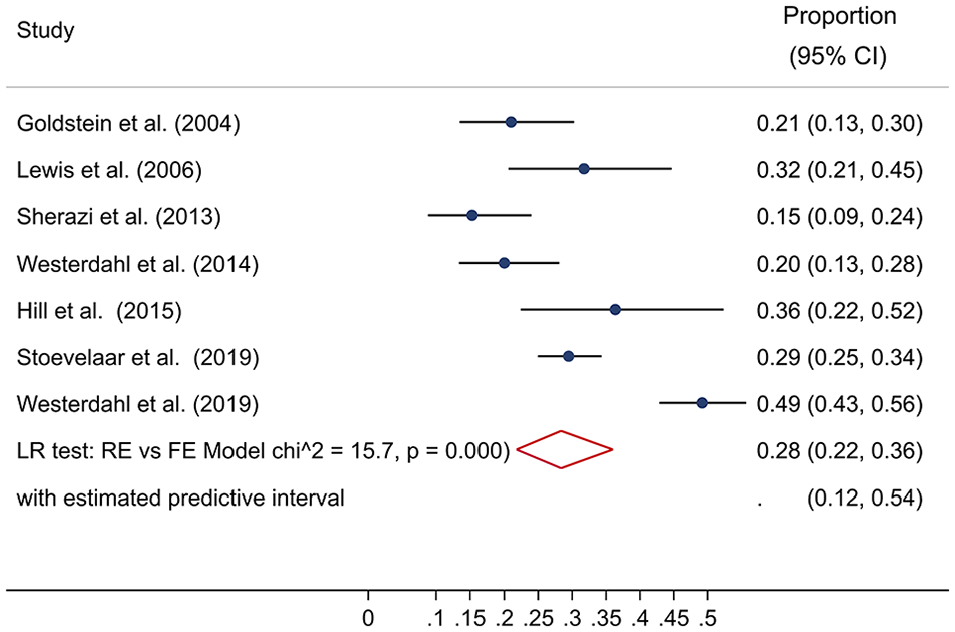
Prevalence of implantable cardioverter defibrillator reprogramming (shocking function deactivation) at the end of life. Forest plot of the studies examining the prevalence of implantable cardioverter defibrillator reprogramming using random-effects meta-analysis. Data presented as proportion and 95% confidence interval (CI, %).
Studies included in the quantitative synthesis
The pooled prevalence of implantable cardioverter defibrillator reprogramming at end of life was 28% (95% CI: 22%–36%), with a tau-square of 0.14 and an estimated predictive interval of 12%–54%. Publication of the EHRA and HRA recommendations explained some of this heterogeneity; when we stratified by outcome assessment date, the estimated predictive interval among studies conducted before the statement publication narrowed to 13%–29% (Supplemental Figure 1).
Five studies included patients treated before the recommendations were published in 2010.16,18,21,23,28 They reported lower reprogramming prevalence (pooled proportion prevalence of 20%, 95% CI: 16%–25%) than articles evaluating the outcome after 2010 (pooled proportion prevalence of 40%, 95% CI: 32%–49%).24,28,29 Two studies included patients treated both before and after 2010 and presented the results separately. Kinch Westerdahl et al. 29 conducted a study in 2019 that included 341 patients from more than 60 hospitals in Sweden and reported the prevalence of implantable cardioverter defibrillator reprogramming at the end of life among patients who died between 2003 and 2010 (34%) and the prevalence of reprogramming among patients who died in 2014 (52%). Stoevelaar et al. conducted a study that included 380 deceased patients from Dutch hospitals; the prevalence of reprogramming among patients who died between 2007 and 2009 was 16% and the prevalence among those who died between 2010 and 2016 was 34%. 28 In both studies, there was a difference in prevalence of 18% over the time, with higher rates after the statements publication.
Studies not included in the quantitative synthesis
Among the three studies that could not be included in the meta-analysis, one assessed the effectiveness of an institutional education campaign about device reprogramming at the end of life and showed that, after the campaign, out of the 13 patients who died, seven had the device reprogrammed. 27 The remaining two studies had a sample size of less than 15 patients for the analysis, due to missing data or because, being a prospective study, most of the patients were still alive.20,25
Prevalence of advance directives among patients with implantable cardioverter defibrillator
Six studies assessed prevalence of advance directives in patients with implantable cardioverter defibrillator. Advance directives included living wills and personal representatives for medical decision-making.
The pooled prevalence was 53% (95% CI: 37%–68%), with a tau-square of 0.58 and an estimated predictive interval of 10%–92% (Supplemental Figure 2).
The presence or absence of advance directives across the studies was assessed in two ways: studies used interviews/self-assessment questionnaires or extracted the information from clinical charts. Due to this difference in the information source, the prevalence of advance directives ranged from 30% to 84%, which explains the broad predictive interval of the pooled estimate. Studies that relied on interviews or self-assessment questionnaires (n = 3) reported higher rates of advance directives than studies that extracted this information from clinical charts (n = 3) (Supplemental Figure 3).
The minimum prevalence of advance directives across the studies was 30%, but the prevalence of advance directives that specifically addressed the implantable cardioverter defibrillator was much lower. Five studies reported that implantable cardioverter defibrillator was rarely explicitly discussed; pooled prevalence was 1% (95% CI: 1%–3%), with a tau-square < 0.001 and an estimated predictive interval from 0% to 4% (Figure 3).20,17,19,22,26 The study by Kirkpatrick et al. had the highest prevalence of advance directives that directly addressed the implantable cardioverter defibrillator: advance directives prevalence was 50.4% (140/278 patients had one), but only 2.1% (3/140) of advance directives addressed the implantable cardioverter defibrillator. 19
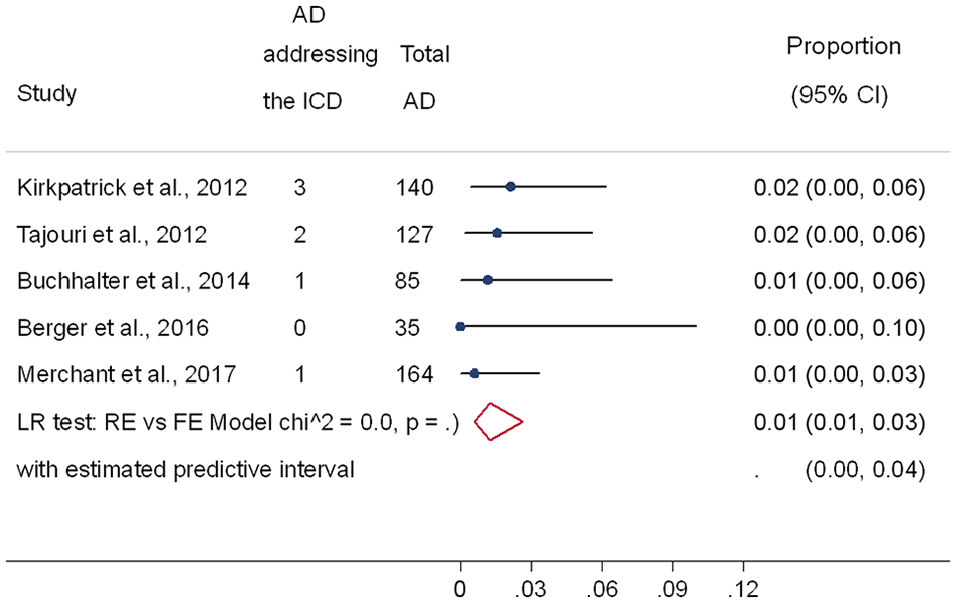
Prevalence of advance directives (AD) explicitly addressing the implantable cardioverter defibrillator. Forest plot of the studies examining the prevalence of advance directives that addressed the implantable cardioverter defibrillator using random-effects meta-analysis. Data presented as proportions and 95% confidence interval (CI, %).
Influence of the presence of advance directives on implantable cardioverter defibrillator reprogramming
Just one study reported an association between advance directives and implantable cardioverter defibrillator reprogramming at the end of life, concluding that patients who had advance directives were no more likely to have had their devices reprogrammed. 20 In this study, data about implantable cardioverter defibrillator reprogramming were missing in 68% of patients (30/44) who died. Since the finding of no association was derived from the remaining 14 patients, there was not enough evidence on the role of advance directives on implantable cardioverter defibrillator reprogramming at the end of life.
Additional outcomes
Prevalence of delivered shocks at the end of life
Five of the studies we included reported prevalence of shocks at the end of life in addition to the main outcomes we defined. Because “end of life” is not a well-defined period, reports on outcomes were so heterogeneous that we could not quantitatively pool prevalence of shocks. Some studies reported prevalence in the last 3 months,18,21 others in the last month,16,18,21,28 last week, 21 last days/day,23,28 or last minutes. 16 Prevalence of shocks in the last months of life ranged from 7% to 27%.
Discussion
Main findings
We found that nearly three out of four patients died with a fully active device, half of the patients with an implantable cardioverter had an advance directive, and few of them had an advance directive that specifically addressed device activity at the end of life; the pooled prevalence estimate was only 1%.
Since the 2010 publication of the EHRA and the HRA statements, others have also recommended discussing implantable cardioverter defibrillator reprogramming in patients nearing the end of life.2,30 Although protocols now guide physicians and healthcare providers through the deactivation process step by step,31,32 our review found only a 28% pooled prevalence of implantable cardioverter defibrillator reprogramming at the end of life. Across studies, prevalence ranged from 15% to 52%, and almost all of the device reprogramming happened in a hospital setting, with fewer in hospices and nursing homes, and very few in patients at home. Our pool estimate is low, given the results of studies that have assessed patients’ desire for reprogramming.17,33,34 In one of these studies, the authors included patients who were not yet at the end of life and told them about the potential benefits and burdens of an active shock function. Then, the authors presented to them hypothetical common scenarios such as incurable disease and permanent inability to get out of bed; 71% of participants wanted the implantable cardioverter defibrillator reprogrammed in at least one of these scenarios. 33
Between 0% and 0.01% of patients with implantable cardioverter defibrillator had an advance directive that explicitly addressed the device. In the subgroup of implantable cardioverter defibrillator patients that had any form of advance directive, the pooled prevalence of advance directives that explicitly addressed device activity at the end of life was 1%. This is very low for any cutoff of percentage indicating a satisfactory use of advance directive and indicates that the advance directive, which was created to increase patients’ autonomy and ensure medical management that aligns with their preferences, is an underused legal document. Berger et al. showed that only 35/57 patients with implantable cardioverter defibrillator had an advance directive and none of them addressed or discussed modifying the activity of the device. However, when they interviewed the patients, 15 of that 35 reported that they wanted the device reprogrammed at the end of life. 17 In this small sample, 15 patients could have suffered shocks against their will at the end of their life; an unwanted outcome that could have been prevented through discussion.
Strengths and limitations
The quality of studies included in this review, assessed by the risk of bias, was compromised mainly by the lack of confidence intervals when reporting prevalence (in all the studies) and insufficient sample size (in 9 out of the 14 studies), suggesting they were underpowered to correctly estimate prevalence. However, since the meta-analysis allowed us to increase power by increasing the sample size, we think our study is not similarly limited. Regarding the statistical analysis, although all studies failed to report confidence intervals, the statistical methods employed were adequate.
As a limitation for the external validity of our study, the hospitals most likely to investigate the prevalence of advance directive and the prevalence of implantable cardioverter defibrillator reprogramming may be those most likely to have implemented them in everyday practice. However, many hospitals and care settings do not implement this proceeding into their routine. Even among the institutions that care for people near death, such as hospices, a few have clear policies on implantable cardioverter defibrillator management. A study of 100 randomly selected hospices among the 3750 in the United States found that only 10% had a reprogramming policy. 35 Thus, the real prevalence of advance directives that specifically addresses implantable cardioverter defibrillator, and the prevalence of implantable cardioverter defibrillator reprogramming at the end of life, might be much lower than our results suggest.
Finally, certain study designs may not be the most appropriate to assess the outcomes included in this review. For our first aim, to assess the prevalence of implantable cardioverter defibrillator reprogramming at the end of life, a prospective design could be appropriate as long as it includes only patients with short life expectancy or in whom a progressive decline has been detected. Otherwise, the follow-up needed to evaluate the outcome would be too long, with the risk of high loss of follow-ups or not obtaining the expected number of events. Such was the case in the study conducted by Kramer et al., 25 in which, at the end of the follow-up, only nine patients had died. Similarly, institutional studies assessing the effectiveness of an intervention in a short period are not adequate as the sample size tends to be very small and the reported estimate would correspond to a specific intervention rather than a prevalence derived from routine clinical practice. 27
The main strength of our study is that ours was the first to summarize and critically assess the prevalence of the implantable cardioverter defibrillator reprogramming at the end of life. On the other hand, without claiming to demonstrate or infer causality, the study showed an improvement in the prevalence of the device reprogramming at the end of life after the publication of the HRS and the EHRA statements in 2010. In addition, we both qualitatively and quantitatively summarized the prevalence of advance directives among patients with implantable cardioverter defibrillator, and the prevalence of advance directives that explicitly addressed the device.
Implications and recommendations
This study shows the underuse of implantable cardioverter defibrillator reprogramming and the underuse of documents that record patients’ preferences for end-of-life device management as are the advance directives. Although the rates of deactivation have been higher following the publication of the statements, they continue to be low. Thus, guidelines alone are not sufficient to bring about a change in practice. It is therefore important to design institutional strategies involving both health personnel and patients with their families. These strategies should focus on informing patients with implantable cardioverter defibrillator and their families about the consequences of dying with an active shock function and encouraging them to discuss this with their treating physician. In addition, healthcare personnel should be trained to discuss this with patients and to know how to proceed if the patients desire the device reprogramming.
Furthermore, clinicians must also consider the timing to reprogram the cardioverter defibrillator, as some evidence suggest that the decision to reprogram the device may have been triggered by the patients’ experience of multiple shocks over the preceding weeks. 21
As a gap, women were underrepresented in these studies (21% of included patients). National registries show that women are less likely to receive implantable cardioverter defibrillator than men (e.g. 15% in France and 21% in Australia).3,36 Registry data from 11 European countries found women made up only 23% of the patients with implantable cardioverter defibrillator. 37 Women are also underrepresented in trials for prevention of sudden cardiac death with the device, including MADIT II (15%) and SCD-HeFT (23%).38,39 It is necessary to increase the participation rate of women in both clinical trials and cardiovascular interventions in order to achieve gender balance in outcome assessment and medical care. 40
Conclusion
Although expert consensus statements and several guidelines recommend discussing deactivating the device shock function with patients who are nearing the end of their lives, and studies that have assessed patients’ preferences have shown that most patients would prefer the shock function deactivation,33,34 we found that nearly 75% of patients die with a fully active device. This reveals a gap both between guidelines and clinical practice and between patients’ preferences and the reality of their last days of life.
If more physicians and other healthcare professionals adhered to the clinical guidelines for discussing with the patient their preferences for managing the device, including recommending and establishing advance directives, fewer patients and families would suffer the stress and distress of repeated and futile shocks in the last weeks of their lives. 7 Healthcare directors could facilitate the compliance with the guidelines by organizing training for physicians to initiate discussions on device reprogramming, as well as by organizing informative talks for patients and their families to inform them about end-of-life device management options and encourage them to take the initiative to discuss this with their treating physicians.
Supplemental Material
Supplemental_material – Supplemental material for Unmet device reprogramming needs at the end of life among patients with implantable cardioverter defibrillator: A systematic review and meta-analysis
Supplemental material, Supplemental_material for Unmet device reprogramming needs at the end of life among patients with implantable cardioverter defibrillator: A systematic review and meta-analysis by Valentina Gonzalez-Jaramillo, Piotr Sobanski, Jose A Calvache, Luisa F Arenas-Ochoa, Oscar H Franco, Lukas Hunziker, Steffen Eychmüller and Maud Maessen in Palliative Medicine
Footnotes
Acknowledgements
We thank Beatrice Minder, Information Specialist, for her valuable help to conduct the literature search. We also thank Dr. Orestis Efthimiou for his advices on statistical analysis of the data.
Finally yet importantly, we would like to thank Dr. Kali Tal, Scientific Editor, for her contributions to the edition of this article.
Author contributions
VG, JC, and LA screened titles and abstracts. VG obtained full text, determined eligibility of articles, and participated in data extraction. VG, JL, and LA assessed the quality of the included studies. VG conducted the data synthesis and analysis. PS, MM, and SE contributed in the interpretation of the data. VG, PS, SE, and MM drafted the final paper. All authors contributed to the critical revision of the paper and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 801076, through the SSPH+ Global PhD Fellowship Programme in Public Health Sciences (GlobalP3HS) of the Swiss School of Public Health.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
