Abstract
Background:
There is evidence that psychosocial and spiritual interventions of short duration, such as reminiscence therapy, provide positive impacts on quality of life and emotional and existential well-being in adults receiving palliative care.
Aim:
To determine (1) the feasibility of integrating ‘LIFEView’, a video-based software with >1600 videos of world destinations, in palliative care settings, and (2) positive, neutral or harmful effects of using ‘LIFEView’ videos.
Design:
A mixed-methods pre–post intervention pilot study was conducted to collect feasibility and preliminary data on physical and psychological symptoms, physiological indicators, spiritual well-being and aspects of quality of life.
Setting/participants:
Adult patients on an inpatient palliative care unit or receiving care from a community palliative care consultation team who were capable of providing consent and completing the outcome measures were eligible participants.
Results:
Overall, 27/41 (66%) participants took part in the study. Feasibility criteria, including participant acceptability, low participant burden, tool completion rate and retention rate, were fulfilled, though challenges were experienced with recruitment. Modest improvements, though non-significant, were shown on preliminary data collected on physical and psychological symptoms using the Edmonton Symptom Assessment System–revised, spiritual well-being assessed by the 12-item Functional Assessment of Chronic Illness Therapy – Spiritual Well-Being scale and physiological measurements. Qualitative analysis revealed five themes: motivations for using ‘LIFEView’, perceptions of the technology, reminiscence, ‘LIFEView’ as an adaptable technology and ongoing or future use.
Conclusion:
A future adequately powered study to investigate the impacts of ‘LIFEView’ on patient well-being and quality of life appears to be feasible.
Keywords
What is already known about the topic?
Patients receiving palliative care often experience psychological and existential distress towards the end of life.
Psychosocial and spiritual interventions, such as reminiscence or life review therapies, may be effective in supporting well-being for patients in palliative care.
What this paper adds?
The findings of this article indicate that ‘LIFEView’, a prototype technology–based reminiscence programme, can be used with patients receiving palliative and end-of-life care with high participant acceptability and low burden.
This pilot study demonstrates that it is feasible to integrate ‘LIFEView’ into clinical care practices on an inpatient palliative care unit and within the community for patients receiving palliative care, provided that volunteer support is available.
Volunteer resources and flexibility in volunteer availability are essential in sustaining the programme within palliative care settings, given participant interest in continued use of the technology.
Implications for practice, theory or policy
This study involved the engagement of seriously ill patients in a palliative care setting in order to facilitate the research-driven development of a technology product geared towards this care context.
In this article, majority of patient and family member impressions of ‘LIFEView’ were positive, illustrating the potential for expanding ‘LIFEView’ and similar technology-based programmes to other palliative and end-of-life care settings.
Background
Psychosocial and spiritual distress is common in adults receiving palliative care and is associated with depression, a desire for hastened death, hopelessness, suicidal tendencies and decreased quality of life.1–5 There is increasing recognition that psychosocial and spiritual interventions, such as reminiscence therapy, may be effective in supporting well-being in palliative care.6–9 Reminiscence therapy focuses on the recall of past memories and experiences aided by memory triggers such as photographs, music or meaningful objects. 10 A recent systematic review on the use of psychosocial and spiritual therapies of short duration (<4 sessions or <21 days), including reminiscence or life review interventions, demonstrated positive effects on patient quality of life, emotional and existential distress in the palliative care context. 11 Reminiscence programmes delivered by care staff and using technology have been demonstrated to improve well-being and quality of life in individuals with dementia in community and inpatient settings.12,13 However, findings from the dementia patient population may not generalize to the specialist palliative care context as the latter involves individuals with life-threatening illness nearing the end of life. 14 To our knowledge, there is limited published evidence describing the use of technology-based reminiscence interventions in a specialist palliative care setting. From our literature searches, we found one study investigating the use of virtual reality technology to facilitate reminiscence with patients receiving specialist palliative care. 15 Further research is necessary to identify technology-based reminiscence programmes that may be suitable for use with patients within this care context.
Motiview™, 16 was developed in 2013 by Motitech®, a Norwegian technology company with a userbase in North America, United Kingdom, Australia and the Nordic countries. Motiview allows older people to bicycle through familiar virtual surroundings and memories using TV-displayed videos, ambient sound and music, combined with a portable user-adapted exercise bicycle. The technology consists of a continuously growing video and music library of over 1600 videos in 30 countries. Motiview aims to increase physical activity and memory stimulation which may help prevent falls, improve management of complex health conditions and cognitive fitness, and prevent unnecessary emergency department visits in older people.
In early 2017, preliminary discussions about adapting Motiview for a palliative and end-of-life care context were held between Motitech and a senior leadership member of Bruyère Continuing Care and the Bruyère Research Institute. A meeting was coordinated between Motitech’s leadership team and the senior author (R.H.) to discuss a partnership to adapt Motiview for the purposes of reminiscence and virtual travel for the palliative care population. Key reasons for considering the adaptation of Motiview for this context included its ease of use for patients, staff and volunteers, minimal need for technological skills on the part of users, low possibility of inducing disorientation or nausea and limited availability of similar technologies with the same advantages.
Thus, the concept of ‘LIFEView’ was created as a prototype technology in development by Motitech and a project team was assembled, including both researchers and members of the palliative care interprofessional team. ‘LIFEView’ solely uses the audiovisual capabilities of Motiview via a touchscreen laptop tablet connected to a large-screen TV to facilitate reminiscence. This technological set-up was selected to enable interactivity and minimize risks (e.g. nausea or disorientation) to the vulnerable palliative care population.
Feasibility studies are recommended in palliative care due to unforeseeable changes in disease course, recruitment issues and attrition rate from death and change in functional status.17,18 The primary objective of this study is to explore the feasibility of using ‘LIFEView’ as a reminiscence programme in palliative care. The secondary objective is to collect preliminary qualitative and quantitative data on physical, psychological, physiological and spiritual effects of its use.
Methods
A prospective, convergent mixed-methods pilot study was undertaken using a pre–post intervention research design. The study was approved by local research ethics boards. Participants were recruited between 29 August 2018 and 30 April 2019. Quantitative data were collected using patient-rated questionnaires and a wireless blood pressure monitor prior to and immediately after the intervention and with an observer-rated scale during the ‘LIFEView’ session. Qualitative data were collected via field notes throughout study participation and using semi-structured interviews after the intervention.
Intervention
The intervention consisted of participants using ‘LIFEView’ for approximately 1 hour in the presence of the study’s research coordinator (M.K.). A spiritual care professional or social worker was also available on-site to provide support in the event of participant distress.
The research coordinator was trained by the technology company, Motitech, in use of the technology. The research coordinator assembled the TV and touchscreen laptop tablet in the patient’s room or residence and provided participants with an in-person demonstration of how to use the technology. If necessary, the research coordinator offered assistance throughout the ‘LIFEView’ session.
Participants were given the option to use the touchscreen laptop tablet to operate the software’s interface, unless they were unable to do so due to physical limitations (e.g. vision loss, reduced upper limb mobility). In such cases, the research coordinator assisted participants with selections. Participants selected videos within the ‘LIFEView’ interface for virtual tours of destinations of their choice. They were also able to play music to accompany videos from within the software interface, pause, rewind and fast forward through videos and stop the session at their discretion using the touchscreen laptop tablet.
Setting and participants
Eligible participants included adults (⩾18 years) receiving specialist palliative care within an inpatient palliative care unit at a subacute teaching hospital, or within the community through interprofessional palliative care consultation service home visits. Based on clinical care team assessments, participants who were able to complete the study outcome measures and provide written informed consent were included. Family members of participants were also included in interviews provided that they were ⩾18 years and present during the ‘LIFEView’ session. Patients were excluded if they were experiencing delirium, severe cognitive impairment, known behavioural abnormalities or a Palliative Performance Scale score 19 of <30% at the time of consent, based on assessments by the clinical care team.
Sample size
We aimed for a study sample of 30 participants, which is in alignment with recommended sample sizes of between 24 and 50 for pilot studies.20–22 The research team felt that this sample size was sufficient for reaching data saturation 23 for the qualitative component of this study.
Outcome measures
Primary outcome: feasibility of intervention
Available guidance on conducting feasibility studies in palliative care indicates the need to assess recruitment rates, retention rates, participant acceptability and burden of the intervention and outcome measures, given the complexities and unpredictability of this care context.17,18 Completion of assessment tools and recruitment and retention rates were calculated to determine the feasibility of conducting a future larger trial using ‘LIFEView’ and its impacts on health outcomes.
Indicators of participant acceptability of the technology were assessed by the research coordinator during the ‘LIFEView’ session using the observer-rated Greater Cincinnati Chapter Well-Being Observation Tool©.24–26 This tool is commonly used to assess psychosocial interventions in patient populations with cognitive deficits and contains seven domains: interest, sustained attention, pleasure, negative affect, sadness, self-esteem and normalcy. Each domain was rated on a 0–4 scale (0 = never, 1 = rarely, 2 = some of the time, 3 = most of the time, 4 = always).
To further address acceptability, demand, implementation and practicality aspects of feasibility, 27 the research coordinator compiled process-based observational field notes throughout the course of the study to assess:
Facilitators or barriers to using ‘LIFEView’;
Physical set-up issues;
Differences between inpatient and community settings;
Participant burden based on adverse effects noted by participants or informal caregivers;
Overall participant perceptions of the technology or assessment tools;
Feedback from the participant’s clinical care team;
Indications of participant interest in ‘LIFEView’ based on duration of video viewing, perceived length of video, family member or caregiver participation and requests for volunteer-led ‘LIFEView’ session(s) after study participation.
The feasibility of using volunteer services to support the long-term sustainability ‘LIFEView’ was assessed based on the number of participant requests for additional sessions and volunteer availability to meet the demand until patient death or decline in condition and recorded in the research coordinator’s field notes.
Secondary outcomes
The following outcome data were collected by the study’s research coordinator at least 24 h before (pre-‘LIFEView’) and immediately after using ‘LIFEView’ (post-‘LIFEView’). If necessary, reading and writing assistance was provided by the research coordinator:
Change in psychological and physical symptoms. The Edmonton Symptom Assessment System–revised (ESAS-r),28,29 a nine-item symptom assessment tool evaluating pain, tiredness, drowsiness, nausea, lack of appetite, shortness of breath, depression, anxiety, well-being and sleep, was completed by participants. Each item is rated by patients on a 0–10 scale (0 = best possible, 10 = worst possible).
Change in physiological indicators. The research coordinator used a wireless cuff to measure participant heart rate and blood pressure.
Change in spiritual well-being. The 12-item Functional Assessment of Chronic Illness Therapy – Spiritual Well-Being scale (FACIT-Sp-12) 30 was completed by participants. Each item is rated by patients on a 0–4 scale (0 = not at all, 4 = very much).
These outcome measures were selected for preliminary data collection in this pilot study to determine the value of further exploration in a future adequately powered study. It was surmised that use of ‘LIFEView’ may impact participants’ physical and psychological symptoms, spiritual well-being and physiological indicators. The ESAS-r and FACIT-Sp-12 were selected as assessment tools given their validity for use in palliative and end-of-life care contexts.28–30 Blood pressure and heart rate were measured to determine whether participants experienced calming effects after use of the intervention.
A semi-structured, audio-recorded interview was conducted by the research coordinator within 24 h post-‘LIFEView’ with each participant, and if present, their family members. These interviews explored the overall participant and family experience of ‘LIFEView’. The procedures for collecting these outcome measures are outlined in Figure 1.

Process map of study procedures from initial contact with the research team.
Statistical analysis
Quantitative data were analysed using SAS 31 software. Feasibility data, participant characteristics and outcomes were described using descriptive statistics. Changes in pre- and post-‘LIFEView’ outcomes were analysed using the Wilcoxon signed rank test, with a p value of <0.05 considered statistically significant.
Qualitative analysis
Audio-recorded interviews were transcribed verbatim and analysed using NVivo 12. 32 Qualitative data collected from interviews and observational field notes were analysed using an iterative, inductive thematic analysis approach.33,34 Codes and themes were generated from a subset of the data using group open coding. 35 Open coding was considered complete when the group (M.K., B.G. and M.D.) agreed that there were minimal new concepts or themes emerging from the data. Subsequent to open coding, two researchers (M.K. and B.G.) independently coded the full qualitative data set in accordance with emerging patterns or themes, refining themes as needed. Disparities in coding were discussed and negotiated with a third individual (M.D.) to ensure analytical rigour.
Results
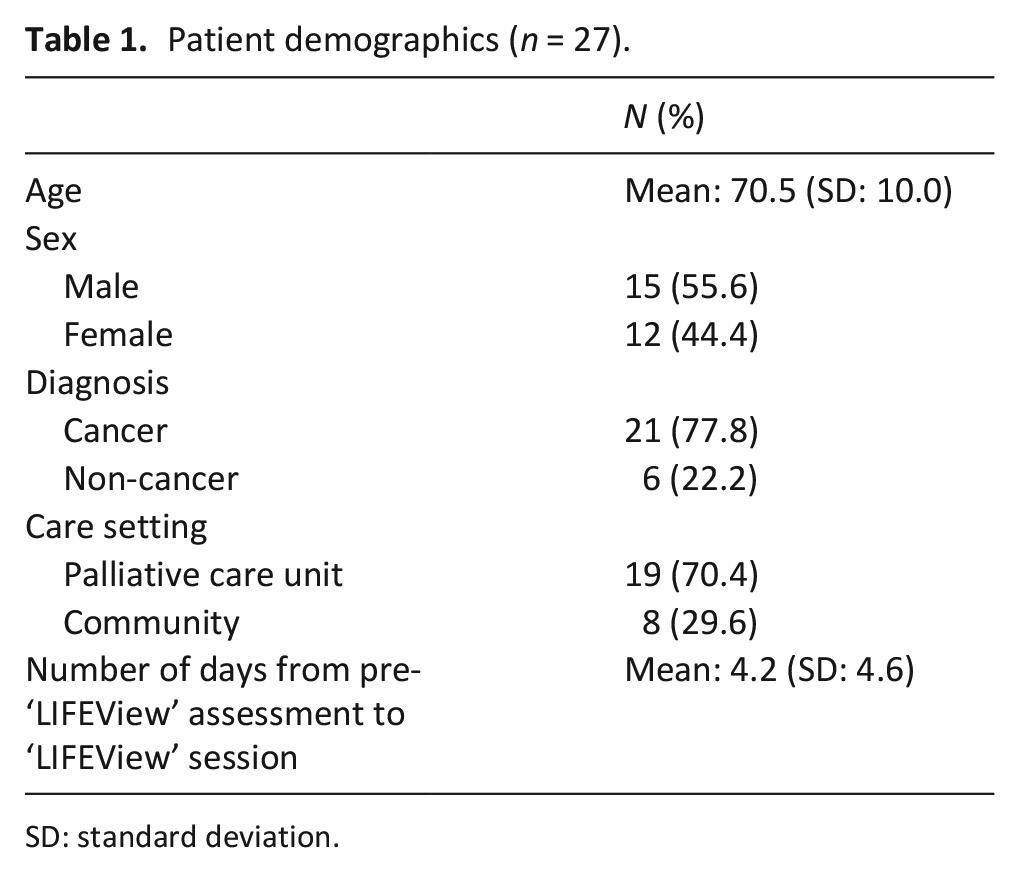
Patients admitted to the inpatient palliative care unit (30/394; 8%) or referred to a single community palliative care service (11/673; 2%) were approached by their clinical care team for verbal assent to be contacted by the research team. Of these individuals, 31/41 (76%) were recruited to the study after consenting to participate. Subsequent to consenting, three participants requested their withdrawal from the study due to feeling ‘foggy’ (n = 1) and progressive illness or fatigue (n = 2). One participant died prior to taking part in the study. These participants were excluded from data collection and analysis. The remaining 27/41 (67%) participants took part in the intervention with their family members (n = 16). Detailed information on patient participation is provided in Figure 2. Demographics for included participants are presented in Table 1.
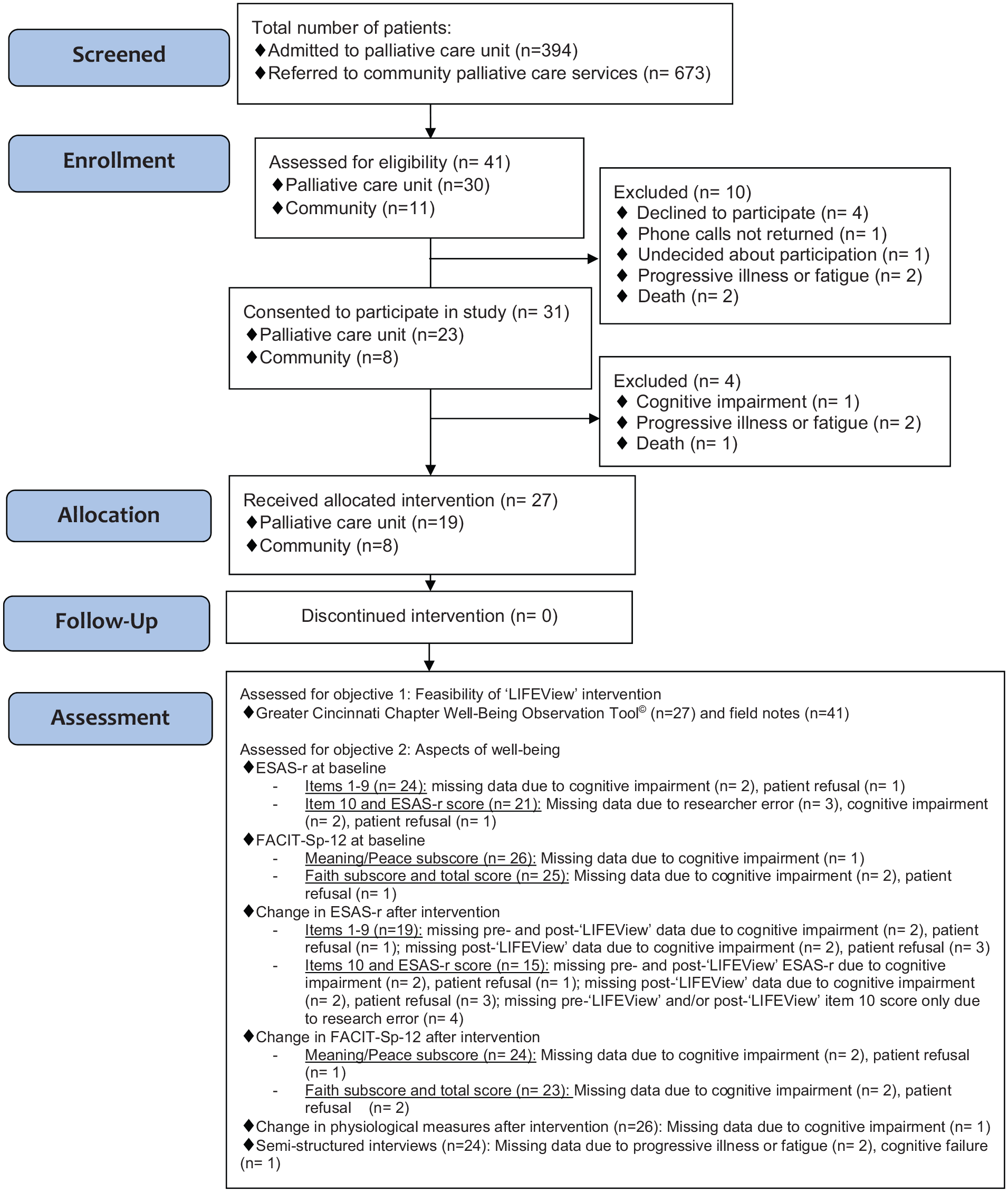
Modified CONSORT extension-pilot and feasibility trials 36 flow diagram.
Patient demographics (n = 27).
SD: standard deviation.
Feasibility of ‘LIFEView’ intervention
For the 27 participants who used ‘LIFEView’, the mean viewing time was 60.6 min (SD: 17.4) and 23 (85.2%) verbalized enjoying the session. The Greater Cincinnati Chapter Well-Being Observation Tool© was completed for all 27 participants (Table 2). High levels of participant interest (mean: 3.70/4, SD: 0.72), attention (mean: 3.74/4, SD: 0.71) and pleasure (mean: 3.56/4, SD: 0.85) were observed during ‘LIFEView’ sessions, while minimal negative effects were noted. Largely positive participant perceptions of ‘LIFEView’ from observational field notes, interview data and overall completion of tools (15/27; 56%) provided further evidence for participant acceptability of the technology and study procedures.
Results of Greater Cincinnati Chapter Well-Being Observation Tool© for all participants (n = 27) during their ‘LIFEView’ sessions, as assessed by a single observer.
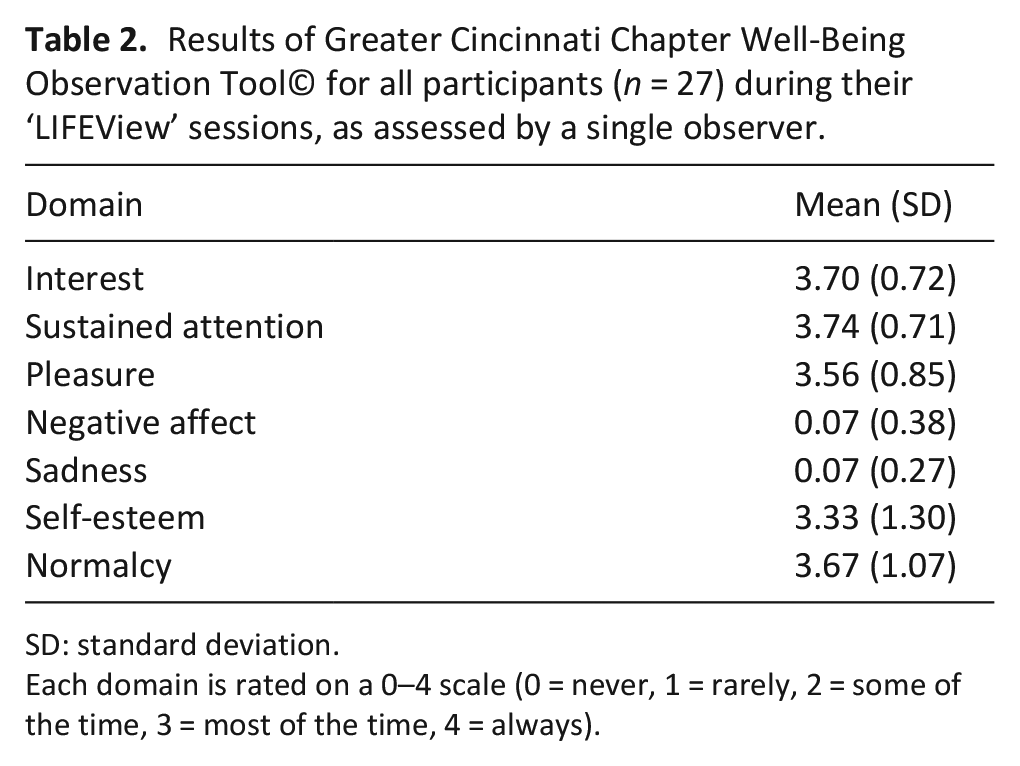
SD: standard deviation.
Each domain is rated on a 0–4 scale (0 = never, 1 = rarely, 2 = some of the time, 3 = most of the time, 4 = always).
Majority of missing data for tool completion was due to changes in cognitive function as assessed by the participant’s clinical care team or participant refusal. Minimal occurrences of missing data occurred due to researcher error (n = 4) where ‘Sleep’ was not entered in the blank space for an additional item on the ESAS-r form. No major barriers were encountered with respect to the physical set-up of the technology based on field notes collected.
Participant burden was minimal; there were no adverse effects specific to the use of ‘LIFEView’ and few concerns noted by participants or their caregivers. Specific concerns included participant fatigue potentially obstructing participation (n = 1), uncertainty of their enjoyment of the session (n = 1) and the potential for future ‘LIFEView’ users experiencing sadness due to their reminiscences (n = 1). Participants who were concerned about their fatigue and enjoyment of the session took part in the study fully. The participant who noted that other users may feel sadness elaborated that the benefits offset the harms.
After participation in the research study, participants were given the option to request continued access to ‘LIFEView’. Following a participant’s request for additional sessions, volunteers visited patients with the technology. Additional volunteer-led sessions were requested by 18/27 (66.7%) participants, 7/27 (25.9%) declined this offer and 2/27 (7.4%) had unknown preferences for additional sessions. Overall, 23 volunteer-led sessions were provided to 8/18 (44.4%) of those who requested continued access to ‘LIFEView’, for a mean of 2.88 additional sessions per participant (SD: 2.48). The 10/18 (55.6%) participants who requested but did not receive volunteer-led sessions were no longer responsive, somnolent or deceased by the time a volunteer was available for a subsequent session.
Aspects of participant’s well-being and overall experience of ‘LIFEView’
Quantitative results
Pre-‘LIFEView’ ESAS-r and FACIT-Sp-12 scores are presented in Supplementary Table 1. No significant differences were found in the change in ESAS-r ratings (Supplementary Table 2), FACIT-Sp-12 scores (Supplementary Table 2) or blood pressure and heart rate measurements (Supplementary Table 3) between pre- and post-‘LIFEView’ assessments. Of note, minor improvements were demonstrated for the change in ESAS-r and FACIT-Sp ratings, particularly for ESAS-r rated well-being, tiredness and anxiety items and meaning/peace and faith subscales of the FACIT-Sp. Heart rate measurements pre- to post-intervention showed a negative mean difference.
Qualitative results
Overall, 24 participants and 16 family members participated in interviews (mean duration = 13.76 min, SD: 7.23). Analysis of interview transcripts (n = 24) and observational field notes (n = 41) yielded five major themes encompassing the participant experience of ‘LIFEView’. The qualitative data reflected an overarching theme related to the temporality of reminiscence and meaning-making, with a specific focus on participant creations of meaning from the past, and into the present and future. ‘LIFEView’ offered participants the potential to explore meaning-making by enabling experiences of destinations they were motivated to visit, but had not had an opportunity to see in person, or to reminisce about places they had visited in the past. Challenging present-day experiences of illness and use of the technology as an escape were also woven into participant motivations, allowing participants to make meaning and reminisce to a time and place prior to, or beyond their present-day experience.
Positive, neutral and negative impressions of the technology illustrated participant experiences of ‘LIFEView’. Majority of participants (14/16; 88%) who noted one or more negative perceptions of ‘LIFEView’ had predominantly positive experiences across the qualitative data and reported enjoying the session overall. Participants engaged in reminiscing and creating meaning from these past experiences, particularly in the presence of family members. Other participants discussed leaving a legacy for their loved ones. Other themes included participant views of ‘LIFEView’ as an adaptable technology, wherein participants noted recommendations or appealing components, and ongoing or future use of the technology. These themes are further delineated in Table 3.
Themes and subthemes of the participant experience of ‘LIFEView’ arising from interviews (n = 24; 796 references) and field notes (n = 41; 675 references).
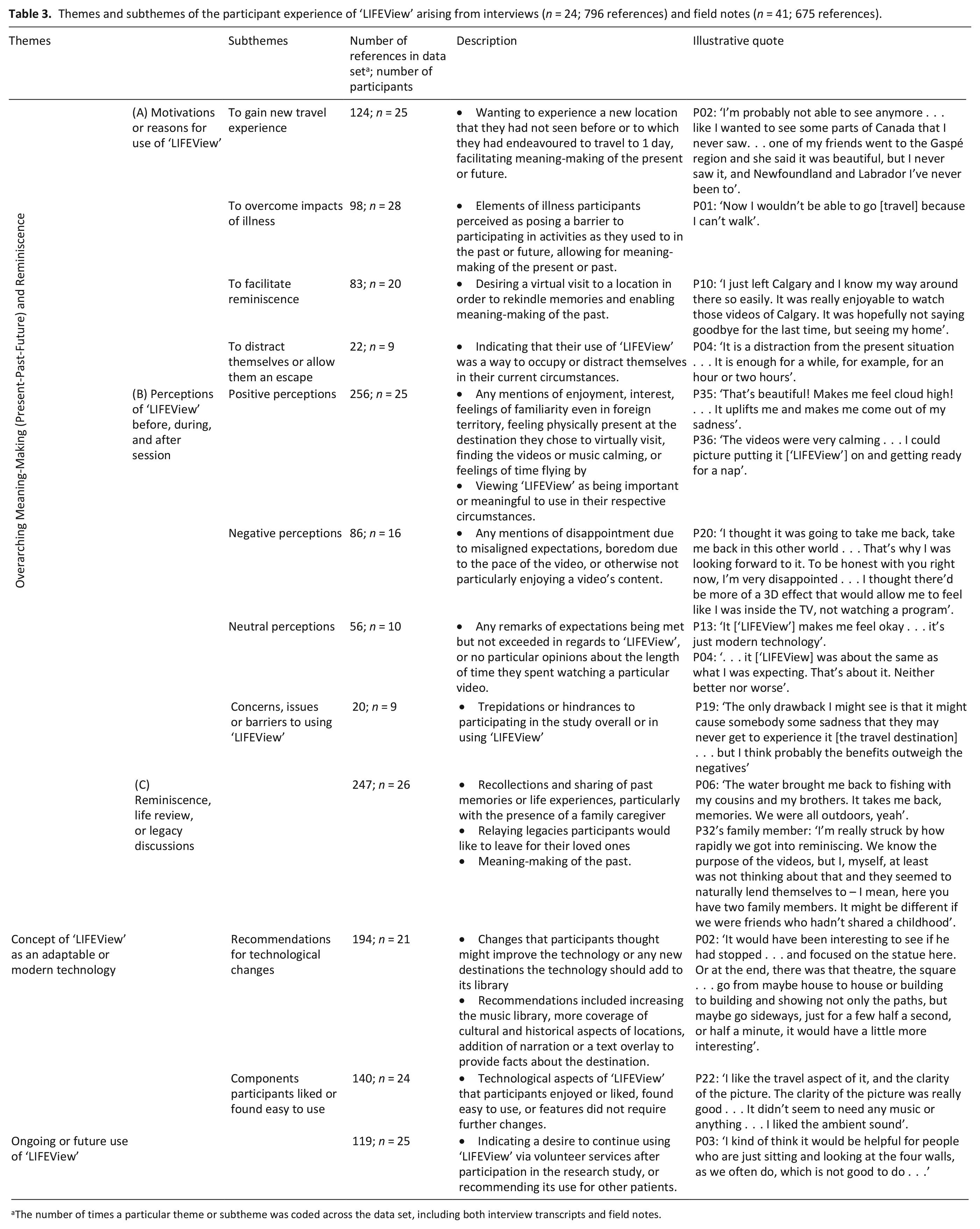
The number of times a particular theme or subtheme was coded across the data set, including both interview transcripts and field notes.
Discussion
Main findings
Overall, this pilot study provides evidence that ‘LIFEView’ technology is feasible to integrate into existing care practices as a reminiscence and virtual travel programme for adults receiving inpatient and community palliative care services. For most participants, use of ‘LIFEView’ videos and accompanying music yielded extensive conversation, reminiscence and meaning-making during sessions and interviews. Correspondingly, previous studies using reminiscence or life review interventions in palliative care contexts demonstrated reminiscence as an effective method of finding meaning in one’s life experiences and also found associated reductions in psychosocial and spiritual distress.7,8 Future research with ‘LIFEView’ should encourage family participation, as individuals with pre-existing relationships to participants may increase instances of reminiscence and meaning-making, and thus potential improvements in well-being.
According to the literature, the ‘well-being’ item on the ESAS-r demonstrates a modest correlation with quality of life in patients with advanced cancer. 37 Our study found a non-significant but potentially meaningful change in the well-being item of the ESAS-r and improvements in spiritual well-being, physical and psychological symptoms and blood pressure measurements. These measures warrant in-depth assessment in a larger study with adequate statistical power, particularly in light of literature showing improved quality of life and psychosocial and spiritual well-being with patient use of similar brief psychosocial interventions.7,8,11 A new study is currently underway to further examine these outcomes. 38 Of note, well-being and quality of life in advanced cancer patients can be multi-faceted; 39 therefore, forthcoming studies should include a comprehensive assessment of patient well-being and quality of life without increasing patient burden.
Of note, majority of negative participant perceptions of ‘LIFEView’ appeared to coincide with unmet initial expectations, particularly misperceptions of the technology (e.g. use of virtual reality headsets or videos enabling extensive visits of cultural aspects of each destination). In future work using ‘LIFEView’, participants should be provided with a clear explanation and/or a picture of the technology to reduce misconceptions.
A key challenge encountered in this study was a low recruitment rate across both inpatient and community settings, which reflects the well-documented difficulties of recruiting in palliative care.40–45 For future work, application of established strategies may improve recruitment, such as communication and outreach to the clinical care team, 45 increased promotion of the study to participants,45,46 using simple study methods43–45 and ongoing recruitment rate monitoring. 41
Our study’s completion rate of patient-reported assessment tools (56%) is comparable to prior pilot and feasibility studies in palliative care (50%–60%).47–49 The use of a mixed-methods study design may have contributed to participant completion of outcome measures through minimizing participant burden, 44 while also accounting for a multitude of perspectives.
The availability of volunteer resources is fundamental for sustainability of the technology within care settings after the culmination of the research study. Although 66.7% of participants requested additional sessions, only 44.4% were able to access volunteer-led sessions likely due to unpredictable or rapid changes in patient condition and limited volunteer availability. Ongoing efforts should address the accessibility and flexibility of volunteer resources for the provision of ‘LIFEView’ sessions.
Based on this current work, considerations for future research on psychosocial and existential programmes in palliative care include increasing researcher, and if applicable, volunteer flexibility in delivering the intervention due to unpredictable changes in patient prognosis, and ensuring the prevention of missing data. Use of novel technology in research may result in participants’ expectations being unmet due to misperceptions of the equipment. Researchers using such technology in future work should make efforts to clearly explain the technology being used, possibly even providing a pictorial or other visual cue to reduce the potential for participant misconceptions. Forthcoming studies should endeavour to enhance recruitment rates using strategies outlined in the literature41,43–46 and increase the scope of the study sample to include other care contexts where patients may have palliative and end-of-life care needs.
Strengths and limitations of the study
Given high participant acceptability of the intervention and study procedures, and low participant burden, this study demonstrates that it is feasible to use ‘LIFEView’ as a technology-based reminiscence programme in palliative care. Considering the complexities and high attrition rates (30%–50%) typical of the palliative care context,44,50 the retention rate (67%) of participants in this study is noteworthy but must be interpreted with caution due to the small sample size.
Key limitations of this pilot study include lack of blinding and the use of a small sample size across a single inpatient setting and one community service. The use of a single-arm study design reduces the generalizability of results, while also potentially introducing bias. In addition, poor recruitment rates and selection bias may have been caused by (1) gatekeeping, the previously documented hesitance well-intentioned clinicians may experience in approaching patients for research studies, 46 and (2) clinicians overlooking approaching eligible patients amid their clinical duties. Targeted contact with clinical team members may improve recruitment rates in future research. The observer-rated scale and field notes used in this study are a possible source of bias due to inherent difficulties of remaining fully objective in observational data collection. Furthermore, the presence of the research coordinator during the ‘LIFEView’ session and data collection procedures may have introduced observer bias.
Conclusion
This pilot study illustrates the feasibility of using ‘LIFEView’ technology as a reminiscence and virtual travel programme within inpatient and community palliative care settings. Preliminary quantitative data demonstrated areas for future exploration in a larger study. Qualitative results demonstrated the patient and family caregiver receptiveness to the technology, supporting its use within the palliative care context.
Supplemental Material
Supplementary_Table_1_Pre-LIFEView_scores_for_ESAS-r_and_FACIT-Sp-12 – Supplemental material for A mixed-methods pilot study of ‘LIFEView’ audiovisual technology: Virtual travel to support well-being and quality of life in palliative and end-of-life care patients
Supplemental material, Supplementary_Table_1_Pre-LIFEView_scores_for_ESAS-r_and_FACIT-Sp-12 for A mixed-methods pilot study of ‘LIFEView’ audiovisual technology: Virtual travel to support well-being and quality of life in palliative and end-of-life care patients by Monisha Kabir, Jill L Rice, Shirley H Bush, Peter G Lawlor, Colleen Webber, Pamela A Grassau, Bahareh Ghaedi, Misha Dhuper and Rebekah Hackbusch in Palliative Medicine
Footnotes
Acknowledgements
The authors would like to thank Motitech AS® for providing ‘LIFEView’ licences and technological support for the duration of this study, the volunteers (Misha Dhuper, Wafaa Kaouache and Layla Ziha) who contributed their time to providing additional ‘LIFEView’ sessions to participants and their informal caregivers after their participation in the research study and the many clinical care team members across both the inpatient and community palliative care settings who contributed their time to assist with recruitment. This study would not have been possible without the contributions of all of the above individuals.
Author contributions
M.K. contributed to ethics submissions, data collection and analyses, manuscript preparation; J.L.R. to methodological support and critical appraisal of manuscript; S.H.B. to methodological support and critical appraisal of manuscript; P.G.L. to methodological support and critical appraisal of manuscript; C.W. to methodological support, data analyses, critical appraisal of the manuscript; P.A.G. to methodological support, data analyses, critical appraisal of the manuscript; B.G. to data analyses, critical appraisal of the manuscript; M.D. to data analyses, critical appraisal of the manuscript; and R.H. to methodological support, critical appraisal of the manuscript.
Data sharing
The study data are held at the Bruyère Research Institute. Requests for additional information about study data or results can be forwarded to the corresponding author (M.K.).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
The study was approved by the Bruyère (Protocol #M16-19-019) and Ottawa Health Science Network (Protocol #20180589-01H) research ethics boards on 9 July 2018 and 24 August 2018, respectively.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a Spark 2017 Centre for Aging and Brain Health Innovation (CABHI) grant.
Technology details
‘LIFEView’ is a prototype technology. As per discussions with Motitech AS®, the software developer, the technology has been referred to within quotations throughout this article. The name of the technology is subject to change with future iterations following development and trademarking.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
