Abstract
Background:
Psychological distress is highly prevalent among patients with metastatic colorectal cancer.
Aims:
To perform an economic evaluation of a combined screening and treatment program targeting psychological distress in patients with metastatic colorectal cancer in comparison with usual care.
Design:
Societal costs were collected alongside a cluster randomized controlled trial for 48 weeks. A total of 349 participants were included.
Setting:
Participants were recruited from oncology departments at 16 participating hospitals in the Netherlands.
Methods:
Outcome measures were the Hospital Anxiety and Depression Scale and quality-adjusted life-years. Missing data were imputed using multiple imputation. Uncertainty was estimated using bootstrapping. Cost-effectiveness planes and cost-effectiveness acceptability curves were estimated to show uncertainty surrounding the cost-effectiveness estimates. Sensitivity analyses were performed to check robustness of results.
Results:
Between treatment arms, no significant differences were found in Hospital Anxiety and Depression Scale score (mean difference: –0.058; 95% confidence interval: –0.13 to 0.011), quality-adjusted life-years (mean difference: 0.042; 95% confidence interval: –0.015 to 0.099), and societal costs (mean difference: –1152; 95% confidence interval: –5058 to 2214). Cost-effectiveness acceptability curves showed that the probability of cost-effectiveness was 0.64 and 0.74 at willingness-to-pay values of €0 and €10,000 per point improvement on the Hospital Anxiety and Depression Scale, respectively. The probability that the intervention was cost-effective compared to usual care for quality-adjusted life-years was 0.64 and 0.79 at willingness-to-pay values of €0 and €20,000 per quality-adjusted life-year, respectively.
Conclusion:
The intervention is dominant over usual care, primarily due to lower costs in the intervention group. However, there were no statistically significant differences in clinical effects and the uptake of the intervention was quite low. Therefore, widespread implementation cannot be recommended.
Keywords
Distress negatively affects the healthcare system because of its association with reduced quality of life.
Routine screening for distress is recommended by major cancer organizations on the assumption that identification of elevated distress levels will result in an increased uptake of care and, consequently, reduction of distress.
Although the intervention program was dominant over usual care, it did not significantly improve distress and quality of life.
In addition, the uptake of the intervention was quite low.
Therefore, widespread implementation of the intervention program cannot be recommended based on the results of this study.
Psychosocial needs of patients in end-of-life care are often already met through support by relatives and primary caregivers.
Introduction
Colorectal cancer is the third most common cancer in males and second most common in females.1,2 More than 1.3 million patients are diagnosed with colorectal cancer worldwide each year. 3 Approximately 40%–50% of those diagnosed with colorectal cancer develop metastatic colorectal cancer and for most of them curative treatment is not possible anymore. Patients with an advanced state of disease have a high prevalence of distress.4,5 Distress negatively affects the healthcare system because of its association with reduced health-related quality of life,6,7 reduced treatment adherence, and poor prognosis.8,9 Furthermore, psychological distress among cancer patients is associated with an economic burden. A recently published paper showed, for example, that higher psychological distress in patients with cancer is associated with higher mental healthcare and primary care costs. 10
Routine screening for distress is recommended by major cancer organizations since it is assumed that identification of elevated levels of distress will result in increased uptake of psychosocial services and, as a consequence, reduction of distress.11–14 However, implementation of such programs requires the use of scarce healthcare resources. Therefore, it is important to assess not only the effectiveness but also the cost-effectiveness of screening and treatment programs in cancer patients. 15 Cost-effectiveness is an important factor in deciding whether such a program should be implemented into hospital routine care. Stepped-care approaches have been advocated as a potentially cost-effective approach to providing mental care and support. 16 Combining routine screening for distress among metastatic colorectal cancer patients with a stepped-care treatment approach could result in a more efficient way of providing psychological support to this group of patients.
An intervention program was, therefore, developed to detect and reduce distress in patients with metastatic colorectal cancer who were mainly receiving palliative care. This intervention program, the TES program, combined targeted selection (T), enhanced care (E), and referral to evidence-based interventions using a stepped-care (S)-oriented approach (hereafter referred to as intervention program). Although no evidence was found that the intervention program resulted in improved patient outcomes for distress, it showed improved patient satisfaction with care and better self-reported cognitive functioning, 17 making it important to also estimate the joint uncertainty surrounding the cost and effect differences between the treatments. 18 Therefore, the aim of this study is to assess the cost-effectiveness and cost-utility of the intervention program in comparison with usual care among patients with metastatic colorectal cancer scheduled to start palliative treatment with first-line chemotherapy from a societal perspective.
Methods
Study design
The economic evaluation was conducted from a societal perspective alongside a multi-center, cluster randomized controlled trial (RCT) comparing the intervention program with usual care among patients with metastatic colorectal cancer. 19 Effect and cost outcomes were evaluated at baseline, and after 3, 10, 24, and 48 weeks, meaning that the time horizon was 48 weeks. The trial was approved by the Medical Ethics Committee of the VU University Medical Center and is registered in the Dutch trial registry (http://www.trialregister.nl/trialreg/index.asp, identifier: NTR4034). The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Detailed information on the study design and the intervention program is provided elsewhere.17,19
Population and setting
Between July 2013 and October 2016, participants were recruited from oncology departments at 16 participating hospitals in the Netherlands. All participants provided written informed consent. Participants were eligible if (a) they had a diagnosis of metastatic colorectal cancer, (b) they were scheduled to start palliative treatment with first-line chemotherapy, and (c) they had a life expectancy of more than 3 months. Exclusion criteria for participation were (a) younger than 18 years or older than 85 years, (b) insufficient command of the Dutch language, (c) recent psychotherapy (i.e. in the past 3 months, at least one session every 2 weeks), and (d) severe psychopathology.
Randomization and sample size
This study was a cluster RCT, in which participating hospitals were used as the unit of randomization to avoid contamination between treatment groups. The randomization procedure was performed by a blinded statistician, prior to participant recruitment. Due to the nature of the intervention, oncologists, nurses, psychologists, and patients could not be blinded. However, statistical analyses were performed blinded for group status.
The overall expected effect size was 0.18. To demonstrate this effect on the Hospital Anxiety and Depression Scale (HADS), a minimum of 302 participants was needed. This was based on a longitudinal design with four follow-up measurements, a within-subject correlation of ρ = 0.30, a two-sided α ⩽ 0.05, and a β = 0.80. In a cluster RCT, individuals within a cluster (i.e. hospital) cannot be assumed to be independent. 20 Consequently, an intra-cluster correlation (ICC) of 0.005 was assumed, meaning 715 participants were needed.19,21,22 During the recruitment period, the actual proportion of participants treated for psychological distress in the intervention program group was lower than expected. The observed low uptake of the intervention program triggered the need to pursue a futility analysis, resulting in halted recruitment. 17
Intervention
Participants in hospitals assigned to the intervention program were screened for psychological distress by a trained nurse/clinical nurse specialist before the start of chemotherapy, and at 10 and 18 weeks after the start of chemotherapy using the HADS and the “Lastmeter” (Distress Thermometer and corresponding Problem List).23,24 A score of ⩾13 on the HADS or ⩾5 on the Lastmeter was seen as an indicator for psychological distress. In routine practice, a considerable number of patients scheduled for palliative treatment do not receive adequate psychological support, because psychological needs are under-recognized in clinical oncology practice.25–27 Therefore, within the intervention program, regular screening for psychological distress took place. After evaluation of the psychological distress scores, trained nurses proposed treatment in the form of stepped care to participants scoring above the thresholds. In addition, the intervention program was also offered to participants who expressed the need to talk to a professional. The steps of the intervention program consisted of (a) watchful waiting, (b) a guided self-help program via Internet or a booklet, (c) face-to-face problem-solving therapy offered by a trained nurse, and (d) referral to specialized psychosocial services and/or psychotropic medication. The rationale behind the first step, watchful waiting, is that psychological distress often decreases or disappears over time (natural course). The second and third steps in the intervention program are based on problem-solving therapy which has been shown to be effective in treating psychological distress.28–32 The last step in the intervention program is psychotherapy, medication, or referral to other services. This step is only initiated if psychological distress persists. More detailed information about the intervention program is provided elsewhere.17,19
In the control group, participants received usual care. Treatment of psychological distress in this group of participants was only initiated when psychological distress was recognized by healthcare providers. Care was not restricted in any way.
The intervention program aimed to improve usual care in several ways.17,19 These improvements include the following: formal screening at frequent intervals for distress versus ad hoc interviews if participants mention problems or when an oncologist or nurse identifies problems; enhanced care provided by a collaborative team which is coordinated by a trained nurse versus regular care provided by an oncologist and nurse; diagnostic evaluation with standardized interviews assessing distress and problem analysis by psychologists versus non-standardized interviews by oncologists and nurses; and provision of a guided self-help program, individual face-to-face counseling, medication, or planned referral to other treatments versus ad hoc support, advice, and referral to other treatments when needed.
Clinical outcomes
The clinical outcomes of this economic evaluation were the HADS and quality-adjusted life-years (QALYs). The HADS was measured at baseline, and after 3, 10, 24 weeks, and 48 weeks. 33 The HADS is a 14-item self-assessment scale (range: 0–42) for measuring psychological distress. The HADS has shown to be reliable, valid, and responsive. 34 QALYs were calculated based on health-related quality-of-life measurements at baseline, and after 3, 10, 24, and 48 weeks using the EuroQol (EQ-5D-5L). 35 The EQ-5D-5L assessed quality of life on five dimensions (mobility, self-care, activities of daily living, pain/discomfort, and depression/anxiety) with five answer levels (no problems to severe problems). The EQ-5D-5L health states were converted to utility scores using the Dutch tariff, where 0 refers to death and 1 to full health (range: –0.446 to 1, where negative utilities indicate that a health state is valued worse than death). 36 Using the area-under-the-curve method, QALYs were calculated by multiplying the amount of time a participant spent in a specific health state with the utility score associated with that health state. Transitions between health states were linearly interpolated.
Costs
Costs were collected from the societal perspective and included the following: oncological treatment costs, medication costs, other healthcare costs, informal care costs, lost productivity costs due to absenteeism from paid work, and intervention costs. Healthcare utilization outside the hospital, informal care, and absenteeism from paid work were measured using an adapted version of the Trimbos and iMTA questionnaire for Costs Associated with Psychiatric illness (TiC-P, adapted version). 37 The adapted cost questionnaire was administered at 10, 24, and 48 weeks after baseline with a recall period of 3 months. Costs were valued using standard costs from the Dutch costing guideline. 38 Medications were valued using prices from www.medicijnkosten.nl. 38 Costs related to oncological treatment were based on treatment costs of metastatic colorectal cancer reported by Farkkila et al. 39 and converted using purchasing power parities (PPPs). 40 The shadow price of informal care is assumed to be equal to the tariff for cleaning work. Lost productivity costs due to absenteeism from work were calculated using gender-specific income values of the Dutch population and calculated according to the friction cost method. The friction cost method assumes that after a specific period of time (i.e. friction period: 85 days), the sick employee is replaced and that there are consequently no productivity losses anymore after this period. 41 All costs were expressed in euros for the year 2016 using consumer price indices. 42 Discounting was not necessary because follow-up was shorter than 52 weeks.
Intervention program costs were estimated using a bottom-up approach. Resources involved in the implementation of the intervention program were based on a logbook, which was prospectively completed by the research nurses. The following costs were included: (a) costs of development of the intervention program, (b) costs related to training the nurses and psychologists, and (c) costs related to resources used in the implementation of the intervention program for each participant. Costs of development of the intervention program included the costs of printing booklets and building a website for the guided self-help program. Website costs were based on a similar web-based intervention 43 and converted using PPP. 40 Costs for training of nurses and psychologists were based on their hourly wage. Costs related to resources used in stepped care for each participant were based on the time a participant spent with a health professional (i.e. screening time of 5 min with trained nurse), a face-to-face problem-solving therapy session (i.e. 30 min with a trained nurse), and referral to specialized psychosocial services and/or psychotropic medication (i.e. standard price of a consultation with a psychologist or psychiatrist from the Dutch costing guideline). A detailed table with all prices that were used to value costs is provided in Supplemental Appendix S1.
Statistical analyses
Cost-effectiveness and cost-utility analyses were performed according to the intention-to-treat principle. Missing cost-and-effect data were replaced using multiple imputation with chained equations (MICE). 44 Cost and effect data were assumed to be “Missing At Random.” 45 The imputation model included variables that differed at baseline, were related to missing data, or were associated with the outcome. To deal with the skewed distribution of cost data, predictive mean matching (PMM) was used in MICE. 46 The number of imputed datasets was increased until the loss of efficiency was less than 5%, resulting in 20 imputed datasets. 46 Each of the imputed datasets was analyzed separately as described below. Results from the multiple datasets were pooled using Rubin’s rules. 47
Multilevel regression models were used to estimate differences in costs and effects between the treatment groups while accounting for the hierarchical structure of the data. For costs, a two-level structure was used where hospital and participants represented the first and second level, respectively. For the difference in HADS, an additional level was used to account for repeated observations (i.e. HADS scores at different time points). The multilevel regression model for the difference in HADS included three levels; that is, repeated observations (third level) are clustered within participants (second level) and participants are clustered within hospitals (first level). Thus, the analysis accounted for a longitudinal hierarchical structure. Progression of metastatic colorectal cancer was added as a covariate in the regression models to adjust for possible confounding, as was performed in the effectiveness analysis. 17 Incremental cost-effectiveness ratios (ICERs) were calculated by dividing the difference in total costs by the difference in effects. Bias-corrected bootstrapping was used to estimate 95% confidence intervals around the mean cost and effect differences (5000 replications). Statistical uncertainty surrounding ICERs was illustrated by plotting the bootstrapped cost-effect pairs on a cost-effectiveness plane (CE plane). Cost-effectiveness acceptability curves (CEACs) were also estimated, which demonstrated the probability that the intervention program is cost-effective compared to usual care for a range of different ceiling ratios (i.e. the willingness-to-pay for one point decrease in HADS or for one additional QALY). 48 In the Netherlands, the informal threshold of cost-effectiveness for medical interventions ranges between €20,000 and €80,000 per QALY gained. 49 For outcome measures such as the HADS, no formal cost-effectiveness thresholds have been determined. Analyses were performed in IBM SPSS Statistics 24® (IBM Corp., Armonk, NY, USA), StataSE 16® (StataCorp LP, CollegeStation, TX, USA), and MLwiN® version 2.36 (University of Bristol, Bristol, UK). 50
Sensitivity analysis
Four sensitivity analyses (SAs) were performed. First, the economic evaluation was performed from the healthcare perspective (SA1). Second, the Human Capital approach was used to estimate lost productivity costs (SA2). With the human capital approach, all lost productivity hours were included in the cost estimates. In the third sensitivity analysis (SA3), training costs of healthcare providers in the intervention program were added to total societal costs. In the last sensitivity analysis (SA4), the regression models in the cost-effectiveness analysis were not adjusted for possible confounding by disease progression.
Results
Population
Sixteen oncology departments consented to participate and were allocated to the intervention program (n = 8) or usual care (n = 8). In total, 393 participants met the inclusion criteria and were willing to participate. Of these 393 participants, 5 died before the start of the study, 13 were too ill to participate, and 25 withdrew their consent or could not be contacted. Thus, 349 participants were included, of whom 184 participated in the intervention program group and 165 received treatment as usual. Table 1 presents baseline characteristics of the two study groups. There was a clinically relevant difference in HADS score at baseline between the groups: the usual care group scored higher on the HADS than the intervention program group.
Baseline characteristics.
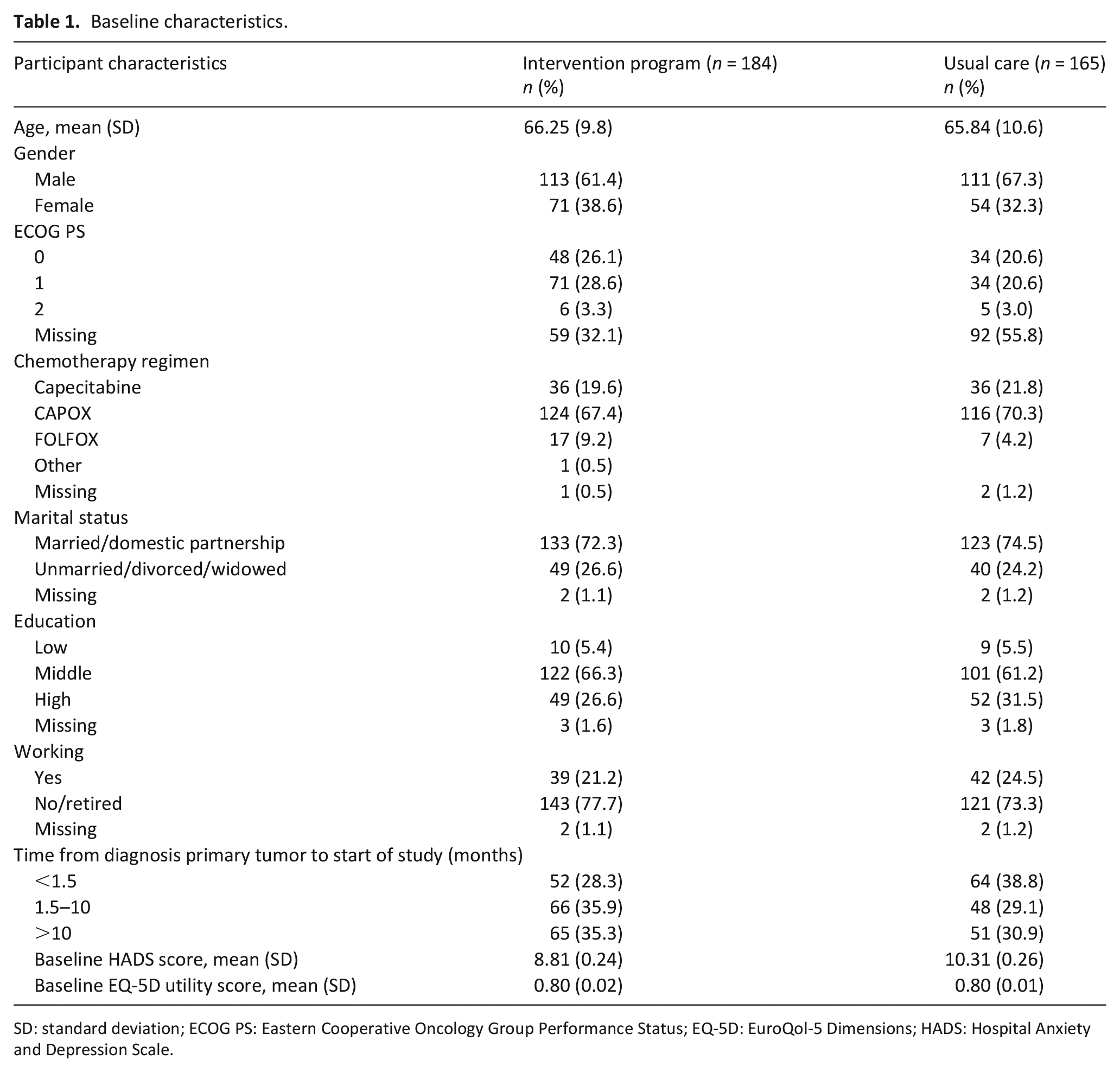
SD: standard deviation; ECOG PS: Eastern Cooperative Oncology Group Performance Status; EQ-5D: EuroQol-5 Dimensions; HADS: Hospital Anxiety and Depression Scale.
In the intervention program group, 47 participants (25.5%) died versus 51 participants (30.9%) in the usual care group. On average, participants in the intervention program group survived 1 week longer than participants in the usual care group, but this difference was not statistically significant (intervention program 43 vs usual care 42 weeks, 95% confidence interval (CI): –1.6 to 3.2). During follow-up, 110 (40%) participants in the intervention program group and 98 (41%) participants in the usual care group completed the study. Of these, 141 (40%) had missing data. Participants with incomplete data significantly differed from those with complete data regarding gender (higher rate of incomplete data for males), progression of disease (higher rate of incomplete data for participants with progression of metastatic colorectal cancer), smoking (higher rate of incomplete data for smokers), and primary location of tumor (higher rate of incomplete data for participants with left-sided tumor).
Overall, 48 participants entered stepped care after screening and a total of 62 interventions were used. Watchful waiting was used most frequently (n = 41; 66.1%). The guided self-help program was chosen two times by participants, but neither one of those two participants actually started this treatment due to disease progression. Face-to-face problem-solving therapy was given to one participant, and a referral to specialized psychosocial services and/or psychotropic medication was made 18 times.
Clinical outcomes (HADS and QALY)
The mean score on the HADS at different time points is summarized in Table 2. The overall difference in HADS over time between the intervention program and usual care was –0.058, indicating that the intervention program resulted in a decrease of 0.058 points more on the HADS than usual care; however, this difference was not statistically significant (95% CI: –0.13 to 0.011). The mean difference in QALYs between the groups was 0.042, which was also not statistically significant (95% CI: –0.015 to 0.10).
Multiple imputed effects and costs for the intervention program group (n = 184) and usual care group (n = 110) after 48 weeks.
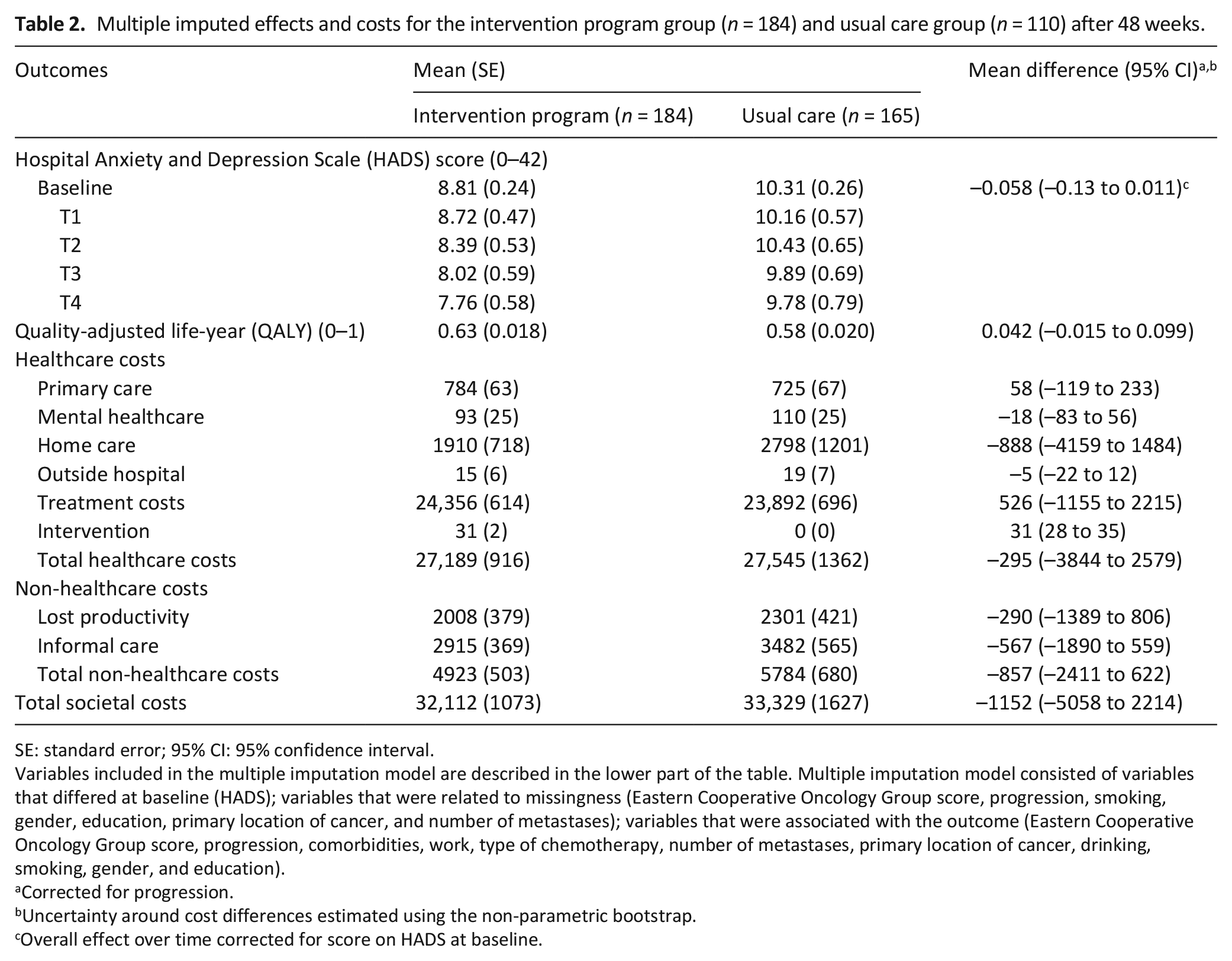
SE: standard error; 95% CI: 95% confidence interval.
Variables included in the multiple imputation model are described in the lower part of the table. Multiple imputation model consisted of variables that differed at baseline (HADS); variables that were related to missingness (Eastern Cooperative Oncology Group score, progression, smoking, gender, education, primary location of cancer, and number of metastases); variables that were associated with the outcome (Eastern Cooperative Oncology Group score, progression, comorbidities, work, type of chemotherapy, number of metastases, primary location of cancer, drinking, smoking, gender, and education).
Corrected for progression.
Uncertainty around cost differences estimated using the non-parametric bootstrap.
Overall effect over time corrected for score on HADS at baseline.
Costs
The mean total intervention costs amounted to €31 per participant (see Table 2). There were no statistically significant differences in total healthcare costs per participant between the intervention program group and the usual care group (–€295; 95% CI: –3844 to 2579), non-healthcare costs (–€857; 95% CI: –2427 to 615), or total societal costs (–€1152; 95% CI: –5058 to 2214). The main contributor to total societal costs was the metastatic colorectal cancer treatment costs (76% vs 72% of total societal costs).
Cost-effectiveness and cost-utility
For the HADS, the ICER was €19,991 per point of improvement on the HADS (see Table 3), indicating that improvement of one point on the HADS scale was associated with a saving of €19,991 in the intervention program group as compared to the usual care group. Thus, the intervention program was dominant over usual care. The CEAC (Figure 1(b)) shows that the probability of cost-effectiveness at ceiling ratios of €0, €20,000, and €80,000 per point improvement on the HADS and the probability that the intervention program is cost-effective compared to usual care were 64%, 82%, and 95%, respectively.
Results of the cost-effectiveness analysis, cost-utility analysis, and sensitivity analyses.
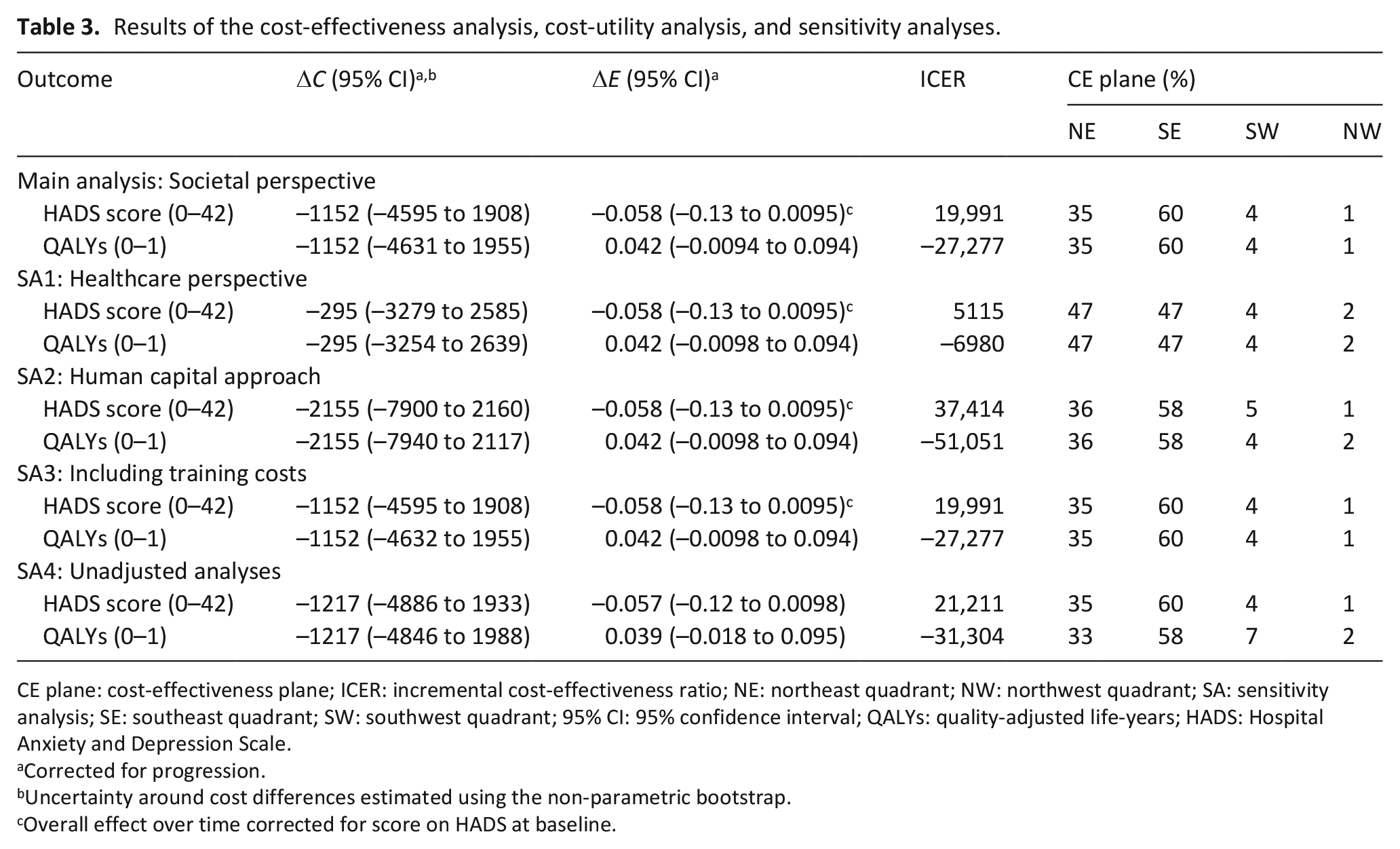
CE plane: cost-effectiveness plane; ICER: incremental cost-effectiveness ratio; NE: northeast quadrant; NW: northwest quadrant; SA: sensitivity analysis; SE: southeast quadrant; SW: southwest quadrant; 95% CI: 95% confidence interval; QALYs: quality-adjusted life-years; HADS: Hospital Anxiety and Depression Scale.
Corrected for progression.
Uncertainty around cost differences estimated using the non-parametric bootstrap.
Overall effect over time corrected for score on HADS at baseline.
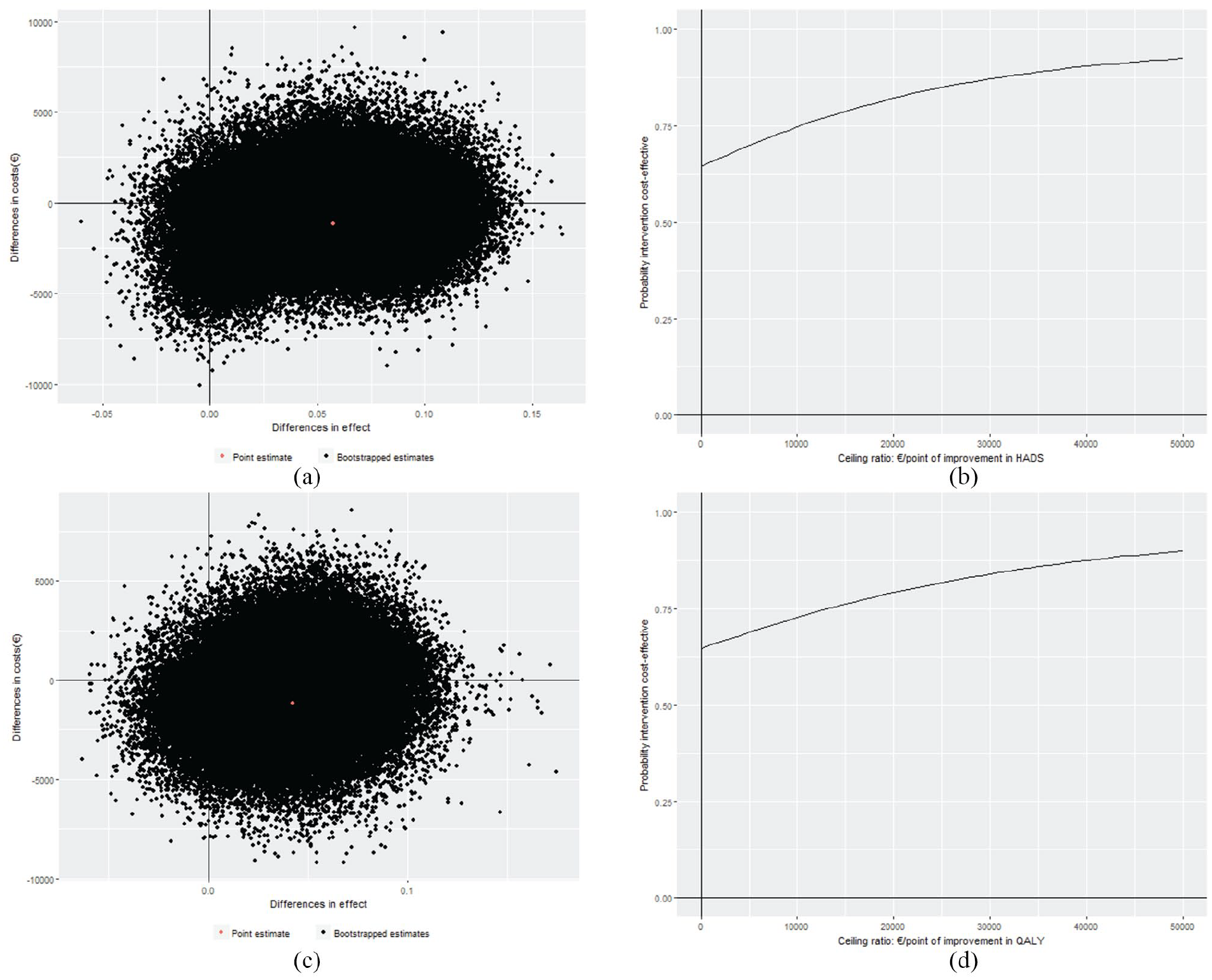
Cost-effectiveness planes and cost-effectiveness acceptability curves: (a) cost-effectiveness plane for difference in HADS, (b) cost-effectiveness acceptability curve for difference in HADS, (c) cost-effectiveness plane for difference in QALY, and (d) cost-effectiveness acceptability curve for difference in QALY.
The cost-utility analysis (CUA) resulted in an ICER of €–27,277 per QALY, meaning that to gain one QALY, €27,277 is saved in the intervention program group as compared to the usual care group, again indicating dominance of the intervention program over usual care. The CEAC (Figure 1(d)) shows probabilities that the intervention program is cost-effective compared to usual care of 64%, 79%, and 94% at ceiling ratios of €0, €20,000, and €80,000 per QALY, respectively.
SA
From the healthcare perspective (SA1) as compared with the main analysis, a smaller total cost difference was found. At ceiling ratios of €0 per additional unit of effect, the probability that the intervention program is cost-effective compared to usual care was 51% both for the HADS and QALYs. This increased to 75% and 71% for a ceiling ratio of €20,000 per additional unit of effect for the HADS and QALYs, respectively.
In comparison with the main analysis, using the Human Capital Approach to calculate lost productivity costs (SA2) resulted in larger cost savings for the intervention program as compared to usual care. However, the difference in societal costs between the groups was not statistically significant (€–2155; 95% CI: –7900 to 2160). The CEACs show that the probability of cost-effectiveness was 63% for a ceiling ratio of €0 per additional unit of effect. At a ceiling ratio of €20,000 per additional unit of effect, the probability of cost-effectiveness increased to 74% and 79% for HADS and QALY, respectively.
Adding training costs of healthcare providers to total societal costs (SA3) did not change the cost difference substantially as compared to the main analysis (€–1152; 95% CI: –4595 to 1908). Probabilities of cost-effectiveness were similar to the primary analysis. This was also the case for the unadjusted analyses (i.e. without adjustment for covariates).
Discussion
Main findings
This economic evaluation showed that there were no statistically significant differences in clinical outcomes (HADS and QALYs) or societal costs between the intervention program and usual care. At a ceiling ratio of €0 per additional unit of effect, the probability of the intervention program being cost-effective compared to usual care was 64% and this increased to 82% and 79% at a ceiling ratio of €20,000 per additional unit of effect for the HADS and QALY, respectively. Although the intervention program showed dominance over usual care, with relatively high probabilities of cost-effectiveness at the Dutch threshold of €20,000 per QALY, this was mainly driven by lower costs (in particular, lower costs of home care), whereas there were no clinically relevant differences in distress scores between groups. Results from the SA showed similar cost-effectiveness results as the main analysis. The exact numbers in this article are slightly different than in the accompanying paper presenting the results of the effectiveness analysis. However, the conclusion was not affected, that is, there was no difference in improvement in HADS score between the study arms. 17 This difference is caused by the use of multiple imputation to handle missing data in the economic evaluation.
Strengths and limitations
An important strength of this study was the pragmatic design which increases the generalizability of the results. Second, a societal perspective was used, meaning that all relevant costs were collected regardless of who payed for them. This makes it possible to identify shifting of costs between sectors. Third, different levels of hierarchy were accounted for in the analysis of costs and effects, namely participants (level 2) nested within different hospitals (level 1). 20 For effects, an additional level was added that took into account the multiple, correlated HADS measurements within participants over time (repeated measures of HADS; level 3). 51 However, there are also some limitations. First, 40% of data were missing which was accounted for by using multiple imputation; this is the preferred method to deal with missing data in economic evaluations. 52 Second, a relatively short time horizon was used (i.e. 48 weeks). However, due to the severe nature of the disease, severe deterioration or death within this period is likely. Thus, this time horizon may be long enough. Third, oncological treatment and website costs were not measured during the trial, but were calculated using data from other studies.39,43 To make these cost estimates comparable between countries, costs were converted using PPPs. 40 Finally, although the EQ-5D is the preferred instrument to estimate utility scores, it may not capture all important aspects of palliative care.53,54 Therefore, to capture psychological distress among this group of terminally ill participants, the HADS was included as an outcome measure as well.23,55
Interpretation of findings and comparison with the literature
A systematic review concluded that screening for distress among cancer patients in itself does not improve psychological distress, but that screening should be combined with pharmacological or behavioral interventions to improve psychological distress. 56 The intervention program combined both of these elements, but found no difference in improvement in HADS score over time between palliative care patients receiving the intervention program and patients receiving usual care. The absence of effect is most likely due to the much lower than expected uptake of the intervention program; only a minority of participants in the intervention program group did actually receive treatment for psychological distress. 17 This low uptake is, however, in line with recent findings in other studies.57–59 Low uptake may be related to the fact that palliative care patients’ psychosocial needs are already being met through support from relatives and primary caregivers (i.e. doctors and nurses). 60 One study found that most cancer patients do not need special psychosocial counseling other than discussing their emotional issues with their caregiver (i.e. doctor) and family. 61 Other potential explanations of the low uptake of the intervention could be the stigma associated with psychosocial care or the stress of additional appointments for patients in the palliative care phase.62,63 Although the intervention program was found to be dominant over usual care, there is much uncertainty as reflected in the 95% CIs and the estimated probabilities of cost-effectiveness. This, in combination with the lack of a clinical effect, makes it difficult to draw a strong conclusion about the cost-effectiveness of the intervention program compared to usual care. The limited effectiveness of the intervention is in line with a study performed by Hollingworth et al. 64 who evaluated an intervention consisting of a face-to-face meeting. The meeting involved a conversation with a trained nurse during which concerns were identified and potential solutions were discussed followed by immediate actions. The intervention was found to be feasible and relatively cheap (i.e. £19). Nevertheless, the authors did not find evidence that the intervention improved physical well-being or quality of life and was not considered cost-effective compared to usual care.
The findings of this study and the aforementioned previous studies should not be construed as evidence that psychological treatment of distress is not effective. On the contrary, a systematic review found that psychological treatment for patients with cancer effectively reduces psychological distress. 65 Moreover, a recent study concluded that a stepped-care procedure similar to the intervention program was effective in reducing distress and highly likely to be cost-effective in comparison with usual care in patients with head, neck, and lung cancer.66,67 Thus, psychological treatment of distress can be effective, but how to identify palliative care patients in need of effective psychological treatment is an important question. The findings of this study indicate that screening and subsequently offering treatment to distressed patients in the palliative care phase is not more effective than selection of patients for psychological treatment by clinicians (case finding).
The number of QALYs gained in the intervention program group as compared to the usual care group was smaller than minimally important differences for QALYs reported in the literature68,69 and were not statistically significant. This minor gain in QALYs is driven by an insignificant increase in both the length of life and quality of life in the intervention program group as compared to the usual care group. The modest positive effect on quality of life of the intervention program is in line with the clinical analyses that showed significantly improved participant satisfaction with care and better self-reported cognitive functioning, which was attributed to the enhanced conversation between participants and healthcare providers about the participants’ emotional concerns in the intervention program group. 17 Both distress and quality of life have been shown to improve after discussing emotional issues with primary caregivers,70–72 regardless of their primary distress scores.
Due to the skewed distribution of costs, economic evaluations alongside clinical trials are generally underpowered which is reflected in wide confidence intervals. 73 In this study, total societal costs in the intervention program group were €1152 lower than in the usual care group. This difference was small as compared to the mean costs in the groups (€32,112 and €33,329) and not statistically significant. However, there were some striking differences in costs. On one hand, our results showed that metastatic colorectal cancer treatment costs in the intervention program group were non-significantly higher than in the usual care group, which is related to the non-significant increase in survival of participants in this group. On the other hand, home care and informal care costs were substantially lower in the intervention program group as compared to the usual care group, although not statistically significant possibly due to the low power of the study for costs. Healthcare costs for patients at the end of their life, just before death, are often higher than for patients at another stage in life. This is not explicitly attributable to the costs of dying, but to the costs of care provided to patients during their last phase, 74 especially in terms of nursing care and informal care. It is unsure, however, whether this observed difference in home care and informal care costs can be explained by this or that this is a chance finding.
Conclusion
Based on the results of this study, the intervention program is considered dominant over usual care, which was driven by lower costs in the intervention program group. However, there was considerable uncertainty surrounding the cost-effectiveness estimates and there were no statistically significant differences in distress between the groups. In addition, the uptake of the intervention was quite low. Therefore, widespread implementation cannot be recommended based on this study.
Supplemental Material
Appendix_1_Cost_categories_with_unit_prices_ – Supplemental material for Economic evaluation of a combined screening and stepped-care treatment program targeting psychological distress in patients with metastatic colorectal cancer: A cluster randomized controlled trial
Supplemental material, Appendix_1_Cost_categories_with_unit_prices_ for Economic evaluation of a combined screening and stepped-care treatment program targeting psychological distress in patients with metastatic colorectal cancer: A cluster randomized controlled trial by Mohamed El Alili, Claudia S.E.W Schuurhuizen, Annemarie M.J. Braamse, Aartjan T.F. Beekman, Mecheline H. van der Linden, Inge R. Konings, Joost Dekker and Judith E. Bosmans in Palliative Medicine
Footnotes
Author contributions
All authors declare that they (1) made a substantial contribution to the concept of design of the work, or acquisition, analysis, or interpretation of data; (2) drafted the article or revised it critically for important intellectual content; (3) approved the version to be published; and (4) participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Alpe d’HuZes Foundation/KWF Kankerbestrijding (VU 2011-5279).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
