Abstract
Background:
Evaluations of complex interventions compared to usual care provided in palliative care are increasing. Not describing usual care may affect the interpretation of an intervention’s effectiveness, yet how it can be described remains unclear.
Aim:
To demonstrate the feasibility of using multi-methods to describe usual care provided in randomised controlled trials (RCTs) of complex interventions, shown within a feasibility cluster RCT.
Design:
Multi-method approach comprising usual care questionnaires, baseline case note review and focus groups with ward staff completed at study end. Thematic analysis of qualitative data, descriptive statistics of quantitative data, followed by methodological triangulation to appraise approach in relation to study aim.
Setting/participants:
Four general medical wards chosen from UK hospitals. Purposive sampling of healthcare professionals for usual care questionnaires, and focus groups. Review of 20 patients’ notes from each ward who died during admission or within 100 days of discharge.
Results:
Twenty-three usual care questionnaires at baseline, two focus groups comprising 20 healthcare professionals and 80 case note reviews. Triangulation of findings resulted in understanding the usual care provided to the targeted population in terms of context, structures, processes and outcomes for patients, families and healthcare professionals. Usual care was described, highlighting (1) similarities and embedded practices, (2) heterogeneity and (3) subtle changes in care during the trial within and across sites.
Conclusions:
We provide a feasible approach to defining usual care that can be practically adopted in different settings. Understanding usual care enhances the reliability of tested complex interventions, and informs research and policy priorities.
Keywords
Usual care provided to patients is rarely described in detail in randomised controlled trials (RCTs) of a complex intervention in palliative care.
To interpret the effectiveness of interventions tested within RCTs, the care provided in the comparison arm must be described.
Approaches including the use of open-ended questions and observations have been used in trials to understand care provided but lack convergent validity.
Usual care provided in an RCT was characterised using a multi-method approach at different time points and from different professional perspectives.
Similarities and variations in the care provided to patients within and across study sites and over time were identified refuting the assumption that all control participants received the same usual care.
This paper provides a method for the classification of the usual care that should be embedded within RCTs of complex interventions.
Assumptions made about the usual care delivered to patients during intervention development may not always remain valid at the testing stage.
Characterising usual care ensures that interpretation of findings of the effectiveness of the tested intervention is more valid.
To avoid incorrect interpretations of complex interventions in palliative care, usual care is best characterised using a multi-method approach embedded within the design of RCTs.
Introduction
Towards the end of life, patients and their families have complex needs. In recent years, palliative care interventions have been developed to improve care.1,2 While essential in all aspects of care, person-centred care, promoting autonomy and choice, is vital in palliative care provision. 3 However, palliative care practices and end-of-life care policies vary across care settings, and different patient populations.4,5 Complex interventions have been evaluated using randomised controlled trials (RCTs) where the tested intervention was compared to ‘standard’ or ‘usual’ care.6,7 Care may also vary at different time points during a trial. Usual care is the care the targeted patient population would be expected to receive as part of the normal practice and, within RCTs, refers to the care the participants who are not receiving the tested intervention receive. 8 Usual care should reflect locally adapted practices. While researchers have paid attention to clearly defining the interventions within trials, 9 scant attention has been paid to describing the characteristics of care provided to controls, and in some instances patients in the test arm in the absence of the intervention. This concern is amplified by international reporting guidelines for RCTs emphasising the importance of providing a detailed explanation of the comparison.8,10
Not detailing the care in comparison presumes all control participants receive a similar standard of care, within and across sites, and that usual care practices remain unchanged during the trial. 11 Taking part in a research study may influence the care provided within the control arm of an RCT. 12 If the usual care is incorporating the latest evidence, it may also resemble the tested complex intervention. 13 Without this information, interpreting the effectiveness of the intervention is challenging. 14 Providing clear descriptions of the intervention and usual care is critical for understanding the fidelity of implementation and delivery of an intervention tested across settings. 11 Defining usual care may also provide valuable information for further development of the complex intervention and inform its scalability to other settings, by benchmarking and identifying areas that could be improved to achieve a better quality of care. 15
Attempts have been made to describe usual care, including open-ended questionnaires to gauge health professionals’ understandings of care provided to patients.9,14,16 Open-ended questions offer a practical approach to understand ‘treatment-as-usual’, yet the use of a single self-report method limits understanding to practitioners’ views. Incorporating multiple data sources and triangulation across them enable exploration of different constructs of usual care.17,18 Multi-method approaches such as direct clinical observations can be time- and resource-intensive. 19 Indirect methods, including using routine data, may be appropriate, particularly when used in multi-centre, large-scale trials.
We aimed to demonstrate how a multi-methods approach can be adopted to describe the usual care provided in RCTs of complex interventions. The usual care provided prior to the implementation of the complex intervention for the chosen intervention sites and the care provided in the control arms throughout the trial are described within the feasibility cluster RCT of the AMBER care bundle. 20 This is a complex intervention aimed at providing better care to patients whose situations are ‘clinically uncertain’, with an irreversible, deteriorating condition, and at risk of dying during their hospital admission.20–23
Methods
Design
Our study is a prospective, longitudinal, multi-method study within a parallel, feasibility cluster RCT of a complex intervention (ISRCTN36040085).5,20,24,25 Data were collected between June 2017 and August 2018.
Setting
Four district general hospitals 26 in England were randomised to the intervention and control arms of the trial (Table 1). Within each of these four hospitals, one general medical ward was purposefully chosen as the study ward based on the number of deaths. The selection process of study wards at each study site is detailed elsewhere. 20 As the usual care questionnaires and case notes reviews were conducted at baseline, prior to the implementation of the intervention, the baseline data from the two intervention study wards were also included for the description of usual care.
Study sites.
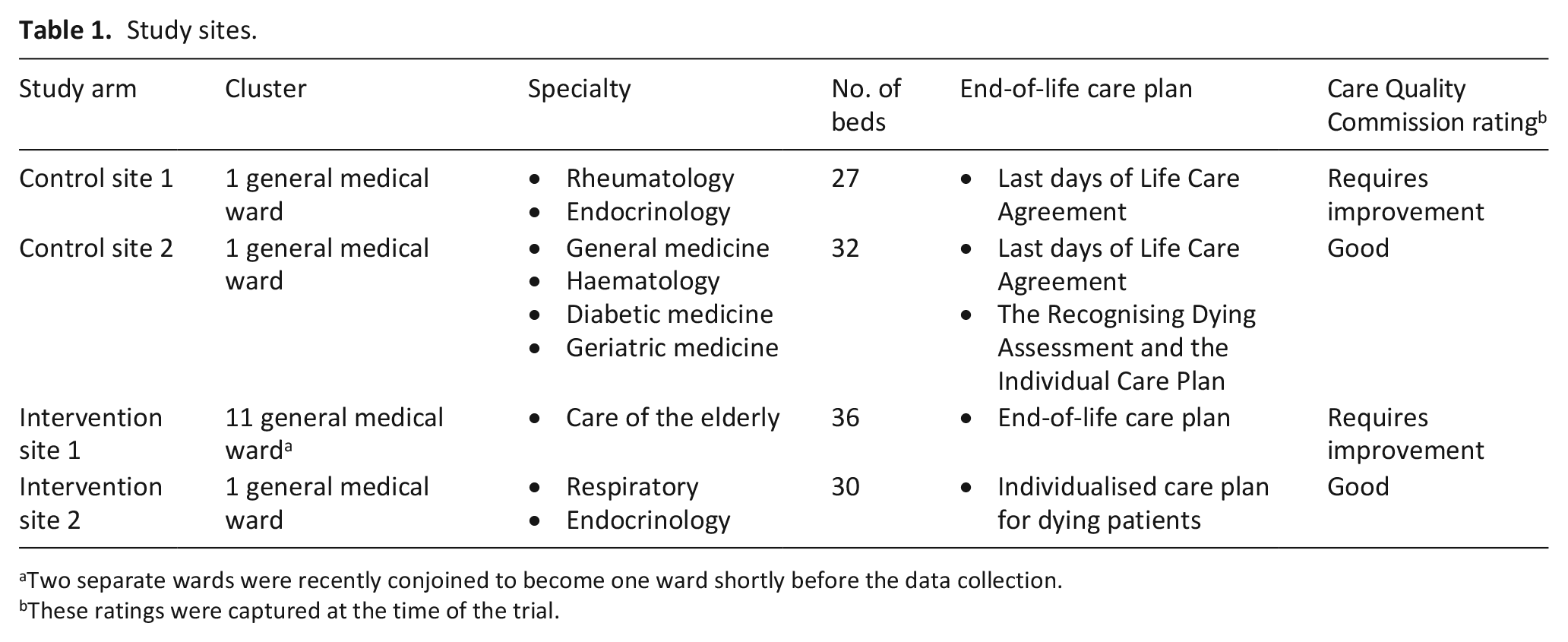
Two separate wards were recently conjoined to become one ward shortly before the data collection.
These ratings were captured at the time of the trial.
Data collection
The AMBER care bundle focuses on managing clinical uncertainty and improving communication within the multidisciplinary teams and with patients and families. 21 Exploration of usual care needed to enable comparison with this complex intervention by understanding the constructs of managing clinical uncertainty in current practice. Literature on existing methods of assessing usual care,9,11 process evaluations15,27,28 and integrative models of relations between the quality of care and health outcomes 29 was reviewed. As a result, we identified important aspects that were required to provide a clear understanding of the usual care provided across study sites. This included the context, healthcare professionals delivering the care, structures and processes in the wards and hospitals, and relevant anticipated outcomes of care for the patients, and their families. We chose methods which can be woven into the design of the cluster RCT, and the clinical context within the constraints of limited resources. We used the following data collection methods: usual care questionnaires, focus groups and case note reviews.
Usual care questionnaires
We used a self-report questionnaire to explore usual care with healthcare professionals. The questionnaire was administered in the control wards and the intervention wards at baseline only. We developed a study-specific questionnaire to document usual care across the whole trial (see Supplementary File 1). This enabled exploration of usual care aligned to key constructs of our intervention 9 and drawing on studies to inform the content and format. The questionnaire included 23 questions, mainly open-ended questions. The questions explored the structures, processes and outcomes of care, 15 including initial care planning, communication with families, recognising dying and clinical uncertainty, referrals and discharge procedures. We piloted the questionnaire at two wards of a London teaching hospital. The pilot explored the questionnaire content and format with questions subsequently re-worded for clarity. Pilot data were not included in the main data.
We purposefully sampled healthcare professionals based on their profession and seniority to aim to recruit five participants from each site with representation from across the multidisciplinary team (e.g. Medical consultant, Ward sister/manager, Junior doctor, Staff nurse, Healthcare assistant). Potential participants were identified and approached by the local research nurses working at the study sites.
Focus groups
Focus groups were undertaken in each site with clinicians after completion of data collection for the main trial outcome. The focus groups intended to explore the experiences of healthcare professionals in caring for, and communicating with, patients whose situations were clinically uncertain, and their families. We explored communicating with patients and their families about clinically uncertain situations, teamwork and practices for enhancing communication of the healthcare professionals who worked on the control sites. The topic guide was informed by studies examining the intervention acceptability and use.22,23 Eligible participants were identified and approached by the research nurses at each study sites, and a poster advertising the focus group for staff displayed on each ward. To enable participation, the focus groups were held at lunchtimes in meeting rooms on the study wards. Focus groups were led by either one of the researchers: J.K. (male) and C.J.E. (female) – both senior researchers experienced in complex interventions, palliative care and qualitative research. Field notes were taken (E.Y. and H.J.). Focus groups were audio-recorded and transcribed verbatim.
Case note reviews
The case note reviews 21 were conducted by a palliative care clinical nurse specialist at baseline in all four sites. The nurse specialist was the facilitator for the AMBER care bundle intervention. Case note reviews enabled exploration of more objective data on usual care to complement the self-report questionnaire data which may be subject to social desirability bias.30,31 The note reviews comprised 10 patients who died during admission and 10 who died within 100 days of discharge. Each case note was randomly selected from the deaths during or after the admission. This intended to include case notes for individuals with a clinically uncertain prognosis during the admission. Case notes of the deceased patients were only accessed by hospital staff with clinical responsibility for the care in the ward. De-identified anonymous information was used as research data and shared with the research team. This method of case note review was part of the AMBER care bundle ‘benchmarking’ process to understand usual care at baseline and monitor changes over time.
Analysis
Qualitative data from the usual care questionnaires and focus groups were analysed separately. For the analysis of the focus group data, thematic analysis informed by the framework approach was conducted, to inductively code, organise and identify emerging themes. 32 The first steps of the coding and analysis were performed by E.Y. (female), who is a research assistant with experience in mixed-methods. To enhance analytical rigour, the researchers (E.Y., J.K., H.J.) reviewed coding and completeness of the framework. Where coding differed, issues were reconsidered by H.J., J.K., and E.Y. until a consensus was achieved. 33 Unusual or non-confirmatory views were examined and unwarranted claims about patterns were avoided. Excerpts were used to illustrate themes.
Usual care questionnaire data were analysed by E.Y. adopting a directed content analysis approach prior to triangulation.34,35 Coding was deductive in terms of pre-determined categories of structures, processes, and outcomes, similarities, variations, and changes over time. This methodology allows flexibility for survey designs which include quantitative, open-ended and closed-ended questions. 35
Identifiable information was removed preserving confidentiality for both the focus groups and the usual care questionnaires. Qualitative data from the focus groups were managed in NVivo 11 36 and data from the usual care questionnaires were managed in SPSS. 37
We used descriptive statistics for the numerical data in the usual care questionnaire and case note review data analysed using SPSS. 37
The findings from three data sources were triangulated at the interpretation stage looking for correspondence (complementary information on the same issue), convergence (findings from different data sources agreeing), divergence (findings from different data sources contradicting each other), and silences (a theme or a finding arising from one data source and not from the others) after all data from different sources had been analysed separately.38–40 The integration of findings from more than one data sources with a different methodology to address the same phenomenon is known as ‘data triangulation’.38,40 We believed that inter-method discrepancies may lead to a better understanding of usual care and in doing so highlight the areas for potential improvement. We also considered silences to be a possibility since, while using a multi-method approach, different methods will have varying strengths about contributing to the description of the usual care.
Research governance and ethical approval
Ethical approval was obtained from the National Research Ethics Committee–Camden and King’s Cross (20 December 2016, REC Reference: 16/LO/2010) and Health Research Authority (25 January 2017). Research governance approvals were obtained from participating hospitals.
Results
Usual care questionnaires
Twenty-three healthcare professionals completed the usual care questionnaire at baseline (Table 2). We were able to purposively sample healthcare professionals across a range of professional groups and seniority. We initially aimed to recruit and obtain five questionnaires from each ward; however, one of the study wards was significantly larger than the others, as it has just been recently conjoined to an adjacent ward. Hence, we collected data from eight instead of five healthcare professionals to have better coverage of the care provided in this ward. Although, initially, the usual care questionnaires were planned to be repeated at consecutive time points, this was not feasible within the short data collection period. Collecting data at baseline from a range of healthcare professionals was feasible. Completeness of the usual care questionnaires was high with a median of 97.6% (range: 58.3%–100%) of the questionnaire completed per participant. The questions within the usual care questionnaire were deemed as completed if the participant provided an answer, or stated that they did not know the answer. The usual care questionnaire took approximately 15 min to complete.
Professions who completed the usual care questionnaire.
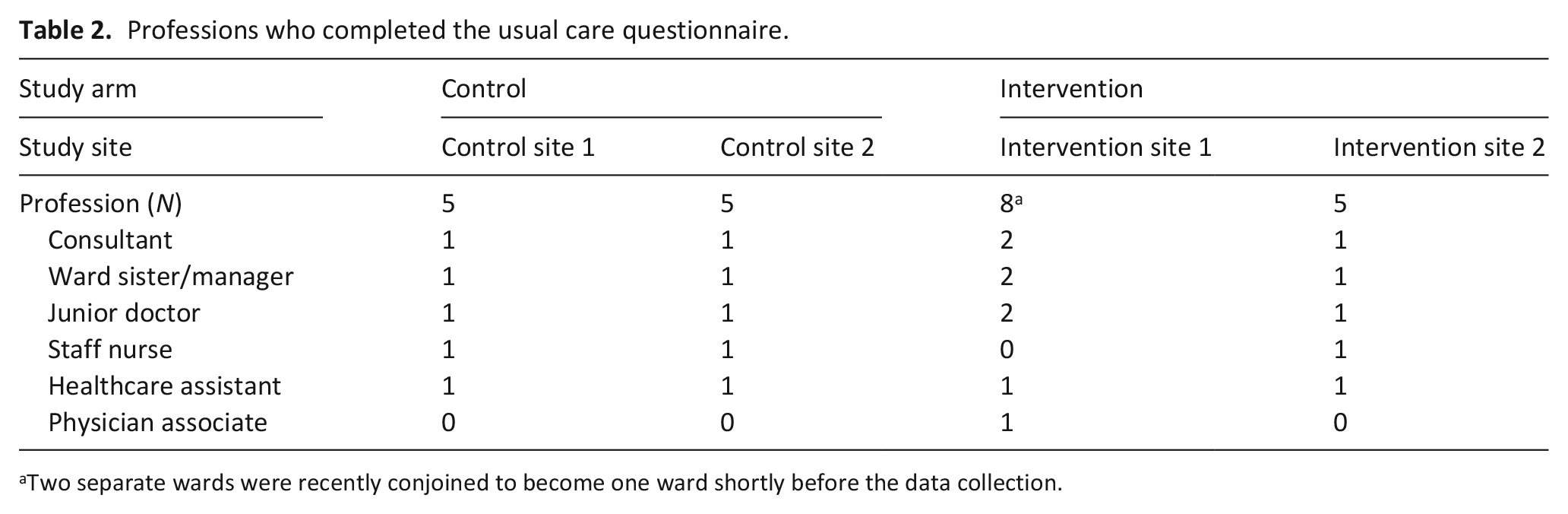
Two separate wards were recently conjoined to become one ward shortly before the data collection.
Focus groups
Two focus groups were conducted with healthcare professionals at two control site wards, attended by 20 healthcare staff (
Focus group participant characteristics.
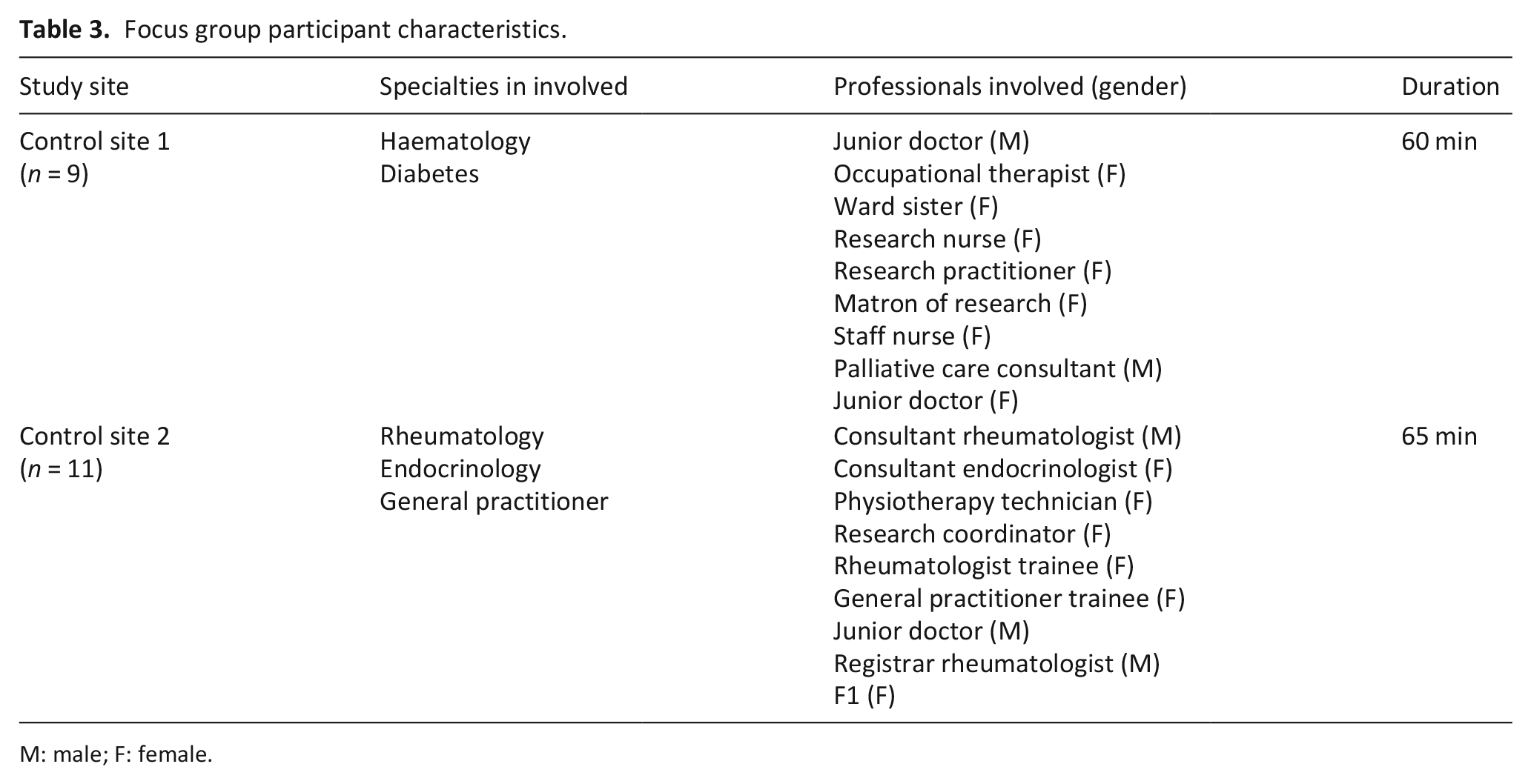
M: male; F: female.
Case note reviews
Eighty case note reviews were completed (Table 4). Most of the decedents were aged above 71 years (55%) and had a primary diagnosis of cancer or respiratory disease.
Baseline case notes review per study site and trial arm (
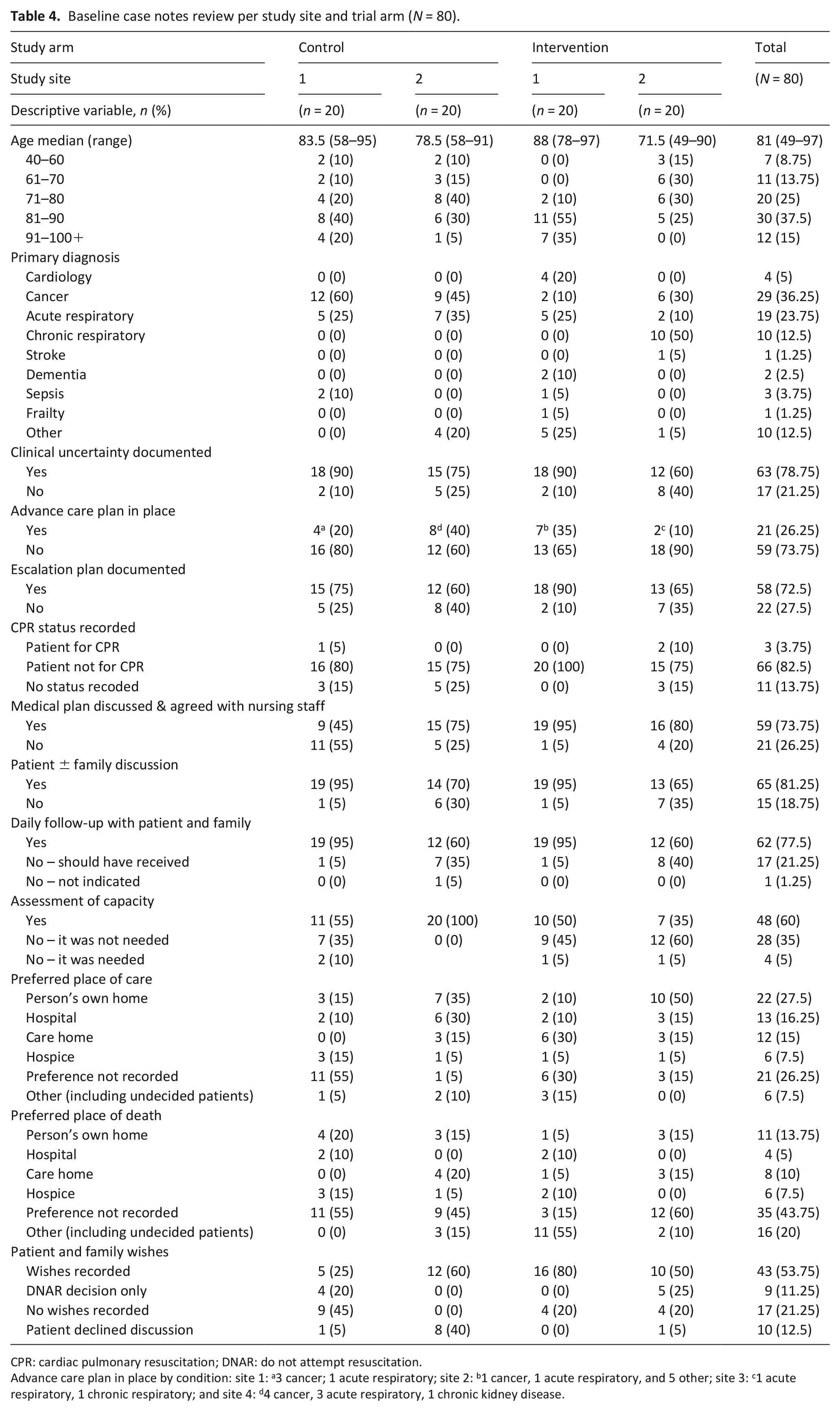
CPR: cardiac pulmonary resuscitation; DNAR: do not attempt resuscitation.
Advance care plan in place by condition: site 1: a3 cancer; 1 acute respiratory; site 2: b1 cancer, 1 acute respiratory, and 5 other; site 3: c1 acute respiratory, 1 chronic respiratory; and site 4: d4 cancer, 3 acute respiratory, 1 chronic kidney disease.
Describing usual care across the study sites
By triangulating data from the focus groups, usual care questionnaires and case note reviews on the construct of usual care, we were able to define and classify usual care for patients whose situations were clinically uncertain. The classification comprised of (1) similarities and embedded practices, (2) heterogeneity within and across study sites and (3) subtle changes in the control arm during the study (Table 5). Within this trial’s study sites, we observed similarities across the domains of admission and current care planning, communication with the patient and family, escalation decisions, recognition of the clinical uncertainty, and the emotional support provided to the staff members, whereas heterogeneity within and across the study sites was observed for documentation of deterioration and specialist involvement, advance care planning, decision-making processes and communication between ward staff, and competence and confidence of the ward staff in communication. Finally, subtle changes in the usual care were observed at the control sites, specifically relating to the changing attitudes towards referral practices to the palliative care team.
The classification of the usual care.

MDT: multidisciplinary team; GP: general practitioner; ITU: intensive therapy unit.
Discussion
We demonstrated how a multi-method approach can successfully be adopted within the financial and time constraints of a trial to describe comprehensively the usual care provided to the patient population targeted by the complex intervention. While widely used statements such as CONSORT 10 and TIDieR 8 call for a description of the care provided in the control group, no guidance is provided for how this information should be obtained, specifically for RCTs of complex, common in palliative and end-of-life care. 9 Building on from the literature and using a multi-method approach, we identified embedded practices and variability in care provided to patients across four sites within a multi-centre RCT, highlighting the importance of reliably collecting information on the quality of care, rather than assuming a similar standard of care. We also identified subtle changes in clinical practices of staff in the control arm from baseline onwards. While in the exemplified trial, changes in the control arm were small, in larger trials, understanding and monitoring for potential changes in the usual care practices hold an imperative value for the complex intervention’s development and implementation.
Within the context of this trial, 20 where the complex intervention was designed to serve patients with a terminal diagnosis, and their families, quality of care and treatments can be highly variable. 4 Complex interventions tend to be subtlety modified during local adaptation, adding on to the heterogeneity of the usual care in clinical practice. 27 To optimise patient and family outcomes, care is expected to be personalised, where the patients and family members are seen as equal partners in decision-making regarding care. 41 Aspects of person-centred care, for example, coordinating and integrating care, ensuring continuity of care and multidisciplinary working, 41 rely on having embedded clinical structures and processes. This requires staff across different professional groups to actively engage with them as part of their usual practice. Understanding contextual aspects of the usual care across sites, variability among healthcare professionals, and triangulation with data from patients’ notes enables researchers and intervention developers to map aspects of care expected to be uniform, and those expected to be heterogeneous. This knowledge assists in identifying the linkages between the mechanisms of a complex intervention and the intended outcomes, compared to the usual care within an RCT. As RCTs are accepted as providing the highest level of evidence, 42 defining the usual care by incorporating a multi-method approach within RCTs should represent a sensible methodological addition to this study design. This maximises the utility of findings on the processes as to how the intervention works to deliver the intended outcomes, and the requirements for use, compared to usual care.
Historically, ensuring high-quality care to individuals involved standardising the usual care, and having interventions in place to deliver treatments and care, with little flexibility. However, there is a danger that these interventions can easily turn into ‘tick-box’ exercises and, in some instances, when not combined with adequate evaluation, may lead to harm instead of benefit to patients.43,44 Increasing evidence points in the direction of a healthcare model which involves interpretation of research evidence for the delivery of person-centred care in clinical practice.45,46 Describing the usual care within RCTs intends to illuminate understanding on the context by exploring differences between settings and levels of care in clinical contexts (e.g. micro, macro and chrono-systems). 47 Understanding context is essential for embedding evidence-based change in clinical care to enhance clinical effectiveness for patients.
Our findings highlight important variability in the manner ‘usual’ care was provided within, and across study sites, having implications for both the way findings from similar RCTs of complex interventions are interpreted in terms of the comparator, and the requirement for successful implementation. 11 While findings converged on prioritising and informing patients and families about their clinical situation, we identified variance in healthcare professionals’ perceptions of their competence in communicating with patients and families when prognosis was uncertain, involvement of each ward staff member in clinical decision-making, and effective communication between ward staff regarding patient’s and family’s knowledge. These findings highlight the lack of similar usual care in the control arm. Some control participants may have been receiving usual care that was different compared to the intervention; others may have received care similar to patients in the intervention arm. Heterogeneity of care received in the control arm, if not successfully contrived with randomisation, may reduce the chance of detecting potential effects of the tested intervention. Not having a clear understanding of the usual care and how it compares to the complex intervention could lead to overinterpretation of its benefits or deprive patients, whose needs remain unmet, of a potentially beneficial intervention.
Strengths and limitations
The use of a multi-method approach enabled detailed exploration of the healthcare professionals’ perceptions on care provision, enhanced through case note reviews to provide a deeper exploration of specific clinical activities. Data triangulation enabled convergence and divergence across the data sets.
When considered in isolation, the findings from the usual care questionnaires and focus groups may be interpreted with caution due to subjectivity. Triangulation with the case note reviews overcomes this concern.
The data collection methods for characterising usual care were easy to implement within the context of a feasibility multi-centre, cluster RCT, with relatively limited resources. Capturing the important aspects of the care within the specified context is valuable, yet there were no available questionnaires which could be adapted for this study. 9 Hence, we developed the usual care questionnaire specifically for this study. While being piloted and proven to be successful in aiding in describing the usual care, this tool was not validated.
We were not able to obtain information on healthcare professionals who approached and those who refused to take part in the focus groups, or the usual care questionnaire; hence, opinions of those who participated in the study might differ from those who did not.
We are mindful of the absence of nursing representation in one of the focus groups. This limits our findings’ transferability to other care settings. We wanted to purposefully include the nurses in the focus groups, proportionate to their integral role in patients’ care. Although several nurses expressed interest and confirmed their availability beforehand, on the day of the focus group, no nurses were able to attend, due to urgent clinical commitments. This highlights the issues faced while conducting research in a real-life context. In future, studies should aim to improve nursing and allied-health professional representation by considering additional flexibility and resources in the study design to accommodate the unpredictable nature of clinical work.
We were not able to conduct direct observations of care due to logistical constraints. Direct observation of clinical practice might not always be feasible or ethically acceptable for vulnerable populations. However, studies should consider incorporating non-participant observation of care delivery to gain a better understanding of usual care, by cross-validating the quality of structure and process of care in RCTs of complex interventions. 15
Case note reviews were completed by only one clinical nurse specialist. Having a single person responsible for data extraction might introduce rater bias. Where possible, a review of patients’ notes should be carried out by two independent researchers, and report corresponding inter-rater reliability.
Conclusions
We have shown it is feasible and advantageous to use a multi-method approach to explore usual care in RCTs of complex interventions for patients nearing the end of life. We have highlighted embedded practices and knowledge, and variability in the usual care depending on healthcare professionals’ skills, patient disease groups and contextual factors. This study makes a methodological contribution to the research field by providing a practical and feasible approach for describing usual care. While there has been a growth in the number of studies that have evaluated complex interventions, to date, there has been a lack of agreement on how usual care can be defined. We successfully addressed this concern. To optimise the design of RCTs and improve evidence-based practice, future studies should adopt and develop the proposed multi-method approach in different settings. Within the context of limited funding opportunities for experimental studies, researchers conducting RCTs of complex interventions should aim to fully understand, and provide a definition of, the usual care. This would provide greater confidence in the study findings. Understanding usual care can strengthen the reliability of complex interventions tested in RCTs and accordingly set research funding and policy priorities.
Supplemental Material
Focus_Group_Topic_Guide – Supplemental material for Understanding usual care in randomised controlled trials of complex interventions: A multi-method approach
Supplemental material, Focus_Group_Topic_Guide for Understanding usual care in randomised controlled trials of complex interventions: A multi-method approach by Emel Yorganci, Catherine J Evans, Halle Johnson, Stephen Barclay, Fliss EM Murtagh, Deokhee Yi, Wei Gao, Andrew Pickles and Jonathan Koffman in Palliative Medicine
Supplemental Material
Usual_Care_Questionnaire – Supplemental material for Understanding usual care in randomised controlled trials of complex interventions: A multi-method approach
Supplemental material, Usual_Care_Questionnaire for Understanding usual care in randomised controlled trials of complex interventions: A multi-method approach by Emel Yorganci, Catherine J Evans, Halle Johnson, Stephen Barclay, Fliss EM Murtagh, Deokhee Yi, Wei Gao, Andrew Pickles and Jonathan Koffman in Palliative Medicine
Footnotes
Acknowledgements
We wish to acknowledge funding for this study from the National Institute for Health Research (NIHR) Health Technology Assessment (HTA) programme, as well as support from the research managers at the NIHR Evaluation Trials and Studies Coordinating Centre. We are very grateful to the AMBER Care Bundle design (Dr. Irene Carey, Dr. Adrian Hopper, Susanna Shouls & Charlene Davis) team who worked with us for adapting the case notes review data extraction tool. We wish to thank Linda Launchbury, the palliative care clinical nurse specialist, who completed the case notes review at all study sites and acted as the clinical facilitator for the study intervention. We wish like thank the clinical staff at each of the four hospital sites for agreeing to be involved in this study and for participating in the focus groups and completing the usual care questionnaires. We would especially like to thank the principal investigators and the clinical teams at each the four hospital sites: Henry Penn – Northwick Park Hospital, London North West University Healthcare NHS Trust; David Brooks – Chesterfield Royal Hospital, Chesterfield Royal Hospital NHS Foundation Trust; Natalie Broomhead – East Surrey Hospital, Surrey and Sussex Healthcare NHS Trust; and Carole Robinson – Tunbridge Wells Hospital, Maidstone and Tunbridge Wells NHS Trust for agreeing to be involved in this trial. We express particular thanks to the research nurses working across each of the four trial sites, who were instrumental in the recruitment and making arrangements for the palliative care clinical nurse specialist to come in and conduct the case note reviews. We also wish to thank the non-clinical staff at each of the four trial sites for their input into the administration for facilitating communication between the research team and research nurses. We wish to thank our study steering committee members Liz Sampson, Morag Farquhar, Joanne Droney and Toby Prevost for their valuable input. We are grateful to Sylvia Bailey and Colleen Ewart who provided expert patient public engagement input throughout the project.
Author contributions
J.K., C.J.E., F.E.M.M., D.Y., W.G., A.P., S.B. and E.Y. made substantial contributions to the conception and design of the study. J.K., E.Y. and C.J.E. contributed to the organisation of the conduct of the study. E.Y., C.J.E., J.K. and H.J. carried out the study (including acquisition of study data). J.K., E.Y. and H.J. contributed to analysis and interpretation of study data. J.K., E.Y., H.J. and C.J.E. drafted the paper. All authors critiqued the output for important intellectual content and read and approved the final manuscript.
Data management and sharing
Data from this study are currently not available online. Further information about the research materials could be accessed from the lead author upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This paper presents independent research funded by the National Institute for Health Research (NIHR) under the Health Technology Assessment Programme (15 October 2017). C.E.J. is funded by a HEE (Health Education England)/NIHR Senior Clinical Lectureship. The views and opinions expressed are those of the authors and do not necessarily reflect those of the NHS (National Health Service), the NIHR, MRC (Medical Research Council), NETSCC (NIHR Evaluation, Trials and Studies Coordinating Centre), the NIHR or the Department of Health. The funders had no role in study design, data collection and analysis of the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
