Abstract
Background:
In many countries, the consumption of opioid medicines is too low to meet population needs. Discussions within the Access To Opioid Medication in Europe project indicated that there may be significant differences in the perception of barriers for their adequate use, depending on the stakeholders.
Aim:
The aim of this study was to examine the perception of barriers and their impact concerning opioid medicines, comparing policy makers, healthcare professionals working in the field of pain management, palliative care or harm reduction and other stakeholders.
Design:
Data were collected using a questionnaire partially constructed from existing surveys, reviewed for content validity by four experts and pilot-tested in Latvia.
Setting/participants:
Participants of the Access to Opioid Medication in Europe national conferences were invited to complete the questionnaire. Stakeholder groups were compared using non-parametric rank-sum tests.
Results:
In total, 199 participants (54%) in seven countries completed the questionnaire. Most frequently rated major barriers included lack of financial resources and inadequate knowledge, skills and training among policy makers (55%–66%). Overall, policy makers perceived issues less often as major barriers or having major impact (29% barrier, 32% impact) compared to other stakeholders (36%–42% barrier, 39%–51% impact). Significant differences were seen on several aspects. For example, excessive regulation or bureaucracy for prescribing was rated as having major impact by 55%–57% of healthcare professionals in contrast to only 20% of the policy makers (p = 0.002).
Conclusion:
Multiple barriers may play an important role, partly depending on the perspective of the stakeholder involved. Hence, when addressing perceived barriers, it is important to include all relevant stakeholder groups. Only then, effective and widely supported solutions can be implemented.
Keywords
What is already known about the topic?
Numerous studies have reported on barriers to access, including patients’ reluctance to use opioids, inadequate staff knowledge of pain management and complicated regulations.
Most studies focused on patients, physicians or nurses; only a few studies examined the perception of barriers among policy makers.
There are no studies comparing policy makers and healthcare professionals working in the field of harm reduction, pain management and palliative care.
What this paper adds?
Most frequently rated major barriers included lack of financial resources and inadequate knowledge, skills and training among policy makers.
Issues were least often seen as a major barrier by policy makers and most often by harm reduction professionals.
Significant differences between stakeholder groups were identified on specific aspects, such as excessive regulation or bureaucracy for prescribing.
Implications for practice, theory or policy
Multiple barriers may play an important role, partly depending on the perspective of the stakeholder involved.
Potential negative consequences of control measures for healthcare professionals and patients may not always be recognized by the stakeholders who draft these policies.
To implement widely supported solutions, it is important to include all stakeholders, taking into consideration their different perceptions.
Background
Opioid analgesics are considered the cornerstone of the treatment of moderate-to-severe cancer pain. Despite their widely recognized analgesic properties, the use of these medicines has been a key topic in international debates over the past decade. Discussions have predominantly focused on the risks associated with these medicines. For example, public media and scientific literature have reported repeatedly about the increase in the number of patients with opioid dependence and opioid-induced death that was seen in the United States since 1999.1,2 This increase in the number of deaths has reached a new peak in 2017 with an estimated 72,000 drug overdose deaths, of which more than two-thirds involved opioids.3,4
To prevent illegitimate use of opioid medicines, governments and policy makers have undertaken control measures which often have a legal or regulatory foundation. This increased level of control may be justified if it aims at controlling traffic of the illicitly produced substances at the root of the problem. However, in discussions addressing the ‘opioid epidemic’, in many cases no clear distinction is made between overdoses from prescribed opioids, use of illicit opioids and illegal diversion or misuse of prescription opioids. This confusion may result in a disproportionate generalized fear of opioids, and additional control measures may, in practice, also reduce legitimate opioid use, limiting access for patients in medical need. 5
Although an increase in use of opioid analgesics has been seen in most high-income countries, on a global scale there is still a high level of inequity.6,7 Data from the International Narcotics Control Board show that 95.7% of the global consumption of opioid analgesics in 2011–2013 was consumed by only 15% of the global population. 6 Only 7.5% of the global population lives in countries where the consumption of opioid analgesics is considered adequate. 8 National drug control systems containing rules that are more strict than required by international drug control conventions are considered to contribute to this inequity, in addition to other factors, such as a lack of knowledge and education, societal attitudes and economic issues. 9 While it is beyond doubt that non-medical use and diversion should be reduced, it is paramount to balance strategies to ensure access to pharmaceutical products that are legitimately on the market for patients in medical need. 9
Identifying barriers to legitimate opioid use is a crucial first step in ensuring access. Numerous studies have reported on barriers to access, mostly focusing on one stakeholder group, such as patients, physicians or nurses. Reported barriers include patients’ reluctance to use opioids, inadequate staff knowledge of pain management, physicians’ reluctance to prescribe opioids and complicated regulations.10–20 A few studies have also examined the perception of barriers among healthcare decision and policy makers.6,19,20 Identified barriers include lack of education and training, fear of dependence or diversion, limited financial resources and problems in opioid manufacturing, storage and distribution.6,19,20 Discussions within the Access To Opioid Medication in Europe (ATOME) project suggested that considerable differences may exist in the perception of barriers between policy makers and healthcare professionals working in various medical fields, that is, pain management and harm reduction. These differences may have major impact, in practice; as national drug control policies are usually drafted and implemented by stakeholders who do not have clinical experience with these medicines, the potential negative consequences of control measures for healthcare professionals and patients in medical need may not always be recognized.
This study aimed to examine the perception of barriers concerning opioid medicines and their impact among various stakeholder groups, comparing healthcare decisions makers, healthcare professionals working in the field of pain management, palliative care or harm reduction and other stakeholders, including patient representatives.
Methods
Sample and data collection
A convenience sample was recruited consisting of participants of the ATOME national conferences in seven countries (Estonia, Hungary, Latvia, Lithuania, Poland, Serbia and Slovakia) between March 2013 (Latvia) and April 2014 (Poland). Participants of the ATOME national conferences included representatives of relevant stakeholder groups and others who might be interested in attending the conference. General guidance was provided by the ATOME team to the national organizers of the conferences on which stakeholders to invite. Potential participants were approached by e-mail using a pre-defined invitation letter. Other (non-invited) participants were recruited through announcements of the national conferences, which were made public through various sources, including the ATOME website and print materials (posters).
Invited participants included representatives from governmental bodies and organizations (e.g. the healthcare inspectorate in charge of controlled medicines and governmental agencies in charge of drug control legislation), healthcare professional organizations, consumer and patient organizations and representatives from non-governmental organizations (e.g. relevant activists or programme staff). Data were collected using a questionnaire that was handed out at conference registration; participants were asked to complete the survey before the official beginning of the conference. No reimbursement was provided for attending the ATOME national conferences or completing the questionnaire.
Instrument
The questionnaire was developed partly based on existing surveys, such as the Abstinence Orientation Scale developed by Caplehorn et al. 21 and the Barriers Questionnaire II developed by Gunnarsdottir et al.22,23 Questions from existing surveys were reformulated to avoid the use of stigmatizing language according to World Health Organization (WHO) policy guidelines 9 and to address all potential barriers which might be experienced by distinct stakeholders (e.g. healthcare professionals and policy makers) working in different fields (pain management, palliative care and harm reduction). The questionnaire comprised four sections: (1) knowledge and attitudes, (2) barriers, (3) feasibility of the questionnaire and (4) personal or professional background (see Annex 1). In this paper, we present the results of Sections 1, 2 and 4.
The knowledge and attitudes section (Section 1) consisted of a series of 19 statements relating to opioid medicines used in pain management, harm reduction and palliative care with a ‘true’, ‘false’ and ‘don’t know/uncertain’ response option. Section 2 consisted of a list of 34 potential barriers covering five areas: (1) knowledge, education and guidance, (2) regulation of opioids, (3) information/guidelines, (4) attitudes/concerns and (5) resources and access to opioid medicines. Participants were asked to indicate the degree to which each item represented a barrier in their practice or country on a scale of 0 (not a barrier) to 3 (= major barrier) with an additional ‘don’t know/uncertain’ option. An open-ended question sought information on additional barriers. Furthermore, to evaluate the extent to which participants were hindered by these barriers, they were asked to rate the level of impact that this aspect has or had in their work or practice, using a scale of 0 (no impact) to 3 (major impact) with an additional ‘don’t know/uncertain’ option. To explore possible changes and developments in each country, participants were asked whether the situation in their view had changed during the past 5 years using a filter question (yes; no; don’t know/uncertain; if yes, has the situation worsened or improved?). Section 4 consisted of a series of questions on background characteristics of the participant, including gender, age, education and occupation (see Annex 1).
Instrument validation
The questionnaire was reviewed for content validity by four experts in pain management, palliative care, harm reduction and policy (authors A.K.M-T., L.R., S.P. and W.S.). A pilot test was conducted in one country (Latvia) to evaluate the feasibility of the questionnaire using an additional set of seven questions. Small changes in wording were made based on the respondents’ feedback to the feasibility questions. As the changes were small, data from Latvia were included in the overall analysis. The questionnaire was translated into the languages of the involved countries by Nova Language Services (Barcelona, Spain) to facilitate response. Translations were back-translated into English on a random basis to verify correct translation.
Data analysis
Respondents were categorized into five different groups based on the question ‘Which group represents your current position best?’ (see Annex 1, Section 4): (1) palliative care/pain management healthcare professionals (PC/PAIN HCP), (2) harm reduction healthcare professionals (HR HCP), (3) other healthcare professionals (HCP OTHER), (4) policy makers (PM) and (5) others (OTHER). The group PM included ‘Government officers’ and ‘Politicians’ and ‘Other healthcare decision makers’. The group OTHER included the remaining answer options: ‘Other (please specify)’ and ‘Patients or patient representatives’. Although only one answer was supposed to be given, seven respondents provided multiple answers to this question. These respondents were categorized based on the following: the answer option PM prevailed in combination with other answer options. In addition, the answer options PC/PAIN HCP and HR HCP prevailed in combination with HCP OTHER as these positions were considered a specialization. A total of 10 respondents were excluded from the sample as they did not state their current position.
Data analysis included descriptive statistics (percentages and mean values) for the total sample and for each stakeholder group. In addition, for the knowledge questions (Annex 1, Section 1), χ²-tests were used for the comparison of frequencies of correct answers between different groups for each statement. The option ‘don’t know/uncertain’ was considered incorrect. For the attitude statements, no statement was considered correct or incorrect. For the barriers and their impact questions (Annex 1, Section 2), non-parametric rank-sum tests were used to compare the five groups. Data on the percentage of respondents that perceived a potential issue as a major barrier or as having major impact were presented in total and for each group separately. An average of 1.7% (range 0%–4.5% per question) of the barriers/impact values was missing. Overall, more data were missing regarding the impact questions: 1.8% impact versus 1.6% barriers questions. Missing data were assumed to be missing at random and were handled with pairwise deletion. Differences between the groups were analysed using Kruskal–Wallis tests.
Data were analysed using IBM SPSS statistics 23.0. A p-value ⩽ 0.05 was considered statistically significant. This study was approved by the Institutional Review Board of the Utrecht Institute for Pharmaceutical Sciences, Utrecht University, The Netherlands. Confidentiality and anonymity were maintained throughout the study.
Results
Demographic characteristics
Data were collected from 199 (54%) of the 366 participants of ATOME conferences in seven countries. Respondents were healthcare professionals (22%), healthcare professionals working in the field of pain management/palliative care (35%), harm reduction (11%), policy makers (19%) and other stakeholders (13%) (Table 1). The group OTHER included medical students (2%), lecturers (2%), representatives from non-governmental organizations (1%), social workers (1%), patient representatives (3%), representatives from pharmaceutical companies (1.5%), a drug monitoring expert (0.5%) and a human rights advocate (0.5%). The sample included more females (74%) than males. The majority of the respondents (32%) were between 41 and 50 years of age and the majority of the respondents (36%) had > 20 years of professional working experience (Table 1).
Characteristics of the sample (n = 199: Estonia (54%), Hungary (53%), Latvia (72%), Lithuania (52%), Poland (49%), Serbia (44%) and Slovakia (53%)).
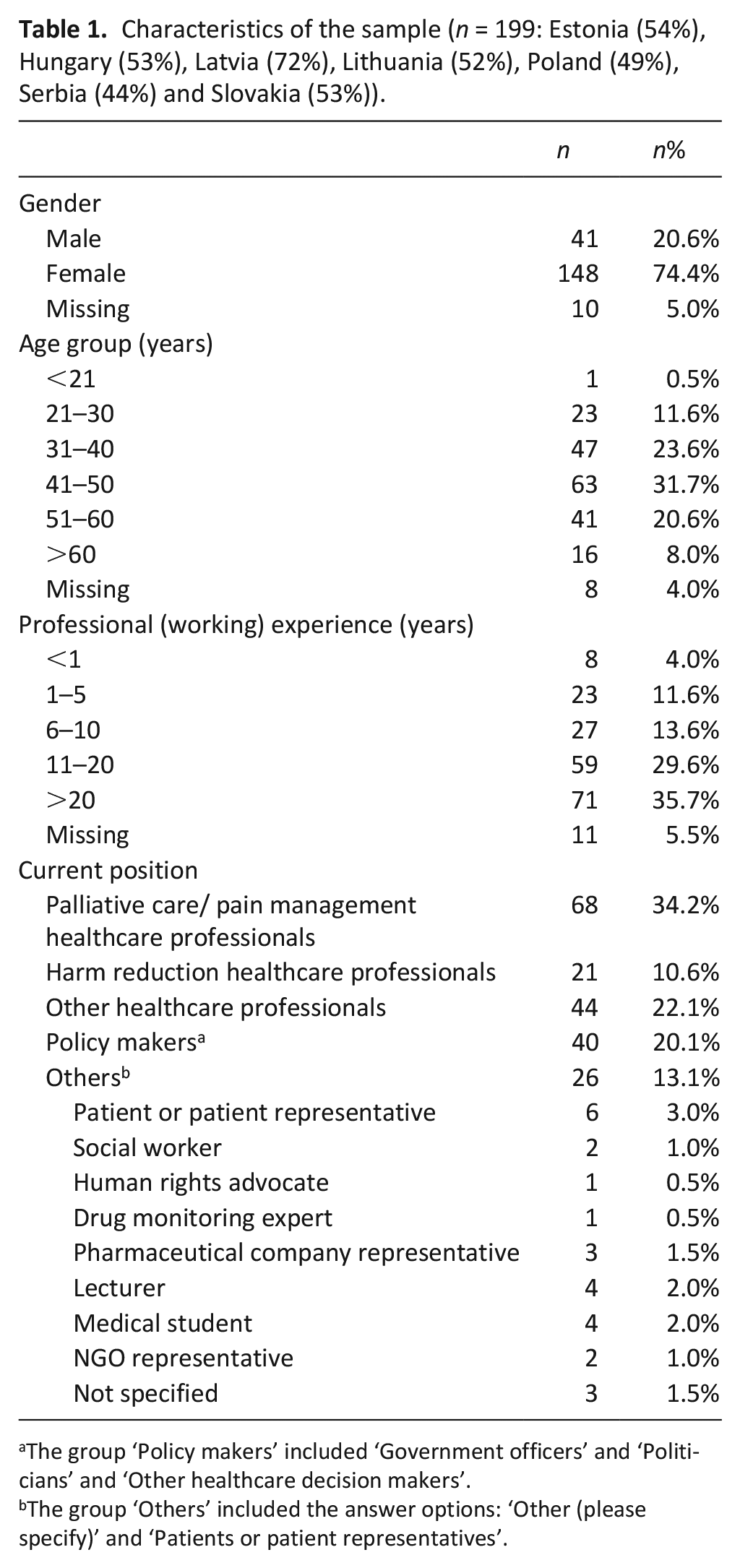
The group ‘Policy makers’ included ‘Government officers’ and ‘Politicians’ and ‘Other healthcare decision makers’.
The group ‘Others’ included the answer options: ‘Other (please specify)’ and ‘Patients or patient representatives’.
Knowledge
For the knowledge statements, only one answer was considered to be correct. In total, 56% of the statements were answered correctly (see Annex 2). The highest percentage of correct answers (72%) was given for the statement ‘When prescribed and administered according to good medical practice, opioid medicines are not likely to cause abuse (true)’, while it was lowest (35%) for ‘Intramuscular administration of opioids is painful (true)’. This statement received the highest rate of uncertainty (38%).
Comparing the stakeholder groups, overall, the highest percentage of correct answers was given by PC/PAIN HCP (74%), while it was lowest for HR HCP (50%) followed by OTHER (51%) (see Annex 2). Overall, the highest level of uncertainty in the knowledge section was found among PM (Annex 2). There were significant differences between groups in three of the nine knowledge statements: ‘Strong opioids should not be used to control pain unless a patient is dying, as these drugs will not be effective if used over a prolonged period of time’ (p = 0.002), ‘Opioids are more efficacious when given intramuscularly, compared to oral application’ (p < 0.001) and ‘Intramuscular administration of opioids is painful’ (p < 0.001), see Annex 2.
Attitudes towards opioid medicine use
In terms of awareness concerning the use of opioids in the context of pain therapy and harm reduction approaches, the highest level of cognizance was found for the statement ‘Focusing on pain therapy will lead to a neglect of curative treatment and to the end of life’, which was disagreed upon by 75% of the respondents, followed by the statement ‘In all places with large numbers of injecting drugs users, needle and syringe exchange programs should be established’ (74% Agreed) (see Annex 3).
Comparing the stakeholder groups, the most striking difference was seen for the statement ‘All patients in my country who need medical treatment with opioid medicines can have access to treatment with these medicines’; 71% of HR HCP disagreed with this statement in comparison to 23% of the group PM. The statement ‘In order to prevent abuse of opioid medicines, the daily dose or the total amount prescribed needs to be limited’ was disagreed with by the majority of PC/PAIN HCP (64%) and HR HCP (62%) in contrast to the group PM (30%). At the same time, in this latter group the highest level of uncertainty was found (23%). The statement ‘Being free of drugs (substances of abuse) is the only reasonable goal of treatment efforts for people with opioid dependence’ was disagreed upon by 76% of the HR HCP, with mixed levels of (dis)agreement among the other stakeholder groups.
Barriers towards opioid medicine use and their impact
In total, 29 potential barriers to access to opioid medicines and their impact on professional practice were rated (Section 2, Annex 1). Overall, 36% of the issues were rated as a major barrier and 43% of the issues were perceived as having major impact on professional practice (see Figure 1). The issues that overall were most frequently perceived as a major barrier or as having major impact were lack of financial resources at an institutional level (66% barrier; 70% impact) and at governmental level (63% barrier; 66% impact), followed by inadequate knowledge, skills and training regarding opioid medicines among policy makers or government representatives (55% barrier; 70% impact; see Figures 2 and 3). Overall, all issues were perceived to have more impact than they were seen as an actual barrier (Figure 1, Annex 4).
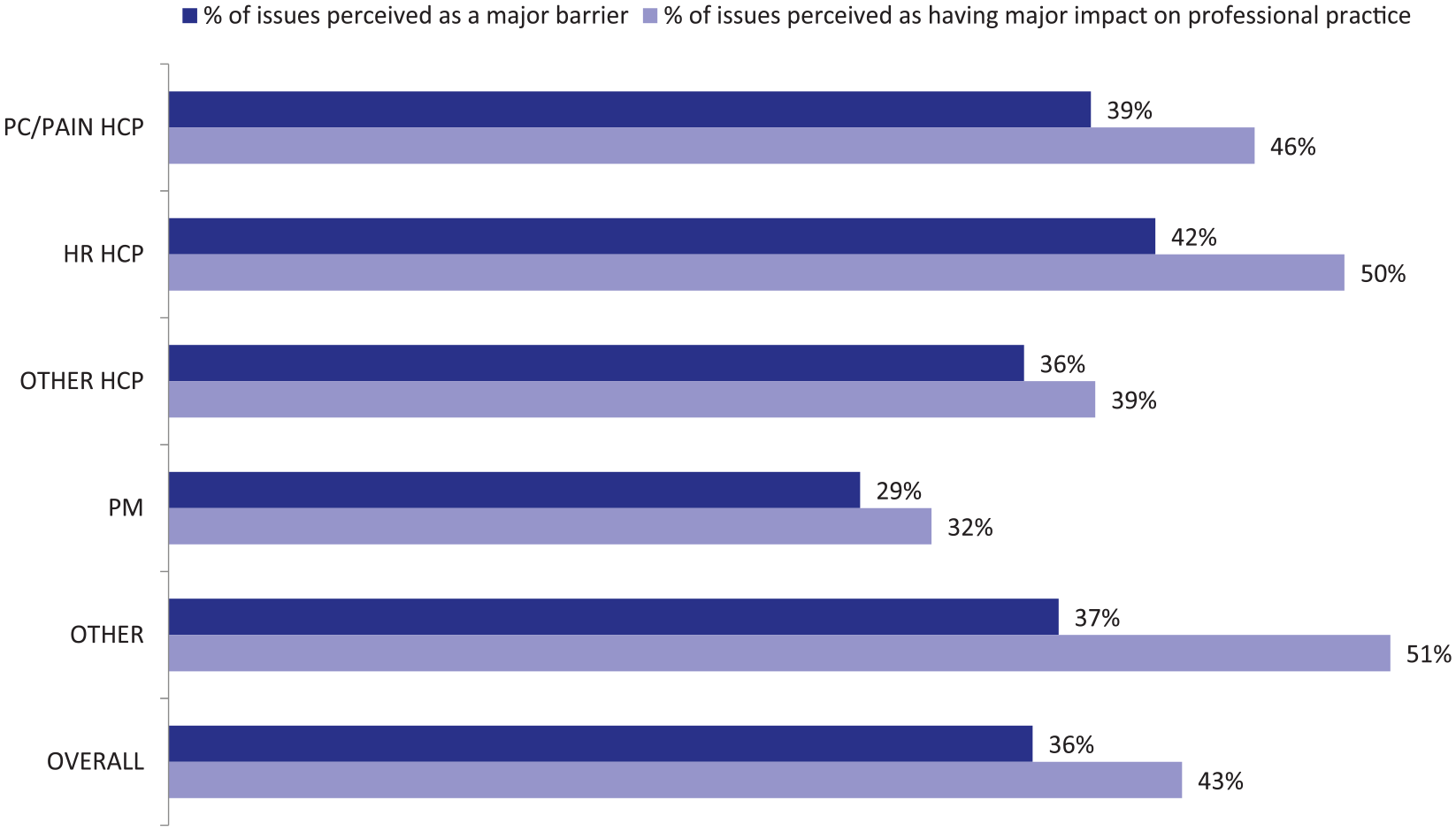
Percentage of issues perceived as a major barrier or as having major impact per stakeholder group and overall (n = 199).a
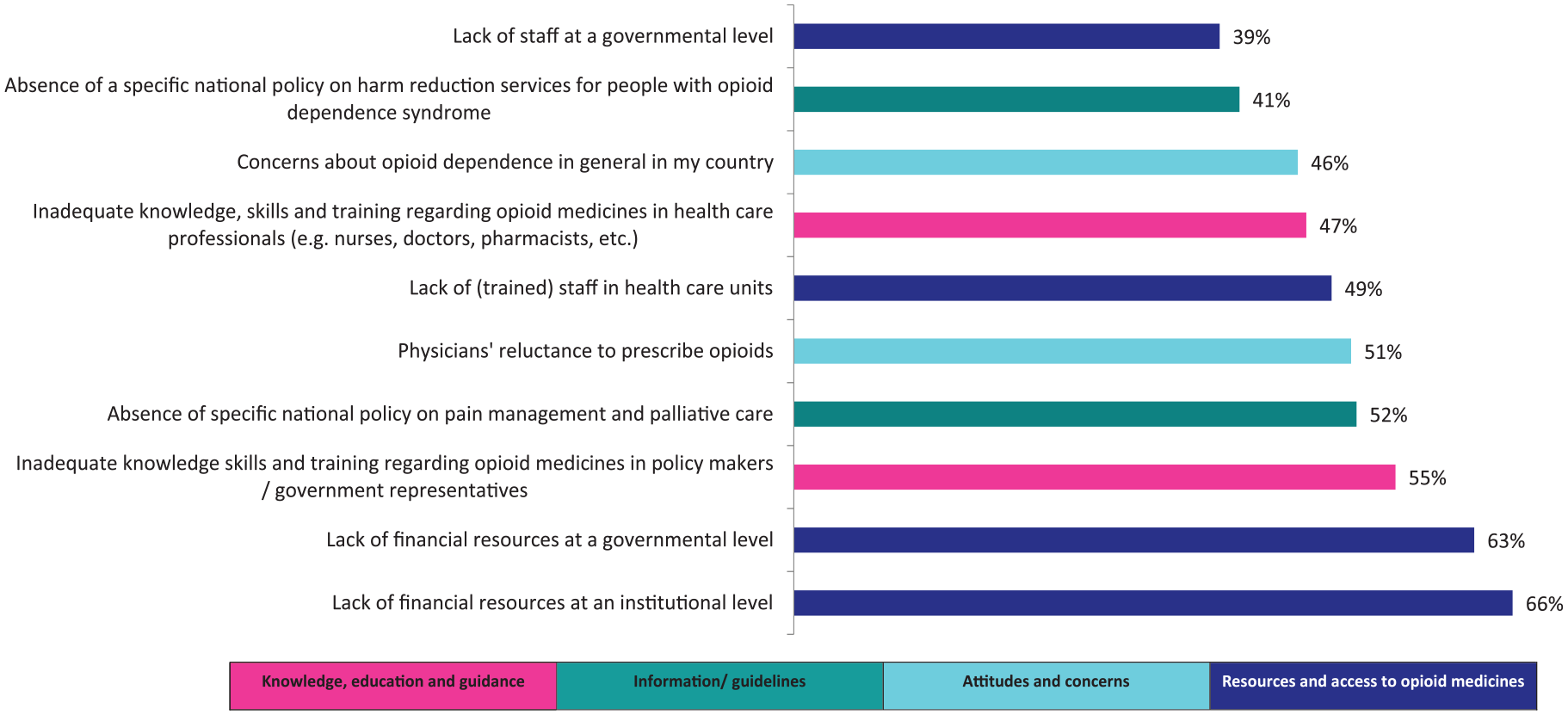
Top 10 issues perceived as major barriers (all stakeholder groups; n = 199).
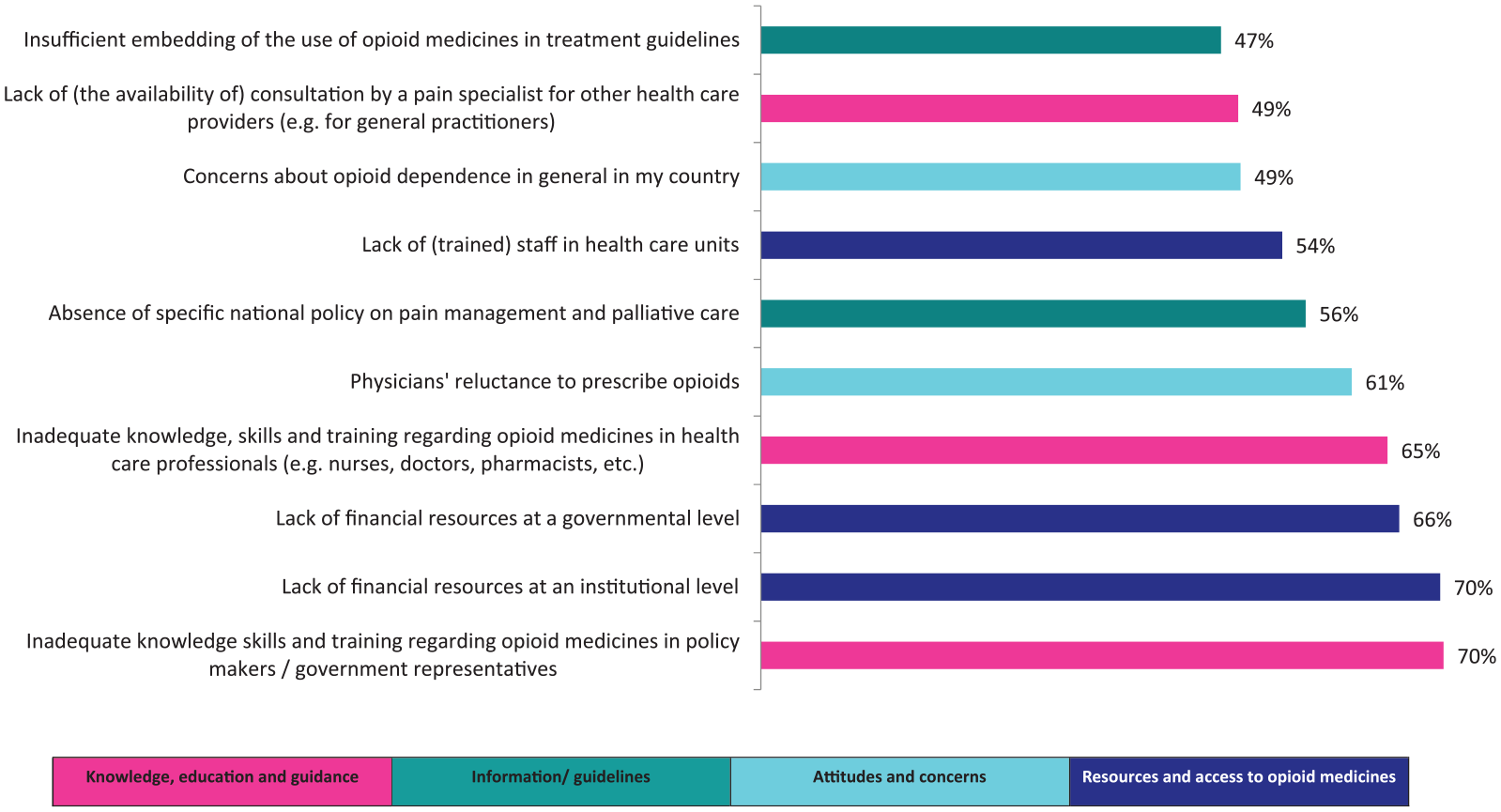
Top 10 issues perceived as having major impact on professional practice (all stakeholder groups; n = 199).
Differences in the perception of barriers between groups
Comparing stakeholder groups, issues were overall least often rated as major barriers or as having major impact by policy makers (PM: 29% barrier, 32% impact) and most often by harm reduction healthcare professionals (HR HCP: 42% barrier, 50% impact) followed by the group OTHER (37% barrier, 51% impact) (Figure 1). Significant differences were seen on specific aspects (see Table 2). For example, physicians’ reluctance to prescribe opioids was less often perceived as a major barrier or as having major impact by PM (32% barrier; 39% impact) in comparison to other stakeholders (48%–62% barrier, p = 0.0230; 57%–76% impact, p = 0.0170). PC/PAIN HCP regarded lack of (trained) staff in healthcare units frequently as a major barrier or as having major impact (65% barrier; 68% impact), while this issue was not as often perceived as a problem by the groups OTHER and HCP OTHER (31%–40% barrier, p = 0.0160; 41%–50% impact, p = 0.0080). Expressed in absolute figures, the largest difference was seen for the impact that excessive regulation or bureaucracy of opioids may have on professional practice (p = 0.020), which was rated as a major barrier by 67% of HR HCP in contrast to only 25% of PM.
Significant differences in the proportion of respondents reporting issues as major barriers or as having major impact (n = 199). A complete overview of all potential barriers (barriers + impact) for each stakeholder group is provided in Annex 2. a .
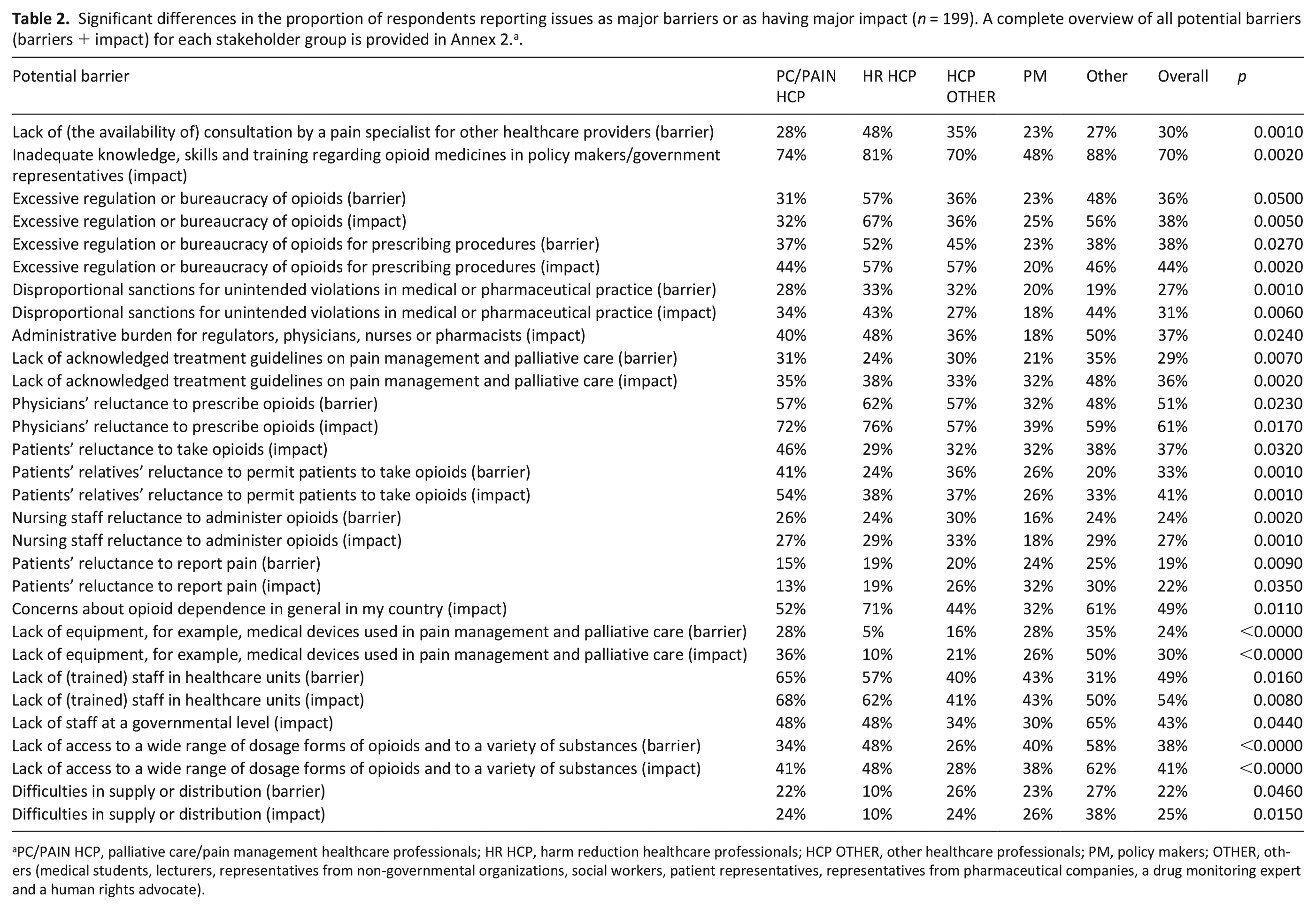
PC/PAIN HCP, palliative care/pain management healthcare professionals; HR HCP, harm reduction healthcare professionals; HCP OTHER, other healthcare professionals; PM, policy makers; OTHER, others (medical students, lecturers, representatives from non-governmental organizations, social workers, patient representatives, representatives from pharmaceutical companies, a drug monitoring expert and a human rights advocate).
Also, the top 10 issues differed between the various stakeholder groups (Annex 5). We observed, for example, that the ‘regulation of opioids’ was not reflected in the top 10 issues of the groups PM and PC/PAIN HCP. In addition, for all groups except PM, lack of financial resources was perceived most frequently as a major barrier (range: 57%–76%), but lack of knowledge, education and training in policy makers/government representatives was more frequently judged to have a major impact on professional practice (range: 70%–88%).
Additional barriers to access
In total, 10 respondents, 9 healthcare professionals and 1 non-governmental organization representative mentioned additional barriers in the open-ended questions. Examples of reported barriers include regulations on the reimbursement of medication (Poland) and the requirement of authorization by a second medical specialist when prescribing opioid analgesics for patients with chronic non-cancer pain (Hungary).
Discussion
This study shows that there are significant differences in the perception of barriers between policy makers, healthcare professionals working in the field of harm reduction, pain management and palliative care, and other stakeholders. The aspects that were most frequently perceived as a major barrier or as having major impact were lack of training, lack of financial resources and physicians’ reluctance to prescribe opioids. In addition, we found that the responses to the knowledge and attitude questions mirror familiarity with specific professional discourses in the diverse stakeholder groups. For example, the statement that being free of drugs is the only reasonable goal of treatment efforts for people with opioid dependence received a strong level of disagreement among HR HCP, reflecting long-standing advocacy work in the field of harm reduction, including awareness for the detrimental effects of a policy exclusively directed at freeness from drugs. Similarly, the high level of correct answers to the knowledge questions among PC/PAIN HCP is associated with medical best practice guidance in the field of palliative care and pain therapy.
Overall, policy makers perceived issues less often as major problems compared to other stakeholders. For example, a high proportion of healthcare professionals (44%–57%) reported that excessive regulation or bureaucracy of prescribing procedures had a major impact on accessing opioids, while this was seen as a major issue by only 20% of the policy makers. The regulatory burden perceived by healthcare professionals may be the reason for the reported reluctance to prescribe opioids. The perception of this regulatory barrier was most prominent among harm reduction professionals (76%). Overall, this latter group perceived issues most frequently as major barriers. This was also clear from the attitudes statements, with 71% of the harm reduction professionals disagreeing with the statement that ‘all patients who need medical treatment with opioid medicines can have access to treatment with these medicines’ in comparison to 23% of the policy makers. This specific finding is in line with the results of a recent analysis of national legislation and regulations that showed that patients with opioid dependence may experience specific barriers to accessing opioids in addition to those experienced by non-dependent patients. 24 In addition, the results of that legislation analysis showed that legal and regulatory documents contain language that contributes to the stigmatization of opioid dependence 24 , which may be one of the reasons why concerns about opioid dependence were frequently rated as a major barrier by harm reduction professionals in the current study.
Lack of financial resources was most frequently perceived as a major barrier, but lack of knowledge was more frequently judged to have a major impact on professional practice. Though lack of financial resources is certainly an omnipresent problem, it does not imply that national action should primarily focus on this problem. Some low- and middle-income countries, for example, have successfully attained constructive solutions to improve access to opioid medicines even with limited financial resources. 25 However, the lack of knowledge with respect to issues related to opioid use, in practice, suggests that higher levels of awareness and cognizance may contribute to sensitization, as well as better healthcare policies and service delivery in the field of pain treatment, palliative care and approaches to opioid dependence. Overall, the sample of the current study was too small to make comparisons between countries with respect to the perception of barriers in different stakeholder groups.
Only a limited number of studies have examined differences in the perception of barriers between healthcare professionals and policy makers. Srisawang et al. 19 examined barriers experienced by policy makers or regulators and healthcare professionals regarding the use of opioid medicines in Thailand. Lack of education and training in cancer pain management for healthcare professionals was regarded the main barrier among physicians, which was also noted among our palliative care/pain professionals. Moreover, Srisawang et al. 19 found that in contrast with our findings policy makers or regulators perceived all potential issues more frequently as serious problems compared to physicians. A possible explanation might be the underreporting of problems by physicians due to selection bias: participating physicians were selected by the hospital director. Another study, conducted by Leon et al. 20 in Colombia, found that insufficient human resources, deficiencies in filling out official forms, fear of expiration of medication and insufficient safety conditions for storage were frequently reported as barriers by officers from Regional Competent Authorities. The main barriers reported by physicians were complicated procedures to authorize medication by the Health Maintenance Organizations, followed by poor accessibility in hospitals or pharmacies due to limited hours for dispensing medicines. The sample size of the Colombian study was very small, and the study was not set up to compare both groups. 20 There are no other studies comparing healthcare professionals working in the field of harm reduction, pain management and palliative care, which is a strength of the current study. Physicians working in different medical specialties and government officials have been shown to experience different types of barriers and also a different magnitude of barriers. A complete understanding of the impediments to access to opioid medicines in a country or region can, therefore, not be obtained by researching only one stakeholder group.
Limitations
Several limitations of this study should be addressed, which include the sampling method used, the validity of the questionnaire and aspects inherent to survey research. First, a convenience sample was recruited for this study consisting of (mostly invited) participants of the ATOME national conferences. Participants were invited based on their interest and/or expertise in aspects that concern the usage of opioid medicines. Moreover, not all conference participants completed the survey, mostly due to organizational reasons (e.g. late arrival and registration, or distraction from questionnaire completion due to different priorities at the outset of the conference). In addition, due to the limited number of patients or patient representatives that attended the conferences and/or completed the questionnaire, this stakeholder group was included in a mixed group with other stakeholders. All aspects mentioned above limit the generalizability of our findings to the target population. Other methods of data generation and sampling strategies have been given consideration but were not regarded a suitable option due to the limited resources and workforce for this study and limited access to (invited) participants. For example, expert interviews would have yielded additional insight into the practical impact of specific barriers; however, it would have been beyond the scope of this project to realize their conduct and analysis. In terms of recruitment, agreements were made with the organizers of the conferences that people could only be contacted to participate in this study provided they had given permission to be approached, which could be obtained upon registration for the conference. To ensure that stakeholder groups were invited in a uniform matter, a guidance document was used advising on the groups of people that could be invited to participate. Second, this questionnaire is a modified version of existing questionnaires and has not been validated, which may limit the validity of our results. Finally, this study relies on self-reporting which may lead to social desirability. By assuring anonymity and confidentiality we expect to have decreased the likelihood of receiving socially desirable answers. Future research should be conducted on a larger scale in a group that is representative of the target population, using a validated questionnaire to confirm the validity of our results. In addition, qualitative interviews or focus group discussions with representatives from different stakeholder groups would allow for a more in-depth understanding of the meaning of certain barriers for people’s everyday life and professional practice.
Despite these limitations, we believe the results to be of significant value in that they provide insight into the barriers that may be encountered by stakeholders. Our results suggest that access to opioid medicines can be improved by providing more education and training for healthcare professionals and policy makers and by ensuring the availability of financial resources at different levels, such as in hospitals and other treatment facilities but also in governmental bodies. In addition, lifting legal and regulatory barriers could contribute to better access, in particular, for patients in need of harm reduction. As these data do not explain the reasons behind the differences in the perception of barriers, intensified dialogue is necessary to develop mutual understanding and effective solutions. Further qualitative studies could facilitate these discussions. The results also show the importance of involving government officials and policy makers in these discussions to increase awareness of the impact of certain legislation, regulations and policies on clinical practice.
Conclusion
This study shows that different barriers may play an important role in access to opioid medicines, depending on the stakeholders involved. When addressing these perceived barriers, intensified dialogue between all stakeholder groups is necessary to facilitate a mutual understanding and develop widely supported solutions to improve access to opioid medicines.
Supplemental Material
Suppl._material_PMJ-18-0410_R2_-_no_tracking – Supplemental material for The perception of barriers concerning opioid medicines: A survey examining differences between policy makers, healthcare professionals and other stakeholders
Supplemental material, Suppl._material_PMJ-18-0410_R2_-_no_tracking for The perception of barriers concerning opioid medicines: A survey examining differences between policy makers, healthcare professionals and other stakeholders by Marjolein JM Vranken, Lisa Linge-Dahl, Aukje K Mantel-Teeuwisse, Lukas Radbruch, Marie-Hélène DB Schutjens, Willem Scholten, Sheila Payne and Saskia Jünger in Palliative Medicine
Footnotes
Acknowledgements
The authors would like to express their gratitude to all Access To Opioid Medication in Europe (ATOME) country team members for their contribution to the ATOME project. The authors would also like to thank Prof. Dr H.G.M. Leufkens for his contribution to this study and the Institutional Review Board of the Utrecht Institute for Pharmaceutical Sciences, Utrecht University, The Netherlands for reviewing the study protocol and questionnaire. This paper was prepared on behalf of the ATOME consortium which was composed of 10 partners from the fields of palliative care (Department of Palliative Medicine, University of Bonn, Germany; Hospice Casa Sperantei, Brasov, Romania; Help the Hospices, London, UK; International Observatory on End of Life Care, Lancaster University, UK; European Association for Palliative Care, Milan, Italy); law/health policy (World Health Organization, Geneva, Switzerland; Utrecht University, Utrecht, The Netherlands) and harm reduction (Eurasian Harm reduction Network, Vilnius, Lithuania; National Anti-Drug Agency, Bucharest, Romania; Harm Reduction International, London, UK). Together, this group consisted of national, Europewide and international organizations with long-standing experience in opioid medicine issues; the 10 ATOME partners worked with the country teams, including government officials and public health and medicines experts, to carry out legislative and policy reviews, leading to recommendations that aimed to facilitate access for all patients requiring treatment with medicines controlled under international drug conventions. Core scientific group/work package leaders: Lukas Radbruch, University of Bonn/Malteser Krankenhaus Bonn/Rhein-Sieg, Germany; Willem Scholten, Consultant – Medicines and Controlled Substances, The Netherlands; Sheila Payne, Lancaster University, UK; Sergey Votyagov, Eurasian Harm Reduction Association, Lithuania; Daniela Mosoiu, Hospice Casa Sperantei, Romania; Paula Frusinoiu, National Anti-Drug Agency, Romania; David Praill, Help the Hospices, UK; Rick Lines, Harm Reduction International, UK; Marie-Hélène Schutjens, Utrecht University, The Netherlands; Sheila Payne, European Association for Palliative Care, Italy. Academic Advisory Board: Snezana Bosnjak, Institute for Oncology and Radiology, Serbia; David Clark, University of Glasgow, UK; Ambros Uchtenhagen, Institut für Sucht- und Gesundheitsforschung Zürich, Switzerland; John Lisman, Lisman Legal Life Sciences, the Netherlands; Stein Kaasa, The Norwegian University of Science and Technology (NTNU), Norway; Per Sjøgren, Rigshospitalet, Copenhagen, Denmark. Project management: Lukas Radbruch (project coordinator); Saskia Jünger (project executive officer); Willem Scholten (scientific coordinator) and Sheila Payne. The Division of Pharma-coepidemiology and Clinical Pharmacology of Utrecht University is designated as a WHO Collaborating Centre for Pharmaceutical and Regulation. The WHO Collaborating Centre for Pharma-ceutical Policy and Regulation receives no direct funding or donations from private parties, including pharma industry. Research funding from public–private partnerships, for example, IMI, TI Pharma (![]() ), is accepted under the condition that no company-specific product or company-related study is conducted. The Centre has received unrestricted research funding from public sources, for example, Netherlands Organization for Health Research and Development (ZonMW), the Dutch Healthcare Insurance Board (ZIN), European Union Seventh Framework Programme (FP7), the Dutch Medicines Evaluation Board (MEB), and the Dutch Ministry of Health, Welfare and Sport.
), is accepted under the condition that no company-specific product or company-related study is conducted. The Centre has received unrestricted research funding from public sources, for example, Netherlands Organization for Health Research and Development (ZonMW), the Dutch Healthcare Insurance Board (ZIN), European Union Seventh Framework Programme (FP7), the Dutch Medicines Evaluation Board (MEB), and the Dutch Ministry of Health, Welfare and Sport.
Authors’ contribution
All authors made a substantial contribution to the design of the study and the collection of data. L.L.-D. and M.J.M.V. analysed the data. S.J., A.K.M-T. and M-H.D.B.S. revised the analysis and participated in the interpretation of the data. M.J.M.V. drafted the manuscript, and all authors revised the manuscript critically for important intellectual content. All authors approved the version of this manuscript to be published and have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: W.S. provides consulting services as an independent consultant on regulation of and policies related to psychoactive substances. Examples of these include conducting workshops on availability of pain management, providing an overview of importation and exportation rules, providing information on controlled substance policies, the review of cannabis and the application of the International Nonproprietary Name. This has included work for, inter alia, Grünenthal and Mundipharma. M-H.D.B.S. provides legal services as an independent legal consultant for several pharmaceutical companies, including Takeda and Medice.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Union Seventh Framework Programme (FP7/2007–2013, Grant No. 222994), with the overall aim to improve the access to opioid medication in Europe.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
