Abstract
Background:
Screening for problematic opioid use is increasingly recommended in patients receiving palliative care.
Aim:
To identify tools used to assess for the presence or risk of problematic opioid use in palliative care.
Design:
Scoping review.
Data sources:
Bibliographic databases (inception to January 31, 2020), reference lists, and grey literature were searched to find primary studies reporting on adults receiving palliative care and prescription opioids to manage symptoms from advanced cancer, neurodegenerative diseases, or end-stage organ diseases; and included tools to assess for problematic opioid use. There were no restrictions based on study design, location, or language.
Results:
We identified 42 observational studies (total 14,431 participants) published between 2009 and 2020 that used questionnaires (n = 32) and urine drug tests (n = 21) to assess for problematic opioid use in palliative care, primarily in US (n = 38) and outpatient palliative care settings (n = 36). The questionnaires were Cut down, Annoyed, Guilty, and Eye-opener (CAGE, n = 8), CAGE-Adapted to Include Drugs (CAGE-AID, n = 6), Opioid Risk Tool (n = 9), Screener and Opioid Assessment for Patients with Pain (SOAPP; n = 3), SOAPP-Revised (n = 2), and SOAPP-Short Form (n = 5). Only two studies’ primary objectives were to evaluate a questionnaire’s psychometric properties in patients receiving palliative care. There was wide variation in how urine drug tests were incorporated into palliative care; frequency of abnormal urine drug test results ranged from 8.6% to 70%.
Conclusion:
Given the dearth of studies using tools developed or validated specifically for patients receiving palliative care, further research is needed to inform clinical practice and policy regarding problematic opioid use in palliative care.
Keywords
Prescription opioids are essential medications for symptom management in palliative care.
Opioids can cause serious adverse effects, including problematic opioid use, overdose and death.
In response to growing concerns about problematic opioid use, palliative care experts and organizations are recommending the use of tools to screen for problematic opioid use.
This scoping review identified 42 observational studies reporting on tools used to assess for problematic opioid use in palliative care.
The tools identified were urine drug tests and questionnaires (CAGE, CAGE-AID, ORT, SOAPP, SOAPP-SF, SOAPP-R), which reported mostly on research conducted in outpatient settings in the US.
No studies identified in this review adequately evaluated the effectiveness of these tools to assess for problematic opioid use in palliative care.
There is a lack of tools to assess for problematic opioid use that are developed or validated specifically for palliative care populations.
Research on development and validation of tools to assess for problematic opioid use in palliative care is urgently needed.
Introduction
Prescription opioids are essential medicines in palliative care because they effectively reduce pain and dyspnea. 1 There is likely no other class of drug that is as instrumental in reducing suffering at the end-of-life.2–4 Guidelines for opioid use in palliative care have traditionally emphasized proactive treatment to prevent pain, and it has long been held that addiction is both extremely rare and irrelevant for those with a short life expectancy.5–7 However, health care professionals and organizations are increasingly recognizing that a significant minority of patients receiving palliative care may demonstrate problematic opioid use, both relating to their own use and to diversion of these medications.8–11 This is particularly relevant given the ongoing international crisis of opioid-related deaths.12–16 As well, there is advocacy for and evidence supporting early involvement of palliative care in serious but not imminently life-threatening illnesses, which can lead to increased and prolonged exposure to opioids.12,13,17–19
Identification of patients who are at high risk and have problematic opioid use is an important step in measuring the extent of opioid-related harms. Tools have been developed for patients with chronic non-cancer pain, including questionnaires and urine drug tests.20–22 In response to the growing concerns about problematic opioid use, experts and organizations are recommending the use of these tools in palliative care.17,21,22 However, these tools may not be suitable for palliative care due to differences between palliative care and chronic non-cancer pain. Traditionally, all chronic pain disorders outside of pain related to cancer and end-of-life are known as “chronic non-cancer pain.” 23 By definition, chronic non-cancer pain is present for longer than 3 months 24 but most patients will experience pain for years. 25 These patients can therefore receive chronic opioid therapy and be at risk of significant opioid-related harms, including problematic opioid use,26,27 overdose, 28 and death.29,30 In contrast, patients receiving palliative care have advanced, progressive illnesses that are usually associated with worsening symptoms and functional deterioration.31,32 Opioids are titrated to balance pain control and adverse effects. 33 Opioid therapy duration and the development of opioid-related harms are naturally limited by patients’ life-expectancy. 34 Given these significant differences, knowledge about the available evidence that supports the use of these tools in palliative care is critical before widespread adoption into clinical practice. Therefore, we conducted a scoping review to answer the research question, “what tools are used to assess the presence or risk of problematic opioid use in studies conducted in the palliative care context?”
Methods
The purpose of scoping reviews is to identify and map available evidence, rather than to produce critically appraised answers to research questions. 35 We used the Arksey and O’Malley methodological framework, which was enhanced by Levac et al. and the Joanna Briggs Institute, to conduct our scoping review.36–39 Key stakeholders informed our research question and search strategy, provided input on the results and analysis, and helped formulate the key recommendations and messages. These partners were representatives from the Health Canada, Canadian Hospice Palliative Care Association (CHPCA), Canadian Society of Palliative Care Physicians (CSPCP), Canadian Virtual Hospice (CVH) and the University of Toronto Safer Opioid Prescribing continuing professional development course.40–43 The PRISMA extension for Scoping Reviews (PRISMA-Scr) was used as a guide for reporting this review. 44 Our review protocol was not registered online but is available upon reasonable request.
Study eligibility criteria
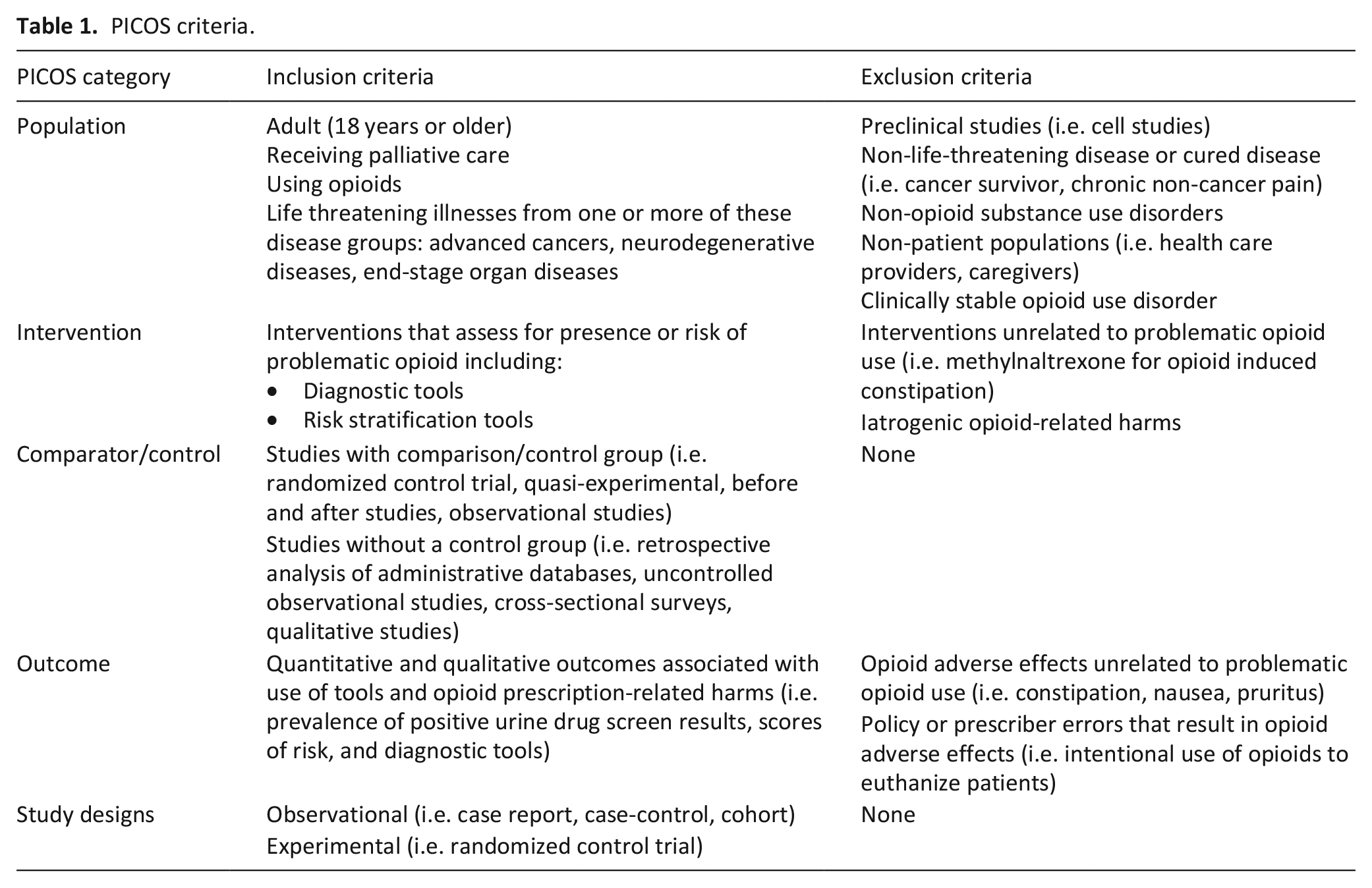
We used the Population, Intervention, Comparison/Control, Outcome, Study design (PICOS) framework to structure our eligibility criteria (Table 1). 45 We focused on patients who were receiving palliative care and were prescribed opioids for symptom management related to advanced cancers, neurodegenerative diseases, or end-stage organ failure. These three disease groups have been identified by CSPCP as needing palliative care. 46 There were no restrictions according to design, publication year, location, or language. Exclusion criteria included studies related to chronic non-cancer pain (such as from accidents or other non-life-threatening illnesses), chronic pain in survivors of adult cancers, and non-opioid substance use disorders as well as studies that used surrogate markers for opioid-related harms (e.g. alcohol use disorder).
PICOS criteria.
Search strategy
In collaboration with an information specialist and our key stakeholders, we developed a comprehensive search strategy that involved searching bibliographic databases from inception to January 31, 2020 (Supplemental Appendix A: Bibliographic databases searched and Ovid Medline® search strategy). Where available, controlled vocabulary terms and text words were used to maximize the number of potentially relevant studies identified. The search strategies were limited to human studies and adult populations (age ⩾ 18 years) where the limits were applicable. To identify additional relevant studies, we conducted hand searches of reference lists for all potentially eligible full text articles. Furthermore, we conducted a grey literature search using grey literature databases and websites of prominent health and medical organizations (Supplemental Appendix B: Grey literature search terms and sources).
Guiding definitions
The following established definitions were used to guide this review: misuse was defined as “opioid use not in accordance with prescribed directions, regardless of the presence or absence of harm resulting from use;” 47 abuse was “intentional opioid use for non-medical purposes;” 47 diversion was defined as “unlawful channeling of regulated pharmaceuticals from legal sources to the illicit marketplace;” 48 and, problematic opioid use was used to encompass the concepts of opioid misuse, abuse and diversion.47,49 Palliative care was defined as “an approach that improves the quality of life of patients and their families facing the problem[s] associated with life-threatening illness, through the prevention and relief of suffering by means of early identification and impeccable assessment and treatment of pain and other problems, physical, psychosocial, and spiritual.” 50
Data management and synthesis
The information specialist in our team oversaw the information management process. We used EndNote X8.2™ bibliographic software to store the records retrieved from all literature searches and to remove duplicate entries. 51 We used Covidence, a web-based literature review software platform, to screen all records. 52
Our PICOS criteria were applied in duplicate and independently to the titles and abstracts of the records (Table 1). For articles with no available abstracts, categorization was based on the title alone. If the two reviews conflicted, then a third review of the record was conducted to determine the final inclusion. This same screening process was applied to retrieved full texts. Eligible conference proceedings and abstracts were included in full-text review and we contacted study authors for additional information, as necessary. We expanded on the reasons for exclusion for each of the PICOS criteria and developed a hierarchy of importance for the identified reasons for exclusions (Supplemental Appendix C).
Each full text record was independently documented and coded using standardized forms in duplicate. The data extraction forms were piloted and revised throughout the review process. The following data were extracted: first author, title, publication year, record type (abstract or full text article), language, location, setting, study design, duration, population, inclusion/exclusion criteria, sample size, data collection methods, and comparison or control. Specific data related to measurement tools that were extracted were: administration of the tools, use of the tool, cut-off values, and results. Sources of evidence, key concepts and findings, and gaps in knowledge were synthesized and presented as narrative descriptions and tables.
Results
A total of 42,510 records were identified by bibliographic databases, grey literature, and hand searching of references (Figure 1 for PRISMA diagram). After removing duplicates and screening for relevance, we included 42 studies published between 2009 and 2020 with a total of 14,431 participants. The majority of the studies were published in the US (n = 38; 91%) and conducted in outpatient palliative care clinics (n = 36; 86%). Of these studies, 15 (36%) were abstracts, and 27 (64%) were cross-sectional studies. Table 2 presents the general characteristics of included studies. The grey literature search did not yield any results that met our study eligibility criteria. We contacted authors of 13 studies53–59,68–73 and received responses from six authors,54,55,69–72 who provided additional data for four studies.54,55,69,72
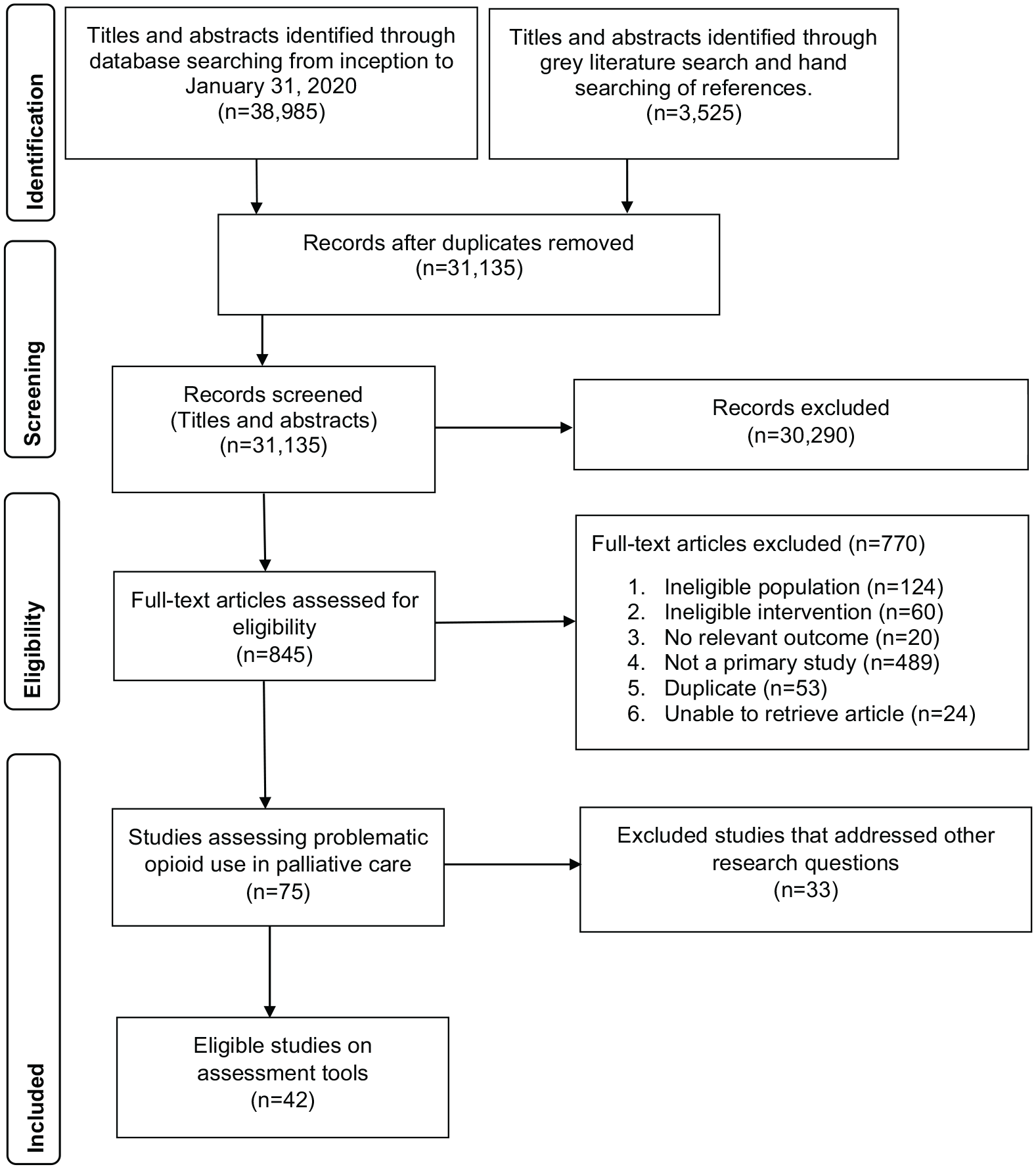
PRISMA flow diagram.
General characteristics of studies included in this review.
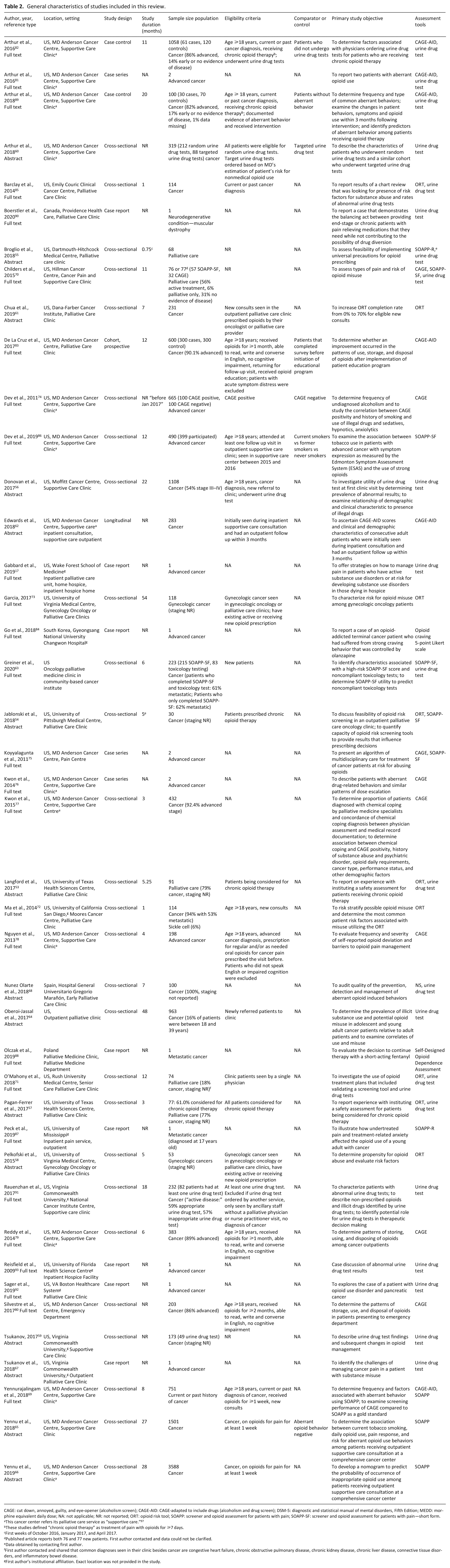
CAGE: cut down, annoyed, guilty, and eye-opener (alcoholism screen); CAGE-AID: CAGE-adapted to include drugs (alcoholism and drug screen); DSM-5: diagnostic and statistical manual of mental disorders, Fifth Edition; MEDD: morphine equivalent daily dose; NA: not applicable; NR: not reported; ORT: opioid risk tool; SOAPP: screener and opioid assessment for patients with pain; SOAPP-SF: screener and opioid assessment for patients with pain—short form.
This cancer center refers its palliative care service as “supportive care.” 97
These studies defined “chronic opioid therapy” as treatment of pain with opioids for ⩾7 days.
First weeks of October 2016, January 2017, and April 2017.
Published article reports both 76 and 77 new patients. First author contacted and data could not be clarified.
Data obtained by contacting first author.
First author contacted and shared that common diagnoses seen in their clinic besides cancer are congestive heart failure, chronic obstructive pulmonary disease, chronic kidney disease, chronic liver disease, connective tissue disorders, and inflammatory bowel disease.
First author’s institutional affiliation. Exact location was not provided in the study.
We identified two types of tools used to assess for problematic opioid use in palliative care: questionnaires (n = 32, 76%)53–55,57,58,61–63,65,66,68–89 and urine drug tests (n = 21, 50%),17,53,55,56,57,59,60,63,64,67,68,70,71,81,82,85,89–93 with an overlap of both types in 11 studies (26%).53,55,57,63,68,70,71,81,82,85,89 Two studies did not specify which questionnaire they used.68,88 The primary study objective of 22 studies (52%) was to evaluate the use of one or more assessment tools in palliative care.53–63,66,68–73,82,85,91,93 In the remaining studies, the primary objectives were to describe patients with problematic opioid use who were receiving palliative care (n = 17, 40%);60,64,67,74–81,84,86–88,90,92 and to evaluate the impact of interventions related to problematic opioid use in palliative care (n = 3, 7%)17,83,89 (Table 2). Though evaluation of the tools was not the primary objective in these studies, they reported on use of these tools to assess for problematic opioid use. Tables 3 and 4 present the reported characteristics and findings related to the use of these tools in palliative care.
Reported characteristics and findings of problematic opioid use questionnaires used in palliative care.
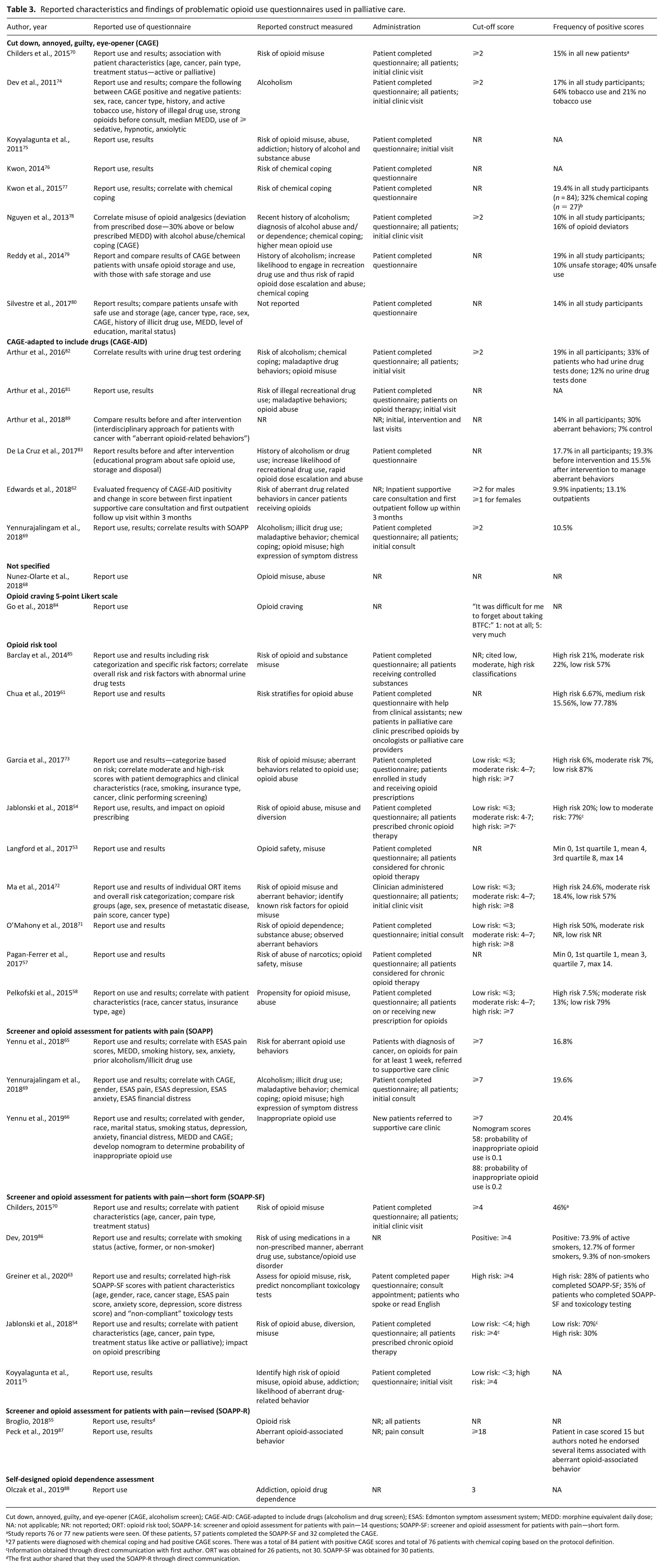
Cut down, annoyed, guilty, and eye-opener (CAGE, alcoholism screen); CAGE-AID: CAGE-adapted to include drugs (alcoholism and drug screen); ESAS: Edmonton symptom assessment system; MEDD: morphine equivalent daily dose; NA: not applicable; NR: not reported; ORT: opioid risk tool; SOAPP-14: screener and opioid assessment for patients with pain—14 questions; SOAPP-SF: screener and opioid assessment for patients with pain—short form.
Study reports 76 or 77 new patients were seen. Of these patients, 57 patients completed the SOAPP-SF and 32 completed the CAGE.
27 patients were diagnosed with chemical coping and had positive CAGE scores. There was a total of 84 patient with positive CAGE scores and total of 76 patients with chemical coping based on the protocol definition.
Information obtained through direct communication with first author. ORT was obtained for 26 patients, not 30. SOAPP-SF was obtained for 30 patients.
The first author shared that they used the SOAPP-R through direct communication.
Reported characteristics and findings of urine drug tests used in palliative care.
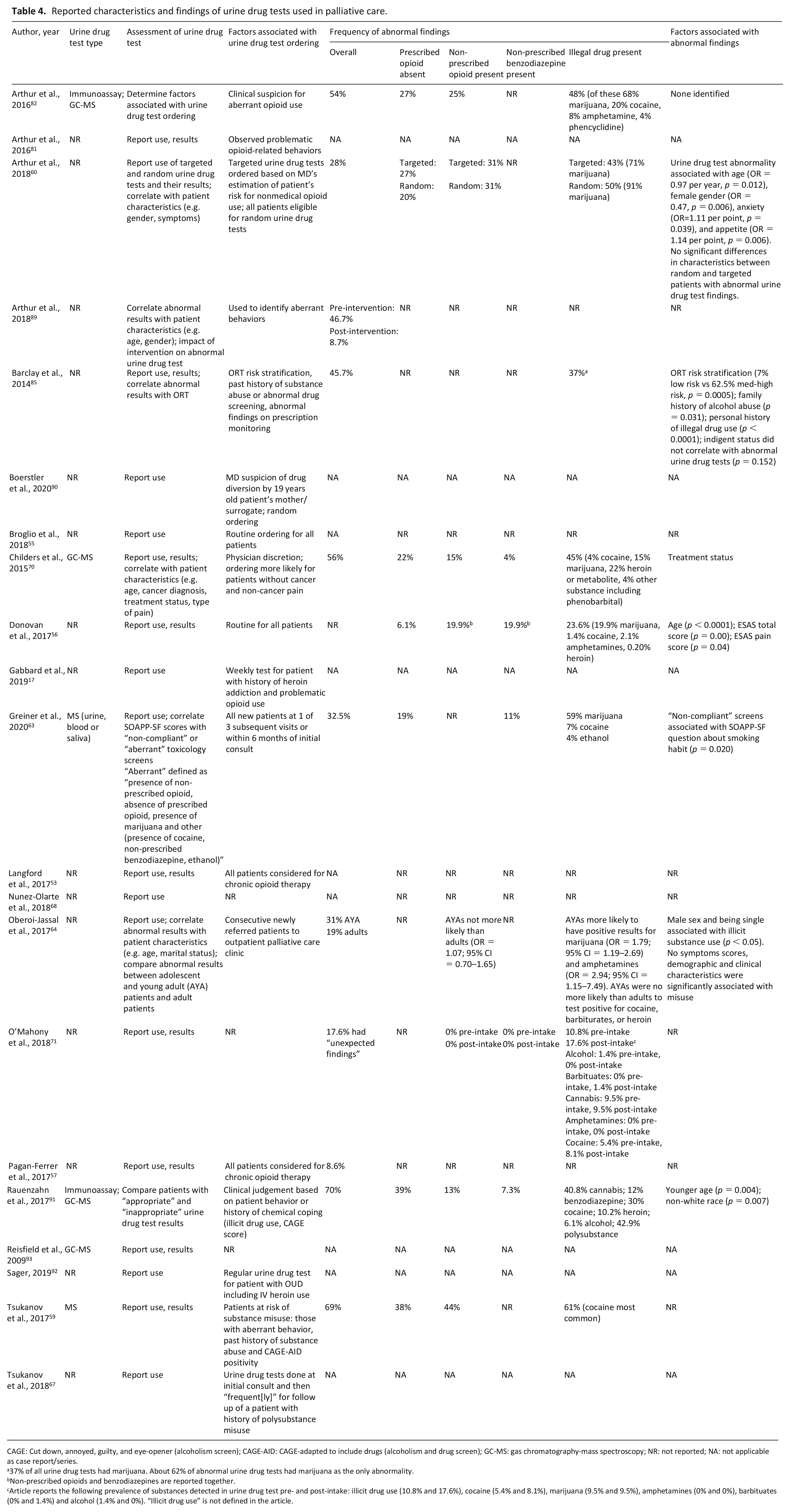
CAGE: Cut down, annoyed, guilty, and eye-opener (alcoholism screen); CAGE-AID: CAGE-adapted to include drugs (alcoholism and drug screen); GC-MS: gas chromatography-mass spectroscopy; NR: not reported; NA: not applicable as case report/series.
37% of all urine drug tests had marijuana. About 62% of abnormal urine drug tests had marijuana as the only abnormality.
Non-prescribed opioids and benzodiazepines are reported together.
Article reports the following prevalence of substances detected in urine drug test pre- and post-intake: illicit drug use (10.8% and 17.6%), cocaine (5.4% and 8.1%), marijuana (9.5% and 9.5%), amphetamines (0% and 0%), barbituates (0% and 1.4%) and alcohol (1.4% and 0%). “Illicit drug use” is not defined in the article.
Problematic opioid use questionnaires (n = 32)
The following questionnaires were used in the studies included in our review: Cut down, Annoyed, Guilty, and Eye-opener (CAGE, n = 8, 25%),70,74–80 CAGE-Adapted to Include Drugs (CAGE-AID; n = 6, 28%),62,69,81–83,89 Opioid Risk Tool (ORT) (n = 9, 28%),53,54,57,58,61,71–73,85 Screener and Opioid Assessment for Patients with Pain (SOAPP) (n = 3, 9%),65,66,69 SOAPP-Revised (SOAPP-R; n = 2, 6%),55,87 and SOAPP-Short Form (SOAPP-SF; n = 5, 16%)54,63,70,75,86 Four studies (13%) used two different questionnaires;54,69,70,75 one study (3%) used a self-designed opioid dependence assessment; 88 one study (3%) used a 5-point Likert scale to measure opioid craving; 84 and one study (3%) did not specify the questionnaire that they used. 68 Supplemental Appendix D3 provides information about the development and validation studies for CAGE, CAGE-AID, ORT, SOAPP, SOAPP-SF, and SOAPP-R questionnaires.
Most studies (n = 30, 94%) were conducted in outpatient clinics53–55,57,58,61–63,65,66,68–79,81–83,85–89 and in the US (n = 28, 88%),53–55,57,58,61,62,63,65,66,69–79,81–83,85–87,89 and published between 2009 and 2020. The same tertiary cancer center conducted many of the studies (n = 16, 50%): CAGE (n = 7, 44%),74–80 CAGE-AID (n = 6, 37.5%),62,81–83,89,94 SOAPP (n = 3, 19%),65,66,69 and SOAPP-SF (n = 2, 13%).75,86 This cancer center refers to its palliative care service as “supportive care.” 95 All studies were observational study designs with the most common being cross-sectional studies (n = 22, 69%).53–55,57,58,61,63,65,66,68–74,77–80,85,86 Most studies (n = 30, 94%) focused on patients with cancer, with reported proportion of participants with cancer ranging from 75% to 100%.53,54,57,58,61–63,65,66,68–70,72–79,81–83,85–89 Of these studies, 10 studies (31%) reported that 7.6% to 46% of their participants had early stage or no evidence of cancer.56,63,70,77,79,80,82,83,89,91 Fourteen studies (47%) did not report the stages of their patients’ cancer diagnoses.53,54,57,58,61–63,65,66,68,69,72,73,85 Regarding the two studies that did not focus primarily on cancer, one study did not provide information about their patients’ diagnoses. 55 The other study reported only a small proportion (18%) of its patients had current or past history of cancer. 71 The diagnoses of the remaining patients were not reported in the article. 71
Regarding assessment of the questionnaires, 13 studies (40%) only reported on use of the questionnaires in palliative care.53,55,57,61,62,68,71,75,76,81,84,87,88 The remaining studies assessed the following: relationship between questionnaire results with patient characteristics (n = 9, 28%),58,63,65,66,69,70,72,73,86 specific problematic opioid use (n = 4, 13%),77–80 and urine drug tests (n = 3, 9%);63,82,85 questionnaire results before and after implementing problematic opioid-use related interventions (n = 2, 6%);83,89 comparison of characteristics of patients with normal and abnormal questionnaire (n = 2, 6%);72,74 and, impact of questionnaire results on opioid prescribing (n = 1, 3%). 54 Of these studies, only two evaluated the psychometric properties of the questionnaires that were used.63,69 One study (3%) developed a nomogram based on SOAPP to determine probability of inappropriate opioid use. 66
A majority of the studies routinely administered paper questionnaires to patients (n = 21, 66%),53,54,57,58,61–63,65,66,69–72,73,74,75,78,81,82,85,89 particularly during initial clinic visits (n = 12, 38%),62,63,66,69–72,74,75,78,81,89 or to specific patients being considered for (n = 2, 6%)53,57 or receiving opioids (n = 7, 22%).54,58,61,65,73,81,85 One study delivered the ORT through verbal, face-face interviews; 72 and, in one study, clinical assistants helped patients complete the ORT. 61 Twelve studies (38%) did not provide information about how they administered questionnaires to patients.55,68,76,77,79,80,83,84,86,87–89 Furthermore, the studies used the questionnaires to measure multiple constructs related to problematic opioid use including ones that the questionnaires have not been validated to assess. Detailed findings about use of these questionnaires in palliative care are presented in the following sections and Table 3.
Cut down, annoyed, guilty, and eye-opener (CAGE) (n = 8)
We identified eight studies that adopted the CAGE as a proxy screening tool for problematic opioid use.70,74–80 This questionnaire was previously validated for constructs related to problematic alcohol use, but not opioid use, in populations such as psychiatric inpatients and primary care outpatients (Supplemental Appendix D). Our included studies reported using the CAGE to assess for the following problematic opioid-related constructs in palliative care: misuse,70,75 abuse,75,79 addiction, 75 higher use, 78 and rapid dose escalation. 79 The CAGE was also reported as a measure of general substance abuse,75,79 chemical coping,76–78 and problematic alcohol use (Table 3).74,75,78,79 We identified only one study conducted in a setting (emergency department) other than outpatient palliative care clinics (Table 2). 80 Excluding the two case series,75,76 the sample sizes ranged from 76 to 432 participants with cancer; and, the reported prevalence of positive CAGE scores ranged from 10% to 19% in cross-sectional studies (n = 6).70,74,77–80 One study had reporting errors that made data interpretation and synthesis challenging. 70
CAGE scores were correlated with patient characteristics (age, 70 cancer diagnosis, 70 pain type, 70 treatment status 70 ) and specific problematic opioid use (chemical coping, 77 opioid dose deviation 78 ). Chemical coping was defined as “using prescribed opioids to control non-nociceptive symptoms.”77,96 While opioid dose deviation was defined as when “patient-reported [morphine equivalent daily dose (MEDD)]. . . falls 30% above or below the prescribed MEDD range.” 78 One study compared the characteristics of CAGE-positive and CAGE-negative patients, including history of illegal drug use, use of strong opioids and MEDD. 74 Two studies assessed compared CAGE scores between patients with safe and unsafe opioid use and storage practices.79,80 We identified no studies that evaluated the psychometric properties of the CAGE in the context of palliative care.
Cut down, annoyed, guilty, and eye-opener-adapted to include drugs (CAGE-AID) (n = 6)
We identified six studies that assessed the use of CAGE-AID in a palliative care context.62,69,81–83,89 This questionnaire has been validated for problematic alcohol and general substance use but not opioid use in settings outside of palliative care (Supplemental Appendix D). Our included studies reported using the CAGE-AID to assess for the following constructs in palliative care: alcoholism,69,82,83 general drug use,69,81,83 rapid opioid dose escalation, 83 opioid abuse,81,83 opioid misuse,69,82 chemical coping,69,82 high expression of symptom distress, 69 and maladaptive81,82 or aberrant drug behaviors62,69 (Table 3). All studies were conducted in outpatient palliative care clinics in a single US cancer center (Table 2).62,69,81–83,89 Excluding the case series, 81 the studies had samples sizes ranging 100 to 751 patients with cancer; and the reported frequency of positive CAGE-AID scores ranged from 9.9% to 19.3% in all study participants in the case control (n = 2),82,89 cross-sectional studies (n = 1), 69 longitudinal (n = 1), 68 and prospective cohort (n = 1) 83 studies.
In addition to reporting the CAGE-AID scores, one study correlated CAGE-AID scores with frequency of physicians ordering urine drug tests. 82 Two studies compared the prevalence of positive CAGE-AID scores before and after an intervention for problematic opioid use: one study assessed an education program about safe opioid use, storage and disposal; 83 the other assessed an interdisciplinary approach for patients with cancer with “aberrant opioid-related behaviors.” 89 Only one study evaluated the psychometric properties of the CAGE-AID for identification of patients at risk of “aberrant drug behaviors” in the context of palliative care: specifically, the questionnaire’s validity (sensitivity, specificity, positive, and negative predictive values) but not its reliability, responsiveness, and interpretability. 69 Although the SOAPP has not been validated in palliative care (refer to SOAPP section below), this study used the SOAPP questionnaire as the “gold standard” to determine whether patients had a high risk of “aberrant opioid use.” 69
Opioid risk tool (ORT) (n = 9)
We identified nine studies that assessed the use of the ORT in a palliative care context.53,54,57,58,61,71,72,73,85 Previous attempts had been made to validate this tool as a measure of problematic opioid use in chronic non-cancer pain (Supplemental Appendix D). Our included studies reportedly used the ORT to assess for the following constructs in palliative care: opioid abuse,54,58,61,73 dependence, 69 misuse,53,54,57,58,72,73,85 diversion, 54 aberrant behaviors,71–73 and safety.53,57 Three studies used the ORT to measure general substance abuse 71 and misuse (Table 3). 85 All studies were cross-sectional studies that were conducted in outpatient palliative care clinics in the US (Table 2).53,54,57,58,61,71–73,85 The sample sizes ranged from 30 to 231, with the primary study population in eight studies being patients with cancer.53,54,57,58,61,72,73,85 One study was conducted in a geriatric palliative care clinic and only 18% of participants had current or prior cancer diagnoses. Details about the remaining participants’ medical diagnoses were not provided. 71
ORT administration in palliative care varied between the studies. Though the ORT was mainly self-administered by patients (n = 7);53,54,57,58,71,73,85 one study had clinicians administer the questionnaire 72 and another had clinical assistants help patients complete the ORT. 61 Three studies administered the ORT for participants who were being considered for53,57 or already receiving chronic opioid therapy; 54 whereas, three studies administered the ORT to participants who had existing active or were receiving new opioid prescriptions.58,61,73 One retrospective study collected data on ORT administered to all participants who received a controlled substance. If ORT scores were missing, medical records were reviewed to calculate it. 85 Information about study participants’ opioid histories were not provided in two studies.71,72
Based on their ORT scores, patients were categorized into low, medium, or high risk of developing aberrant behavior if prescribed chronic opioid therapy (Table 3 provides cut-off values used in the studies). The reported frequency of high-risk individuals in seven studies ranged from 6% to 50%.54,58,61,71,72,73,85 The ORT scores in two studies were not interpretable as they were reported as interquartile ranges.53,57 Additionally, the ORT scores were correlated with urine drug test results, 85 participant demographics (e.g. age,58,72 sex, 72 race,58,73 insurance type,58,73 smoking 73 ) and clinical characteristics (e.g. cancer diagnosis,72,73 presence of metastatic disease, 72 pain score 72 ). We identified one study that performed the ORT to determine if the results influenced opioid prescribing decisions for outpatient palliative care patients who were already on chronic opioid therapy. 54 Of the 30 patients, 20% were deemed high-risk on the ORT and opioid prescribing decisions were changed for 33% of these patients. However, the most common prescribing decision for the high-risk participants was to maintain the same opioid dose. 54 We did not identify any studies that examined the psychometric properties of the ORT in the context of palliative care.
Screener and opioid assessment for patients with pain (SOAPP) (n = 10)
We identified ten studies that assessed the use of SOAPP in palliative care: three on the SOAPP,65,66,69 five on the SOAPP-Short Form (SOAPP-SF),54,63,70,75,86 and two on the SOAPP-Revised (SOAPP-R).55,87 The SOAPP was originally developed to assess risk or predict problematic opioid use; it was subsequently shortened (SOAPP-SF) and revised (SOAPP-R) (Supplemental Appendix D). Our included studies reportedly used the SOAPP and its variants to assess for the following constructs in palliative care: opioid misuse,54,63,69,70,75 abuse,54,75 diversion, 54 risk 63 ; inappropriate opioid use 66 ; risk of aberrant opioid-related behaviors65,87 and opioid use disorder 86 ; and, general drug-related aberrant behaviors (Table 3).65,69,75,86 One study used a total SOAPP score of ⩾7 as their “gold standard” to identify participants with a high risk of “aberrant opioid use.” 69 All studies were conducted in outpatient palliative care clinics in the US (Table 2). Excluding the case series, 75 the sample sizes ranged from 30 to 1051 patients with cancer; and, the reported frequency of positive or “high-risk” scores on the SOAPP and its variants ranged from 9.3% to 46% in the cross-sectional studies.54,55,65,69,70,75
In addition, the scores from SOAPP and its variants were correlated with patient demographics (e.g. age,63,70 gender 63 ), clinical characteristics (e.g. cancer diagnosis,63,70 cancer treatment status, 70 pain type 70 ), Edmonton Symptom Assessment System scores,63,69 and concurrent substance use (alcohol, 65 tobacco, 86 illicit drug use 65 ). One study assessed the impact of SOAPP-SF scores on opioid prescribing. 54 We identified one study that found SOAPP-SF score of ⩾4 was not significantly associated with “noncompliant” toxicology tests (p = 0.83); however, a threshold of ⩾3 was most associated with “aberrant toxicology tests” in an outpatient palliative care clinic in a cancer institute (OR 5.14, 95% CI [1.86 to 14.26], p = 0.002; sensitivity 0.75, specificity 0.64). 63 “Aberrant toxicology tests” were defined as the absence of prescribed opioid, presence of non-prescribed opioid, presence of cannabis, and other substances (presence of cocaine, non-prescribed benzodiazepine, ethanol). 63 When the presence of cannabis was not considered aberrant, SOAPP-SF ⩾3 was still found to be significantly associated with abnormal urine drug tests. 63
Urine drug tests (n = 21)
Urine drug tests are clinical tools that are used to monitor compliance with prescription opioid therapy, development of problematic opioid use and use of non-prescription drugs. 97 Two common types of urine drug tests are: immunoassays, which are used at the point of care, and gas-chromatography/mass-spectroscopy (GC-MS), which are processed at a laboratory. 97 We identified 21 studies that examined urine drug test use in a palliative care setting.17,53,55–57,59,60,63,64,67,68,70,71,81,82,85,89–93 The majority of studies were conducted in the US (n = 19, 91%),17,53,55–57,59,60,63,64,67,70,71,81,82,85,89,91–93 and in outpatient clinics (n = 20, 95%) (Table 2),53,55–57,59,60,63,64,67,68,70,71,81,82,85,89–93 and published between 2016 and 2020 (n = 18, 86%).17,53,55–57,59,60,63,64,67,68,71,81,82,89–92 The main population was patients with cancer, but two studies (14%) did not specify their participants’ medical diagnoses.55,71 One case report was about the ethical challenges of opioid prescribing for a young patient with muscular dystrophy where clinicians were concerned about drug diversion by his mother. 90 The most common study design was cross-sectional (n = 13, 62%) (Table 2).53,55–57,59,60,63,64,68,70,71,85,91
The reported primary study objectives were: to identify factors associated with ordering urine drug tests; 82 to report on the use of urine drug tests;17,53,55,57,59,64,67,68,71,85,90,92,93 and to correlate abnormal urine drug tests with patient characteristics, including age,64,70,89 gender,64,60 marital status, 64 cancer diagnosis, 70 type of pain, 70 symptoms (e.g. anxiety, appetite), 60 treatment status (active or palliative), 70 and problematic opioids risk tools (ORT, 85 SOAPP-SF 63 ) (Table 2). One study compared the characteristics of patients with “appropriate” and “inappropriate” urine drug tests. 91 “Inappropriate” urine drug tests were defined as the presence of non-prescribed drugs (e.g. opioid, benzodiazepine, cannabis, cocaine) and absence of prescribed opioids. 91 Another study examined the impact of an interdisciplinary approach for patients with cancer on their aberrant opioid-related behavior, which included abnormal urine drug tests. 89
The type of urine drug tests used to detect prescription and illegal drugs was reported in six (29%) studies (Table 4).59,63,70,82,91,93 Two studies (10%) conducted in the same supportive care clinic first performed immunoassays and then confirmed the results with GC-MS.82,91 The other four studies (19%) reported use of only GC-MS.59,70,93 The urine drug test type used in the remaining 15 studies (71%) was not reported.17,53,55–57,60,64,67,68,71,81,85,89,90,92
The 21 studies varied in terms of how they incorporated urine drug tests into clinical care (Table 4). Six studies (29%) routinely used urine drug tests in clinical care: four studies ordered urine drug tests for all clinic patients;55,56,63,64, and, the two other studies ordered urine drug tests for all patients being considered for chronic opioid therapy.53,57 In contrast, 11 studies (52%) ordered urine drug tests based on clinician discretion and the presence of specific patient factors.17,59,60,70,81,82,85,89–92 These factors included: problematic behavior (e.g. early refill requests),59,82,90,91 history of substance use disorder,17,59,85,91,92 high-risk classification based on ORT 85 results, CAGE positivity,59,91 and abnormal findings on prescription drug monitoring programs. 85 One study did not specify how clinicians estimated patients’ risk for non-medical use to order targeted urine drug tests. 60 Urine drug test ordering was also associated with the following patient characteristics: non-cancer diagnoses, 70 early-stage cancer or no evidence of disease, 82 non-cancer pain, 70 active treatment status, 70 younger age, 82 African American race, 82 less education, 82 high pain intensity, 82 and less fatigue. 82 Three case reports reported on the use of urine drug tests to help manage patients with a history of OUD.17,67,92
The overall frequency of abnormal urine drug test results ranged from 8.6% to 70% (Table 4). These abnormal results can be classified into four categories: (1) prescribed opioid absent from the urine (reported frequency 6.1% to 39%);56,59,60,63,70,82,91 (2) opioid not prescribed for the patient present in the urine (reported frequency 0% to 44%);56,59,60,70,71,82,91 (3) benzodiazepine not prescribed for the patient present in the urine (reported frequency: 0% to 12.2%);56,63,70,71,91 (4) illegal drug present in the urine (reported frequency: 18% to 61%).56,59,60,63,70,71,82,85,91 The most common drug considered illegal in the studies was cannabis, with a reported frequency of 9.5% to 68%.56,60,63,70,82,91 Other drugs detected were alcohol (1.4% to 4%),63,71 cocaine (1.4% to 24.4%),56,63,70,82,91 amphetamines (0% to 8%),56,82 heroin (0.20% to 22%),56,70,91 and phenobarbital. 70 One study found that adolescents and young adult patients with cancer compared to adult patients with cancer have higher prevalence of positive urine drug tests for cannabis (OR = 1.79; 95% CI = 1.19–2.69) and amphetamines (OR = 2.94, 95% CI = 1.15–7.49). 64 Besides young age,64,91 other factors associated with abnormal urine drug test results were non-white race,56,91 cancer treatment status, 70 ORT risk stratification, 85 SOAPP-SF question about smoking 63 and family history of “alcohol abuse.” 85 One study found that if the presence of cannabis was not considered aberrant, then abnormal urine drug test results were also associated with distress, smoking, and SOAPP-SF question about not taking medications as prescribed. 63
Discussion
Using established methodology, our scoping review of literature published up until January 31, 2020 identified 42 studies that reported on the use of tools that assessed for the presence or risk of problematic opioid use in palliative care. The main types of tools used were questionnaires and urine drug tests. The primary objectives of only half (n = 22; 52%) of the studies were to evaluate the use of these tools in palliative care. The remaining studies only reported on the use of these tools in the context of patients with problematic opioid use. Most of the studies (n = 34, 81%) were published within the last 5 years and were conducted in US outpatient palliative care clinics for patients with cancer. A minority of participants across ten studies had early-stage cancers, and only two studies focused on patients without cancer. These findings underscore the need for high-quality studies about assessment tools for problematic opioid use in palliative care, particularly in countries outside of the US and for patients with early stage, advanced cancers, and non-malignant conditions.
Synthesis of knowledge about questionnaires used to assess for problematic opioid use
Of the 42 included studies, we identified 32 studies that used questionnaires to assess for problematic opioid use in palliative care. The main questionnaires used were CAGE and its variant (CAGE-AID), ORT, SOAPP and its variants (SOAPP-SF, SOAPP-R). The following is an appraisal of these questionnaires in the context of previously published literature.
CAGE and CAGE-AID
The CAGE and CAGE-AID questionnaires were originally developed to identify patients with problematic alcohol and general substance use98–100 and have not been validated to specifically assess for problematic opioid use. 101 Yet we identified 14 palliative care studies that used the CAGE70,74–80 and CAGE-AID62,69,81,82,83,89 to measure problematic opioid-related constructs including opioid abuse,75,79,82,83 misuse,69,70,75,81 aberrant drug related behaviors, 62 and chemical coping.69,76–78,82 All the studies about CAGE-AID were conducted in one tertiary cancer center’s palliative care clinic.60,62,69,81–83 Additionally, one study evaluated the validity of the CAGE-AID to identify patients at risk for “aberrant opioid and drug use behaviors” by comparing it to the SOAPP. 69 However, the measurement properties of SOAPP have not yet been formally evaluated in palliative care and therefore should not be used as a “gold standard” 69 to determine whether patients are at high risk for problematic opioid use. Based on these findings, there is limited evidence to support the use of CAGE and CAGE-AID to measure problematic opioid use in palliative care.
ORT
The ORT is a patient-completed questionnaire developed to identify patients prescribed opioids for chronic pain who are at risk of “aberrant behaviors.” 20 Though the original study reported that the ORT has a high degree of sensitivity and specificity (c = 0.82 males, c = 0.85 females), follow-up studies did not demonstrate that the clinician- nor the patient-completed ORT were predictive of the presence of problematic opioid-related behaviors in patients receiving chronic opioid therapy for non-cancer pain.20,102,103 Despite this weak evidence to support its use, nine studies in our review used the ORT in outpatient palliative care clinics53,54,57,58,61,71,72,73,85 to assess for problematic opioid-related constructs, such as abuse,54,58,61,73 dependence, 71 and diversion. 54 One study found that patients stratified as moderate-high risk on the ORT were more likely to have abnormal urine drug test results. 85 However, none of the studies evaluated the questionnaire psychometric properties.
SOAPP
The SOAPP questionnaires were designed to predict which patients with chronic pain would develop “aberrant behaviors and substance misuse” while on long-term opioid therapy.104,105 We identified use of multiple versions of the SOAPP to evaluate risk of problematic opioid use in palliative care: the original, conceptually-derived SOAPP, and its shortened version, SOAPP-SF, and the empirically-derived, revised version, SOAPP-R. We identified one study that evaluated the psychometric properties of the SOAPP-SF: SOAPP-SF threshold of ⩾3 was significantly associated with abnormal urine drug test results in a US cancer institute’s outpatient palliative care clinic. 63 However, the correlation between SOAPP-SF scores, abnormal urine drug test results and problematic opioid use by the patients receiving palliative care was not evaluated.
Synthesis of knowledge about urine drug tests used to assess for problematic opioid use
Urine drug tests were another tool used to identify patients who have or are at risk of problematic opioid use in palliative care. Our review found 21 studies that examined the use of urine drug tests in palliative care. The reported overall prevalence of abnormal urine drug tests in palliative care ranged from 8.6% to 70%. Possible explanations for this high reported prevalence in palliative care include patient selection bias and inclusion of cannabis as an abnormal urine drug result. Regarding patient selection bias, most of the included studies (n = 11, 52%) reported that urine drug tests were ordered at the discretion of the clinician and specific factors were present, such as observed aberrant behavior,59,82,90,91 history of substance use disorder,17,59,85,91,92 and patient characteristics, particularly younger age, 82 African American race, 82 and less education. 82 These results raise concern of the role of stigma, racism, and stereotyping in patient selection for targeted testing. They highlight the need for evaluation of this practice, which can contribute to inequitable access and delivery of palliative care and further stigmatization of marginalized populations. Adoption of an universal precautions approach where all patients are tested, similar to what has been advocated for in chronic non-cancer pain, can potentially reduce stigma, improve patient care and manage opioid-related risk. 106
Another potential contributing factor to the high prevalence of abnormal urine drug test results in palliative care is the inclusion of cannabis as an “illicit drug.”56,60,63,70,82,91 Cannabis use is common among patients with cancer: 66% have used cannabis at some point in their lives and 24% consider themselves to be active cannabis users. 107 It is legal for medicinal purposes in many high-income countries, including several states in the US, where most of these studies were conducted. 108 Therefore, use of urine drug tests to identify patients who have or are at risk of problematic opioid use need to be validated. Future studies about urine drug testing in palliative care should clearly define “abnormal” results and consider categorizing these results as how we presented them in our review: prescribed opioids absent, non-prescribed opioid present, non-prescribed benzodiazepine present and illegal drugs present.
Strengths and limitations
Strengths of our study included use of an established methodology for scoping reviews to identify and map existing knowledge.36,37,39 In addition to conducting comprehensive searches of bibliographic databases from inception to January 31, 2020 and of grey literature, we performed hand searches of references. Study eligibility criteria were broad to capture the extent, range, and nature of the available evidence. We did not restrict study inclusion based on language, location, or study design. The reporting guideline PRISMA-Scr was used to standardize reporting of this review.
Our study also has limitations. Our review focused on patients receiving palliative care who are at risk of or have problematic opioid use, including those with active opioid use disorder. We excluded studies that focused on patients with clinically stable opioid use disorder (e.g. long-term remission) and non-patient populations, such as caregivers and health care providers. Therefore, our study does not capture the entire scope of problematic opioid use in palliative care. Further, our search would not have identified studies that were not indexed as “palliative care,” which may have included studies about patients receiving primary (non-specialist) palliative care. This may have contributed to the lack of studies including patients with non-malignant life-threatening conditions. 109 Additionally, we did not conduct a critical appraisal of the included studies as this was beyond the remit of our scoping review. We did not find studies in non-English languages, which may reflect the databases that we searched.
What this study adds
To our knowledge, this study is the first scoping review that identifies tools that have been used to assess for the presence or risk of problematic opioid use in palliative care. We retrieved mainly observational studies that adopted use of questionnaires that were originally developed in the chronic non-cancer pain population. No identified studies adequately assessed the psychometric properties of these questionnaires in palliative care, and none were identified that reported on tools developed specifically for palliative care populations. Despite the paucity of evidence, palliative care experts, and organizations are recommending the use of tools developed for chronic pain populations17,21,22 in response to increasing concerns about harms associated with opioids;8–11 our findings suggest that these recommendations are premature. Similarly, palliative care services are increasingly adopting the use of urine drug tests. However, the purpose, administration and impact of urine drug tests on patients receiving palliative care are not clear from the current evidence.
This review highlights significant gaps in knowledge. The duration of palliative care involvement can now range from hours to years, given the increasing and earlier integration of palliative care in life-threatening non-malignant and malignant conditions. Therefore, patients receiving palliative care may be at risk of opioid-related harms that differ from those of chronic pain populations, and that may vary according to country, palliative care setting and stage of illness. The development and validation of tools to assess for problematic opioid use in palliative care are the first critical steps to measure the extent of this problem, and to inform prevention and management strategies in this diverse population.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163211015567 – Supplemental material for Assessment tools for problematic opioid use in palliative care: A scoping review
Supplemental material, sj-pdf-1-pmj-10.1177_02692163211015567 for Assessment tools for problematic opioid use in palliative care: A scoping review by Jenny Lau, Paolo Mazzotta, Rouhi Fazelzad, Suzanne Ryan, Alissa Tedesco, Andrew J. Smith, Abhimanyu Sud, Andrea D. Furlan and Camilla Zimmermann in Palliative Medicine
Footnotes
Acknowledgements
The authors would like to thank Canadian Virtual Hospice, Canadian Hospice Palliative Care Association (Ms. Sharon Baxter) and Canadian Society of Palliative Care Physicians (Dr. David J. Henderson) for their roles as key stakeholders in this review. The authors would also like to thank Dr. Christopher Blake for his assistance with record screening.
Authorship
The conception and design of the study was conducted by JL, PM, ADF, CZ, and RF. Searches of the bibliographic databases and grey literature were performed by RF. Records were screened by JL, PM, SR, ADF, and AT. Data analysis was completed by JL, PM, RF, and AS. ADF and CZ provided methodological support throughout the study. JL drafted the manuscript, which was then revised critically for intellectual content by PM, SR, AS, AJS, AT, RF, ADF, and CZ. All authors approved the final version and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy and integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institute of Health Research [grant number OCK_156774] for Dr. J Lau.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
