Abstract
Background:
The European Association for Palliative Care White Paper defined optimal palliative care in dementia based on evidence and expert consensus. Yet, we know little on how to achieve this for people with dementia living and dying at home.
Aims:
To examine evidence on home palliative care interventions in dementia, in terms of their effectiveness on end-of-life care outcomes, factors influencing implementation, the extent to which they address the European Association for Palliative Care palliative care domains and evidence gaps.
Design:
A systematic review of home palliative care interventions in dementia.
Data sources:
The review adhered to the PRISMA guidelines and the protocol was registered with PROSPERO (CRD42018093607). We searched four electronic databases up to April 2018 (PubMed, Scopus, Cochrane library and CINAHL) and conducted lateral searches.
Results:
We retrieved eight relevant studies, none of which was of high quality. The evidence, albeit of generally weak quality, showed the potential benefits of the interventions in improving end-of-life care outcomes, for example, behavioural disturbances. The interventions most commonly focused on optimal symptom management, continuity of care and psychosocial support. Other European Association for Palliative Care domains identified as important in palliative care for people with dementia, for example, prognostication of dying or avoidance of burdensome interventions were under-reported. No direct evidence on facilitators and barriers to implementation was found.
Conclusions:
The review highlights the paucity of high-quality dementia-specific research in this area and recommends key areas for future work, for example, the need for process evaluation to identify facilitators and barriers to implementing interventions.
There is an urgent need to find effective strategies to improve home-based care for people with dementia.
The European Association for Palliative Care (EAPC) White Paper defined optimal palliative care in dementia based on evidence and expert consensus.
We know little about the evidence base on how to achieve optimal palliative care in dementia for people living and dying at home.
There is evidence, albeit limited and of generally weak quality, that shows the potential benefits of home palliative care interventions in dementia in improving end-of-life care outcomes, such as the management and reduction of behavioural disturbances in people with dementia.
The interventions focused mainly on symptom management, continuity of care and psychosocial support, with less attention paid to four other EAPC domains considered important for people with dementia, for example, prognostication of dying or avoidance of burdensome interventions.
The paper highlights several gaps in the evidence, including the limited evidence on facilitators and barriers to implementing the intervention and the lack of consensus on outcome measures used.
The EAPC’s definition of optimal palliative care in dementia provided a useful framework for a systematic assessment of the range and focus of evidence of what is effective for people with dementia living and dying at home.
The review highlights the paucity of high-quality dementia-specific research in this area and recommends key areas for future work, such as the need for process evaluations to identify facilitators and barriers to implementing interventions or for a standard outcome set to facilitate comparisons and meta-analyses.
High-quality dementia-specific research is required to further support the evidence base for palliative care interventions to be a routine care for people with dementia living and dying at home.
Background
Dementia is a life-limiting illness characterised by widespread physical, cognitive and behavioural impairment, resulting in severe disabilities that persist until death.1,2 The global prevalence of dementia is projected to increase to almost 132 million by 2050. 3 The high demand for dementia care, the individual’s preference to stay at home for as long as possible combined with limited supply and rising costs of institutional long-term care services highlight an urgent need to find effective strategies to improve home-based care for people with dementia, including those with advanced dementia requiring end-of-life care.4–7
To improve the quality of life of people with dementia and their families, a palliative care approach has been widely recommended.8–10 In 2014, van der Steen and colleagues published the European Association for Palliative Care (EAPC) White Paper defining optimal palliative care in dementia based on evidence and expert consensus. These experts achieved consensus on 57 salient recommendations that fall under 11 important domains of palliative care: applicability of palliative care; person-centred care, communication, and shared-decision making; setting care goals and advance planning; continuity of care; prognostication and timely recognition of dying; avoiding overly aggressive, burdensome or futile treatment; optimal treatment of symptoms and providing comfort; psychosocial and spiritual support; family care and involvement; education of the health care team; and societal and ethical issues (Supplementary file 1). 11 The majority of this evidence draws on work in long-term care and institutional settings. Less well known is the evidence on the effectiveness of palliative care interventions for people with dementia living at home.
A Cochrane review on palliative care interventions in advanced dementia found only two low-quality studies, neither of which was conducted in the home setting. 12 In a systematic review that aimed to identify populations appropriate for palliative care and effective palliative care models, they found improvements in pain and depressive symptoms in people with dementia. However, this study did not indicate whether the population with dementia was living at home. 13 Another Cochrane review evaluated palliative care services for people living at home with advanced incurable illnesses, including those with dementia. They found reliable evidence that these services could reduce symptom burden and increase the chance that people with terminal diseases will die at home. However, the evaluated services were only for people with cancer and organ failure, rather than for people with dementia. 14
In the last 5 years, there have been an increasing policy commitment to improving dementia care with concomitant increase in research funding.15,16 In order to guide efforts to improve the care for people with dementia living and dying at home, we conducted a systematic review to examine evidence on palliative care interventions for this population. Our overall aim was to synthesise evidence on the effectiveness of palliative care interventions on end-of-life care outcomes (e.g. patient death at home or pain) for people with dementia living at home. In addition, we reviewed facilitators and barriers to implementing these interventions, assessed the extent to which the interventions reflected optimal palliative care in dementia as defined in the EAPC White Paper and identified gaps in evidence.
Methods
Design
The systematic review adhered to the PRISMA guidelines, and the protocol was registered with the PROSPERO international prospective register of systematic reviews (www.crd.york.ac.uk/prospero/-CRD42018093607). The PRISMA Checklist is available in Supplementary file 2.
Eligibility criteria
We included peer-reviewed quantitative studies evaluating palliative care interventions for people with any type of dementia living at home. This included randomised controlled trials (RCTs), controlled clinical trials (CCTs), non-randomised controlled studies, controlled and uncontrolled before and after studies, interrupted time series (ITSs) and case studies published in either English or Dutch. To provide a comprehensive overview of existing research in this area, we included specialist palliative care services and non-specialist palliative care interventions, that is, interventions that were not labelled as ‘palliative care’ but described as aiming to improve care at the end of life for people with dementia.
Specialist palliative care included services with the following four elements: (1) designed primarily for people with dementia living at home, (2) aim to support people outside hospital and other institutional settings for as long as possible and to enable people to stay at home, (3) be provided by specialists in palliative care or intermediate palliative/hospice care, and (4) provide comprehensive care addressing different physical and psychosocial components of palliative care. 14 Non-specialist palliative care included interventions that focused either on people with advanced/severe/late-stage dementia living at home or on people with dementia living at home with the potential impact on palliative care or death and dying or end-of-life care outcomes.
End-of-life care outcomes included patient death at home as the primary outcome. Secondary outcomes included time the patient spent at home, pain, dyspnoea, depressive symptoms, behavioural symptoms common at the end of life, existential or spiritual concerns, communication or care planning, experience or satisfaction, functional status, health-related quality of life, and resource use.
14
In addition to the outcomes registered in Prospero, we added institutionalisation as a secondary outcome, as it had been reported in two of the studies included. Studies that did not focus entirely on the home setting or dementia (e.g. studies on primary care or advanced incurable illnesses) were also included, provided that the majority of the participants (
Search strategy
The search strategy was undertaken in two phases from April to June 2018 to search for literature relating to specialist palliative care services (Phase 1) and non-specialist palliative care interventions (Phase 2). Two search strategies were developed by the research team with advice from an information specialist. In Phase 1, we used a combination of MESH headings, controlled vocabulary and free-text terms to cover palliative/end-of-life/terminal care, dementia, and the home setting. In Phase 2, we covered the home setting combined with either advanced/severe/late-stage dementia or dementia with outcome measures relating to palliative care or death or dying. We searched four electronic databases: PubMed, Scopus, Cochrane Library, including the Cochrane Central Register of Controlled Trials (CENTRAL) and Cochrane Database of Systematic Reviews, and Cumulative Index to Nursing and Allied Health Literature (CINAHL) (from inception to April 2018). The search strategies were validated in PubMed and translated for use in the other databases (Table 1). We also performed hand-searching of relevant journals and reference lists of included and relevant articles and citation tracking in Google Scholar). We also contacted the author of a relevant study protocol to ask for update about their study. 17
Search syntax for the database search.

Study selection
The first author (R.M.) removed duplicates and screened the titles and abstracts for relevance. Studies considered potentially relevant were marked as ‘include’ or ‘uncertain’. A random 20% of articles were independently screened by two co-authors (F.B. and J.L.). Full texts were retrieved for studies deemed as ‘included’ or ‘uncertain’. These were screened by R.M. against the eligibility criteria and checked by F.B. and J.L. Discrepancies were discussed and resolved among the three authors. A PRISMA flowchart was created to describe the selection procedure and the rationale for exclusion was compiled. Mendeley citation management software was used for deduplication and management of references. Multiple reports about a similar study were collated to ensure that each study rather than each paper is the unit of interest.
Data extraction
The data were extracted to a specially designed form in MS Excel version 16 (© Microsoft 2018). This form was pilot-tested on three articles to ensure consistency and was approved by the research team. Characteristics of the included studies were extracted by R.M. and checked for accuracy by F.B. Study characteristics included country, study design, data collection method, research question (aim), setting, participants and intervention type. R.M. and F.B. independently extracted data on outcomes. Discrepancies were resolved by discussion and consensus between three reviewers (R.M., F.B., J.L.). Qualitative data on intervention components and potential barriers and facilitators to implementing the interventions were also extracted.
Quality appraisal
Quality appraisal was conducted by R.M. and F.B. using the ‘Quality Assessment Tool for Quantitative Studies’ developed by Effective Public Health Practice Project. 18 Studies were rated as either strong, moderate or weak on the following components: selection bias, study design, confounders, blinding, data collection methods, withdrawals/dropouts, intervention integrity, and appropriate data analyses used. Two authors discussed any discrepancies and reached consensus. The quality was considered as either strong (no weak ratings), moderate (one weak rating), or weak (two or more weak ratings).
Data synthesis
We described the included studies in terms of country of origin, design, data collection method, intervention components and participants. Due to heterogeneity in interventions and outcomes, data were not pooled in a meta-analysis. Instead, the outcomes of the interventions were presented separately in a table with an indication of whether the effects of the intervention were positive, negative or statistically insignificant. The available data did not allow us to calculate effect sizes. Hence, we presented data in the way it was reported in the study (e.g. P values). Qualitative data on facilitators and barriers were synthesised thematically and presented in a narrative way. We mapped the components of each of the interventions according to the 11 EAPC White Paper domains. This ensured that data synthesis was focused on aspects of care identified by international experts as important in palliative care for people with dementia. This provided insights on potential gaps and room for improvement that could better inform developers of home palliative care interventions in dementia. No subgroup analysis was conducted to look at the difference between specialist palliative care services and non-specialist palliative care interventions due to low number of articles relating to specialist palliative care.
Results
In all, three articles met the inclusion criteria for specialist palliative care services and six for non-specialist palliative care interventions. The overview of the study selection is depicted in Figure 1.
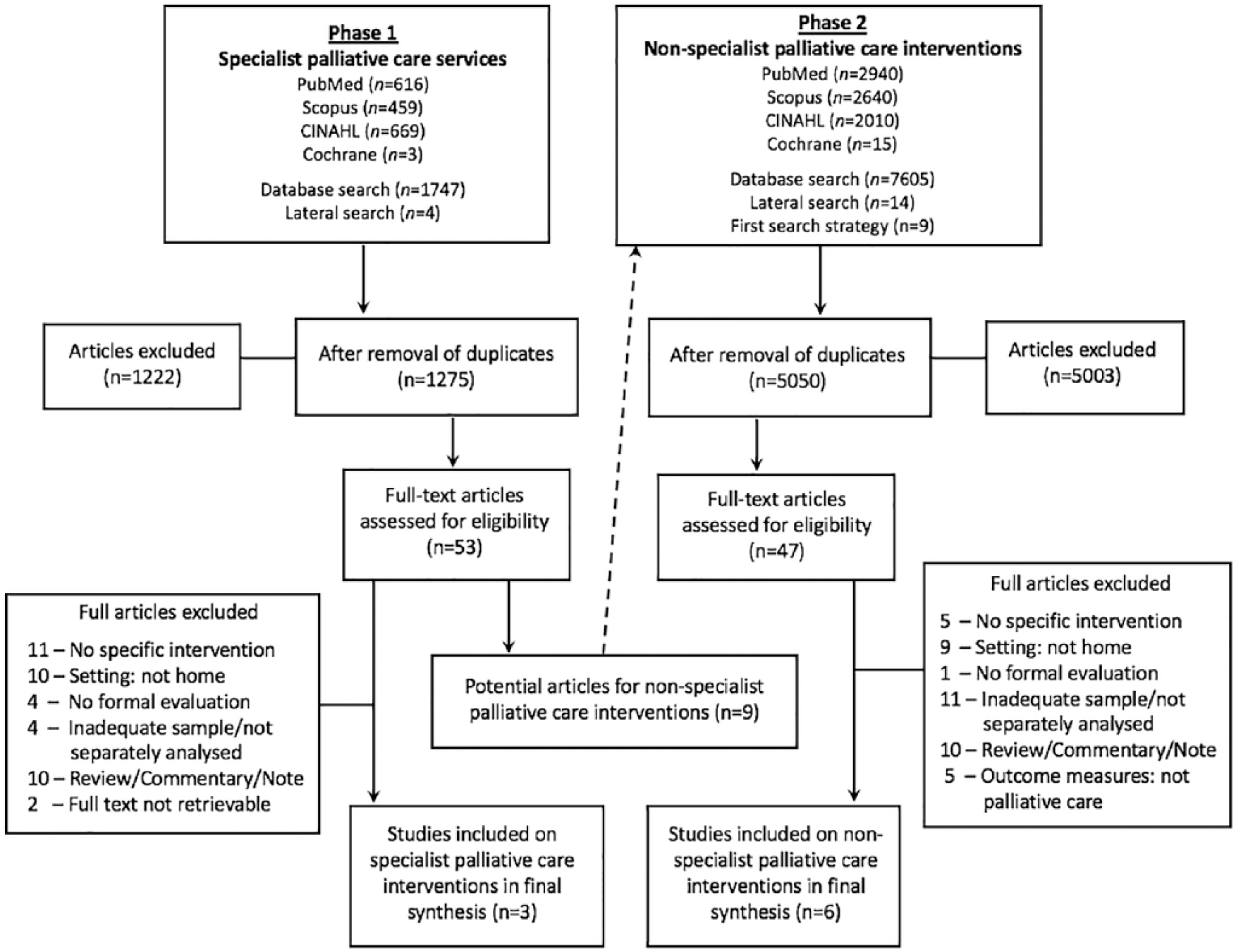
PRISMA flowchart of selection process.
General overview of the studies
We identified three studies evaluating specialist palliative care services19–21 and five evaluating non-specialist palliative care interventions (Table 2).22–27 One study on non-specialist palliative care intervention was reported in two articles.22,23 Four of the studies presented evidence from the United States,19,20,24,25 two from Italy,22,23,27 and one each from the United Kingdom 26 and Japan. 21 Of the studies, four were RCTs,21–24 two retrospective case-control studies,19,25 one retrospective cross-sectional study 20 and one with an unclear study design. 27 All studies used quantitative methods, one of which also used qualitative methods. 20 All studies included both male and female participants, with the majority in their 80s and women. Seven studies included people with dementia and one included terminally ill participants, 64% of whom had dementia. 20 All interventions aimed to improve end-of-life care for people with dementia living at home, while one offered additional support to family caregivers. 24
General overview of the included studies on palliative care interventions (
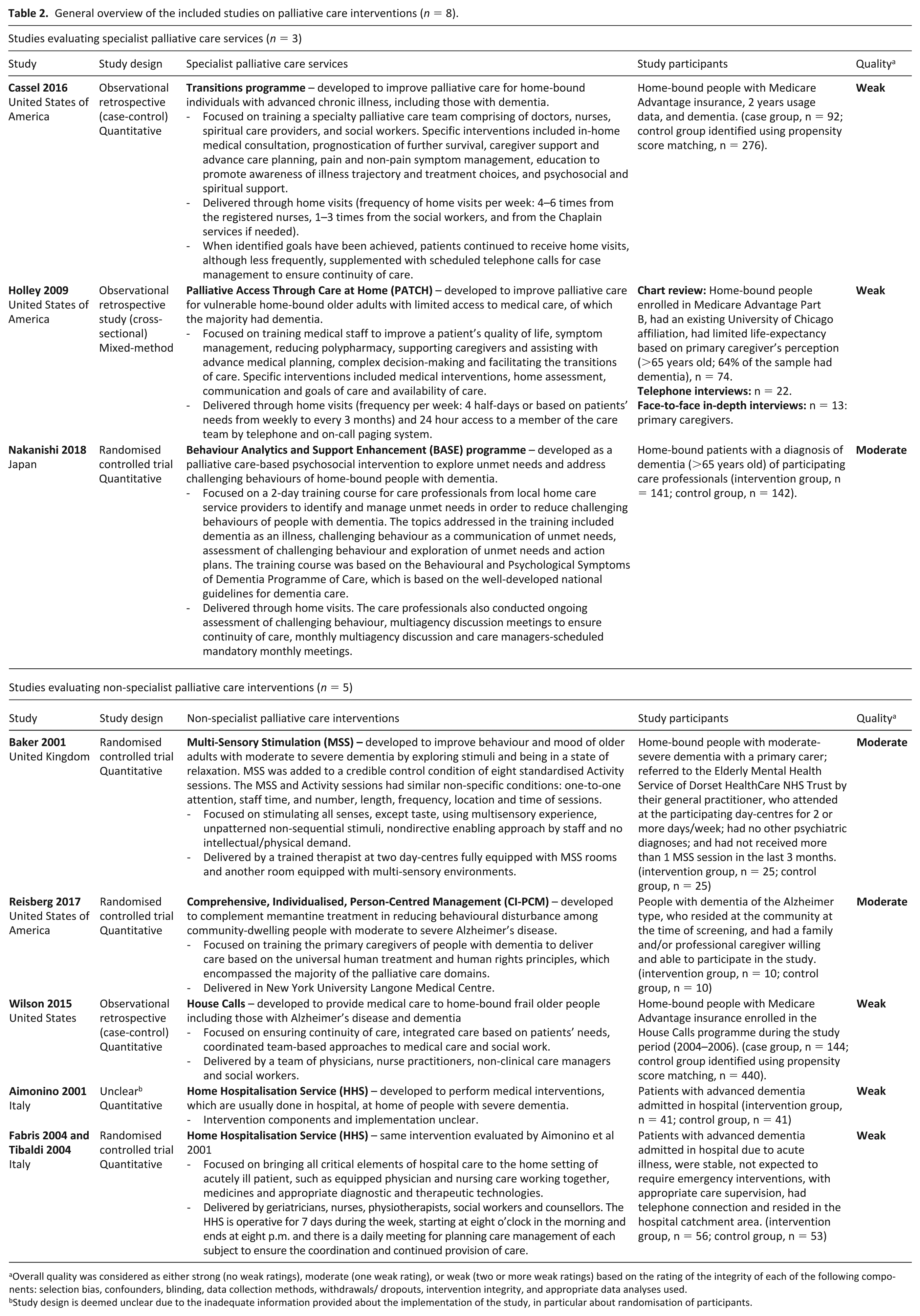
Overall quality was considered as either strong (no weak ratings), moderate (one weak rating), or weak (two or more weak ratings) based on the rating of the integrity of each of the following components: selection bias, confounders, blinding, data collection methods, withdrawals/ dropouts, intervention integrity, and appropriate data analyses used.
Study design is deemed unclear due to the inadequate information provided about the implementation of the study, in particular about randomisation of participants.
Quality of the evidence
Overall, five of the eight studies were considered of weak quality, particularly due to high risk for selection bias, inability to achieve blinding and inability to clearly measure and/or report the integrity and consistency of the intervention (Table 2).19,20,22,23,25,27 Of these studies, three did not control for confounders and used inappropriate data analysis techniques,20,22,23,27 while the other two studies took potential confounders into account by using propensity score matching to identify control group and analysed the data appropriately.19,25 These five studies received moderate ratings for using health/medical records as the source of data, but the procedures for data collection were inadequately reported.
The three other studies were assessed as being of moderate quality, two received a weak rating for not achieving blinding,21,26 while the other one received a weak rating for selection bias due to a small sample size. 24 These studies received strong ratings for study design, confounders, data collection, withdrawals/drop-outs, intervention integrity, and the data analysis techniques used. The quality of the qualitative data from the mixed-methods study was not assessed, as these data were used solely to identify potential barriers and facilitators to implementing the intervention. 20 (See Supplementary file 3 for the composite component ratings for each study).
Palliative care interventions for people with dementia living at home
Overview of specialist palliative care services
We identified three specialist palliative care services, all of which focused on educating the multi-disciplinary healthcare team (Table 2).19–21 Transitions programme 19 and Palliative Access Through Care at Home (PATCH) 20 were specialist palliative care services delivered by trained specialist palliative care team comprising typically of geriatricians, nurses and social workers. Behaviour Analytics & Support Enhancement (BASE) is a palliative care-based psychosocial intervention delivered by trained professional home care providers, who aimed to explore unmet needs and address challenging behaviours of people with dementia in coordination with a multidisciplinary healthcare team. 21 These interventions were delivered through home visits, ranging from four to six times per week or based on patients’ needs. Each intervention had multiple components, such as symptom management, medical consultation, reduction of polypharmacy, assistance with advance care planning and transitions of care, and psychosocial and spiritual support.
Overview of non-specialist palliative care interventions
Non-specialist palliative care encompassed a diverse range of interventions for people with advanced dementia (Table 2). Two studies aimed to address behavioural disturbances, one by using multi-sensory stimulation (MSS) provided in day-care centres 26 and one by training and supporting primary caregivers, that is, formal or family caregiver, to use the Comprehensive, Individualised, Person-Centred Management (CI-PCM) approach. 24 The other three studies aimed to bring care that is usually offered in institutions or hospitals to patients’ homes. One of these studies evaluated the medical care offered by House Calls 25 and two evaluated the diagnostic and therapeutic treatments for acute illnesses offered by Home Hospitalisation Service.22,23,27
Outcomes of home palliative care interventions in dementia
The outcomes of home palliative care interventions for people with dementia are summarised in Table 3.
Outcomes of home palliative care interventions for people with dementia.
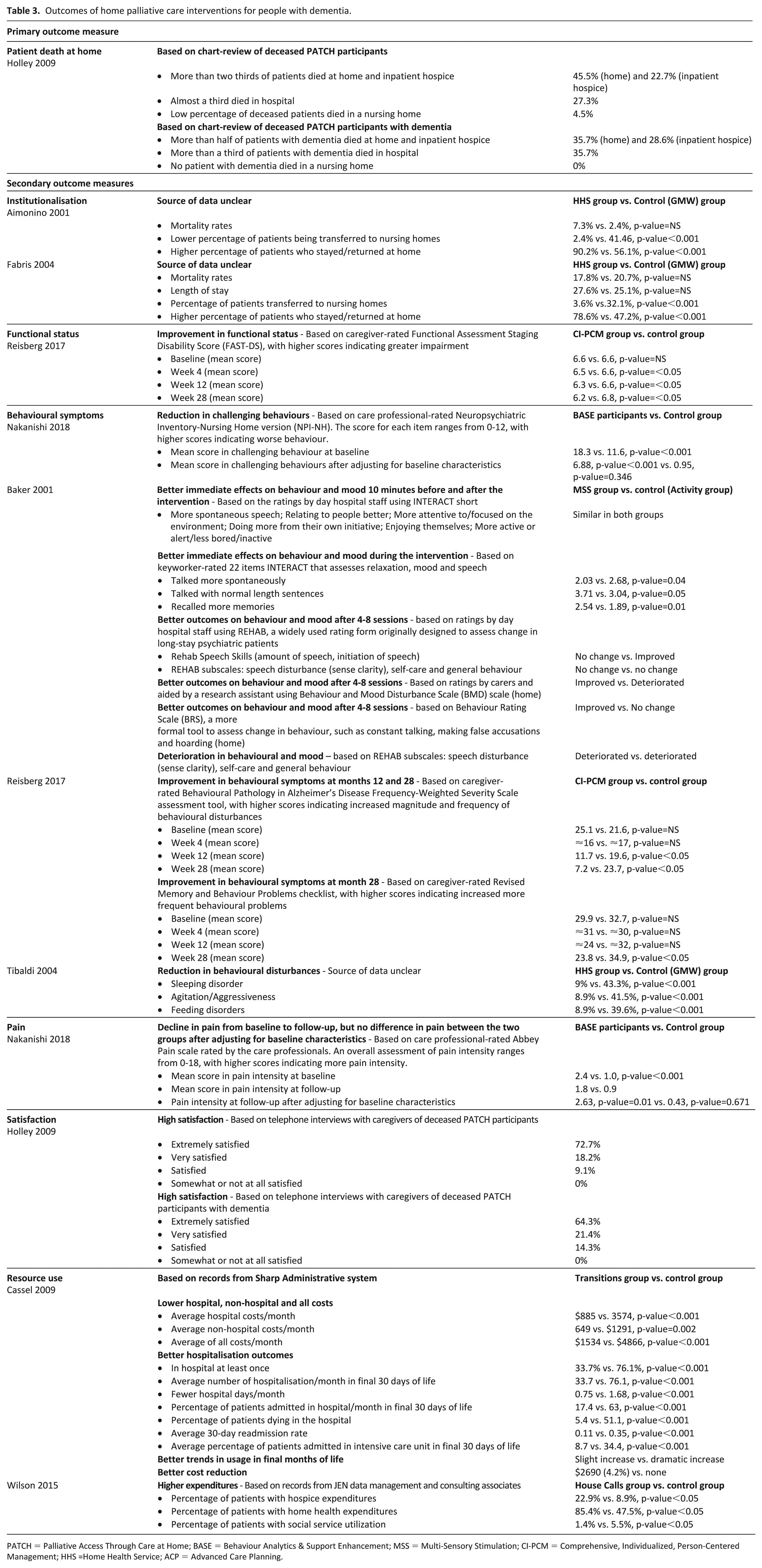
PATCH = Palliative Access Through Care at Home; BASE = Behaviour Analytics & Support Enhancement; MSS = Multi-Sensory Stimulation; CI-PCM = Comprehensive, Individualized, Person-Centered Management; HHS =Home Health Service; ACP = advanced care planning.
Patient death at home
One of the studies (retrospective cross-sectional study of weak quality) reported outcome on patient death at home. 20 This study showed that about two thirds of deceased participants with dementia who received palliative care from the trained specialist care team died at home and inpatient hospice. However, this evidence on effectiveness was inconclusive, as it relied exclusively on cross-sectional data collected through chart review and thus there was no control group or before and after data.
Institutionalisation
Two of the studies reported outcome on institutionalisation. These studies (one RCT and one unclear study design, both studies of weak quality) evaluated Home Hospitalisation Service.22,27 They suggested that people with advanced dementia who received diagnostic and acute care at their own homes were less likely to be transferred to nursing homes and more likely to stay at home.
Functional status
One of the studies (RCT of moderate quality) evaluated functional status as an outcome. 24 This study showed that the functional status of people with dementia who received care from the trained primary caregivers on CI-PCM approach were more likely to improve than those who did not at all follow-up measurements (months 4, 12 and 28).
Behavioural symptoms common at the end of life
Four of the studies assessed behavioural symptoms, all of which suggested that home palliative care interventions are more effective than usual care in reducing behavioural disturbances.21,23,24,26 Three studies (all RCTs of moderate quality) suggested positive effects of home palliative care interventions on behavioural disturbances of people with dementia. These interventions included the MSS sessions offered in day-care centres 26 and the training courses on BASE for professional home care providers 21 and on CI-PCM approach for primary caregivers. 24 The fourth study (RCT of weak quality) reported marginally significant effect in the same direction on sleeping disorder, agitation/aggressiveness and feeding disorders. 23 However, evidence on the duration of effects was conflicting. One study found long-term positive effects of the training course on CI-PCM approach for primary caregivers on behavioural symptoms, 24 whereas another study found that behavioural symptoms deteriorated after the MSS sessions had ceased. This deterioration has been attributed to potential withdrawal effect from the one-to-one session and the appropriate stimulation. 26
Pain
One of the studies (RCT of moderate quality) reported pain as an outcome, but results were inconclusive. 21 This study evaluated the training course on BASE, a palliative care-based psychosocial intervention offered to home care professionals who were responsible for assessing the pain of participants with dementia and discussing this assessment with the multidisciplinary team. This study suggested that compared with control group, the participants in the intervention group had significant pain reduction from baseline to follow-up assessment. However, after controlling for baseline characteristics at follow-up, no significant difference in pain was found between the two groups potentially due to the higher pain score in the intervention group at follow-up. This higher pain score was attributed to the enhanced pain assessment conducted by the trained care professionals in the intervention group, whereas the care professionals who did not receive the training course may have underestimated or remained unaware of the need for pain assessment in dementia.
Satisfaction
One of the studies (retrospective cross-sectional study of weak quality) reported outcome on satisfaction. This study suggested generally high satisfaction rate of primary caregivers of people with dementia with the care provided by the trained specialist palliative care team. 20 However, this evidence was inconclusive, as it relied exclusively on a cross-sectional survey of 22 primary caregivers of deceased people with dementia who received the intervention.
Resource use
Two of the studies (both retrospective case-control studies of weak quality) reported outcomes on resource use based on estimated costs.19,25 Compared to usual care, the transitions programme training of specialist palliative care team resulted in lower hospital, non-hospital and all costs. It also resulted in better hospitalisation outcomes, in terms of frequency, length of stay in hospital, patient deaths in hospital, hospitalisation rates, readmission rates and admission in the intensive care unit in the final 30days of life. They also found that in the final 6 months of life, resource use in the intervention group had only increased slightly, compared with a larger increase in the control group. Net cost reduction was also better in the intervention group than the control group. 19 The other study on House Calls showed that compared to control group, patients with dementia who received medical care in their own homes were more likely to have home health and hospice expenditures, but less likely to have social health expenditures. The authors attributed the relatively low social services utilisation to the limited financial reimbursement available for social services. 25
Facilitators and barriers to implementing the interventions
None of the studies systematically investigated facilitators and barriers to implementing the interventions. Instead, we identified information in the discussion sections of five of the studies, which could be considered facilitators and barriers to implementation.19–21,24,27 Some intervention components may have facilitated the implementation of the interventions, including the 24-hour access to a medical practitioner 20 and the continuing provision of tailored interventions. 21 Some formal caregiver characteristics were also discussed as facilitators to implementation, such as their active engagement, 21 reliability 24 and dedicated and highly qualified teamwork. 27 The limited time allocated to implementing the interventions was considered a barrier to implementation, because it may have hindered behavioural change and the achievement of long-term effects.19,21
Studies mapped according to the EAPC White Paper domains
All of the interventions aimed to optimally manage symptoms at the end of life, such as pain or acute medical illnesses (Table 4). The majority of them also ensured the continuity of care by either directly facilitating the transitions of care between settings19,20,25 or conducting multidisciplinary discussions and collaboration21–23,27 and caregiver support meetings. 24 They also addressed the psychosocial domain by providing psychosocial support19,20,22,23,25,27 or managing behavioural symptoms.21,24 Four EAPC White Paper domains were rarely addressed by the interventions: applicability of palliative care, societal and ethical issues, prognostication and timely recognition of dying, and avoidance of overly aggressive, burdensome or futile treatments. In addition, ‘setting care goals and advance care planning’ was addressed by only three of the eight evaluated interventions.19,20,22,23 Compared to non-specialist palliative care interventions, specialist palliative care services focused more on training the multidisciplinary healthcare team and addressed four to nine domains at once.19–21
Studies mapped according to the EAPC White Paper domains.
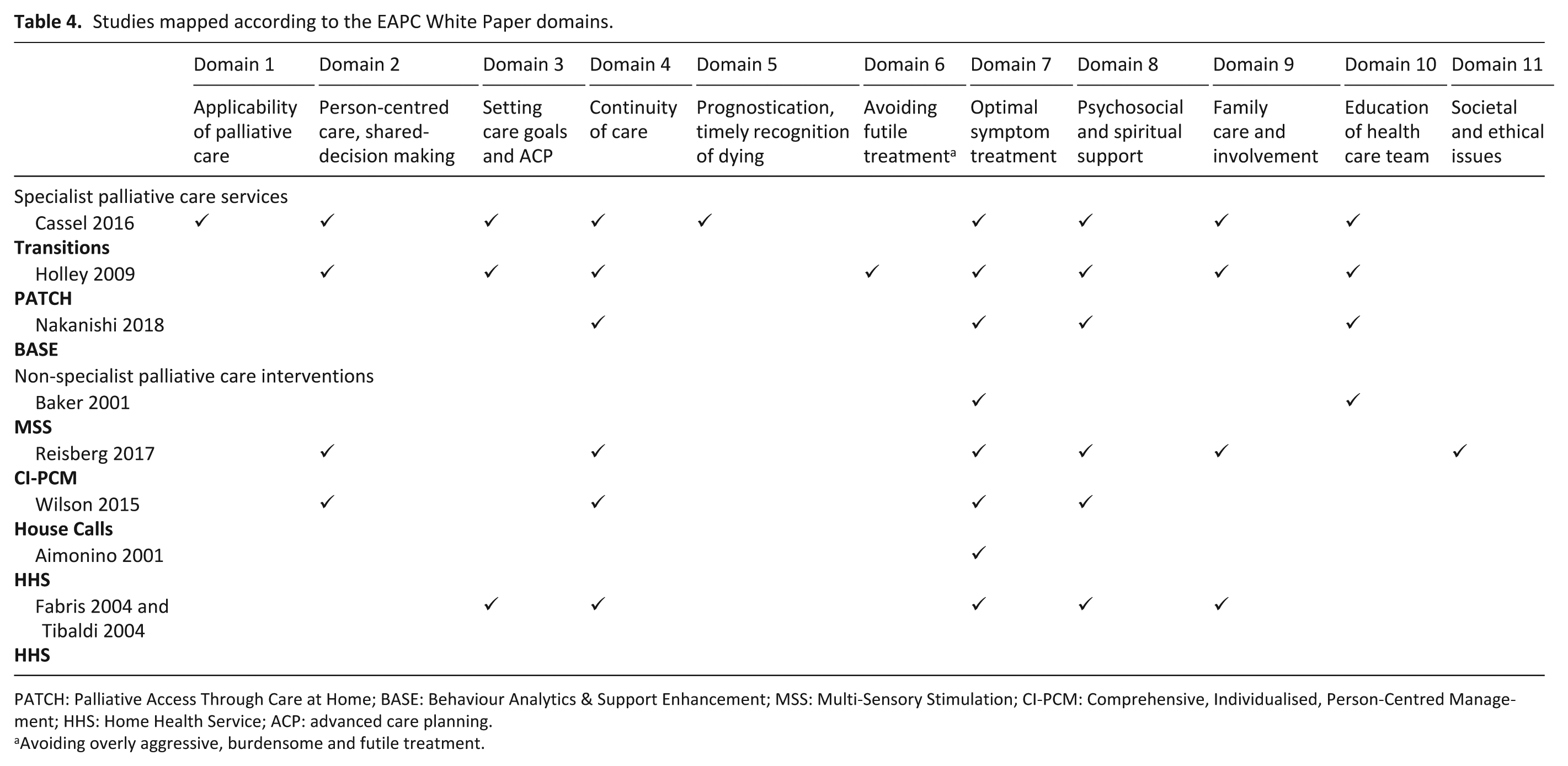
PATCH: Palliative Access Through Care at Home; BASE: Behaviour Analytics & Support Enhancement; MSS: Multi-Sensory Stimulation; CI-PCM: Comprehensive, Individualised, Person-Centred Management; HHS: Home Health Service; ACP: advanced care planning.
Avoiding overly aggressive, burdensome and futile treatment.
Discussion
We retrieved eight studies which evaluated interventions aimed at improving end-of-life care outcomes for people with dementia living at home; three involving specialist palliative care services and five non-specialist palliative care interventions. None of the studies were of high quality, mostly due to high risk for selection bias and the inability to achieve blinding and to measure/report intervention integrity. We found weak evidence showing that home palliative care interventions in dementia can improve end-of-life care outcomes relating to institutionalisation, estimated resource use and functional status. There was moderate evidence of beneficial effects on behavioural symptoms arising from the person’s cognitive and communication problems, but the evidence on whether these effects would last was contradictory. Although the evidence on pain reduction was limited and inconclusive, there was some evidence of enhanced pain assessment. Evidence on facilitators and barriers was not systematically investigated and our findings are based on limited information provided in the discussion sections of the included studies. The mapping of the studies according to the EAPC domains highlighted the main preoccupations and focus of the interventions reviewed.
The existing evidence base is insufficient and generally too weak to robustly assess the effects of home palliative care interventions in dementia. Nonetheless, the potential benefits of the interventions in improving behavioural symptoms and enhancing pain assessment are consistent with earlier findings13,14 and address key issues in dementia care.28–30 Healthcare practitioners are often uncertain how to support people with dementia whose behaviours they find challenging. 31 Pain is also highly prevalent in older adults but poorly assessed in those with dementia,30,32 which may result in patient distress and related behaviours. 33 Managing behavioural symptoms and assessing pain are not only important for patients with dementia. It may also contribute to reducing the burden of family caregivers who are central to enabling patients to stay at home.34,35 While we know that promoting caregiver well-being is important, 36 it is notable that only one of the identified interventions offered additional support to address the emotional and mental needs of family caregivers.
The facilitators and barriers identified are in line with existing evidence37,38 that highlights the importance of networks of support and easy access to tailored interventions. For example, the 24-hour access to medical practitioners and the active engagement of caregivers could enhance the accessibility of the interventions, while the tailoring of interventions based on patients’ needs may increase their applicability for users.
The three most frequently addressed EAPC domains in the studies reviewed (optimal symptom management, continuity of care and psychosocial support) reflect clinician priorities and the core values of palliative care, irrespective of the reason for dying. 39 However, another EAPC priority ‘person-centred care, communication and shared-decision-making’ was only modestly addressed by the interventions. This is surprising when some of the most influential writing on person-centred care is situated in the dementia literature. 40 The studies failed to map according to ‘applicability of palliative care’ and ‘societal/ethical issues’ even though this is a patient population that is increasing but poorly understood by commissioners and policymakers and stigmatised in society. 41 Other domains that were under-represented in the evidence reviewed: prognostication and timely recognition of dying, avoidance of overly aggressive, burdensome or futile treatments and setting of care goals and advance care planning are similarly problematic areas in dementia care. For instance, despite the fact that most older people prefer to die at home, 7 there may still be a substantial proportion of older people with dementia dying in hospitals, 30 which is likely to occur if the palliative phase is not promptly recognised. Also, progressive cognitive decline is inherent in advanced dementia, which hinders the communication between healthcare providers and people with dementia. 1
Implications for research and practice
The lack of evidence suggests that palliative care interventions have been given a low priority in dementia care, particularly in the home setting. 12 This may also be because most of the evidence on palliative care interventions for people with dementia is still situated in the nursing home setting, 12 where in many countries, the majority of people with dementia spend their final months or years of life. 42 Findings from the mapping of the studies according to the EAPC domains raised a question about whether research to date had given adequate attention to the specific issues and challenges experienced by people with dementia. It also highlighted the problems of transferring learnings about symptom management, continuity of care and psychosocial support from one specialty (e.g. cancer care) to the experience of dying with and from dementia. Symptom management should always be a main priority. However, the EAPC domains where there are gaps, are the domains that are very particular to the experiences of living and dying with dementia. Dementia is a socially stigmatised condition, with a highly variable and protracted dying trajectory and one where decisions to treat and/or avoid burdensome treatments involve multiple stakeholders.43,44 The review thus demonstrates that it may still be the early days in research for people with dementia living and dying at home. High-quality dementia-specific research is therefore required to further support the evidence base for palliative care interventions to be a routine care for people with dementia living and dying at home. 10 In designing such palliative care interventions, it is crucial to also consider strategies that would address the specific needs of home-dwelling people dying with and from dementia, for example, prognostication of dying, avoidance of overly aggressive treatments or advance care planning, as well as interventions to assess and address the individual needs of family caregivers.
This systematic review also highlights a number of gaps in the evidence. We found a lack of consensus on the outcomes used, which limited comparisons and meta-analysis. Important end-of-life care outcomes, for example,. patient death at home and quality of life were also not consistently measured.7,39 These issues around outcome measures may be addressed by two ongoing studies aiming to develop standard core outcome sets in dementia, one of which focuses on the home setting, while the other study involves people with dementia in research.45,46 Although the core outcome sets to be developed can be relevant and applicable for people with dementia, it is still important to further explore the extent to which these outcomes would be relevant and applicable in evaluating home palliative care interventions in dementia. Guidelines on how to develop such core outcome sets exist, such as the handbook developed by the Core Outcome Measures in Effectiveness Trials (COMET) initiative. The COMET initiative aims to guide the development of core outcome sets by bringing together relevant key stakeholders, including patients. 47 Most of the studies were also appraised to have weak quality in part due to their inability to measure/report intervention integrity. This has been found as a persistent issue in clinical trials in palliative care, 48 which could be addressed by using standard reporting tools, for example, CONSORT statement. 49 Evidence on cost-effectiveness is also scarce. While some studies reported resource use, it was solely based on estimated costs while disregarding health benefits. Performing concurrent cost-effectiveness analysis using existing guidance could provide a better view of interventions that could potentially yield the greatest improvement in dementia care for the least resources. 50 Finally, evidence on facilitators and barriers was limited. This could be addressed by undertaking process evaluation, which is increasingly being recognised as an integral part of designing and evaluating complex interventions such as palliative care interventions. Performing process evaluations in accordance with the Medical Research Council (MRC) guidance would allow the critical exploration of factors and causal mechanisms that could explain variations in observed outcomes. 51
Strengths and limitations
We systematically and rigorously searched the literature for existing studies on home palliative care interventions in dementia. It was a strength of the study that we drew on the EAPC White Paper to structure the data extraction and analysis. This demonstrated how the evidence was distributed according to an international consensus on what should be the key activities and focus of palliative care in dementia. However, we only found eight relevant studies, none of which were of high quality. The evidence of effectiveness therefore needs to be interpreted with caution. We adopted a broad definition of non-specialist palliative care interventions which may mean that relevant studies were missed. However, our searches were systematic and electronic database searching was supplemented with lateral searching. Our decision to only include quantitative studies, and associated process evaluations, may explain the limited evidence on facilitators and barriers to implementation. However, forward citation tracking of the included studies also did not reveal any relevant studies. To better identify studies that could yield valuable information on factors influencing the implementation of home palliative care interventions in dementia, future reviews should consider the inclusion of both qualitative and quantitative studies. We would also recommend a regular update of the systematic review, as new scientific evidence on factors influencing the implementation of home palliative care interventions emerges, such as the follow-up study of Nakanishi et al. 52 Finally, subjectivity may have been introduced in the mapping of the intervention components according to the EAPC White Paper domains. We dealt with this challenge by discussion within the research team.
Conclusion
The review offers evidence on palliative care interventions for people with dementia living at home and highlights the paucity of high-quality studies in this area. The review emphasises the need for more rigorous and comprehensive research which considers the identified gaps in the evidence and addresses the specific issues and challenges that dying at home with or from dementia poses. The EAPC’s definition of optimal palliative care in dementia provided a useful framework for a systematic assessment of the range and focus of evidence of what is effective for people with dementia living and dying at home.
Footnotes
Acknowledgements
The authors thank Angela Dimond for her assistance in developing the search strings for this systematic review. All authors meet the criteria for authorship as stated by the International Committee of Medical Journal Editors authorship guidelines. All authors have given the final approval of the manuscript to be submitted for publication.
Declaration of conflicts of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Marie Curie Innovative Training Network (ITN) action, H2020-MSCA-ITN-2015 (under grant agreement number 676265). The funding body did not have any role in data analysis, interpretation, and the writing of the manuscript.
