Abstract
Background:
The effectiveness of cardiology-led palliative care is unknown; we have insufficient information to conduct a full trial.
Aim:
To assess the feasibility (recruitment/retention, data quality, variability/sample size estimation, safety) of a clinical trial of palliative cardiology effectiveness.
Design:
Non-randomised feasibility.
Setting/participants:
Unmatched symptomatic heart failure patients on optimal cardiac treatment from (1) cardiology-led palliative service (caring together group) and (2) heart failure liaison service (usual care group).
Outcomes/safety:
Symptoms (Edmonton Symptom Assessment Scale), Kansas City Cardiomyopathy Questionnaire, performance, understanding of disease, anticipatory care planning, cost-effectiveness, survival and carer burden.
Results:
A total of 77 participants (caring together group = 43; usual care group = 34) were enrolled (53% men; mean age 77 years (33–100)). The caring together group scored worse in Edmonton Symptom Assessment Scale (43.5 vs 35.2) and Kansas City Cardiomyopathy Questionnaire (35.4 vs 39.9). The caring together group had a lower consent/screen ratio (1:1.7 vs 1: 2.8) and few died before approach (0.08% vs 16%) or declined invitation (17% vs 37%).
Data quality:
At 4 months, 74% in the caring together group and 71% in the usual care group provided data. Most attrition was due to death or deterioration. Data quality in self-report measures was otherwise good.
Safety:
There was no difference in survival. Symptoms and quality of life improved in both groups. A future trial requires 141 (202 allowing 30% attrition) to detect a minimal clinical difference (1 point) in Edmonton Symptom Assessment Scale score for breathlessness (80% power). More participants (176; 252 allowing 30% attrition) are needed to detect a 10.5 change in Kansas City Cardiomyopathy Questionnaire score (80% power; minimum clinical difference = 5).
Conclusion:
A trial to test the clinical effectiveness (improvement in breathlessness) of cardiology-led palliative care is feasible.
Compared with people with cancer, those with advanced heart failure have equivalent palliative care needs with poorer access to specialist palliative care.
A palliative care approach led by cardiology teams and access to specialist palliative care according to need rather than prognosis is recommended but poorly implemented.
The evidence base for palliative care in heart failure is less robust than that for cancer and there are no published prospectively collected data for cardiology-led palliative care services.
Novel prospectively collected data for cardiology-led palliative care services which show that a future trial of cardiology-led palliative care is feasible in terms of recruitment to a clinical study (note: willingness to randomisation was not tested), data quality and sample size.
Data generating a hypothesis that cardiology-led palliative care may safely improve advance care planning and patients’ understanding of their condition, enable care at home and reduce costs.
Findings should be tested in an adequately powered randomised controlled trial.
Future studies should identify and randomise patients with more severe or complex palliative care needs to ensure optimal use of finite specialist palliative care resources.
Introduction
Compared with people with cancer, those with advanced heart failure have equivalent palliative care needs with poorer access to specialist palliative care.1–6 Palliative care is recommended,7–10 but poorly implemented.11–15
Phase III trials of specialist palliative care for people with heart failure16–19 report cost-effective20,21 benefit with specialist palliative care without compromising survival. However, most palliative concerns in people with heart failure can be managed by the usual care team using a patient-centred approach with access to specialist palliative care for complex issues only.22,23 Previous integrated service descriptions do not include patient-reported outcomes,24–27 and the effectiveness of cardiology-led palliative care is untested.
A partnership between two charities (Marie Curie and British Heart Foundation) and a Scottish NHS health board developed cardiology-led models of heart failure palliative care (‘The Caring Together (CT) Programme’ 28 ).
The study objectives were to assess the feasibility of a future randomised controlled trial (RCT) in terms of (1) recruitment/retention, (2) data quality, (3) variability/sample size estimation and (4) safety.
Methods
Expanded methods are available online.
Study design
This prospective feasibility cohort single-site study (data collection at baseline, 2 and 4 months) used two heart failure outpatient groups. The participants continued 2 monthly measures until the last recruit completed 4 months’ follow-up.
Ethical (NRES Committee London – Camberwell St Giles. REF 14/LO/1813; 07.10.2014) and institutional approvals were obtained prior to recruitment. The participants gave written informed consent.
Participants
Participants were adults with symptomatic heart failure (reduced or normal left ventricular ejection fraction; HFrEF, HFnEF) and family carers. Participants with at least one CT clinic attendance formed the caring together group (CTG). A convenience unmatched usual care group (UCG) was formed from heart failure liaison nurse service patients fulfilling CT criteria but who had not been referred. The risk of contamination was deemed to be minimal. 29
Caring together intervention
Box 1 shows the referral criteria and components. Collaborative primary and secondary care is led by a consultant cardiologist and a heart failure palliative care nurse consultant, through a palliative cardiology multi-disciplinary team (MDT).
Caring together components (adapted, Caring Together Manualisation Report, 2014). 28

Patients with reduced or normal left ventricular function are eligible.
Usual care
The liaison nurse service and other consultant cardiologists’ clinics provide education, self-care promotion, medical therapy up-titration and patient/family support (HFrEF only).
Study measures
Recruitment: screen-to-consent ratio, reasons for non-consent, recruitment rate, retention.
Data quality of protocol measures. (a) Demographic and clinical measures at baseline: New York Heart Association (NYHA) status;
30
Australian-modified Karnofsky Performance Status (AKPS);
31
Charlson et al.’s
32
comorbidity index; medication-optimised cardiac treatment; pulse, heart rate, presence/absence of oedema and jugular venous pressure; urea and electrolytes; and echocardiogram (HFrEF and HFnEF; for HFrEF, ‘mild, moderate, severe’, ejection fraction). (b) Patient measures at baseline and follow-up: AKPS, Edmonton Symptom Assessment Scale – revised (ESAS-r),
33
Kansas City Cardiomyopathy Questionnaire (KCCQ-12),34–36 EuroQol EQ-5D-5L,
37
Hospital Anxiety and Depression Scale (HADS),
38
patient understanding (Likert), health service utilisation and costs (including medication) and Zarit 6 caregiver burden (carers).
39
(c) Clinical measures at baseline and follow-up: survival, clinical documentation of anticipatory care planning (ACP).
Candidate primary outcome data variability for power estimation.
Safety: survival (6 months) and outcomes (4 months).
Data were not collected on willingness to be randomised.
Statistical analysis
Data are presented using descriptive statistics. Sample size estimations were conducted based on variability data around candidate primary outcomes.
Safety assessment: (1) Cox proportional hazards models were used for between-group survival differences and (2) Group 4 month outcomes comparison by study entry group used two-sample t tests (continuous) and Fisher’s exact tests (categorical), unless otherwise stated. No adjustments were made for multiple comparisons. Missing data were not imputed. A ‘per-protocol’ (PP) repeated-measures model analysis excluded UCG participants referred to CT. Two random-effects repeated-measures regression models with (1) the KCCQ-12 and (2) the EQ-5D-5L, as the dependent outcomes adjusted for age, sex and baseline ESAS-r scores, were used. The estimated between-group differences were reported with 95% confidence intervals (CIs) and p values. PP analysis was completed for the initial repeated-measures model (data available).
Incremental costs and outcomes associated with CTG were determined. Resource use items included health service utilisation and medication. Analyses were conducted using SAS for Windows version 9.3. Formal power calculation was inappropriate.
Results
A total of 77 participants (43 CTG and 34 UCG) were recruited (8 April to 18 December 2015). A follow-up of 4 months is presented except for the repeated-measures model, survival and health service utilisation (8 months) and study flow (10 months).
Feasibility measures
Table 1 shows the patient flow. One UCG participant consented for every 2.8 screened versus 1 per 1.4 CTG screened. Fewer in the CTG died before approach (0.08% vs 16%) or declined invitation (17% vs 37%).
Patient flow.
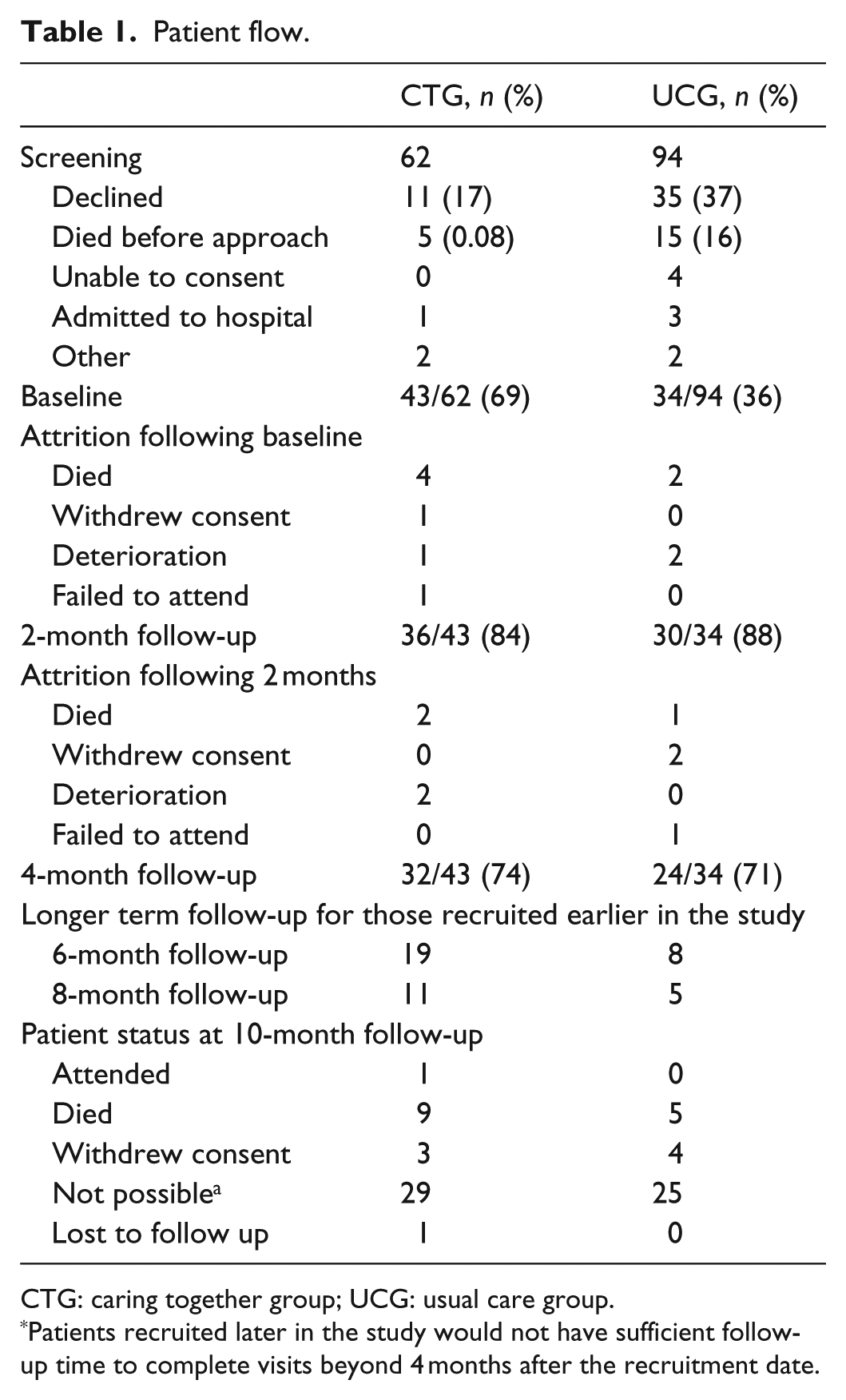
CTG: caring together group; UCG: usual care group.
Patients recruited later in the study would not have sufficient follow-up time to complete visits beyond 4 months after the recruitment date.
Most attrition was due to death or deterioration; those providing follow-up data had better performance status, symptom scores and quality of life (QoL) at baseline (data available). Data quality was otherwise good (Tables 2 and 3).
Outcomes at 4 months.
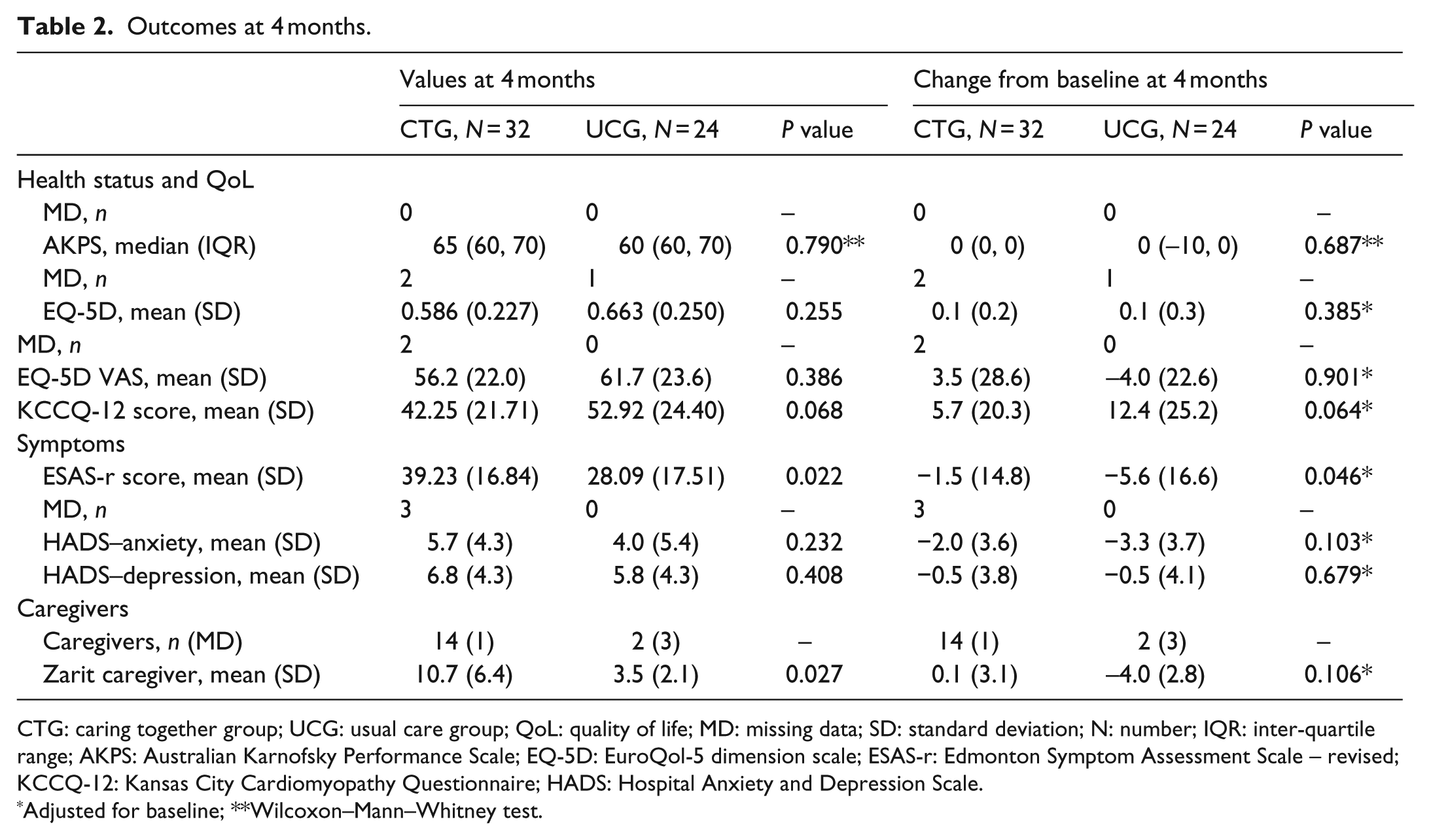
CTG: caring together group; UCG: usual care group; QoL: quality of life; MD: missing data; SD: standard deviation; N: number; IQR: inter-quartile range; AKPS: Australian Karnofsky Performance Scale; EQ-5D: EuroQol-5 dimension scale; ESAS-r: Edmonton Symptom Assessment Scale – revised; KCCQ-12: Kansas City Cardiomyopathy Questionnaire; HADS: Hospital Anxiety and Depression Scale.
Adjusted for baseline; **Wilcoxon–Mann–Whitney test.
Outcomes at 4 months regarding understanding and anticipatory care planning.
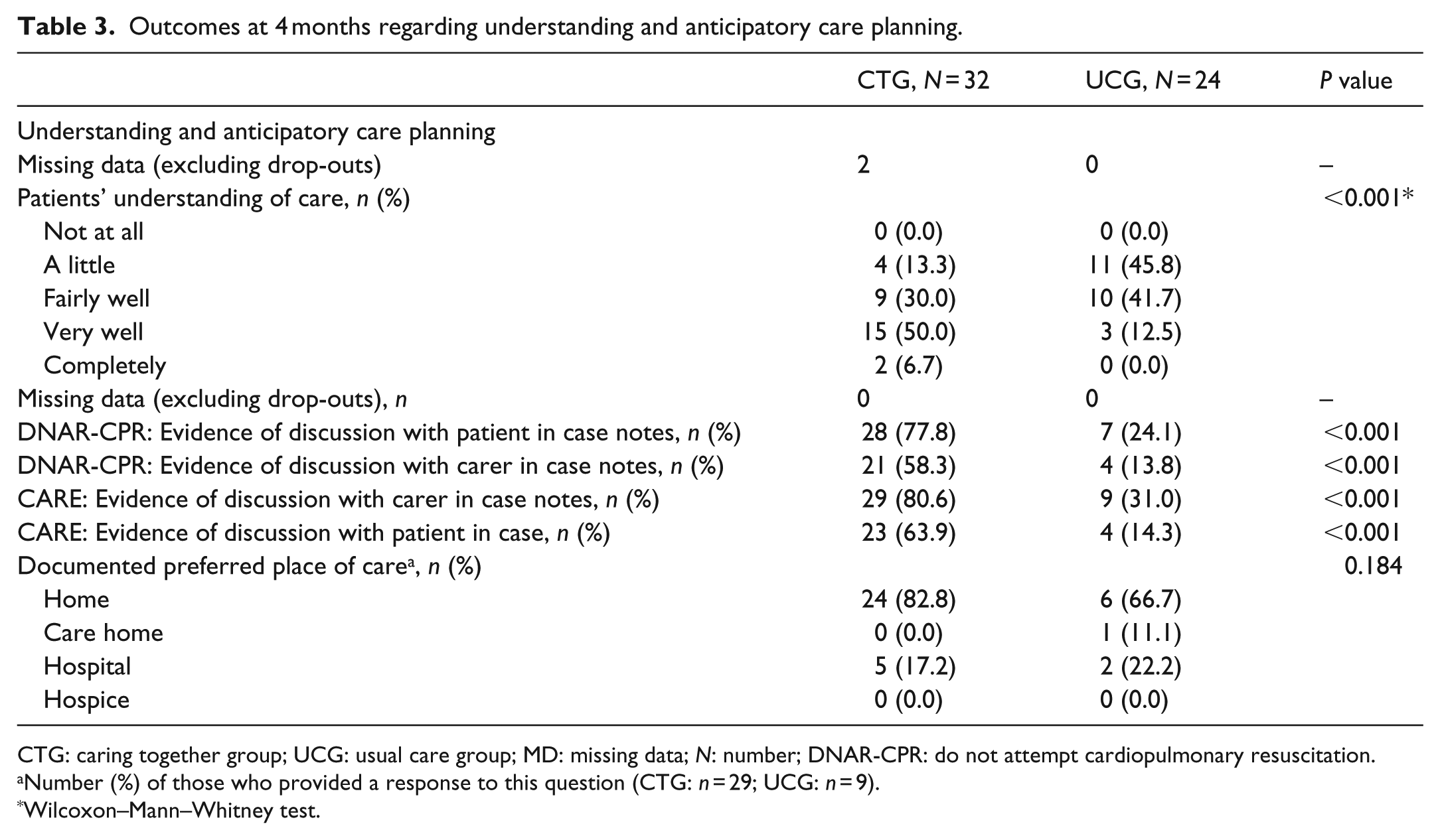
CTG: caring together group; UCG: usual care group; MD: missing data; N: number; DNAR-CPR: do not attempt cardiopulmonary resuscitation.
Number (%) of those who provided a response to this question (CTG: n = 29; UCG: n = 9).
Wilcoxon–Mann–Whitney test.
Baseline characteristics
Both groups were on optimal tolerated cardiac treatment.
There were clinically important between-group differences (Tables 4 and 5). Nearly all UCG participants had HFrEF, but half of the CTG participants had HFnEF (Table 4). CTG patients had worse NYHA class, symptoms (ESAS-r) and quality of life (KCCQ-12), and less deprivation. Most CTG participants had ACP documented and better understanding.
Baseline demographic characteristics.
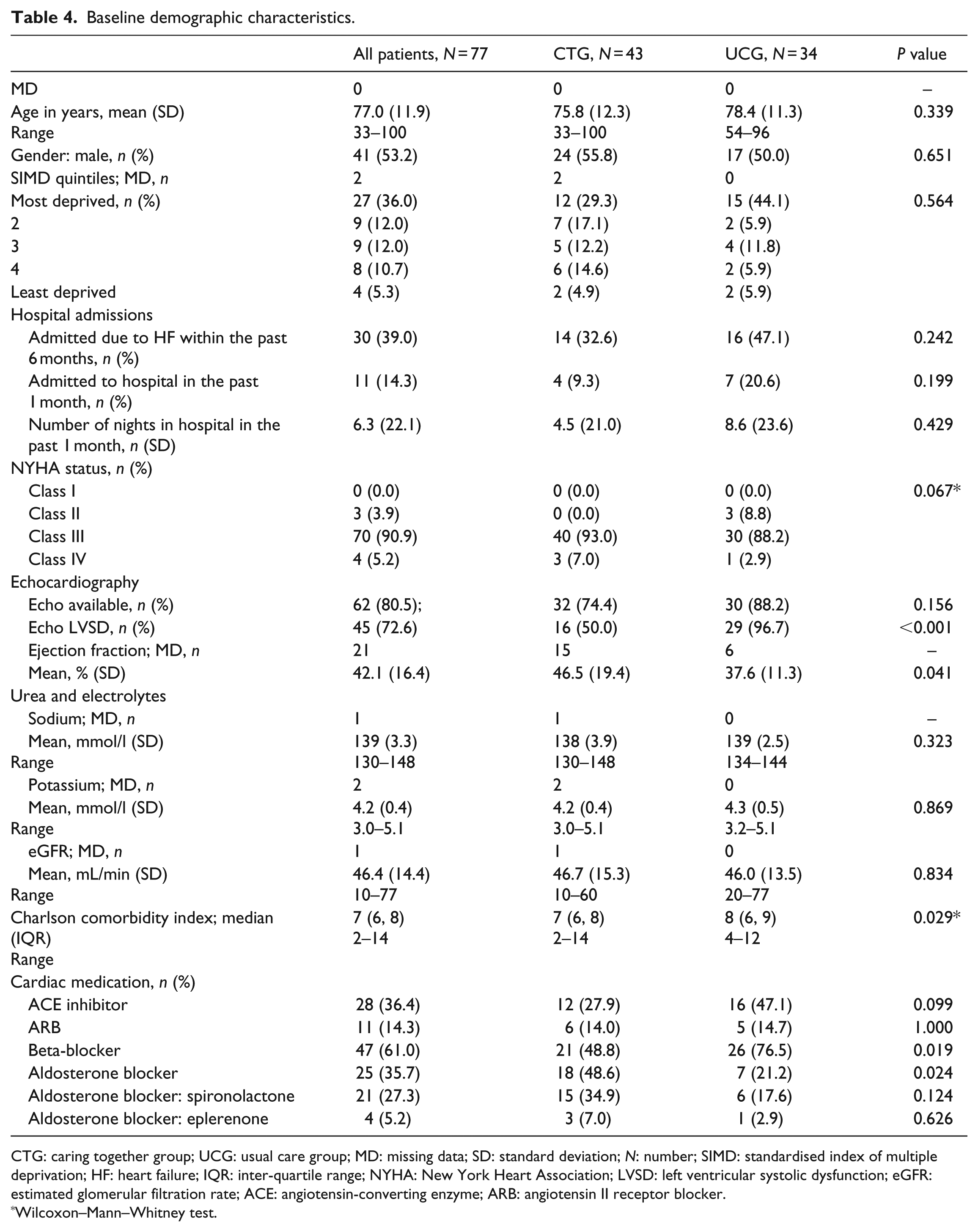
CTG: caring together group; UCG: usual care group; MD: missing data; SD: standard deviation; N: number; SIMD: standardised index of multiple deprivation; HF: heart failure; IQR: inter-quartile range; NYHA: New York Heart Association; LVSD: left ventricular systolic dysfunction; eGFR: estimated glomerular filtration rate; ACE: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker.
Wilcoxon–Mann–Whitney test.
Baseline outcome measures.
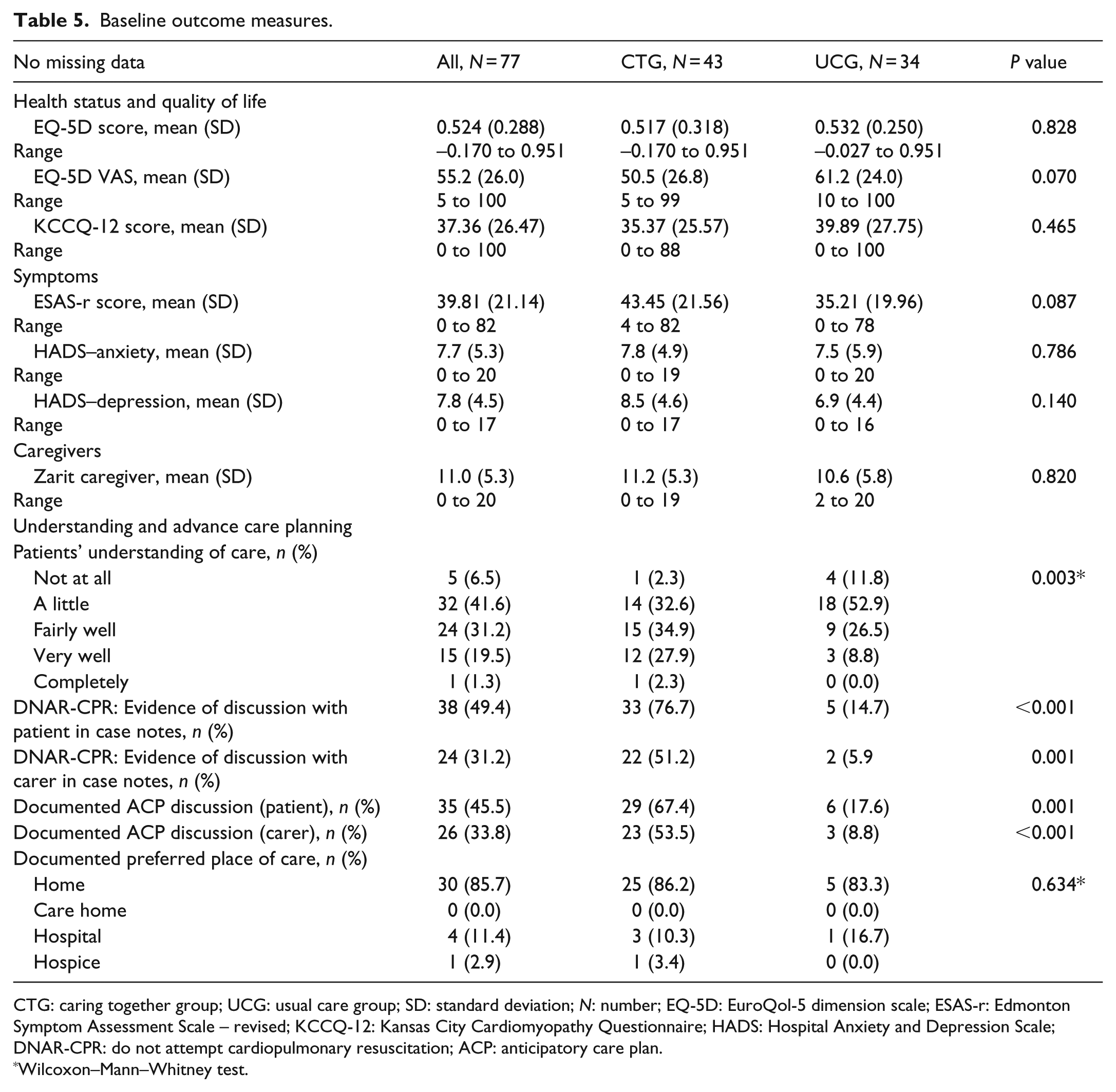
CTG: caring together group; UCG: usual care group; SD: standard deviation; N: number; EQ-5D: EuroQol-5 dimension scale; ESAS-r: Edmonton Symptom Assessment Scale – revised; KCCQ-12: Kansas City Cardiomyopathy Questionnaire; HADS: Hospital Anxiety and Depression Scale; DNAR-CPR: do not attempt cardiopulmonary resuscitation; ACP: anticipatory care plan.
Wilcoxon–Mann–Whitney test.
More UCG participants had prior admissions (47.1% vs 32.6%). All CTG participants had attended CT at least once (median: 8 months; inter-quartile range (IQR): 2–21 months).
Group comparison at follow-up
Overall, the ESAS-r and KCCQ-12 scores improved. At 4 months, adjusted symptom improvement was greater in the UCG (0.046). ACP documentation and understanding remained better in the CTG (p < 0.001). Two UCG participants were transferred to CTG, but there were no differences in the PP analysis findings.
CTG participants had fewer nights in hospital, but more GP visits (eTable 1). CT participants commented on excellent, individually tailored, coordinated care. UCG participants commented on fragmented care, poor communication and different unfamiliar doctors.
There was no between-group difference in survival (eFigure 1 online; time to death).
Repeated-measures model
There were no between-group differences in EQ-5D-5L (p = 0.50) or KCCQ-12 (p = 0.08) at 4 months after adjustment for age, sex or baseline individual ESAS-r (eFigure 2 online).
Health service use and costs
Differences in health service use are shown in eTable 1. Estimated differences in costs show that the average healthcare costs reduced by £785 in CTG. This is statistically insignificant and subject to considerable uncertainty.
Sample size calculation for the main trial
Important clinical differences for KCCQ-12 40 and ESAS (breathlessness) 41 were used. UCG was taken as the population of interest and ESAS (breathlessness) as the symptom most highly correlated with KCCQ-12 (data available). eTable 2 shows the estimated sample sizes (80% and 90% power; alpha 0.05). To detect a difference of 10.5 points (KCCQ-12) and 1 point (ESAS breathlessness), the sample sizes of 176 and 141, respectively, are needed (30% attrition; 252 and 202).
Discussion
Feasibility outcomes
It is feasible to recruit and collect data for a clinical study. As reported previously in palliative studies, 42 attrition was high (27% at 4 months) mostly due to death or deterioration. Further feasibility work should test the willingness to be randomised.
Strengths and limitations
This the first study to provide patient report and cost-effectiveness data from a cardiology-led palliative heart failure service. Other studies report the components only (ACP, patient-centred decisions) 43 or historical controls. 27
The major limitations are as follows: (1) non-randomisation, (2) cost neutrality was assumed and opportunity costs were not considered, (3) missing data and (4) CT participants were already receiving cardiology-led palliative care (possible underestimated benefit).
What this study adds
This study provides useful feasibility and early safety data; the UCG did not appear to be disadvantaged in terms of survival or 4-month outcomes. Willingness to be randomised was not tested but could be built into a pilot embedded into a phase III trial. Recruitment appeared to be easier from patients known to the research team (UCG). This should be taken into account when researchers are dependent on other clinical teams to identify potential participants although eligible patients may be more inclined to participate in an intervention trial.
A recent US statement 7 encourages problem-driven palliative care independent of prognosis, citing benefits as improved patient/carer understanding, symptoms/suffering relief, patient-centred decision-making, improved communication, better ACP and bereavement support. Although the study was not designed to assess effectiveness, the data on patient understanding, ACP documentation, fewer nights in hospital and being shifted from secondary to community care show promise – consistent with Component 5 (Box 1).
Conclusion
A clinical trial investigating the cost-effectiveness of cardiology-led heart failure palliative care is feasible. A future trial should recruit from usual community-based care eligible for but not referred to cardiology palliative care, and identify those needing more intensive palliative care.
Footnotes
Acknowledgements
The authors wish to thank Professor Fliss Murtagh, University of Hull, for valuable advice with regard to the final revision. M.J., K.H. and Y.M. conceived the study, M.J., P.M., A.M., C.G. and A.B. designed the protocol. P.M., A.M. and C.G. conducted the analysis. All the authors contributed to the interpretation of findings. M.J. wrote the first draft and all the authors contributed to subsequent drafts and approved the final manuscript.
Data sharing
The author for correspondence may be contacted regarding access to anonymised data. Data that are not shown in the paper but are available are provided in the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: K.H. and Y.M. were funded by CTPartners. The other authors declare no conflict of interest.
Ethics and consent
Ethical (NRES Committee London – Camberwell St Giles. REF 14/LO/1813; 07.10.2014) and institutional approvals were obtained prior to recruitment. Participants gave written informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the British Heart Foundation and Marie Curie.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
