Abstract
Background:
Recruitment challenges contribute to the paucity of palliative care research with advanced chronic heart failure patients.
Aim:
To describe the challenges and outline strategies of recruiting advanced chronic heart failure patients.
Design:
A feasibility study using a pre–post uncontrolled design.
Setting:
Advanced chronic heart failure patients were recruited at two nurse-led chronic heart failure disease management clinics in Ireland
Results:
Of 372 patients screened, 81 were approached, 38 were recruited (46.9% conversion to consent) and 25 completed the intervention. To identify the desired population, a modified version of the European Society of Cardiology definition was used together with modified New York Heart Association inclusion criteria to address inter-study site New York Heart Association classification subjectivity. These modifications substantially increased median monthly numbers of eligible patients approached (from 8 to 20) and median monthly numbers recruited (from 4 to 9). Analysis using a mortality risk calculator demonstrated that recruited patients had a median 1-year mortality risk of 22.7 and confirmed that the modified eligibility criteria successfully identified the population of interest. A statistically significant difference in New York Heart Association classification was found in recruited patients between study sites, but no statistically significant difference was found in selected clinical parameters between these patients.
Conclusion:
Clinically relevant modifications to the European Society of Cardiology definition and strategies to address New York Heart Association subjectivity may help to improve advanced chronic heart failure patient recruitment in clinical settings, thereby helping to address the paucity of palliative care research this population.
Recruitment of advanced chronic heart failure (CHF) patients is challenging due to high illness burden.
Palliative care needs assessment research is lacking for advanced CHF patients.
The New York Heart Association (NYHA) Functional Classification does not capture the fluctuating health status of advanced CHF patients.
The NYHA classification as a stand-alone inclusion criterion can act as a recruitment barrier to palliative care studies.
The 2012 European Society of Cardiology definition of advanced CHF used in this study is too narrow to reflect the multi-morbidity and fluctuating status frequently associated with advanced CHF.
The 2016 European Society of Cardiology definition better reflects indicators of worsening CHF and has removed NYHA as a criterion for end-of-life care.
More emphasis is required on a palliative care approach throughout the disease trajectory in this definition.
Strategies to address NYHA subjectivity may help to improve advanced CHF patient recruitment in clinical settings including the American College of Cardiology Foundation (ACC)/American Heart Association (AHA) CHF staging system.
Introduction
Chronic heart failure (CHF) is a progressive condition with a comparable mortality rate to cancer, 1 predominantly affecting older adults with multi-morbidity. 2 It is a condition with a risk of sudden or premature death 3 and can necessitate therapeutic decision-making between life-prolonging treatments and quality of life. 4 Older CHF patients with multi-morbidity particularly require a holistic patient-centred approach to care 5 to facilitate therapeutic decision-making around quality of life versus life prolongation. 6 Many CHF patients experience palliative needs. A palliative care needs assessment presents one way to integrate palliative care into CHF management and potentially improve patient care. 7
Recruitment challenges contribute to the paucity of CHF palliative care needs assessment research. Recruitment is difficult for many reasons: advanced CHF is difficult to define; 8 it has unclear inconsistent descriptive nomenclature; 9 it has a variable progression rate 10 and unpredictable prognosis; 11 there is high patient attrition due to illness burden and high mortality; 12 and gate-keeping often occurs. 13
This article describes recruitment challenges experienced in a feasibility study of a palliative-specific patient-reported outcome measure (PROM) intervention and how these challenges were addressed. It provides recruitment solutions for future studies involving CHF patients.
Methods
Study design
This parallel mixed-methods feasibility study 14 involved a pre–post uncontrolled longitudinal study design. This study examined the feasibility and acceptability of the palliative-specific PROM intervention to improve identification and management of palliative symptoms and concerns in advanced CHF patients. As part of this feasibility study, we examined our ability to recruit participants.
Setting
Study settings were CHF disease management clinics in two tertiary referral centres in Ireland led by heart failure nurses (HFN) and supported by CHF physicians.
Inclusion criteria development
Inclusion criteria (Figure 1) were developed by the study lead (P.M.K.), the HFNs and the research team. The criteria were informed by the advanced CHF definition in the 2012 European Society of Cardiology (ESC) Heart Failure Association position statement 11 and by the ESC palliative care position statement.15,16 Although not included in the 2012 ESC definition, 11 a definition of Heart Failure with preserved Ejection Fraction (HFpEF) as per the 2016 ESC definition17,18 was included in the criteria at the HFNs’ recommendation as this constituted over 50% of their patient population. Of note, HFpEF is included in the 2016 ESC definition which was not published at the time of this study. 18 Once recruitment commenced, P.M.K. and the HFNs frequently reviewed the inclusion criteria. Subsequently, the requirement for a heart failure–related hospitalisation in the preceding 6 months was removed because it was not appropriate in a setting where nurse-led clinics work to avoid heart failure–related admissions.
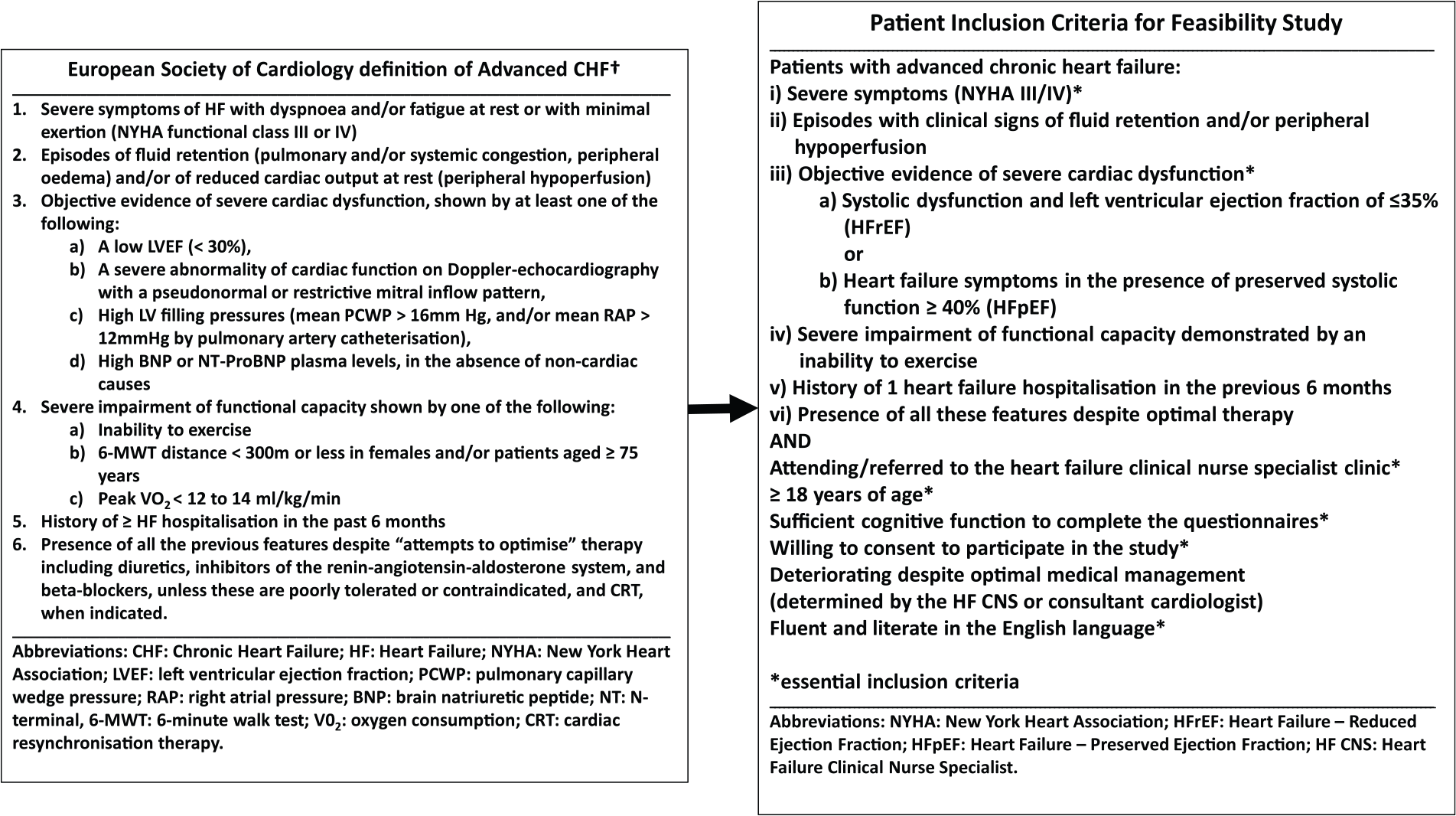
Adaptation of the European Society of Cardiology’s advanced chronic heart failure definition for the study inclusion criteria.
Ethics
Ethical approval was obtained from the Research Ethics Committees: Ref: 13/70, Ref: 1/378/1579 and Ref: BDM/13/14-25. Written informed consent was obtained from participants.
Intervention
The intervention involved administration of the Integrated Palliative care Outcome Scale (IPOS) 19 together with HFN training. Training involved patient-centred education, the role of PROMs to facilitate patient-centred care, training in IPOS interpretation and use in consultation. The IPOS is a 20-item questionnaire measuring patient’s physical symptoms, psychological, emotional, spiritual, information and support needs.
Strategies to address recruitment challenges
To increase engagement, HFNs and service users helped develop study participant documentation. Posters advertising the study were placed in study sites. Screening logs were developed with HFN input. HFNs reviewed the clinical roster daily to identify eligible patients and completed weekly logs recording: numbers of screened and eligible patients, number approached, number recruited, reasons why patients were deemed ineligible, were not approached or declined participation.
Patients were given information on three occasions before written consent was requested. HFNs were provided with summary cards outlining key study points for eligible patients. 20 They provided patients with documentation and obtained permission for the researcher to explain the study in depth. Potential participants returned signed consent forms. The researcher arranged a telephone appointment to complete the first set of questionnaires, which otherwise acted as a patient reminder for those who had not returned consent forms.
Analysis
Analysis of recruited patients was undertaken to determine whether the inclusion criteria modifications identified the same population (advanced CHF patients). We applied the Meta-analysis Global Group in Chronic Heart Failure (MAGGiC) heart failure mortality risk score 21 to calculate the median 1-year mortality risk for each recruited patient 21 to determine participant homogeneity. The Kruskal–Wallis Test was applied to selected clinical markers to test for difference between participants assigned different New York Heart Association (NYHA) classes.
Results
Recruitment
Patients were recruited from September 2014 until February 2015 at the nurse-led clinics. Of 372 patients screened, 81 were approached, 38 were recruited (46.6% conversion to consent 22 ) and 25 completed the IPOS. Figure 2 shows the modified CONSORT flow diagram. 23
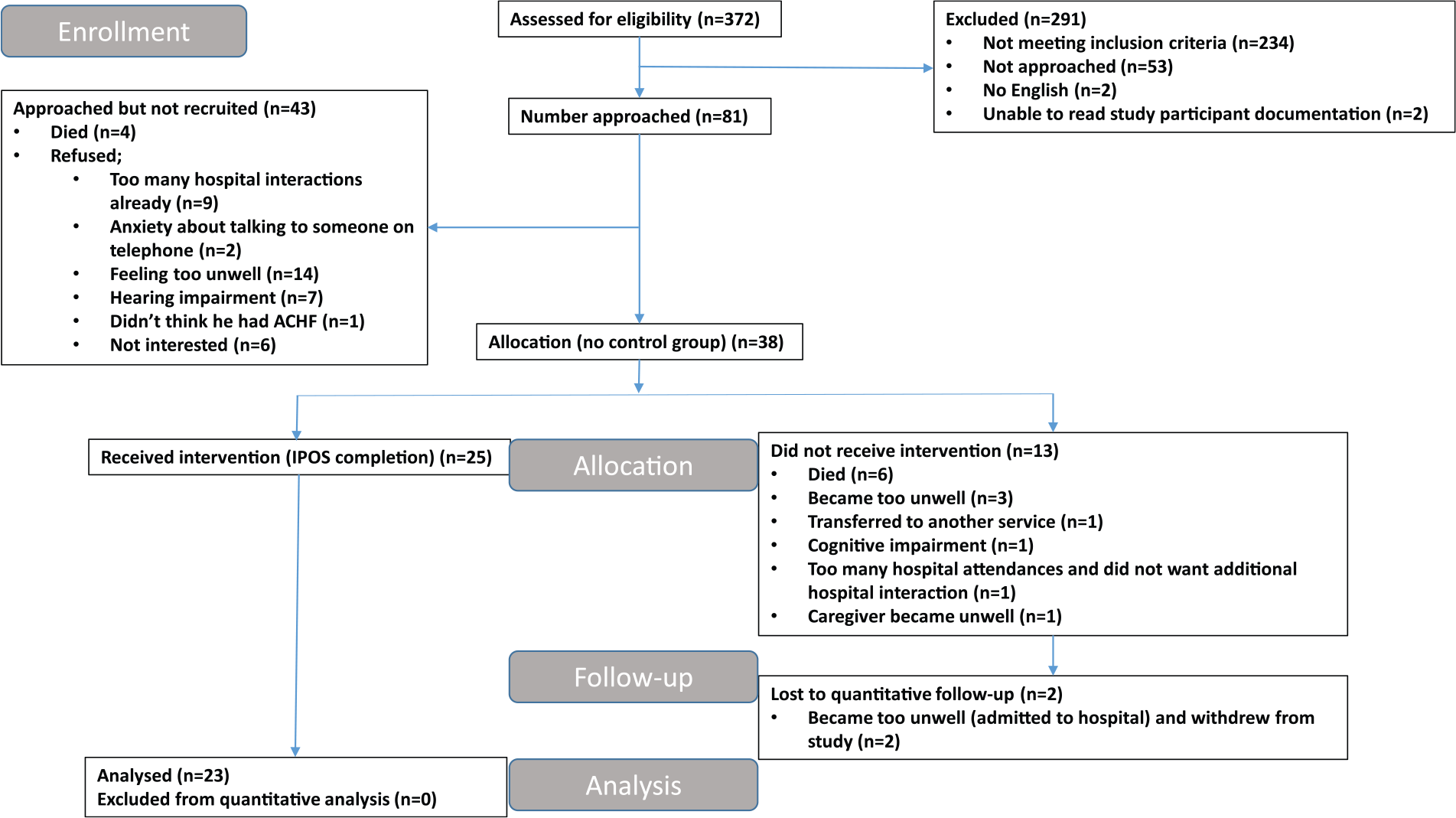
Modified CONSORT flow diagram showing patient recruitment.
Recruited patients’ characteristics
Table 1 reports the demographic and clinical data for participants. To ensure a representative sample of advanced CHF patients, comparison was made with data from Pocock et al.’s 21 study. This demonstrated comparability with deceased patient data (Table 1).
Comparative demographic and clinical patient data between this feasibility study and living patient data from Pocock et al.’s 21 study.
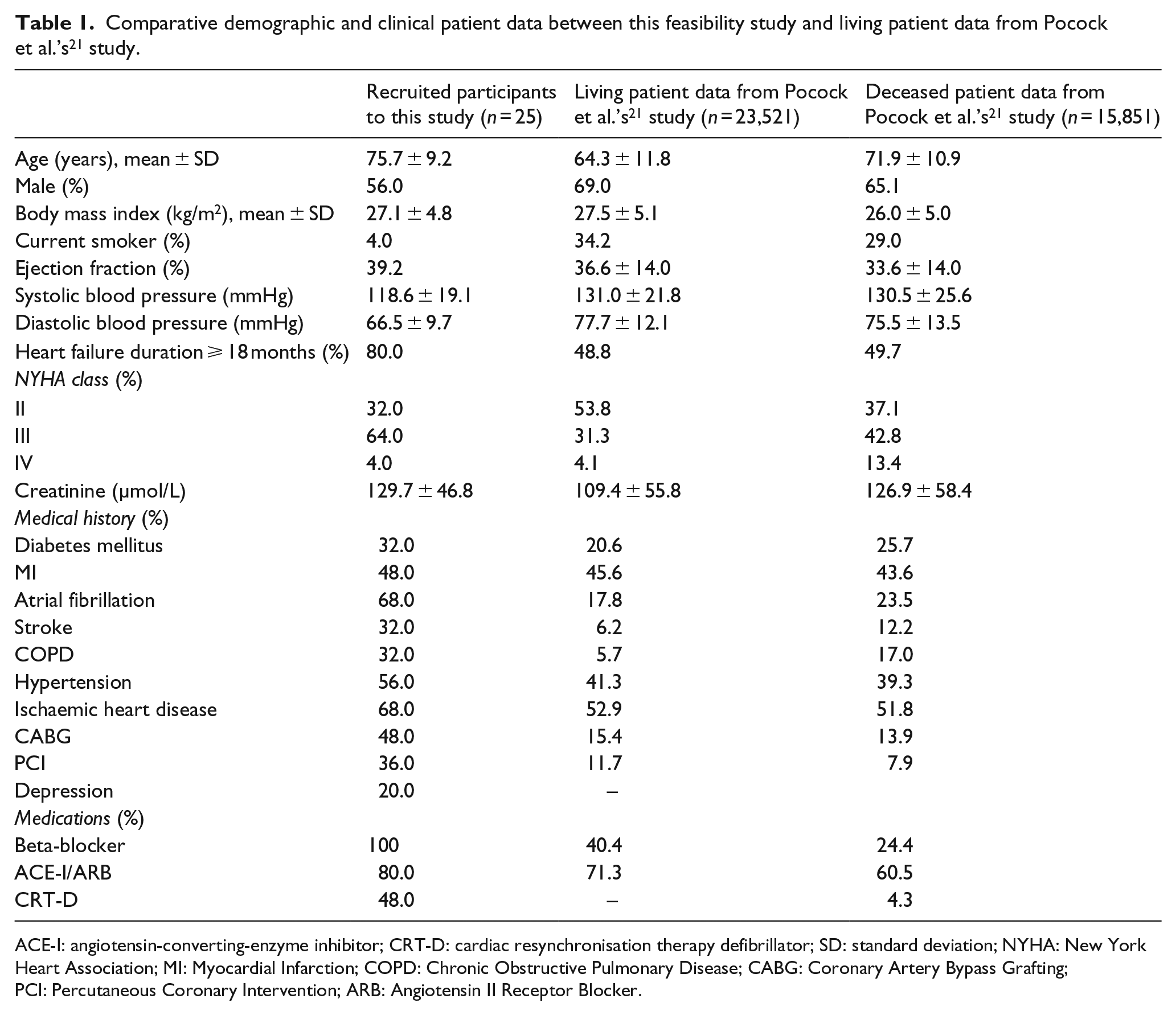
ACE-I: angiotensin-converting-enzyme inhibitor; CRT-D: cardiac resynchronisation therapy defibrillator; SD: standard deviation; NYHA: New York Heart Association; MI: Myocardial Infarction; COPD: Chronic Obstructive Pulmonary Disease; CABG: Coronary Artery Bypass Grafting; PCI: Percutaneous Coronary Intervention; ARB: Angiotensin II Receptor Blocker.
A review of recruitment activity showed that Site A recruited low numbers. Two issues were identified. First, patients were deemed too unwell to be approached for recruitment. Second, patients frequently declined to participate. On exploration, a difference was identified in the manner in which Site A and Site B applied NYHA classification. Site A patients classified as NYHA III frequently had lower functional ability than Site B patients classified as NYHA III. Where Site B classified patients as NYHA III, Site A classified comparable patients as NYHA II. This resulted in Site A identifying a more unwell population of NYHA III patients than Site B limiting recruitment. To ensure recruitment homogeneity, eligibility criteria were modified to allow Site A to recruit patients who were currently classified as NYHA II, who had been NYHA III/IV in the previous 6 months. This resulted in an increase in the total median number approached monthly from 8 to 20, and in the median numbers recruited monthly, from 4 to 9 (Figure 3).
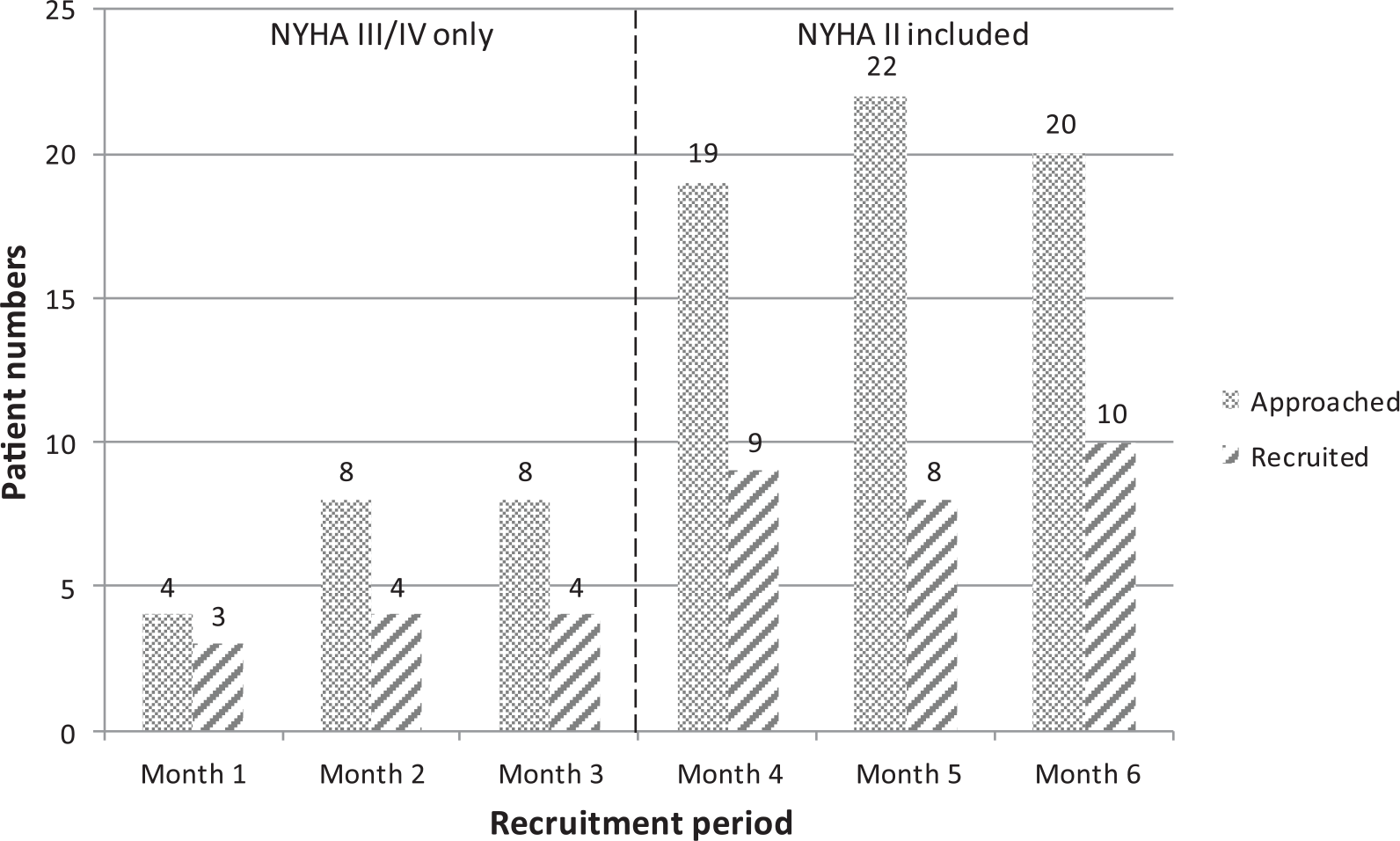
Number of patients recruited per month before and after change in NYHA inclusion criteria.
The MAGGIC mean integer mortality risk score was 27.9 ± 4.9 (median: 29.0) with a mean 1-year mortality proportional risk of 22.2 ± 9.2 (median: 22.7). A Kruskal–Wallis Test demonstrated no statistical difference in creatinine levels between patients assigned NYHA II (n = 8) or assigned NYHA III/IV (n = 17), χ2 (20, n = 25) = 17.75, p = 0.63 between the two sites (Table 2). A Kruskal–Wallis Test demonstrated no statistical difference in 1-year mortality risk between patients assigned NYHA II (n = 8) or assigned NYHA III/IV (n = 17), χ2 (12, n = 25) = 17.75, p = 0.12 between the two sites (Table 2). Median scores for creatinine and 1-year mortality were 117 and 14.7 for the NYHA II group and 122 and 24.8 for the NYHA III/IV group.
Kruskal–Wallis Test for difference between NYHA II or NYHA III/IV grade using grouping variables Creatinine and 1-year mortality risk score.

NYHA: New York Heart Association.
Kruskal–Wallis Test revealed a statistically significant difference in NYHA classification across sites (Site A, n = 10; Site B, n = 15), χ2 (1, n = 25), 16.94, p = 0.0001 (Table 3). The median NYHA class for Site B was 3, that is, NYHA III, while in Site A, it was 2, that is, NYHA II. There was no statistical significant difference in systolic blood pressure, Charlson comorbidity index, 24 creatinine or 1-year mortality risk 21 between patients recruited at each site (Table 3). 21
Kruskal–Wallis Test for difference between Site-A participants and Site-B participants using grouping variables: creatinine and 1-year mortality risk score NYHA II or NYHA III/IV grade.
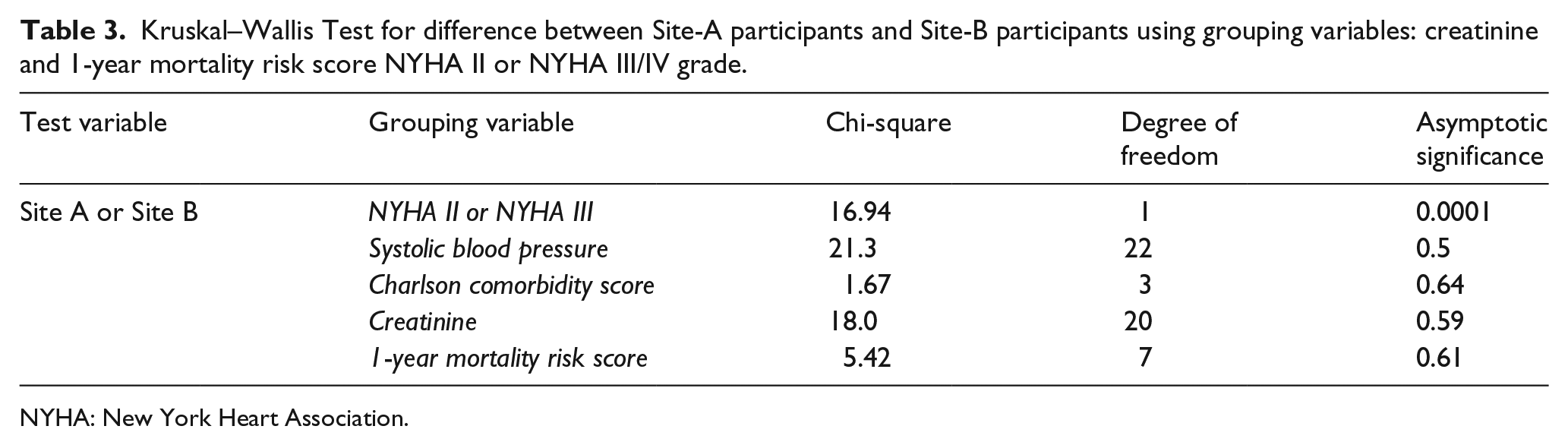
NYHA: New York Heart Association.
Discussion
This study highlights that commonly used advanced CHF descriptors do not comprehensively identify advanced CHF patients, necessitating a different approach in palliative care study inclusion criteria development. The 2012 ESC advanced CHF definition 15 which we used to inform our inclusion criteria (Figure 1) was too narrow to capture fluctuating health status. 25 Patients who were initially too unwell to be approached were too well post-treatment to fulfil inclusion criteria based on this definition. This study demonstrates that the subjectivity of the application of NYHA classification and heterogeneity of this population mean using NYHA as a stand-alone inclusion criterion risks excluding patients from studies and reducing generalisability. Restricting participation to NYHA III/IV excludes more stable patients classified as NYHA II at the time of study recruitment, yet these patients may have palliative care needs.
The NYHA classification was designed to classify heart disease patients based on functional capacity, in the absence of more objective evidence.26,27 Its lack of inter-rater reliability, 28 poor reproducibility and validity have been acknowledged. 29 Yet it continues to play a role in inclusion criteria11,30 and as a determinant for palliative care access. 31 To address NYHA limitations, the American College of Cardiology Foundation (ACC)/American Heart Association (AHA) CHF staging system describes CHF in stages of disease progression, 32 while contextualising NYHA class to provide a more objective and reliable approach to identification (Figure 4). 32
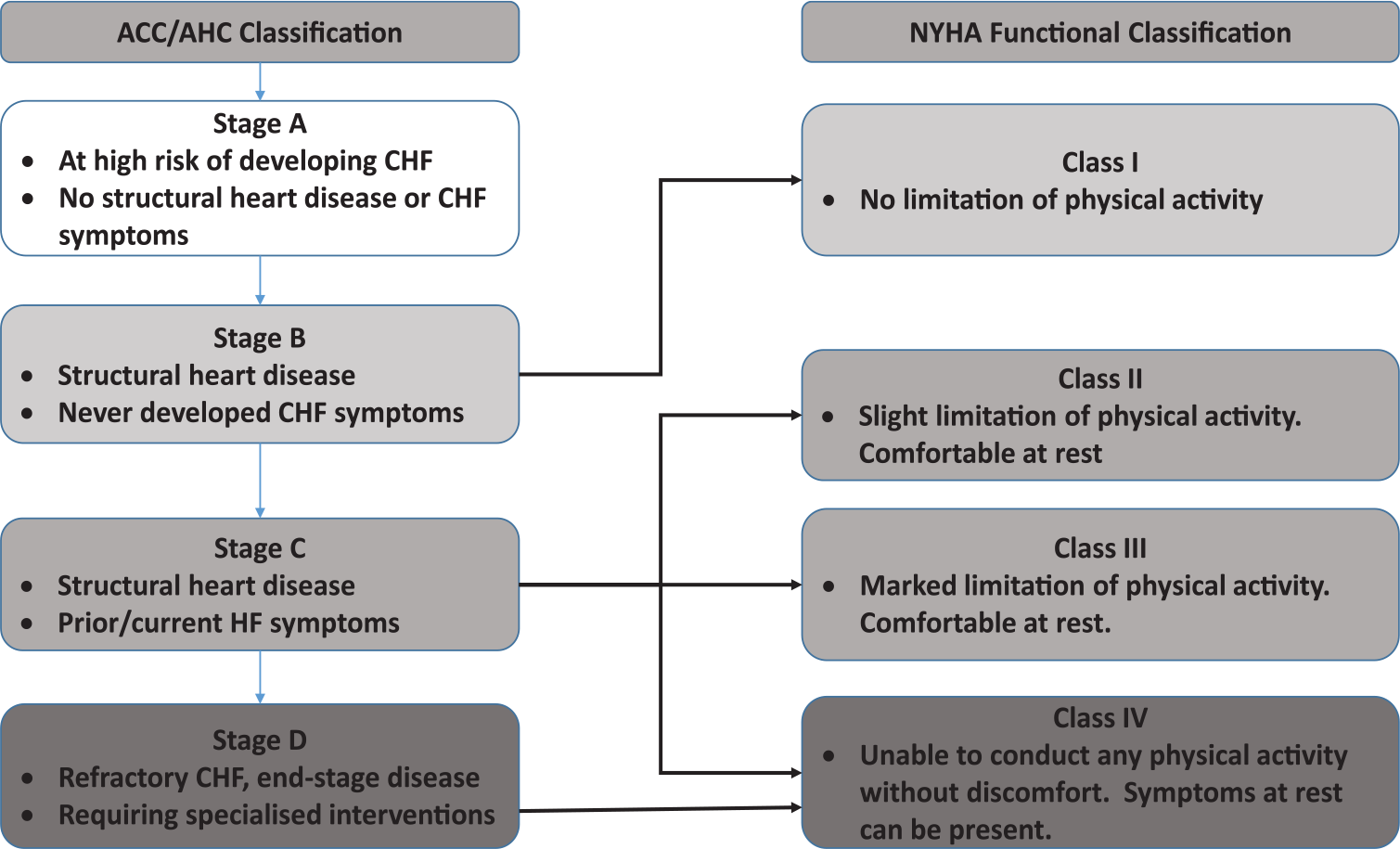
American College of Cardiology–American Heart Association (ACC-AHA) CHF stage and NYHA functional classification of Heart Failure.
CHF is characterised by symptom volatility meaning NYHA class can change over short time periods 33 affecting eligibility for study inclusion criteria applied at a single time-point. We extended recruitment to patients classified as NYHA II, with a NYHA III/IV history. There was no statistical difference in clinical parameters between these patients and those recruited as class NYHA III/IV. Limiting recruitment to NYHA III/IV excludes symptomatically stable NYHA II patients who have been NYHA III/IV previously and are at risk of deterioration from underlying heart disease or multi-morbidity. 3
A limitation of this study is that it may not reflect CHF patients without access to nurse-led clinics. Additionally, we did not conduct quality assurance of the HFNs’ assessment of eligibility and information provision to patients.
Conclusion
The recently updated 2016 ESC definition 18 better reflects the interplay of comorbidities with CHF but more emphasis is required on a palliative care approach throughout the disease trajectory. Despite its shortcomings, NYHA classification is widely used. Strategies to address NYHA subjectivity including the ACC/AHA CHF staging system may improve advanced CHF patient recruitment and advance palliative care research.
Footnotes
Acknowledgements
The views expressed are those of the authors and not necessarily those of the National Health Service, the NIHR or the Department of Health. CLAHRC South London is part of the NIHR, and a partnership between King’s Health Partners, St. George’s University of London and St. George’s Healthcare NHS Trust. The authors thank all collaborators and advisors including service users.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This paper presents independent research funded by BuildCARE, and part-funded by the National Institute for Health Research (NIHR) under the Programme Grants for Applied Research Programme (RP-PG-1210-12015 C-CHANGE) and the NIHR Collaboration for Leadership in Applied Health Research & Care (CLAHRC) through CLAHRC South London. BuildCARE is supported by Cicely Saunders International (CSI) and Atlantic Philanthropies, and led by King’s College London, CSI, UK.
