Abstract
Background:
World Health Organization’s essential drugs list can control the highly prevalent HIV-related pain and symptoms. Availability of essential medicines directly influences clinicians’ ability to effectively manage distressing manifestations of HIV.
Aim:
To determine the availability of pain and symptom controlling drugs in East Africa within President’s Emergency Plan for AIDS Relief–funded HIV health care facilities.
Design:
Directly observed quantitative health facilities’ pharmacy stock review. We measured availability, expiration and stock-outs of specified drugs required for routine HIV management, including the World Health Organization pain ladder.
Setting:
A stratified random sample in 120 President’s Emergency Plan for AIDS Relief–funded HIV care facilities (referral and district hospitals, health posts/centres and home-based care providers) in Kenya and Uganda.
Results:
Non-opioid analgesics (73%) and co-trimoxazole (64%) were the most commonly available drugs and morphine (7%) the least. Drug availability was higher in hospitals and lower in health centres, health posts and home-based care facilities. Facilities generally did not use minimum stock levels, and stock-outs were frequently reported. The most common drugs had each been out of stock in the past 6 months in 47% of facilities stocking them. When a minimum stock level was defined, probability of a stock-out in the previous 6 months was 32.6%, compared to 45.5% when there was no defined minimum stock level (χ2 = 5.07, p = 0.024).
Conclusion:
The data demonstrate poor essential drug availability, particularly analgesia, limited by facility type. The lack of strong opioids, isoniazid and paediatric formulations is concerning. Inadequate drug availability prevents implementation of simple clinical pain and symptom control protocols, causing unnecessary distress. Research is needed to identify supply chain mechanisms that lead to these problems.
Introduction
During 2011, there were an estimated 23.5 million individuals in sub-Saharan Africa living with HIV infection and 1.8 million with new infections and 1.2 million HIV-related deaths. 1 There is an increasing body of evidence that people living with HIV infection endure significant burdensome symptoms, including pain,2–5 fatigue, weight change, cough and skin problems, 6 and that symptoms may persist alongside antiretroviral treatment (ART). 7 Infections such as tuberculosis (TB), pneumonia and candidiasis are a major cause of morbidity. Although recent guidance has raised the cluster of differentiation 4 (CD4) count at which ART should be initiated, 8 patients with advanced disease will experience greater morbidity due to immune reconstitution syndrome. 9 Greater pain and symptom burden are associated with reduced ART adherence5,10 and with a greater probability of switching ART regimens in settings where this is a possibility. 11 Suboptimal adherence limits immunological and virologic responses to ART and increases the risk of mortality. 12 Therefore, symptom management with optimal prevention and treatment of infections are required for the maximum impact on quality of life, morbidity and mortality for patients with HIV.
The pharmacological prevention and management of pain and opportunistic infections requires an effective health care delivery infrastructure, clinical skills and patient access to care. 13 It also requires supplies that allow clinicians to prescribe and pharmacies to dispense those drugs known to be most effective in the prevention and control of common problems. Although data have suggested that stock-outs are a barrier to implementation of clinical guidance with respect to co-trimoxazole (CTX) and isoniazid, 14 the availability of these and other essential drugs and the implementation of minimum stock levels (i.e. the specified lowest amount of a named drug available to dispense in the pharmacy before a re-order should be made) in line with standards for pharmacy and medicine management have not been described. 15 This is especially true for pain management. In order for pain to be effectively controlled, clinical staff should be skilled and able to access three ‘steps’ of analgesia according to the World Health Organization (WHO) pain ladder: non-opioids (such as paracetamol for mild pain), moving up to weak opioids (such as codeine with adjuvants as appropriate) and then to strong opioids (such as morphine with adjuvants as required). 16 Furthermore, analgesics should be not only given ‘by the ladder’ but also ‘by the clock’ and ‘by the mouth’. Therefore, effective pain control requires oral formulations that can be regularly taken by the patient wherever they may be – including at home and in the community – potentially placing a legislative burden on smaller and community-based providers who must adhere to legislative regulation of opioid storage and prescribing. 17
The constrained economic resources of countries within Africa have been identified as obstacles to drug availability. 18 These issues relate directly to infrastructure challenges, such as the number of qualified pharmacists and the efficiency of drug procurement and distribution. Further evidence suggests that supply of analgesics is unreliable among HIV care providers in sub-Saharan Africa and that legislation is overly restrictive for the stock and prescribing of opioids with regard to storage, duration of permissible prescription and designation of health professionals with the legal mandate to prescribe.17,19 However, although drug availability is reliant on a number of policy, regulatory and supply system factors, no studies to date have surveyed the availability within sub-Saharan Africa at the pharmacy level of drugs required by HIV patients to prevent and manage common problems. In the absence of such data, it is unlikely that the goals of HIV-focused health investment in Africa can be fully realised or patient outcomes optimised.
In 2003, the US Government (USG) funded a 5-year, US$15 billion initiative to combat the global HIV/AIDS epidemic: the President’s Emergency Plan for AIDS Relief (PEPFAR). The funds were allocated approximately as follows: treatment (55%), prevention (20%), assisting orphans and vulnerable children (10%) and care and support of individuals with HIV/AIDS (15%). In 2008, PEPFAR was reauthorized for a further 5 years up to US$48 billion. Evaluation of the effect of PEPFAR funding in its target countries has established that there has been a decrease in HIV-related deaths 20 and a reduction in the number of HIV-positive births. 21 While the focus on increased access to ART has achieved results, there has been little investigation from an operational and implementation perspective.22,23 Lack of attention to the dimensions that directly assess the ability to implement simple protocols for effective care, and reduce unnecessary suffering, may undermine and diminish the gains brought by improved ART access. As part of a larger mixed-methods Public Health Evaluation of PEPFAR HIV care and support services in Kenya and Uganda, 24 this investigation aimed to determine within health facilities the availability, minimum stock levels and stock-out frequencies of drugs commonly required for the prevention and management of problems in people with HIV infection.
Methods
Design
The study design was a cross-sectional quantitative pharmacy review in a stratified random sample of HIV facility pharmacies in two sub-Saharan African countries.
Setting and sampling
The study was conducted among HIV care facilities in Kenya and Uganda. In each country, of around 600 HIV care facilities receiving PEPFAR funding, 60 were selected by stratified random sampling. In order to sample a range of facility sizes within the study population, facilities were stratified by number of patients seen for HIV care, using locally provided partner activity data from the non-governmental organisations (NGOs) implementing care and support at the facility level. The three strata (specified a priori) were 1–100, 101–500 and >500 patients seen per year. Inclusion criteria were that they received PEPFAR funding to provide HIV care and support, that they provided care to adults and that they operated in an area that was safe for researchers to visit at the time of data collection. This latter criterion was operationalised as unsafe sites were in areas where violent unrest was reported. Facilities which did not meet the inclusion criteria were replaced using the same random process.
Tool development
Tools were developed by a multidisciplinary team across the focus and academic lead countries (the United Kingdom, Kenya, Uganda and the United States). All tools were piloted in one large facility and in one small facility. Following piloting, the wording and structure of the tools were modified and clarified. At each participating site, information was collected on facility type, total number of registered patients and for each drug, the presence, amount currently in stock, whether drug had expired, number of unopened packs present, minimum stock level (in packs) and whether a stock-out had occurred in the previous 6 months. A minimum stock level is the quantity of remaining stock which prompts the facility to make another order. Drugs were organised by delivery method (i.e. tablet form, syrup, powder for suspension and injection).
The selection of drugs for this study focused on those that prevent and treat common and burdensome problems of people with HIV, are present on the most recent WHO essential drugs list 25 and are in line with current prescribing practice, that is, analgesia according to the WHO pain ladder: CTX, isoniazid and fluconazole. CTX is a simple, well-tolerated and cost-effective intervention, 26 which has been proven to reduce HIV morbidity and mortality in developing countries.27–30 WHO 16 recommends CTX for all HIV-infected persons at WHO clinical stages 2, 3 or 4 or for persons with CD4 < 350 per mm3, as well as for all infants exposed to HIV as an integrated component of HIV chronic care. 31
TB is the most common life-threatening opportunistic infection for those with HIV disease, and preventive isoniazid therapy can reduce the risk of TB by 33% overall and by 64% for those with a positive tuberculin skin test. 32 Joint United Nations Programme on HIV/AIDS (UNAIDS) and WHO have designated isoniazid as part of an essential HIV care package, 33 even alongside ART as it also has been shown to reduce morbidity 32 and has fewer side effects than its alternatives. 34 The WHO recommends isoniazid preventive therapy for all HIV-positive individuals at high risk of TB. 35
Oral or intravenous (IV) fluconazole is used in the treatment of oropharyngeal, oesophageal and vulvovaginal candidiasis in HIV patients, and infected patients may benefit from long-term suppressive or maintenance therapy with fluconazole to prevent recurrence or relapse of infections such as coccidioidomycosis, cryptococcosis, histoplasmosis, mucocutaneous candidiasis and cryptococcal meningitis. It is also active in HIV-positive adults and children. Fluconazole is also being tested as a drug for treatment or prophylaxis for other fungal diseases such as aspergillosis, coccidioidomycosis and histoplasmosis36–39 and is recommended as part of essential HIV care in Kenya. 40
Due to the high prevalence of pain at all stages of the HIV trajectory and alongside treatment, the analgesics from the essential drugs list (i.e. non-opioid, weak opioid and strong opioid) ensure that the WHO pain ladder 16 can be adhered to and pain effectively controlled. The pain ladder has been demonstrated to be highly effective in pain control, and good clinical practice requires drugs to be available at all three levels of the ladder.
Data collection
All sampled facilities were approached for participation by the countries’ Ministry of Health (although some sites were NGOs and mission facilities, they were contacted by the Ministry through their country coordinating function). Project-specific local researchers attended the sampled facilities to complete the data collection tools. This was done without prior notice to the facilities so that the data could be collected from pharmacies without any potential facility effort to enhance the organisation, stock levels or supply of drugs. All pharmacy data were researcher-collected with the attending pharmacist, with facility information being obtained from staff on the day of the visit. Data were recorded on two identical recording sheets. One copy was left with the facility and the other returned to the local research office for data entry.
Data management and analysis
Data were transferred from sampled facilities to the local study offices immediately following collection. Data were double-entered by two different researchers and subsequently validated using EpiData v3.1 (EpiData Association, Odense, Denmark; 2000–2008). Errors in data entry and data recording were identified using consistency and logic checks and followed up by manual checking of questionnaires. Stata v10 (Stata Corp LP, College Station, TX, USA; 2007) was used to undertake descriptive analysis and tests of association.
Ethical approval
Ethical approval to undertake the study was received from the Uganda National Council for Science and Technology (UNCST; Ref SS 1964), the Kenyan Medical Research Institute (Ref KEMRI/RES/7/3/1) and the College Research Ethics Committee at King’s College London (Ref CREC/06/07-140). Subsequent tool changes following initial piloting were also approved.
Results
Sample facility characteristics
Of the original selected facilities (60 Kenya, 60 Uganda), 11 (3 facilities in Kenya and 8 facilities in Uganda) were found not to meet the inclusion criteria. Five sites could not be found, four were in an area of civil unrest, one was exclusively paediatric and one was a duplicate. Therefore, a further 11 equivalent facilities were selected from the original stratified random sample to make a sample size of 120 facilities visited.
The types of health facilities visited, as described by senior staff, are presented in Table 1. Referral hospitals/institutions are defined as those at national/regional or provincial level which provide surgery, specialised care and sometimes training. District hospitals offer basic inpatient services only and not necessarily surgery. Health centres offer predominantly outpatient care, lower level health centres (commonly ‘Health Posts’ in Kenya) offer only a limited range of outpatient-only services and home-based care (HBC) services offer little clinical care. These definitions were locally determined by the Ministry of Health–led Country Teams, and the categorisation was determined by the participating facilities.
Facility type and number of registered HIV patients.
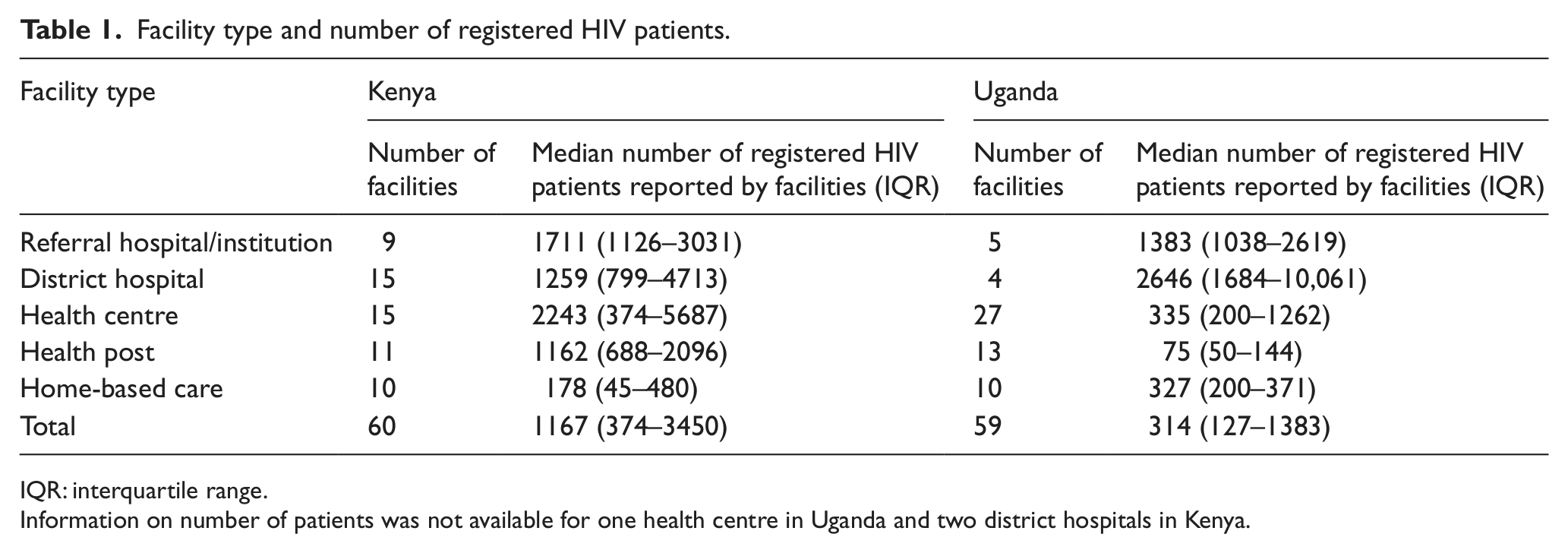
IQR: interquartile range.
Information on number of patients was not available for one health centre in Uganda and two district hospitals in Kenya.
One HBC facility in Uganda was found to have no drug dispensing facilities although it had met the inclusion criteria for a care and support provider. As a result, it was removed from the analysis, leaving 59 facilities in Uganda.
The largest proportions of Kenyan sites were district hospitals and health centres, and the largest proportion of Ugandan sites was health centres. Uganda had a larger number of health centres which were generally smaller (with respect to patient numbers) than the average size for Kenya. Lower level health centres/health posts were also smaller in Uganda than Kenya. Table 1 shows the median number of patients in the previous year (as reported by facility staff) by facility type and country.
Although the sampling frame was stratified by number of patients according to routine reported data, the median number of patients per facility by facility type as reported by facility staff on the day of survey was found to be very different between countries (Table 1). Consequently, further analysis was conducted by facility type rather than patient population number in each country.
Drug availability
Non-opioid analgesics (73%) and adult CTX (64%) were the most commonly available drugs, and morphine (7%) and isoniazid (23%) were the least commonly available drugs (Table 2). Fluconazole and paediatric CTX were each available in over half of facilities. Drug availability was generally higher in hospitals and lower in smaller facilities (health centres, health posts and HBC facilities). Only a few HBC facilities stocked drugs, which were non-opioid analgesics and CTX.
Drug availability by facility type.
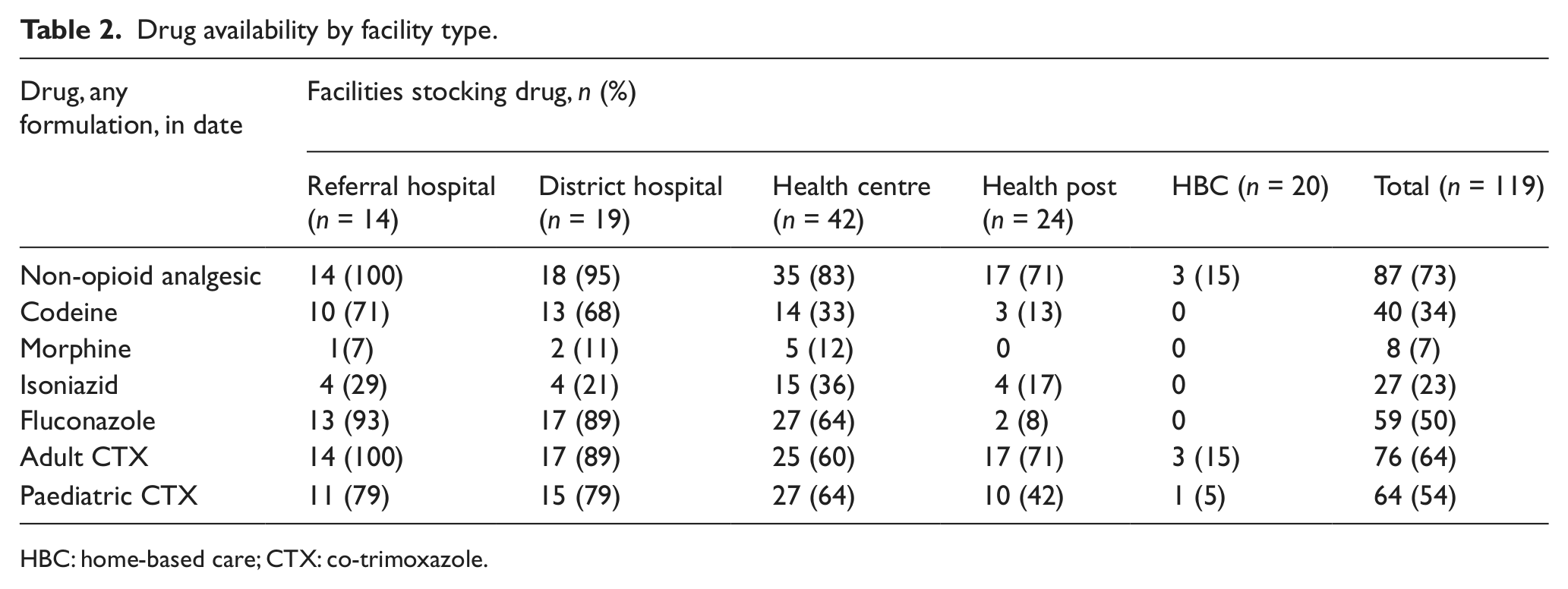
HBC: home-based care; CTX: co-trimoxazole.
Drug stocks and formulations
The type and quantity of stock are represented in Table 3. Data were collected by formulation, but information on strength and dosage was not collected. Therefore, it is not possible to amalgamate the different formulations into an overall quantity and they are presented separately. The most commonly available formulation for every drug (except paediatric CTX) was tablets. Syrup formulation was rarely available for the drugs examined except for non-opioid analgesics (49 facilities) and paediatric CTX (56 facilities). As paediatric CTX is the only drug in the study specifically for children and the only drug most commonly available in syrup form, findings for this drug are reported separately.
Formulations and amounts of drugs in stock at pharmacies.
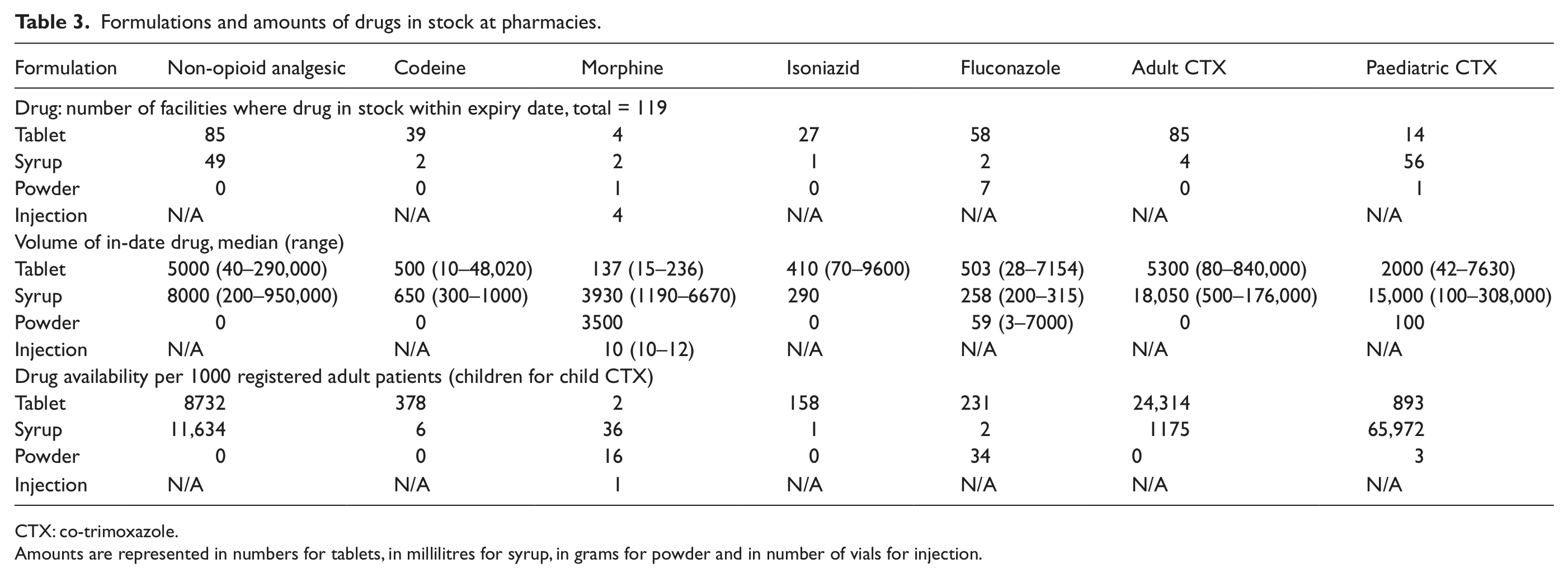
CTX: co-trimoxazole.
Amounts are represented in numbers for tablets, in millilitres for syrup, in grams for powder and in number of vials for injection.
Overall, there was great variation between facilities in the amount of drugs in stock. The most commonly available drugs were also those with the highest amount in stock on the day of the survey. The amount of in-date non-opioid analgesics (median: 5000 tablets and 8000 mL of syrup) and adult CTX (median: 5300 tablets and 18,050 mL of syrup) in stock on the day of the survey was far greater than all the other drugs examined. As the stock levels for all facilities were examined together, the amount of drugs available per 1000 registered patients was calculated. Again, the amount of drugs available per 1000 patients was far greater for non-opioid analgesics (8732 tablets and 11,634 mL) and for adult CTX (24,314 tablets) than for the other drugs (ranging from 2 tablets per 1000 patients for morphine to 231 tablets per patient for fluconazole). Nearly half (56) of facilities stocked paediatric CTX in syrup form, with a large amount found in stock on the day of survey (median: 15,000 mL) and a high availability per child registered.
Minimum stock levels, stock-outs and expired drug stocks
Facilities generally did not have minimum stock levels for the drugs studied (Table 4). The proportion of facilities with a minimum stock level ranged from 11% of those stocking paediatric CTX syrup to a high of 50% of facilities stocking morphine (tablets or syrup) or adult CTX syrup. Minimum stock levels were used for at least one drug at 21% of referral hospitals, 32% of district hospitals, 33% of health centres and 17% of health posts. None of the HBC facilities used minimum stock levels.
Stock levels, recorded stock-outs and expired drugs found in pharmacies.
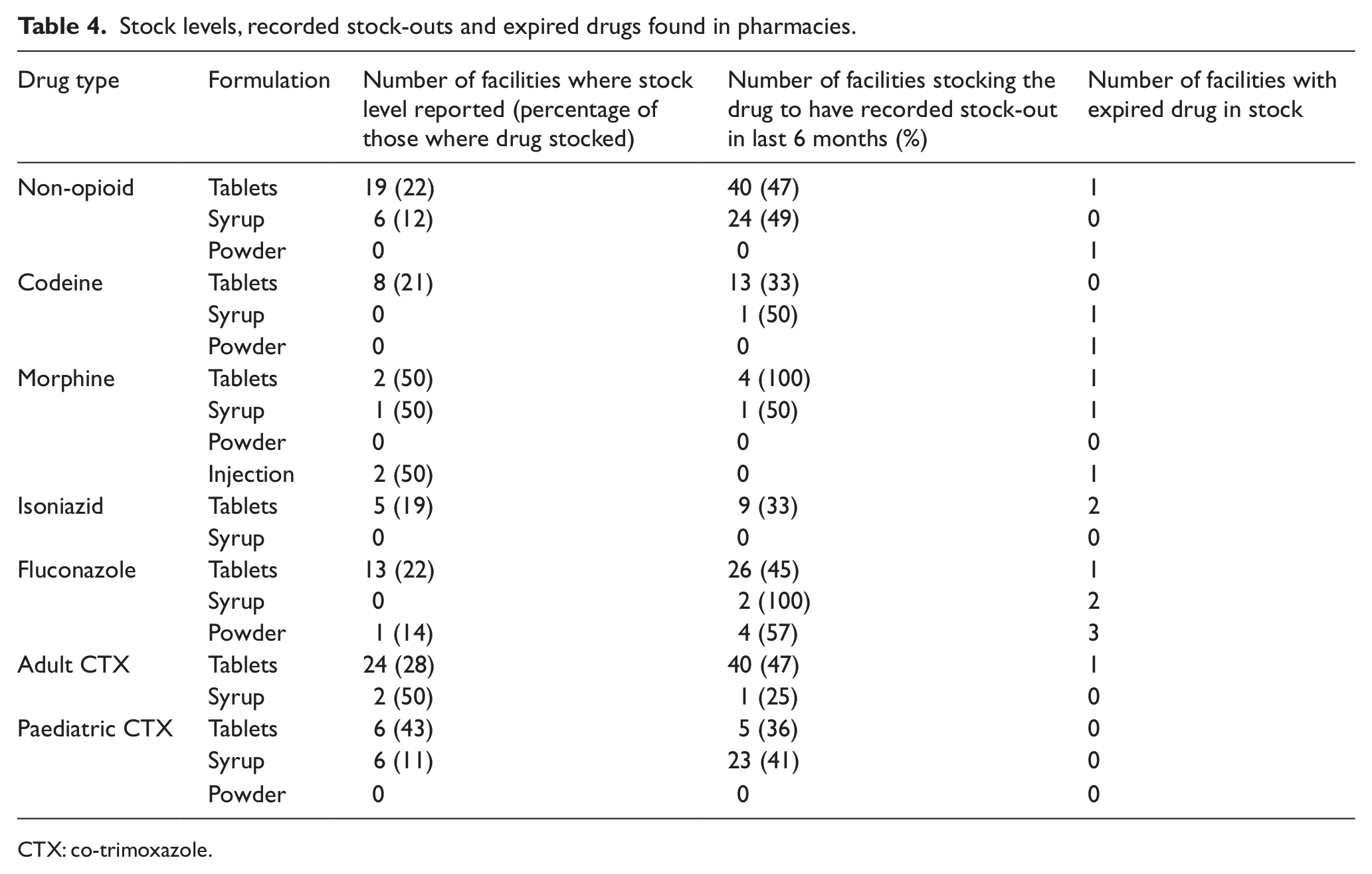
CTX: co-trimoxazole.
Stock-outs were frequently reported. The frequency varied by type of drug. The most commonly stocked drugs (non-opioid tablets and adult CTX tablets) had each been out of stock in the past 6 months in 47% of facilities stocking them. Morphine tablets and fluconazole syrup had been out of stock in the previous 6 months in all the facilities that stocked them (4 and 2 facilities, respectively), whereas morphine powder, injectable morphine, isoniazid syrup and paediatric CTX powder had not been out of stock in the last 6 months at any of the facilities stocking them (found in 1, 4, 1 and 1 facilities, respectively).
When a minimum stock level was defined, the probability of a stock-out in the previous 6 months was 32.6%, compared to 45.5% when there was no defined minimum stock level. This difference in the probability of a stock-out was statistically significant (χ2 = 5.07, p = 0.024).
Discussion
This study measured the availability and stocks of seven commonly required drugs for the management of prevalent HIV-related conditions, such as pain and opportunistic infections in line with WHO essential drug lists and the WHO pain ladder, in PEPFAR-funded HIV care and treatment facilities in Kenya and Uganda. The results demonstrate that facility drug stocks for the management of pain and the prevention and treatment of infections in patients with HIV are inadequate in both range and quantity. This drastically compromises the ability of facility staff to manage symptoms and to adhere to basic care protocols. 40 The results were particularly poor for the peripheral health facilities. Health posts are often the first health system contact for many HIV patients, yet these findings suggest that the majority surveyed were unable to dispense the drugs for the most common symptoms and conditions.
With respect to pain control, it is concerning that even Step 1 (i.e. non-opioid) analgesics were not available in a large number of facilities and were commonly reported as having stock-outs. Step 3 analgesia (i.e. morphine) was only stocked in 8 of the 119 facilities, although the restrictive legislation would preclude many lower level health providers from stocking opioids. Furthermore, a substantial contribution to the availability of morphine was the injection form, and when examined more closely, stocks were found to consist of only between 10 and 12 vials. This formulation and quantity of morphine is not suitable for the management of severe pain in HIV. The WHO pain ladder stipulates analgesia by the mouth and by the clock; 16 therefore, injectable opioids are not appropriate, particularly for patients in the community. Our findings on the availability of analgesia are concerning in the light of the evidence of pain prevalence, which in African home-care HIV patients has had a reported prevalence of 89% and has been found to be associated with great physical and psychological distress. 41 Furthermore, 41% of outpatients in Tanzania with full ART access reported non-neuropathic pain which was not treatable with paracetamol (i.e. Step 1 analgesia), 7 and the prevalence of pain in advanced AIDS is 98%. 2 Stocks of codeine and morphine found in our study are insufficient to manage this prevalence and intensity of pain. Our finding that opioids were expired within pharmacies supports earlier data from a single East African pharmacy and qualitative data that propose under-treatment of severe pain due to ‘opiophobia’ among clinicians. 19
This study found that it was much more common for facilities to suffer stock-outs than to have expired drugs in the pharmacy. This suggests that facilities were more likely to have too few drugs supplied, rather than too many. The exception was morphine; there were only 11 instances of in-date morphine being found (some within the same facility), five stock-outs and three facilities with expired morphine. Thus, it appears that not only was morphine rarely available but also the little that was present was not used. This finding supports previous studies.17,19
Although pharmacies rarely had minimum stock levels, facilities which used minimum stock levels to govern the quantity of stock held were less likely to report stock-outs than facilities which did not. Therefore, health system strengthening may be achieved by simple efforts to implement the established pharmacy and medicine management guidance on minimum stock levels, 15 although our data also suggest that minimum stock levels alone will not prevent all stock-outs.
The study has several strengths and limitations. Data were collected by research staff visiting each facility without prior notice, so the results were not based on staff reports and are likely to reflect the situation faced by pharmacies and patients. As the study was supported by a funder of the facilities surveyed, we did not want to influence the organisation, stock level or supply of drugs by giving warning or the pharmacy review, thereby potentially biasing our data. All visited facilities consented to participate in the study, and data were collected on different formulations as well as different drugs. In order to simplify the study data collection and reporting, we did not collect data on the strength of each formulation, although this limits the depth to which the data can be analysed with regard to patient supply. It may be that available stocks were only in low dose formulations, and the lack of detailed data limits the possibility to further interrogate the data on availability by patient population. In this study, we chose not to collect data on antiretroviral (ARV) supplies as there is an established literature researching the accessibility and sustainability of ARV delivery.42,43 This study therefore focused on the essential WHO supportive care drugs and WHO pain ladder that are frequently required alongside ARVs to maximise quality of life for patients with HIV. It was beyond the scope of this study to explore drug supply chain issues that may explain some of the observed results. Further study is urgently required at the policy, regulatory and central supply levels. Such data will give important context to interpret our data on the availability at pharmacy level and give greater ability to determine at which point in the supply chain problems are occurring. An example of where more research is needed is a greater understanding of why, in our study, stock-outs are experienced even in those facilities that apply minimum stock levels. Prescribing practice is also an important factor in understanding appropriate stock levels. If decentralisation of treatment and care is to be pursued at the policy level, 44 then greater focus is needed to understand the challenges to drug availability at rural and community levels.
When ART and routine monitoring are free of charge to the patient, as they were in the surveyed facilities, HIV care still quadruples family health care expenditure. 45 The major costs to patients are diagnosis and treatment of morbidity events and transportation to health services. When drugs are unavailable, patients are more likely to need to visit other health facilities or pharmacies, thus increasing transport costs. The extra effort involved is also an unmanageable burden, given the high prevalence of fatigue in HIV. 46 It is therefore unacceptable to require patients to ‘shop around’ to maintain a steady supply of essential drugs. Further study to understand the medicine-seeking behaviour of patients would enhance the ability to provide reliable and adequate supplies of drugs in appropriate facilities and access points. Finally, the political economy of drug availability requires attention in light of these data. As noted in the ‘Background’ section, the allocation of ring-fenced funds to ART under PEPFAR has been associated with reduced HIV mortality and improved mother to child transmission. Despite the varying complexities of ART procurement, storage, prescribing, patient monitoring and adherence, there have been great successes in roll-out of these complex and expensive drugs 47 (although systemic and operational challenges persist). 48 The cheaper and simpler pain and symptom preventing and controlling drugs, which are also seen as essential in the presence of ART and have clearly demonstrated benefits for mortality and morbidity, may benefit from equal effort to enhance supply and access.
This study demonstrates that there is a great need to improve health facility pharmacy stocks and the availability of essential drug formulations for the management of HIV-related symptoms and to minimise costs and inconvenience to patients. Not only do funders and governments need to ensure that the facility pharmacy supply chain is secure but also the use of minimum stock levels and local management of supplies need to be promoted at the facility level. Attention to these simple procedures would greatly enhance the control of unnecessary pain and symptom burden.
Footnotes
Acknowledgements
The authors retained full control of the primary data. This study benefited from the participation of a wide range of partners, medical professionals, HIV specialists and palliative care researchers. The authors are grateful for the guidance provided by the US Government Palliative Care Technical Working Group and to the Kenyan and Ugandan Country Teams. Finally, the authors are grateful to the staff and patients at the participating facilities and to Danielle Leivers for manuscript management. R.H. and I.J.H. designed the study and revised this article. V.S. and S.P. analysed and interpreted the data and drafted this article. J.D., R.A.P. and E.N. contributed to the study design, managed data collection and revised the manuscript. S.M. contributed to study design. N.G., M.A., J.K., C.N., G.M. and G.B. collected and managed the data. All authors read, commented on and approved the final manuscript.
Declaration of conflicting interests
The funders had no involvement in the writing of this manuscript or the decision to submit it for publication. The authors are not aware of any competing interests.
Funding
This study received financial support from the United States Agency for International Development under a sub-agreement GPO-A-00-03-00003-00 made under the authority provided to the University of North Carolina.
