Abstract
Background:
There is little guidance on the particular ethical concerns that research raises with a palliative care population.
Aim:
To present the process and outcomes of a workshop and consensus exercise on agreed best practice to accommodate ethical issues in research on palliative care.
Design:
Consultation workshop using the MORECare Transparent Expert Consultation approach. Prior to workshops, participants were sent overviews of ethical issues in palliative care. Following the workshop, nominal group techniques were used to produce candidate recommendations. These were rated online by participating experts. Descriptive statistics were used to analyse agreement and consensus. Narrative comments were collated.
Setting/participants:
Experts in ethical issues and palliative care research were invited to the Cicely Saunders Institute in London. They included senior researchers, service providers, commissioners, researchers, members of ethics committees and policy makers.
Results:
The workshop comprised 28 participants. A total of 16 recommendations were developed. There was high agreement on the issue of research participation and high to moderate agreement on applications to research ethics committees. The recommendations on obtaining and maintaining consent from patients and families were the most contentious. Nine recommendations were refined on the basis of the comments from the online consultation.
Conclusions:
The culture surrounding palliative care research needs to change by fostering collaborative approaches between all those involved in the research process. Changes to the legal framework governing the research process are required to enhance the ethical conduct of research in palliative care. The recommendations are relevant to all areas of research involving vulnerable adults.
Keywords
Introduction
There is limited research investment in the development and evaluation of interventions in palliative care.1,2 Research is required to develop quality palliative care.3–7 This will provide evidence to address deficits in care, to establish whether unproven therapy helps patients and to ensure that palliative care can secure the resources required. 4
Research in palliative care raises particular ethical concerns, 6 which have generated intense debate with little consensus of views.8,9 The ethical decisions regarding the design and conduct of research determine the nature and quality of the research undertaken. Whether patients at the end of their lives should be invited to participate in research is a key issue widely discussed in the literature. 10 On this issue hinges the moral justification of research in palliative care. 11 These views further impact research ethics committees (RECs), which are known as Institutional Review Boards (IRBs) in the United States,12–14 whose judgments determine the quality and safety of interventions detailed in researchers’ ethics applications. 12 Informed consent is challenging in palliative care research as one has to work in circumstances where time is restricted, and patients experience fluctuating/declining physical and mental capacity. 15 In addition, the concerns of family members require consideration as their views are likely to influence patient participation. 16 Given the divergent ideas about the ethical conduct of palliative care research and the lack of empirical evidence, there is a need to identify and arrive at best practices in this field.
The MORECare project aims to identify, appraise and synthesise ‘best practice’ methods to develop and evaluate palliative care, particularly focusing on complex service-delivery interventions and reconfigurations. 17 A main intended output is Methods Guidance on Developing and Evaluating Palliative Care (http://www.csi.kcl.ac.uk/morecare.html). This guidance is underpinned by systematic reviews of the evidence10,18 and, in areas of contentious evidence, expert consensus on best methodological practice. A series of expert ‘think tank’ workshops were held to generate recommendations and build consensus on best practice on identified gaps in the evidence base and issues of particular significance. We held such a workshop in the summer of 2011 regarding ethical issues in research in this area. This article aims to present the process and outcomes of the workshop and consensus exercise on agreed best practice to accommodate ethical issues in research on palliative care.
Methods
The workshop
Design
A one-day closed workshop with invited experts, who were identified through the published literatures on ethical issues in palliative care, the networks of the members of the Project Advisory Group, and searches on the Internet. The participants included senior researchers (epidemiologists, ethicists, social scientists and lawyers), service providers (palliative care consultants and allied health professionals in palliative care), commissioners (from hospital trusts and hospices), members of RECs or research governance bodies, and policy makers (national organisations in palliative care). They all had expertise in palliative care and the ethical issues involving research in this area, and they came from different parts of the United Kingdom. The workshop aimed to debate the ethical challenges of undertaking research in palliative care and to generate recommendations on best ethical practices. A systematic literature review on participation in palliative care informed the design of the workshop. 10
Content
The workshop focused on three topics that were identified as particularly challenging in palliative care research: research participation, informed consent, and applications to RECs. These topics were derived and informed by the systematic review, which was undertaken in preparation of the workshop. Their suitability for the focus of the workshop was discussed in a meeting with the Project Advisory Group. Participants were asked to consider specific questions on these areas (see Box 1).
Questions discussed in the work groups of the workshop
What is best practice to enable patients to participate in research on palliative and end-of-life care (P&EoLC) and protect them from harm?
How can we capture the willingness of individuals to participate in research on P&EoLC, and how can we balance the interests of families with those of the patient?
How can we overcome the challenges in relation to REC members’ review processes and evaluations of EoLC research?
What is best practice in obtaining and maintaining consent for patients and family caregivers who participate in EoLC research?
How can we enable individuals with fluctuating or declining capacity (or those who lack capacity) to participate in research on P&EoLC?
How should we balance the need to give potential participants time to consider participation with the particular time constraints of research at the end of life associated with nearness to death?
Format
Three presentations were held to provide an overview of the state of the science of these three topics of the workshop, and this was followed by discussion. Participants were divided according to their expertise and interest into three work groups that focused on one of the three topics. We used nominal group techniques 19 to structure the process of generating recommendations and forming a consensus on their priority ordering. The work groups discussed the questions asked and then individually wrote down recommendations. The participants then shared in turn their recommendations presenting the most important first, and their rationales. Recommendations were recorded on a flip chart and discussed by the group combining duplicates. The recommendations written down individually were scanned, and together with the recommendations combined in groups, were sent to the MORECare team members undertaking the analysis. All discussions were audio-recorded and transcribed. We used all data gathered in the workshop for the analysis.
Online consultation
Following the workshop, all the recommendations were entered into an Excel spreadsheet. We then combined duplicates, identified those specific to palliative care research and checked the wording for clarity. Doubts were resolved by discussion among the team members. A set of recommendations was agreed on among the project researchers and posted as an online survey to participants in the workshop and members of the MORECare Project Advisory Group comprising senior representatives from academia, service providers, commissioners, policy makers, the voluntary sector and lay members (see Acknowledgements).
Participants in the online survey were asked to rate how much they agreed with the recommendations on a scale from 1–9, where 1 was strongly disagree and 9 strongly agree. A space for comments was provided for each recommendation, and there was a general comments section. The ratings were analysed using descriptive statistics to permit analysis of consensus and rated importance (see Figure 1). The comments were read and considered in the light of the quantitative results.
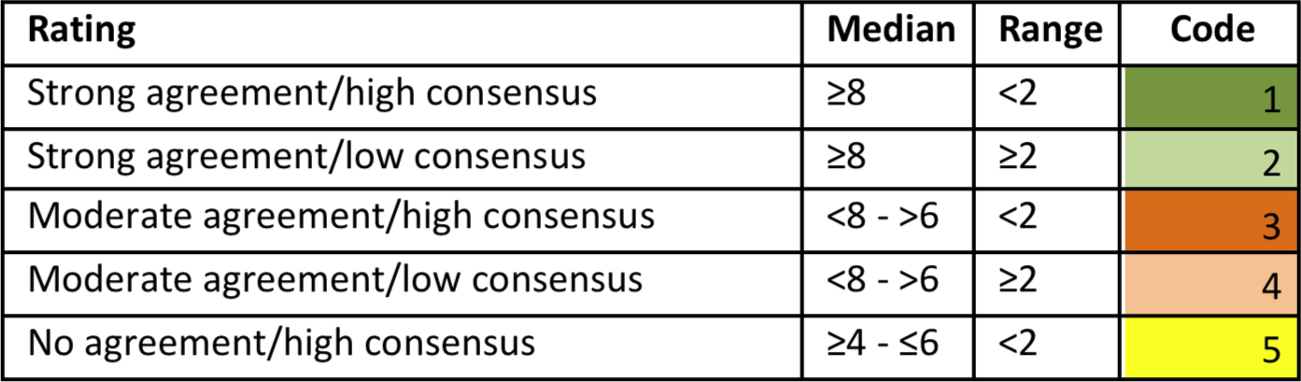
The classification of recommendations.
Ethics approval
The University of Manchester REC approved the MORECare study (ref no. 10328). The online consensus recommendations were presented to the chair for approval prior to posting.
Results
The workshop comprised 28 participants (ethicists, academics in palliative care, members of RECs, service providers and commissioners). In all, 25 participants took part in the work group discussions: eight in group 1, seven in group 2 and eight in group 3. Sixteen recommendations were included in the online survey. From the 61 people (24 attendees and 37 project advisory group members) who were invited to the online consultation, 26 responded.
The workshop
Group 1 discussion: participation in research on palliative care
The starting point of the discussion concerned the general perception that it is inappropriate to
involve people with palliative care needs in research and how this hampers the development of the
evidence base informing palliative care. A workshop participant summed up the prevailing view
regarding research with this population: The culture within our society and especially health care, is that research is extra to
treatment, burdensome and unnecessary. (participant G106)
These ideas can be challenged by integrating research into routine health care. Participants recommended that while conducting their studies, researchers can use the opportunity to document barriers and facilitators to research. This can then provide further evidence of the impact of research on researchers and the experiences of patients and carers. The evidence generated should be inclusive of all research participants who consent to participate.
Participants discussed the specific nature of research in palliative care and identified the following: the research process needs to be sensitive to the changing characteristics of the population associated with advancing disease, there needs to be attention to the challenges researchers face in palliative care research and supervision and support for researchers in the field is essential.
Careful consideration of a study is required to accommodate the problems associated with research in this field (e.g. recruitment, attrition, missing data). Studies need sufficient resources, flexible designs and research procedures, preferably with mixed methods. Collaborative approaches and participative designs were recommended where all the different participants are involved in the research process.
Group 2 discussion: seeking REC approval
This group shared experiences about the difficulties of obtaining ethical clearance as one of the greatest challenges of the research process in palliative care research. RECs apply criteria that are relevant for research in general, but these may not be attuned to the specificities of palliative care. The Health Research Authority’s standardisation of the National Health Service’s ethical review process was considered to exacerbate these challenges. Standardisation was viewed as directed towards protecting RECs from liability and as stifling common sense in concrete research situations.
As researchers need to operate within established ethical frameworks, they should join RECs to
inform decisions and promote understanding of research on palliative care. A way of enhancing
discussion at RECs was for researchers to ensure greater involvement of project user representatives
within the research and application process, for example, by attending REC meetings with
researchers. This could further validate the research proposal, demonstrating the involvement of
users in the study design and the acceptability of the methods proposed to users: … “RECs […] are not unusual in being more risk averse in their decision making for others than
they would be for themselves” […] So they need to be persuaded to balance that ‘natural’ paternalism
with evidence of how […] people who are dying can be treated as responsible autonomous beings
capable of taking risks and burdens. (participant G2010)
Participants recommended that mutual education should be a priority. RECs require training on conducting palliative care research, and researchers require greater knowledge about the legal framework within which these committees operate. Participants noted that RECs should have clear codes, standards and competencies on palliative care. Participants agreed that the establishment of a Research Ethics Network specifically for palliative care could further develop the essential competencies, resources (such as manuals, templates for participation information sheets) and support for researchers.
Group 3 discussion: informed consent
Consent needs to be embedded in a research culture. Participants observed that this could be cultivated in care settings by informing patients on admission that the facility conducts research. At this early stage, ‘consent to consent’ can be sought, which is a way of screening people to identify those who are interested in research and thereby minimise gatekeeping.
Participants agreed that the format of participant information sheets can increase the burden of the consent process. The level of detail needs to be proportional to the risks and burdens involved. Participant information sheet templates for palliative care could be developed and made available from central repositories.
The often fluctuating capacity of the palliative care population presents serious challenges to the consent process. The participants proposed advance consent (early informed consent when the patient still has capacity) as a solution to the problem of fluctuating capacity. Participants recommended that such advance consent should be legally effective for all research, rather than limited to Clinical Trials of an Investigational Medicinal Product (CTIMPs). 20 Currently, the Mental Capacity Act 2005 (the relevant legislation in England and Wales governing health and welfare decision-making in relation to incompetent adults) does not permit research involving incompetent adults on the basis of advance consent. 21 Instead, necessity and risk/benefit criteria are imposed, and assent must be obtained from a non-professional carer based on what the patient’s wishes and feelings about participation in the project would be were the patient competent. 22
As a matter of good practice, participants also recommended that if advance consent were to be relied upon for all trials, contemporaneous assent from someone who is close to the patient and knows whether they would have wished to participate in the research project should also be obtained, as is currently required for non-CTIMPs under the Mental Capacity Act 2005, s. 32.
In the absence of such legal change, participants recommended that when obtaining consent from a competent participant for a non-CTIMP study, the researchers should anticipate the potential loss of capacity and in addition, fulfil the requirements of the Mental Capacity Act 21 by obtaining assent from someone close to them and by meeting the risk/benefit criteria in the Mental Capacity Act. This would provide ‘belt and braces’ (in the words of participant G3018) for consent for patients with fluctuating/declining capacity.
Consent should be a continuous process to ensure sensitivity to changes in an individual’s attitude to participation and signs of distress, especially when capacity is rapidly declining. Further research is necessary on how and when to assess capacity.
Limited time characterises research at the end of life (EoL), while time is also needed to consider consent. In some situations, it is important to allow patients to participate in studies, without having 24 h to consider their decision to enrol, which is often required by RECs. These circumstances require anticipation and specification in the study protocol to avoid coercion.
The online consultation
Table 1 presents the recommendations by subject area. A box and whisker plot shows the ratings of the recommendations in Figure 2.
List of recommendations with mean and medium scores (n = 26).
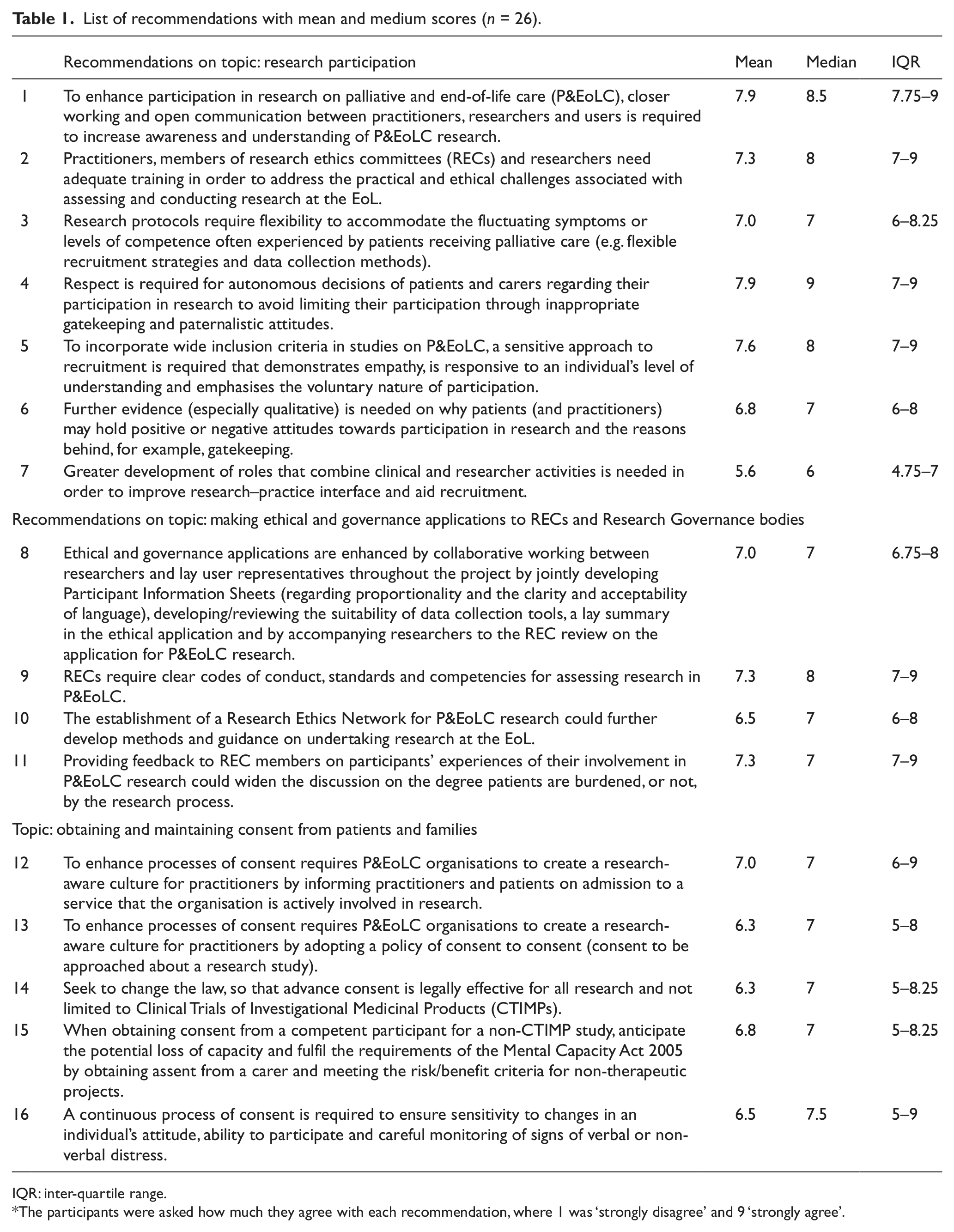
IQR: inter-quartile range.
The participants were asked how much they agree with each recommendation, where 1 was ‘strongly disagree’ and 9 ‘strongly agree’.
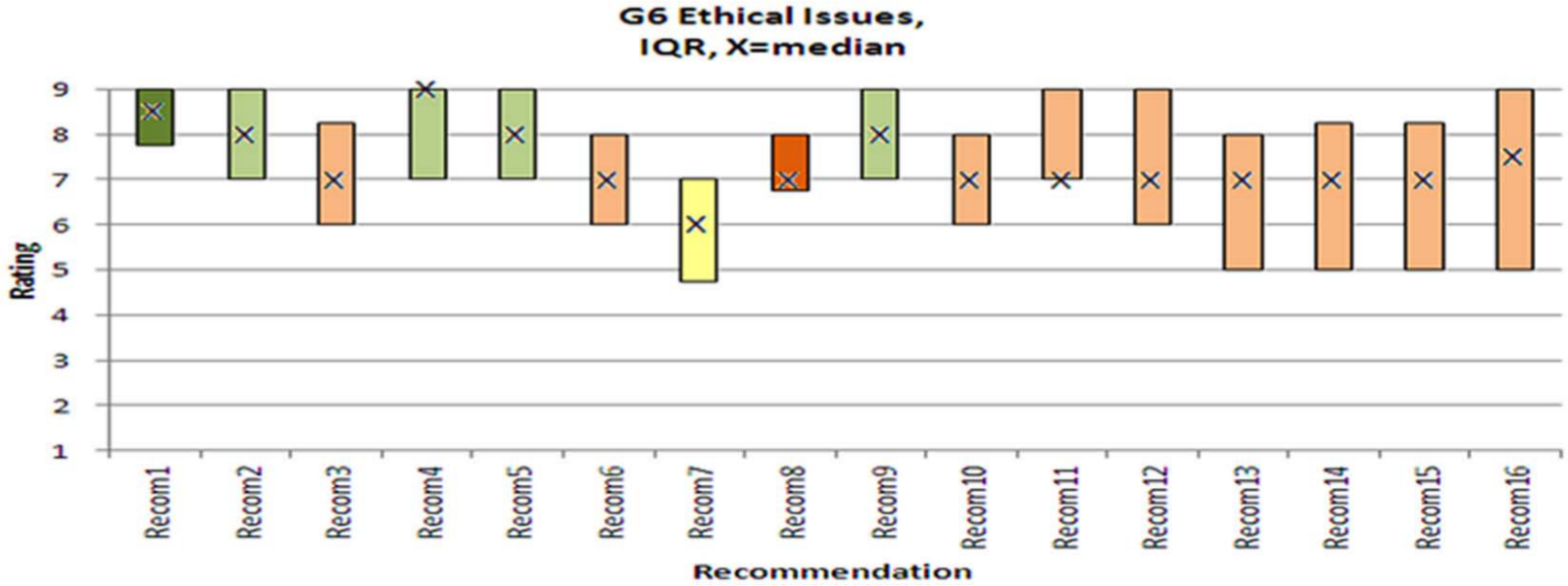
Box and whisker plot of participants’ (n = 26) ratings of the 16 recommendations for the ethical issues in palliative and end-of-life care research.
Research participation
The median scores and narrow inter-quartile ranges show strong agreement on four recommendations of research participation. Especially, recommendation 1 received strong agreement and high consensus. However, recommendations with wider ranges indicate divergence and likely areas of contention. Recommendation 7 triggered most disagreement and low consensus about the combination of clinician–researcher roles.
Applications to RECs
The median scores and range indicate moderate to high agreement on the recommendations in the area of making applications to RECs. However, recommendation 10 triggered only moderate agreement and low consensus about the idea of establishing a research ethics network in palliative care. The concern here expressed in the narrative comments was that research in palliative care could be set apart from other research.
Obtaining and maintaining consent
The recommendations on obtaining and maintaining consent from patients and families were the most contentious. They had the largest divergence in priority ordering. Concerns especially related to recommendation 16 about the continuous process of informed consent, which entails the risk of making research impossible if there would be too many regulatory requirements.
Respondents expressed uncertainty regarding recommendation 14 in terms of its feasibility and implications. This was due to respondents’ lack of expertise on legal issues, which they also admitted with regard to recommendation 15. Here, objections were made to the mechanism of assent. In addition to the requirements of this recommendation, one respondent pointed out that ethical approval would need to be obtained from a REC authorised to approve projects under the Mental Capacity Act.
Table 2 presents the final recommendations, which were refined in light of the narrative comments from the consultation. We did not change all recommendations for the following reasons: when the comments raised questions about operationalisation that went beyond the purpose of the recommendation, if respondents reported a lack of expertise about the recommendation or if issues were already covered by other recommendations.
Summary of narrative comments and final refined recommendations.

Discussion
Although the ethical debate on whether patients near the EoL should be considered ‘too vulnerable’ to be involved in research is far from settled, the participants in this workshop started from the idea that research is needed in palliative care. They therefore concentrated on best practices of research participation to foster scholarship in this area. Participants recognised that the idea that research with this population is not justified underlies the two other issues of this workshop. It determines REC members’ attitudes towards palliative care research and the difficulties of conducting research in care settings that complicate the consent process. A change in culture surrounding palliative care research was viewed as the most important recommendation, which can lead to new ways of thinking about research and can open up new ways of approaching this area. Although the guiding question for this workshop focused on the patient, the workshop participants recognised the involvement of all those involved in the research process. They were not considered as separate entities, but the reality of their interaction was taken into account.
RECs are often especially cautious to grant ethical approval to research applications in palliative care. 12 This relates to societal concerns about involving people with life-limiting conditions in burdensome research with limited individual benefit. RECs may be less familiar with the issues that are particular to this field14,23 and with research designs required to accommodate complexities associated with varying deteriorating illness trajectories.12,14,23 The involvement of a user representative in the process of REC review of the application could greatly enhance mutual understanding between REC members and those conducting the research (recommendation 8). Submitting decisions made by RECs to external peer review could increase accountability and transparency on their decision-making processes (recommendation 10).
The work group on informed consent addressed the importance of undertaking the consent process in a way that enables individuals with fluctuating/declining capacity to participate in research. A way forward is for advance consent to be legally effective for all research, rather than limited to CTIMPs. 20 The recommendation to change the law is a first step in the development of standards for research on palliative care. Such legal change could provide an example for other regulatory contexts relating to the involvement in research of people who have impaired capacity, both in Europe and the United States, where policy guidance is either lacking or diverging in a mixed legal framework.24–26 The wider use of advance consent could reduce attrition and missing data, which are common challenges in palliative care research. A separate recommendation 15 situates this change in the current legal framework for medical research by specifying the requirements from other legal provisions that are directly related to this new measure, such as the Mental Capacity Act, so that it is fully operational.
The recommendations were generated in a think-tank involving discussion and consensus of expert views on undertaking research with individuals requiring palliative care and priority rating. The online consultation allowed anonymous rating and commentary from a broader group of experts. Recommendation 4 concerned the importance of patients’ autonomous decision-making to counter gatekeeping and paternalism. This recommendation had the highest score, showing the priority of allowing and enabling individuals to consider participation in research. This is consistent with most patients’ experiences of participating in research, which can bring direct benefits, shown by a systematic review conducted in preparation for the think-tank. 10
Recommendation 1 on the need to enhance awareness and understanding of palliative care research through closer working between different parties scored second highest and reached greatest consensus. The comment this recommendation received of being ‘self-evident’ underscores that this is the main target to which one should work in this field. The third highest rated recommendation shows the importance given to inclusiveness of a population (older people, the bereaved, those with fluctuating capacity) who are often excluded from designs that generate high-level evidence according to criteria used in evidence-based medicine, 27 but who are of most relevance to palliative care.
Recommendation 7 on the development of combined clinical and researcher roles received the lowest scores. The comments outlined the advantages of working separately within clearly defined roles. This recommendation does not prescribe complete combined training, but rather an understanding of situations when care takes priority, and a sense for research opportunity. Within a collaborative model, people with clinical or researcher roles acquire insight into the aims and procedures of one another’s expertise. Active engagement enhances a sense of ownership of a study, which improves the research–practice interface. The narrative comments to recommendation 13 showed that there was confusion about the term ‘consent to consent’ as it suggests an additional layer of bureaucracy before the actual consent process. However, it refers to a screening process of patients upon admission to a service, by which they show their willingness to be approached to participate into future studies. In addition, the low score of recommendation 14 was due to the commentators’ unfamiliarity with the legal framework. These findings again underscore the need for collaborative interdisciplinary practice and research.
Limitations
The recommendations may not be exhaustive of all the ethical challenges encountered regarding the three topics. This is related to the particular format of a one-day closed workshop with the aim of developing best practice guidance among experts in palliative care research, which limited the number and range of experts who could be invited. Therefore, in addition, the workshop needed to focus on three topics, which were identified as key to the concerns in palliative care research. Only experts from the United Kingdom participated in this workshop. For the online consultation, a 43% response rate was achieved (26/61) following a reminder at 2 weeks. This workshop was the fourth workshop in the project, which took place at the end of July, and the consultation was open for response in the month of August, which is the holiday month for most people. This may be an explanation for the relatively low response rate. The lay members on the Project Advisory Group were not able to attend this workshop, but they were involved in all stages before and after the workshop, and they were invited to the online consultation.
Conclusion
The results of this workshop and consensus exercise provide guidance for ethical issues in palliative care research. The culture surrounding palliative care research needs to change, which will lead to new ways of thinking about research and open up new ways of approaching this area. This can best be realised by fostering collaborative approaches between those involved in the research process. Changes to the legal framework governing the research process are required to enhance the ethical conduct of research in palliative care. The recommendations are relevant to all areas of research involving vulnerable adults.
Footnotes
Acknowledgements
We thank all members of the MORECare project: Principal investigator: Irene J Higginson; co-principal investigator: Chris Todd. The members of MORECare are as follows: Co-investigators – Peter Fayers, Gunn Grande, Richard Harding, Matthew Hotopf, Penney Lewis, Paul McCrone, Scott Murray and Myfanwy Morgan; Project Advisory Group – Massimo Costantini, Steve Dewar, John Ellershaw, Claire Henry, William Hollingworth, Philip Hurst, Tessa Inge, Jane Maher, Irene McGill, Elizabeth Murray, Ann Netten, Sheila Payne, Roland Petchey, Wendy Prentice, Deborah Tanner and Celia A Taylor; Researchers – Hamid Benalia, Catherine J Evans, Marjolein Gysels, Nancy J Preston and Vicky Short. We would also like to thank the participants of the MORECan ethical workshop.
Declaration of conflicting interests
No competing financial interests exist.
Funding
MORECare is funded by the National Institute for Health Research and managed by the Medical Research Council as part of the Methodology Research Programme (project number: G0802654/1).
