Abstract
Objective
To generate a clinical roadmap for managing lateropulsion after supratentorial stroke which integrates theory and practice using a realist review strategy.
Data Sources
Medline Complete, CINAHL Complete, PEDro, Academic Search Complete, Health and Psychosocial Instruments, Health Source: Nursing/Academic Edition, and Embase were searched until December 2025.
Review Methods
Two searches included review and non-review articles about people with contralesional lateropulsion after supratentorial stroke. We excluded articles about people with ipsilesional lateropulsion resulting from brainstem or cerebellar stroke and non-English articles. Guided by Realist and Meta-narrative Evidence Syntheses: Evolving Standards, pairs of authors independently screened titles and abstracts, determining final lists by consensus. Pairs of authors independently extracted data, ranked levels of evidence, and collaborated to synthesise findings following the context-mechanism-outcome configuration of realist reviews. All authors used this foundation to generate the Lateropulsion Clinical Roadmap and clinical recommendations.
Results
The review article search yielded four systematic reviews and one scoping review (of 19 screened). The non-review article search yielded 39 articles (of 167 screened), categorised as neuroimaging, epidemiology, assessment and intervention. Clinical recommendations and a Lateropulsion Clinical Roadmap integrated this information.
Conclusion
A Lateropulsion Clinical Roadmap integrated theory with neuroimaging, epidemiology, assessment and interventions based upon a realist review. Thorough assessment of severity of lateropulsion and neurological impairments should guide clinical decisions about how to leverage sensorimotor systems while considering task difficulty and environment during interventions for lateropulsion. A new hypothesis linking patient presentation with interventions directed at improving sensorimotor performance of the paretic extremities requires further study.
Introduction
People with lateropulsion after supratentorial (hemispheric) stroke remain enigmatic because they demonstrate a contralesional postural tilt as they actively abduct and extend their ipsilesional (stronger) extremities to push themselves in the frontal plane toward their contralesional (weak) side. 1 Clinicians attempting to move the people with lateropulsion toward the ipsilesional side are met by active resistance from the patient, resulting in clinical labels such as pusher syndrome, pushing behaviour, lateropulsion with active pushing, or contraversive pushing.1–4 The current recommended term is lateropulsion.5,6
People with lateropulsion present at lower functional levels, demonstrate more severe lower extremity paresis, experience protracted recovery when sensory, visual, and cognitive impairments co-exist, and pose a higher burden of care.7–13 Clinical practice guidelines for stroke, that are published in English, fail to specifically advise about lateropulsion and evidence to guide clinical management of lateropulsion is limited.14–18 As a result, clinicians, caretakers, and researchers need evidence-based resources to guide targeted rehabilitation for lateropulsion.19,20 A clinical roadmap linking theories about lateropulsion to assessments and interventions may be guided by expert recommendations and prior research. 21
A realist review strategy provides the opportunity to generate a clinical roadmap by reviewing current theories, aligning findings from clinical studies with these theories, and proposing new clinical hypotheses about how to best frame the management of people with lateropulsion.22,23 The objective of this project was to generate a clinical roadmap for managing lateropulsion after supratentorial stroke which integrates theory with practice recommendations using a realist review strategy. Specific aims were to: (1) explore theories related to lateropulsion and their clinical implications; (2) discuss interventions that may be effective, for whom and in what circumstances they would work, and why they may be effective; and, (3) create a Lateropulsion Clinical Roadmap for assessment, intervention and future research.22,23 We hypothesised that our search results will conform with existing theories that people with lateropulsion align their central body axis to an egocentric reference system for verticality which may be tilted away from Earth vertical and/or they are exhibiting a form of neglect called graviceptive neglect.2,24–26
Methods
Realist review strategies emerged in health care policy arenas and can be adapted for clinical research.22,27 The Realist and Meta-narrative Evidence Syntheses: Evolving Standards (RAMESES) methodological and publication guidance served as the foundation for our search and synthesis strategies.22,28–30 We used a two-fold search strategy: first, we examined systematic and scoping reviews of studies related to lateropulsion after supratentorial stroke to summarise past literature; then, we searched for and examined all types of non-review literature that were published after the reviews or might have been excluded by systematic or scoping reviews. Included articles from both searches were categorised as neuroimaging studies, epidemiological research, assessment recommendations and intervention studies.
Medline Complete, CINAHL Complete, PEDro, Academic Search Complete, Health and Psychosocial Instruments, Health Source: Nursing/Academic Edition, and Embase were searched until December 2025.
The target population of this project was adults with lateropulsion following supratentorial (hemispheric) stroke, characterised by contralesional postural tilt in the frontal plane. In order to maintain a focused review on one specific clinical presentation, studies were excluded if they referred to people with traumatic brain injury or stroke in the cerebellum or brainstem who exhibited ipsilesional lateropulsion (as in Wallenberg syndrome), as well as people whose primary symptom was retropulsion (in the sagittal plane). Articles written in a language other than English were excluded.
The first search was conducted to find systematic or scoping reviews that would summarise past evidence for best practice about lateropulsion. Keywords for the first search of reviews were: (Systematic review OR literature review OR scoping review) AND (lateropulsion OR pusher syndrome OR pushing behaviour OR contraversive pushing) AND stroke NOT (medulla OR vermis OR cerebellum OR brainstem). Exclusion criteria for this search included: narrative reviews, historical reviews, randomised control trials, observational studies, case series, cohort studies, and case studies.
The second search targeted more recent articles published after the systematic or scoping reviews or those that met our search criteria but were not discussed in the systematic or scoping reviews. Keywords for the second search of non-review studies were: (Lateropulsion OR pusher syndrome OR pushing behaviour OR contraversive pushing) AND stroke AND (intervention OR examination OR assessment OR rehabilitation OR therapy) NOT (medulla OR vermis OR cerebellum OR brainstem). Exclusion criteria for the second search were: systematic reviews, scoping reviews or other literature reviews or narratives, and letters to the editor. Articles that were reviewed in the systematic and scoping reviews found in the first search were excluded.
Using these respective keywords, one pair of researchers (MWO and SB) independently searched and screened title and abstracts of review articles; another pair (NS and SB) independently searched and screened non-review articles. Pairs compared their findings and agreed to final respective lists consistent with inclusion/exclusion criteria.
The same pair of researchers then independently extracted the following pre-determined information from selected reviews (MWO and SB): type of study, goal of the study, search criteria, main findings, and relationship to theory, if noted by authors. For non-reviews, the second pair of researchers (NS and SB) independently extracted: category (neuroimaging, epidemiology, assessment or intervention), purpose of the study, study design, sample and sample size, outcome measures, method, major findings, what should be known when interpreting results, and relationship to theory, if noted by authors. All review and non-review articles were assessed according to the following levels of evidence: systematic review or meta-analysis of a randomised controlled trial (Level 1); single randomised controlled trial (Level 2); quasi-experimental study or randomised controlled trial (Level 3); cohort study or case-control study (Level 4); systematic review or meta-analysis of a qualitative study (Level 5); single qualitative or descriptive study (Level 6); and, expert opinion (Level 7). 31 Alternate team members reviewed articles authored by any reviewer to maintain integrity and fairness of the review process. Reviewers achieved consensus through discussion in cases of differing initial opinions.
Following suggestions of the Delphi panel for a common terminology, we used the term lateropulsion throughout our summaries, despite original authors using more traditional terms like pusher syndrome or behaviour. 21 The researchers who conducted the searches (MWO, NS, SB) then collaborated to produce a context–mechanism–outcome configuration to integrate the findings from the selected studies and show how postural mechanisms may be triggered within certain contexts or conditions to yield specific outcomes.22,32,33 The focus of the context-mechanism-outcome was to refine the theory and to help clinicians focus on decision points with the format of: If A, then B; If C, then D; and, clinical recommendations. 22 Findings relating to an articles’ purpose statements fell into the ‘If A, then B’ category. Additional findings or findings from the control group or other clinically relevant findings were considered as ‘If C, then D’. Both categories were considered for the clinical recommendations.
The research team examined the context–mechanism–outcome paradigm and looked for other neurophysiological or clinical hypotheses that might be consistent with the literature but outside of the existing theories. The research team developed a systematic framework for assessment and interventions and labelled it the Lateropulsion Clinical Roadmap to guide management of people with lateropulsion after stroke. A Supplemental Guide explains rationale for the Roadmap items and offers a clinical example. Recommendations for future research were based on the clinical recommendations and the Roadmap.
Results
Figure 1 shows the RAMESES diagram illustrating both search processes. 29 Four systematic reviews and one scoping review resulted from the first search of review manuscripts. Comprehensive profiles of neuroimaging, 4 epidemiology,12,13 assessment,4,34 and intervention 17 review research related to lateropulsion after supratentorial stroke are detailed in Supplemental Table 1.
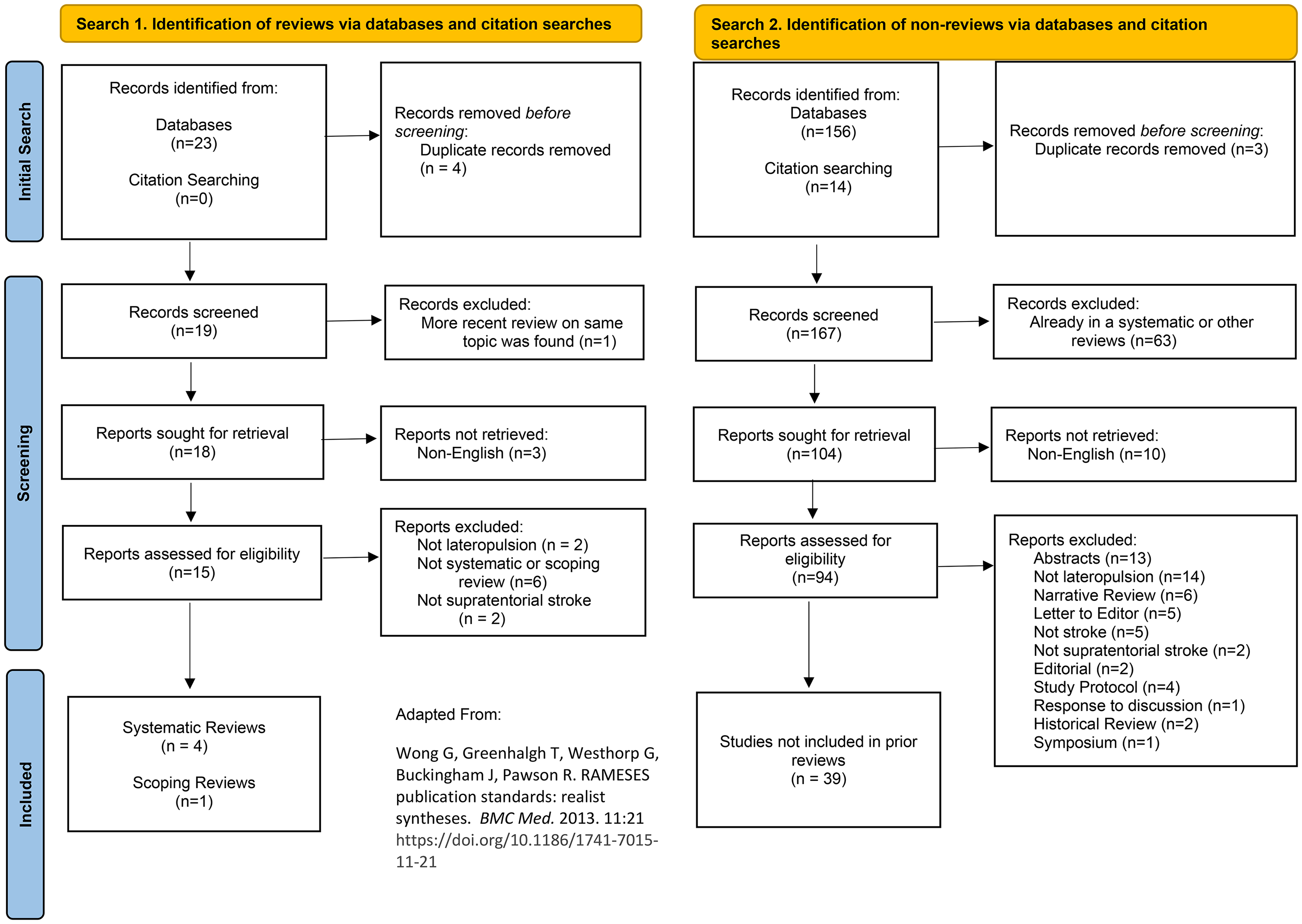
The Realist and Meta-narrative Evidence Syntheses: Evolving Standards (RAMESES) flow sheet showing search results for review articles (left) and articles not included in reviews (right) to July 2025.
The second search yielded 39 articles that met inclusion criteria and were not included in prior reviews. Extracted information for neuroimaging (n = 7, Supplemental Table 2), epidemiology (n = 12, Supplemental Table 3), assessment (n = 12, Supplemental Table 4) and intervention (n = 8) Supplemental Table 5) research were considered for the next step of analysis.
Authors discussed findings from the extracted information and collaborated to generate Table 1. Here, a context (study details)-mechanism (study findings)-outcome (clinical recommendations) framework highlights major findings of reviews and non-review research and offers clinical recommendations. Table 1 uses a format of ‘If A, then B; if C, then D’.
Synthesis of study results and implication for management of people with lateropulsion. Systematic or scoping reviews are presented first in each section, followed by other studies not included in these reviews.

These clinical recommendations served as the basis for the Lateropulsion Clinical Roadmap (Figure 2). A Supplemental Guide outlines the steps found within the roadmap, the rationale for their inclusion, and an example of decision-making during treatment. Figure 3 outlines a hypothetical neurophysiological mechanism/model related to lateropulsion and highlights an approach that may be suitable.
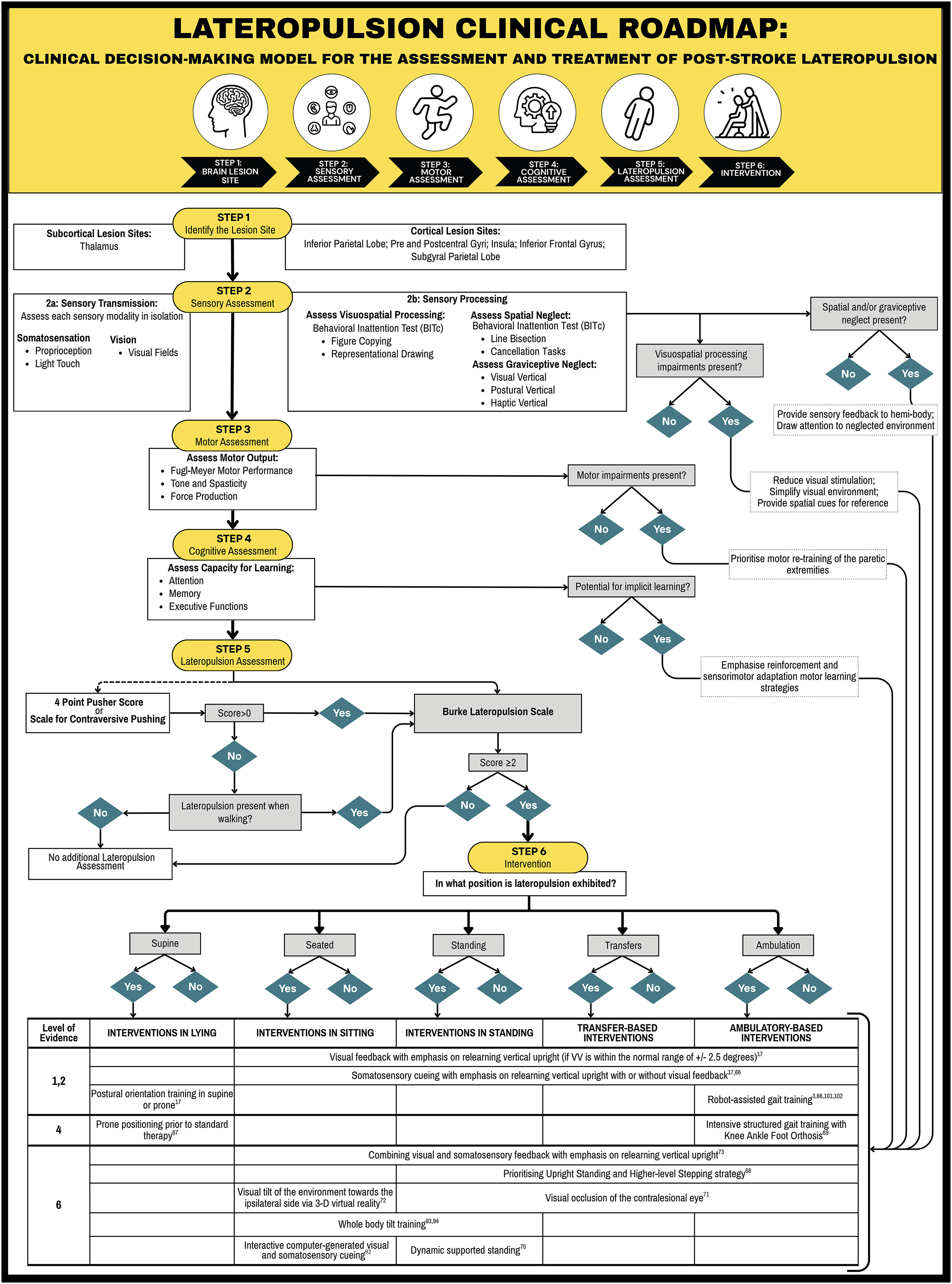
Lateropulsion clinical roadmap: clinical decision-making model for the assessment and treatment of post-stroke lateropulsion based upon realist review.
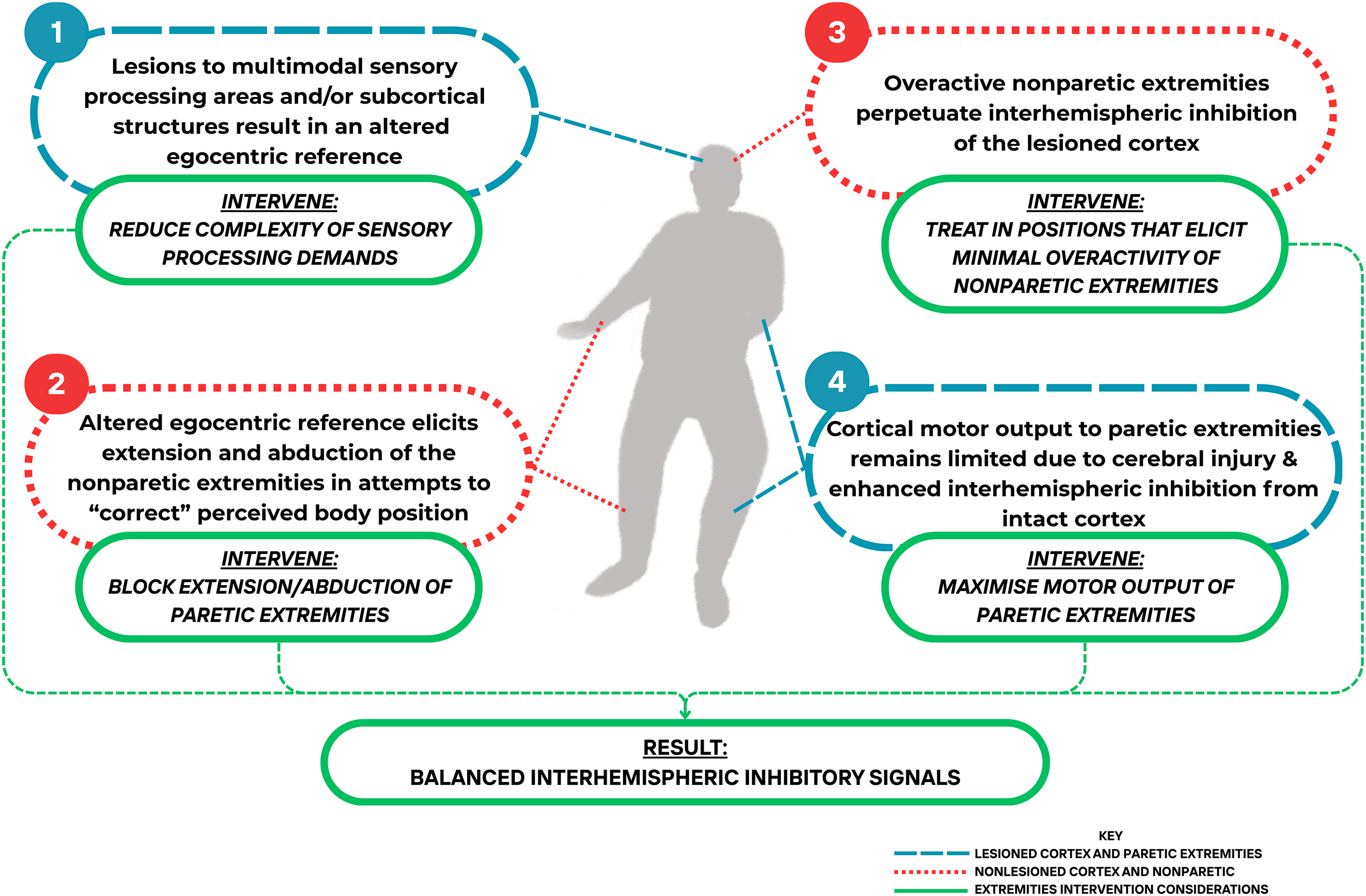
Hypothetical model linking problems associated with lateropulsion with possible interventions.
The following summaries detail the results of both searches for neuroimaging, epidemiology, assessment and intervention categories. Summaries from review articles are followed by those for non-review articles for each category. Key clinical findings that complement Table 1 and the Lateropulsion Clinical Roadmap conclude each summary.
Neuroimaging review and original studies
A systematic review by van der Waal et al. examined publications about the lesion sites associated with lateropulsion (n = 7 articles) as well as the association between verticality perception and lateropulsion (n = 11 articles; n = 1 both topics) until December 2021 (Supplemental Table 1). 4 Prominent lesion sites linked with lateropulsion included the thalamus,2,74–76 inferior parietal lobe,75,77,78 internal capsule,2,76,79 pre and post-central gyri,75,77,80 superior temporal gyrus75,78,79 and insula.4,75,76,78–80
Our second search revealed 6 studies35–40 and 1 case study 41 that were not included in the previous review (Supplemental Table 2). Some studies exclusively examined neuroimaging for people with right brain lesions.35,41 One study compiled data from 3 prior studies 35 and one was a nested sub-study from a prospective longitudinal cohort. 38 One study also included people with brain trauma and brain tumor haemorrhage with and without lateropulsion and only information relevant to supratentorial stroke was extracted. 39 Sue et al. retrospectively analysed the impact of prior stroke or small vessel disease on lateropulsion recovery. 36 Fujino et al. retrospectively studied the relationship of white matter lesions on lateropulsion severity. 37 A case study tracked a person with multiple infarctions at 4 months and 12 months post-stroke using diffusion tensor imaging to examine connectivity. 41
The second search of neuroimaging studies added several right brain sites that may be part of the body orientation network related to lateropulsion: superior occipital gyrus, 35 precuneus and cuneus, 35 superior parietal lobe,35,40 posterior corona radiata, 35 posterior thalamic radiation, 35 anterior and posterior mesial temporal lobe, 40 and, superior frontal gyrus. 35 Left brain sites associated with lateropulsion included the transverse temporal gyrus, supramarginal gyrus, paracingulate and cingulate cortex, and, insula. 40 Studies confirmed the importance of the thalamus38,39 (particularly the posterior thalamus 40 ), the pre and post-central gyri, 38 and, the parietal lobe38,39 (particularly the right inferior parietal lobe 40 ). Moderate to severe right matter lesions, 37 and white matter lesions of the frontal cortex 38 were added to body orientation network.
A key clinical point from the systematic review by van der Waal et al. includes their determination that a complex, multi-centre brain network is responsible for body orientation and control of the body against gravity (see lesion sites listed in Table 1, Lateropulsion Clinical Roadmap, and Supplemental Table 2). 4 These findings agree with the theory that lesions to one or more critical areas result in an altered egocentric reference system caused by impaired processing of both normal or abnormal somatosensory, visual and vestibular input, depending on the lesion site.4,25 Although both hemispheres have the postural orientation network, the right hemisphere network is dominant, having bilateral influence, whereas the left hemisphere network may not exert much ipsilesional influence when lesions occur.4,35,42,81,82 This latter idea links with epidemiological studies which demonstrate more protracted recovery from lateropulsion for people with right brain lesions.12,13 Understanding the history of prior stroke or the extent of white matter lesions or small vessel disease supports the theory that lateropulsion expression depends on the severity of the neural network involvement and may also assist in guiding clinical decisions and predicting a longer duration of recovery from lateropulsion.4,36,37,75,76,83
van der Waal et al. also reviewed verticality perception which addresses the egocentric reference system as a product of somatosensory, vestibular, and visual graviceptive information processing.4,82,84 Specialised tilting chairs or frames measured perceived postural vertical and demonstrated that people with lateropulsion exceeded normal error in either the contralesional 85 or ipsilesional direction.2,86 Consensus about visual vertical perceptual errors has not been reached: some reported that visual vertical was intact for people with lateropulsion,2,63 while others noted ipsilesional 87 or contralesional error76,85,88 in estimating when a visual stimulus was truly vertical. 89 A contralesional bias of haptic vertical was demonstrated for people with lateropulsion tasked with setting a hand-held bar into a vertical position. 85 This information guides clinicians to use simple tests like the bucket test to assess the degree to which a person with lateropulsion can use visual vertical perception to relearn vertical upright. 90 In some cases, explicit visual feedback with a mirror to correct posture may not be effective and more implicit training with cognitive challenges via task and environmental modifications, laser targeting or virtual reality computer gaming may help relearning of verticality.17,91–94 Similarly, if people with lateropulsion demonstrate intact haptic vertical perception, then clinicians may chose more haptic feedback like asking them to touch the ipsilesional shoulder and hip to a wall during gait training. 20
Epidemiological reviews and original studies
Nolan et al. summarised seven studies published until August 2020, which covered factors related to the prediction of the resolution of lateropulsion, rehabilitation length of stay, functional outcomes, and discharge destination after inpatient rehabilitation. 12 Dai et al. conducted a systematic review and meta-analysis of 22 studies to estimate the prevalence of lateropulsion in supratentorial and infratentorial stroke. 13 They also analysed prevalence by side, time after stroke and stroke severity from studies conducted between 1986 and 2021. 13
Supplemental Table 3 outlines 12 epidemiological studies from the second search. Four longitudinal studies,42–45 a cross-sectional observational study, 50 a retrospective observational study, 48 and one longitudinal case study 53 were reviewed. Studies examined participants in the acute and subacute inpatient rehabilitation periods46,47,51,95 and chronic phases of stroke recovery.44,45,49 One case study focused on a person with left basal ganglia and thalamic haemorrhage with subsequent pontine infarct 53 ; one case study included 4 people with parietal lobe lesions. 52
Key clinical messages from epidemiological studies include suggestions to target lateropulsion in the acute and subacute rehabilitation phases, and to plan for protracted inpatient and outpatient rehabilitation if severe lateropulsion is present, especially if right brain lesions exist. 45 The cumulative impact of other stroke deficits should be addressed throughout lateropulsion recovery.
Assessment review and original studies
Koter et al. added to a previous review of assessment tools for lateropulsion to examine 4 tools from 7 manuscripts published until March 2017.34,96 The Scale for Contraversive Pushing, Swedish Scale for Contraversive Pushing, Modified Scale for Contraversive Pushing and the Burke Lateropulsion Scale were analysed. 34
The second search identified 12 studies that focused on assessment. Two studies examined the clinometric properties of the Burke Lateropulsion Scale54,55 and one about the Scale for Contraversive Pushing. 56 Other research has emerged for assessment of longitudinal body axis, 60 weight-bearing asymmetry,57–59 unilateral spatial neglect,61,62 ocular lateral deviation, 64 somatosensation 65 and visual vertical perception 63 (Supplemental Table 4).
A key clinical message resulting from our realist review of assessment and epidemiological studies includes that clinicians should provide a formal, baseline assessment of lateropulsion severity in order to gauge rehabilitation needs, track recovery and predict duration of rehabilitation.13,21 The Burke Lateropulsion Scale 97 and the Scale for Contraversive Pushing 2 have prevailed over time.34,54,56 Although not included in the Koter et al. review, acceptable concurrent validity, convergent construct validity and strong inter and intra-rater reliability have been demonstrated for the 4-Point Pusher Score.13,98 The Lateropulsion Clinical Roadmap used the Burke Lateropulsion Scale with a cut-off score of 2 or above to define lateropulsion because this 17-point scale can track subtle changes in various functional positions. Bergmann et al. determined that a Burke Lateropulsion Scale score of 3 better defined lateropulsion when compared to a balance scale. 55
Other assessment studies focused on the theoretical root of the problem of people with lateropulsion. Barra et al. assessed perceived longitudinal body axis of supine people with and without lateropulsion and observed a contralesional tilt of this axis with lateropulsion. 60 Participants were asked to adjust a luminous 50 × 1 cm bar to align to their longitudinal body axis while in a darkened room. 60 Attempts to assess self-perceived body midline in supine, without reaction to gravity (as in postural vertical testing when upright), may also be appropriate for explicit training for body midline alignment in the early post-stroke period. Assessment of weight-bearing asymmetry with force plates 59 and Wii Balance Board57,58 aligns with egocentric reference system theory. How these tools might assist in later retraining of vertical upright requires further study.
Intervention review and original studies
Paci et al. provided a comprehensive scoping review of 31 articles about interventions for lateropulsion through 2020. Their review included case studies (n = 16), randomised controlled studies (n = 5), single-subject designs (n = 5), non-randomised controlled trials (n = 3), and two case series. 17
The second search revealed one randomised controlled trial, 66 one longitudinal, retrospective study with matching, 69 one retrospective cross-sectional study, 67 four intervention case studies,70–73 and one case series 68 in Supplemental Table 5. Using the Paci et al. 17 categorisation of studies, these additional studies fit into the categories of: visual-somatosensory integration,66,68,69,72,73 somatosensory cues67,70 or visual feedback. 71 Participants in these studies had Burke Lateropulsion Scale scores of 3 or more, 68 Scale for Contraversive Pushing score at or above 1.75, 69 or greater than 0 for each component,66,67 severe lateropulsion,70–72 or severe and protracted lateropulsion. 73
A key clinical message from these intervention studies includes categorising interventions according to the systems that they target. 17 The Lateropulsion Clinical Roadmap suggests a thorough neurological assessment so that clinicians can understand which sensory, motor, perceptual and cognitive systems are available to the patient before deciding which category of intervention would be appropriate. Visual-sensorimotor interventions included tilting the surface upon which the person with lateropulsion sat or stood.17,93,94 Such forced cognitive processing required for these training techniques was similar to the use of motor learning principles to allow the person to self-correct from a supported fall. 99 Specific body alignment retraining and use of a standing frame demonstrated that time spent in upright is important for recovery from lateropulsion.68,70,100 Other sensorimotor studies used knee-ankle-foot orthoses to provide stability during gait training for people with hemiparesis, lateropulsion and unilateral spatial neglect.66,69 The most rigorous studies support robot-assisted gait training which provided dynamic lower extremity stability and postural control when lateropulsion existed.3,17,66,101,102 Abe et al. noted that 30 minutes of daily gait training with either a robotic system or overground training using therapist support and lower extremity orthotics were equally effective in reducing lateropulsion. 66
Discussion
This realist review was appropriate for creating a practical, theory-based Lateropulsion Clinical Roadmap for evaluation and management of lateropulsion in supratentorial stroke (Figure 2, Supplemental Guide). 28 Bridging heterogeneous sources dealing with lateropulsion in a realist review format, we explored theories about lateropulsion and their clinical implications, demonstrating for whom, in what circumstances, and why interventions for lateropulsion may be effective (Table 1).22,23 Our work complements a recent Delphi process, whereby experts showed consensus on common treatment interventions for people with lateropulsion. 21
As we hypothesised, most of the research was grounded in prevalent theories that lateropulsion results from misinterpretation of multi-modal sensory inputs at the brain level yielding a faulty internal, egocentric frame of reference for verticality and/or results from graviceptive neglect whereby the sensory inputs are neglected even though they may be intact.59,88 Realist reviews allow researchers to look at available evidence and identify areas that are not addressed or not fully developed. 22 During this process, we noted that, although epidemiological studies show that people with lateropulsion tend to have moderate to severe contralesional lower extremity weakness, research about specific motor retraining was lacking.8,12,13,20 The overuse of non-paretic extremities to push toward the contralesional side, combined with the lack of use of paretic limbs, allows for a cortical imbalance between sensorimotor feedback from paretic and non-paretic limbs and perhaps for greater than normal interhemispheric inhibition of the damaged motor and sensorimotor transmission centres (Figure 3). 103 Studies using non-invasive brain stimulation over the multisensory temporo-parietal cortex that induced transient graviceptive neglect (verticality misperception) and postural control disturbance in healthy individuals support the present hypothesis of interhemispheric inhibition imbalance associated with lateropulsion.81,90,104–106 Successful forced use of the hemiplegic lower extremity through robotics or bracing aligns with this hypothesis. Protracted recovery from lateropulsion results from prolonged, non-paretic sensorimotor output perpetuating inhibition of the damaged sites of the body orientation network or primary cortices. This hypothesis requires confirmation with connectivity studies and intervention research.107–109
An important corollary of this hypothesis suggests that increased use of the paretic extremities or even electrical stimulation to the paretic side would enhance sensorimotor transmission to and from the lesioned cortex and restore interhemispheric inhibition to the non-lesioned cortex.107,110 Clinically, this concept implies that best practice may involve selecting functional positions for training in which lateropulsion is less strong, limiting over-use of the non-paretic extremities, and increasing use of the paretic extremities (through active, active-assistive or even passive movement), thus promoting balanced interhemispheric inhibition. Research suggests that the focus of interventions might be shifted to evaluating and training the key issue which is the misperception of body centring in positions that are gravity-reduced.59,62,63 Maximally engaging paretic extremities within these gravity-reduced positions, may further enhance cortical signals within the lesioned hemisphere.
Other practical suggestions emerge from this paradigm. Minimising sensory processing demands of the environment, physical challenge, and/or cognitive challenge may further minimise lateropulsion allowing for optimised learning. In contrast, some studies support the addition of a new cognitive challenge by tilting the actual93,94 or virtual support surface 72 from the horizontal in order to bypass faulty somatosensory processing or augment intact somatosensory processing. Clinicians may consider evaluating whether people with lateropulsion respond better to implicit training (e.g., gaming with virtual reality avatars, training for related tasks like side-stepping with body-weight support, using robot-assisted gait training) versus explicit training of body centring.21,35,72,93,111,112 All of these strategies require further research before they can be recommended in routine clinical practice.
Table 1 indicates where weak evidence exists and enumerates areas for future study. We agree with other authors that more randomised, controlled intervention trials are needed to strengthen the evidence-base for lateropulsion rehabilitation.17,21 Structural and functional connectivity studies are required for a better understanding of the neural network related to lateropulsion. Future research may consider investigating the effect and appropriate staging of challenges to the sensory, motor, or cognitive systems under various task and environmental conditions, the impact of sensory feedback and attentional focus, as well as the impact of paretic extremity engagement (with passive or active assistive functional tasks or through functional electrical stimulation) on the severity of lateropulsion. A paradigm shift from an explicit-based functional treatment approach to an implicit-based recalibration of sensorimotor signals and processing and graviceptive reference may be warranted.
This realist review presents with limitations. Selecting only articles published in English may have biased results. Case study and case series evidence was included but future, more robust research would need to confirm these findings. Our foundation for interpreting existing research rested on theories and neuroimaging associated with lateropulsion. Others may look at the findings from different perspectives and derive further hypotheses. Such subjective interpretation is inherent in realist reviews. 22 We do not expect prescriptive generalisability of the findings because people with stroke and lateropulsion have varying degrees of deficit and diverse stroke-related symptoms. We opted for the ‘If A, then B and If C, then D’ paradigm (Table 1) because participants in individual studies often presented or responded differently, depending on lesion side/site and other stroke-related problems, but other ways to examine the literature are possible.
In conclusion, a Lateropulsion Clinical Roadmap (Figure 2, Supplemental Guide) and clinical recommendations (Table 1, Figure 3) were easily derived from a realist review. New ideas include focusing on correcting the egocentric reference system and using implicit learning strategies after a thorough assessment of how stroke-related deficits impact relearning of the vertical upright posture. Clinicians are encouraged to use the Lateropulsion Clinical Roadmap to conduct a thoughtful neurological assessment and to select and stage interventions based on somatosensory processing demands of the training tasks, as well as the level of task challenge that may impact the degree of lateropulsion. This realist review has highlighted current evidence gaps and informed future research to globally progress care for people with lateropulsion.
Clinical Messages
The Lateropulsion Clinical Roadmap, derived from a realist review, suggests consideration of brain lesion sites, sensory transmission and processing / perception, motor performance, and cognition, in addition to the degree of lateropulsion, when deciding a plan of care for people with lateropulsion after supratentorial stroke. Clinicians should evaluate the impact of functional positions and activities in which lateropulsion is present during interventions and select appropriately difficult tasks, environments and sensorimotor challenges during functional training for post-stroke lateropulsion. A key to managing lateropulsion may rest in engagement of paretic extremities when lateropulsion is not exacerbated, thus counterbalancing inhibitory influences of the overactive, non-lesioned cortex. Future research must confirm this hypothesis. Clinicians are encouraged to review cited literature and critically apply findings within the framework of the Lateropulsion Clinical Roadmap when selecting appropriate management strategies for people with lateropulsion after stroke.
Supplemental Material
sj-pdf-1-cre-10.1177_02692155261418609 - Supplemental material for A clinical roadmap for lateropulsion after stroke based on a realist review strategy
Supplemental material, sj-pdf-1-cre-10.1177_02692155261418609 for A clinical roadmap for lateropulsion after stroke based on a realist review strategy by Suzanne Babyar, Nicholas Sheehan, Jessica Nolan, Taiza G.S. Edwards, Jeannine Bergmann, Amy Meyer and Michael W. O’Dell in Clinical Rehabilitation
Supplemental Material
sj-pdf-2-cre-10.1177_02692155261418609 - Supplemental material for A clinical roadmap for lateropulsion after stroke based on a realist review strategy
Supplemental material, sj-pdf-2-cre-10.1177_02692155261418609 for A clinical roadmap for lateropulsion after stroke based on a realist review strategy by Suzanne Babyar, Nicholas Sheehan, Jessica Nolan, Taiza G.S. Edwards, Jeannine Bergmann, Amy Meyer and Michael W. O’Dell in Clinical Rehabilitation
Supplemental Material
sj-pdf-3-cre-10.1177_02692155261418609 - Supplemental material for A clinical roadmap for lateropulsion after stroke based on a realist review strategy
Supplemental material, sj-pdf-3-cre-10.1177_02692155261418609 for A clinical roadmap for lateropulsion after stroke based on a realist review strategy by Suzanne Babyar, Nicholas Sheehan, Jessica Nolan, Taiza G.S. Edwards, Jeannine Bergmann, Amy Meyer and Michael W. O’Dell in Clinical Rehabilitation
Supplemental Material
sj-pdf-4-cre-10.1177_02692155261418609 - Supplemental material for A clinical roadmap for lateropulsion after stroke based on a realist review strategy
Supplemental material, sj-pdf-4-cre-10.1177_02692155261418609 for A clinical roadmap for lateropulsion after stroke based on a realist review strategy by Suzanne Babyar, Nicholas Sheehan, Jessica Nolan, Taiza G.S. Edwards, Jeannine Bergmann, Amy Meyer and Michael W. O’Dell in Clinical Rehabilitation
Supplemental Material
sj-pdf-5-cre-10.1177_02692155261418609 - Supplemental material for A clinical roadmap for lateropulsion after stroke based on a realist review strategy
Supplemental material, sj-pdf-5-cre-10.1177_02692155261418609 for A clinical roadmap for lateropulsion after stroke based on a realist review strategy by Suzanne Babyar, Nicholas Sheehan, Jessica Nolan, Taiza G.S. Edwards, Jeannine Bergmann, Amy Meyer and Michael W. O’Dell in Clinical Rehabilitation
Supplemental Material
sj-pdf-6-cre-10.1177_02692155261418609 - Supplemental material for A clinical roadmap for lateropulsion after stroke based on a realist review strategy
Supplemental material, sj-pdf-6-cre-10.1177_02692155261418609 for A clinical roadmap for lateropulsion after stroke based on a realist review strategy by Suzanne Babyar, Nicholas Sheehan, Jessica Nolan, Taiza G.S. Edwards, Jeannine Bergmann, Amy Meyer and Michael W. O’Dell in Clinical Rehabilitation
Footnotes
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
