Abstract
Objective
To investigate whether presence of aphasia is associated with differences in inpatient stroke rehabilitation care and outcomes.
Design
Observational study of cross-sectional data from hospitals participating in the biennial National Stroke Audit – Rehabilitation Services (Stroke Foundation, 2016, 2018 and 2020). Descriptive statistics and multi-level multi-variable logistic regression were used to examine associations between aphasia status, processes of care, and patient outcomes, with level defined as hospital.
Setting
Australian inpatient rehabilitation hospitals.
Participants
All patients with stroke who had aphasia status recorded.
Main measures
Adherence to processes of care recommended in the Australian and New Zealand Clinical Guidelines for Stroke Management. In-hospital outcomes included complications, independence, survival, and discharge destination.
Results
Overall, 9960 audits were included; 3160 (33%) patients had aphasia (56% male; median age 75; no aphasia: 56% male; median age 76). Patients with aphasia were more likely to have a mood impairment (54% vs. 44%) and less likely to be assessed by a psychologist (40% vs. 49%). Fewer patients with aphasia were involved in goal setting (84% vs. 88%) or development of care plans (91% vs. 96%); or asked about returning to work (67% vs. 74%) or driving (41% vs. 45%). Patients with aphasia had a longer length of stay (median 26 vs. 21 days, p≤0.001) and were less independent on discharge (adjusted odds ratio (aOR) 0.80, 95% CI 0.71, 0.90).
Conclusions
Identified differences in stroke rehabilitation highlight the need for equitable access to care and effective patient-provider communication enabling people with aphasia to more fully participate in their rehabilitation.
Introduction
The importance of rehabilitation in improving post-stroke recovery and outcomes is well-recognised. 1 Principles of effective rehabilitation have been broadly categorised across three domains: context, process and intervention. 2 Stroke and rehabilitation guidelines recommend processes of care, based on best available research evidence, to support the provision of high-quality rehabilitation 1 across these domains. The delivery of safe, high-quality, patient-centred care is dependent on effective patient-provider communication.3,4 Thus, inefficiencies and breakdown in communication can impact on healthcare delivery. 5 This is particularly relevant in stroke rehabilitation, where two-thirds of patients have communication support needs. 6 Approximately one-third of people experience aphasia post-stroke, 7 an acquired language impairment impacting communication. Patients with aphasia experience difficulty communicating and participating in their healthcare,8,9 and healthcare professionals report challenges communicating with people with aphasia. 10 Communication difficulties have been associated with poorer patient outcomes post-stroke, including an increased risk of adverse events 11 and complications 12 ; longer hospital stays7,12; higher mortality rates7,12; variation in discharge destinations7,13 and higher levels of disability.7,14
The provision of high-quality evidence-based stroke care is essential to improve health outcomes following stroke. This is especially pertinent for people with communication impairments due to their poorer outcomes. However, the clinical presentation of patients with aphasia may disrupt the delivery of effective rehabilitation care. 14 This is critical to understand and address, because inpatient rehabilitation provides the most intensive rehabilitation care1,15; is a key transition point between hospital and community 15 and greatest improvements in language are associated with early intervention commenced within 1 month post-stroke. 16 Furthermore, the absence of adequate rehabilitation and support has implications on unmet needs relating to quality of life, social reintegration, self-efficacy and self-identity.1,17
While differences in acute stroke care for patients with and without aphasia have been investigated in recent Australian studies,18,19 the influence of aphasia on stroke rehabilitation is less understood. Hence, the aim of this study was to investigate whether there were differences in the provision of evidence-based stroke care and in-hospital outcomes in Australian rehabilitation hospitals for people with and without aphasia.
Methods
This was a cross-sectional, observational study of retrospective data collected from Australian hospitals participating in the biennial National Stroke Audit – Rehabilitation Services (henceforth the Rehabilitation Audit) conducted in 2016, 2018 and 2020. The Rehabilitation Audit aims to promote evidence-based care and comprises two components: an organisational survey and a clinical audit. 20 The organisational survey collects information to describe the characteristics and resources of hospitals providing stroke rehabilitation. The clinical audit identifies patient-level adherence to clinical guidelines through a retrospective case note audit of up to 40 consecutive stroke admissions at each participating hospital in a pre-determined period. Data abstraction follows a systematic process using a standardised web-based data collection platform with in-built logic checks and manual inter-rater reliability checks.20,21
Data from hospitals participating in any of the three audit cycles were aggregated. The presence of aphasia was determined by a Yes/No response to the clinical audit question ‘Did the patient have aphasia?’. This was determined by a stroke clinician documenting the presence of aphasia in the clinical notes at any stage during the acute admission. Aphasia type and severity were not recorded in the audit. In instances where aphasia status was unknown, these cases were excluded from further analysis. Patient demographics, clinical characteristics on admission, evidence-based processes of care variables and outcome indicators were collected for all included patients.
Patient demographics included information such as age and sex. Clinical characteristics included type of stroke, impairments on admission, level of independence (modified Rankin Scale [mRS] and Cognitive and Motor components of the Functional Independence Measure [FIM]), as well as time post stroke-onset and type of rehabilitation ward. The processes of care collected reflect agreed national clinical guideline recommendations for stroke rehabilitation.20,22 These included allied health assessments and management of impairments (e.g. mobility, neglect, continence, nutrition, mood and activities of daily living), as well as elements of care pertaining to communication, support and discharge planning (e.g. goal setting, carer training, discussions about return to work and driving, secondary prevention education and development of care plans). In-hospital outcomes included complications during rehabilitation admission, mortality, length of stay, discharge destination and independence at discharge (mRS 0–2 23 ; FIM cognitive 30–35; FIM motor 78–91 24 ).
Data analyses
Patient demographics, clinical characteristics, processes of care and outcomes were compared for people with and without aphasia post-stroke. For questions relating to demographics and clinical characteristics, ‘not documented’ and ‘unknown’ responses were excluded from the denominator, and only valid Yes/No responses were included in the analysis. For audit questions relating to processes of care, ‘not documented’ and ‘unknown’ responses were both assumed to be negative (i.e. care process not provided) and were included in the denominator for the analysis. These approaches are consistent with previous stroke audit studies.25,26 Records with missing responses for specific questions were considered incomplete and excluded from the analysis of those questions.
Chi-square and Kruskall–Wallis tests were used to compare patient demographics and clinical characteristics by aphasia status. Adherence to processes of care were compared by aphasia status using multi-level logistic regression. It was considered that all patients, regardless of their clinical profile, should receive all recommended processes of care for which they were eligible. Accordingly, these models were only adjusted for patient variation within hospitals, consistent with previous studies exploring differences in processes of care.27,28 The denominators for each process of care accounted for ineligible patients (e.g. processes of care only recommended for ischaemic stroke were measured only for patients with this type of stroke). Additional sensitivity analyses were also undertaken adjusting process of care adherence models for patient and clinical factors (as per outcome analysis outlined below) and audit year.
Logistic and quantile/median multi-level multivariable regression were used to compare in-hospital outcomes by aphasia status. These models were adjusted for potential confounders known to be associated with stroke outcomes, 29 including patient demographics (age and sex) independence on admission (mRS 0–2; FIM cognitive ≥30; FIM motor ≥78) and type of stroke (haemorrhagic). Arm impairment was also included as a confounder indicative of stroke severity, based on a validated prognostic model for predicting stroke outcome. 29 Inability to walk and speech impairment on admission were correlated with mRS and aphasia status, respectively, and were therefore not included in the models as stroke severity indicators. All variables were tested for collinearity before inclusion. Models were also adjusted for hospital variation, with sensitivity analysis undertaken for audit year.
Data were analysed using Stata/SE 18.0. Level of significance was p < 0.05. The study and manuscript conform to the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guideline. 30 Ethical approval was granted by Monash University Human Research Ethics Committee (Project ID 35037) and ratified by the University of Queensland Ethics Committee (Project No. 2023/HE000015). The stroke services of participating hospitals volunteered to participate in the Rehabilitation Audit.
Results
Patient demographics and clinical characteristics on admission
There were 10,007 rehabilitation admissions audited from 133 participating hospitals across the three audit cycles. Of these, 9960 (>99%) had a recorded response regarding presence or absence of aphasia and were included in the analyses, with 3160 (33%) reported to have aphasia and 6500 (67%) reported not to have aphasia.
Patients with aphasia were more likely to be younger and were admitted to inpatient rehabilitation a median of one day later post-stroke than patients without aphasia (Table 1). Compared with patients without aphasia, patients with aphasia were more likely to experience a haemorrhagic stroke (19% vs. 16%) and present with impairments such as sensory, cognitive and perceptual deficits, and hydration and nutrition problems. On admission, patients with aphasia were less likely to be independent on the mRS (9% vs. 11%) or FIM cognitive (12% vs. 37%), when compared to those without aphasia. Patients with aphasia were more likely to be able to walk independently and were more independent on the FIM motor (12% vs. 8%). Although numbers were small, patients with aphasia were also more likely to present with certain complications on admission.
Participant demographics and clinical characteristics by aphasia status.
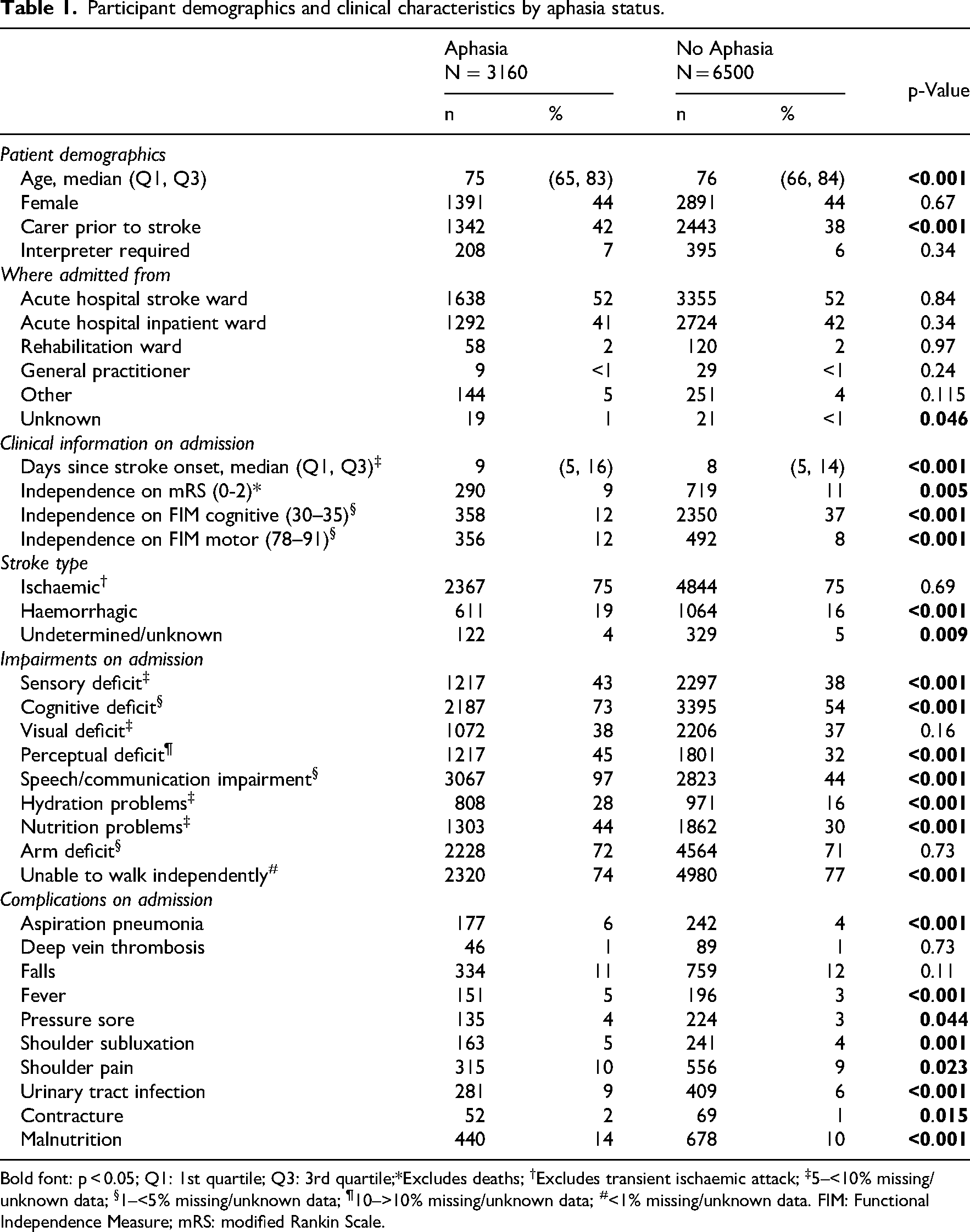
Bold font: p < 0.05; Q1: 1st quartile; Q3: 3rd quartile;*Excludes deaths; †Excludes transient ischaemic attack; ‡5–<10% missing/unknown data; §1–<5% missing/unknown data; ¶10–>10% missing/unknown data; #<1% missing/unknown data. FIM: Functional Independence Measure; mRS: modified Rankin Scale.
Processes of care
Compared to patients without aphasia, patients with aphasia were more likely to be seen by a speech pathologist (99% vs. 93%; aOR 11.57; 95% CI 7.47, 17.91), social worker (89% vs. 87%; aOR 1.27; 95% CI 1.09, 1.49) and dietitian (82% vs. 77%; aOR 1.34; 95% CI 1.16, 1.54), with high rates of assessment across both groups (Table 2). Patients with aphasia were less likely to be assessed by a psychologist (overall: 24% vs. 27%; aOR 0.79; 95% CI 0.68, 0.92; if mood impairment: 40% vs. 49%; aOR 0.59; 95% CI 0.46, 0.75), despite an increased likelihood of having a mood impairment if assessed (54% vs. 44%; aOR 1.47; 95% CI 1.30, 1.66). In terms of mood management, patients with aphasia were more likely to be treated with anti-depressants (61% vs. 57%; aOR 1.19; 95% CI 1.00, 1.42) and less likely to receive psychological cognitive-behavioural interventions (40% vs. 44%; aOR 0.82; 95% CI 0.68, 0.99). There were no differences in being treated on a dedicated stroke rehabilitation unit between the two groups.
Regression analyses of associations between aphasia status and processes of care relating to assessment and management of impairments.
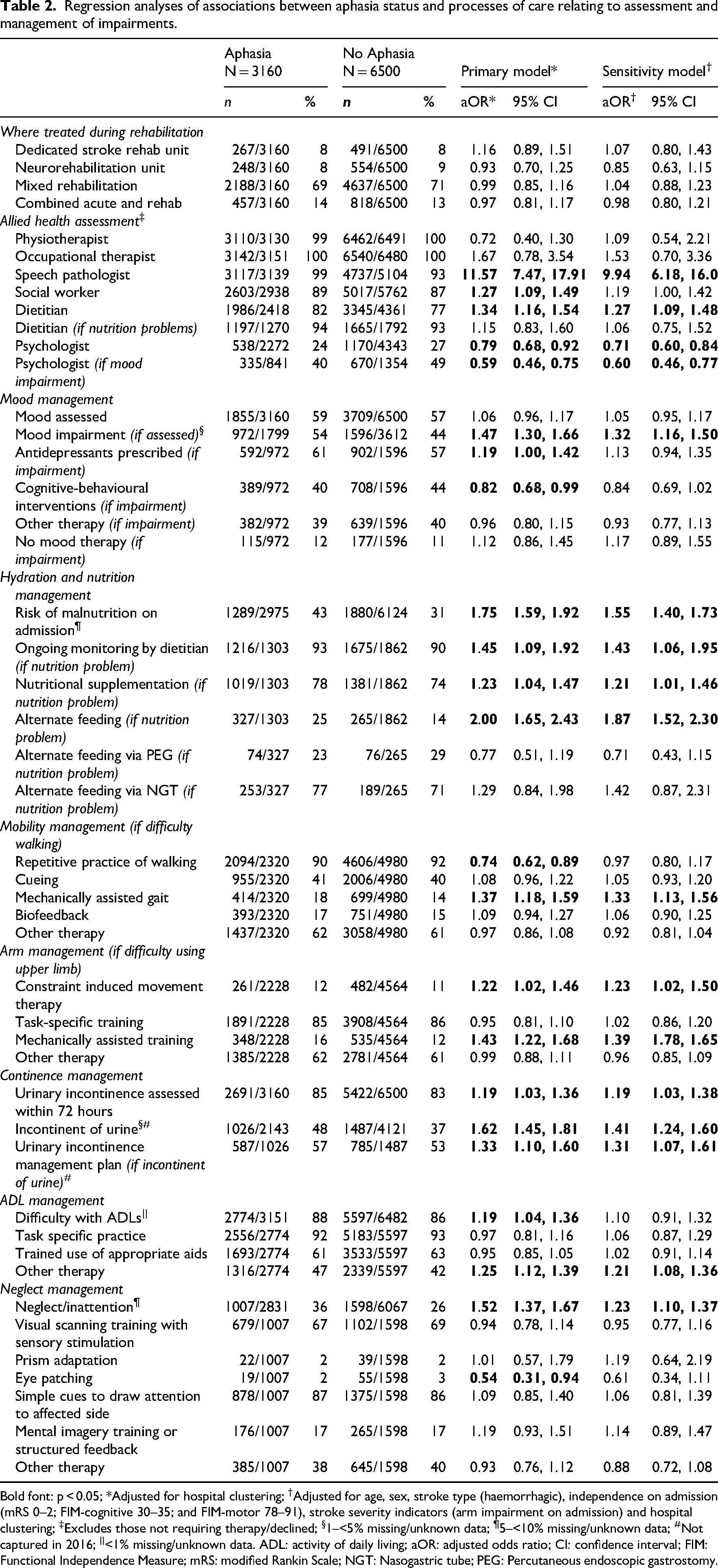
Bold font: p < 0.05; *Adjusted for hospital clustering; †Adjusted for age, sex, stroke type (haemorrhagic), independence on admission (mRS 0–2; FIM-cognitive 30–35; and FIM-motor 78–91), stroke severity indicators (arm impairment on admission) and hospital clustering; ‡Excludes those not requiring therapy/declined; §1–<5% missing/unknown data; ¶5–<10% missing/unknown data; #Not captured in 2016; ||<1% missing/unknown data. ADL: activity of daily living; aOR: adjusted odds ratio; CI: confidence interval; FIM: Functional Independence Measure; mRS: modified Rankin Scale; NGT: Nasogastric tube; PEG: Percutaneous endoscopic gastrostomy.
Differences in adherence to processes of care related to management of impairments are outlined in Table 2. Of note, there were differences in nutrition management, whereby patients with aphasia were more likely to receive nutritional supplementation (78% vs. 74%; aOR 1.23; 95% CI 1.04, 1.47) and were twice as likely to receive alternate feeding (25% vs. 14%; aOR 2.00; 95% CI 1.65–2.43). There were also differences in continence management by aphasia status. Patients with aphasia were more likely to be assessed for urinary incontinence, although assessment rates for both groups were high (85% vs. 83%; aOR 1.19; 95% CI 1.03, 1.36). They were more likely to be incontinent (48% vs. 37%; aOR 1.62; 95% CI 1.45, 1.81), and a continence plan was documented more often for patients with aphasia compared to those without (57% vs. 53%; aOR 1.33; 95% CI 1.10, 1.60).
In terms of communication and support (Table 3), adherence to goal setting was high across both groups, with goals more likely to be set for patients with aphasia than those without (93% vs. 92%; aOR 1.29; 95% CI 1.08, 1.54). Patients with aphasia were less likely to be involved in the goal setting process (84% vs. 88%; aOR 0.74; 95% CI 0.65, 0.85) and there was a greater reliance on family only involvement (8% vs. 4%; aOR 2.47; 95% CI 2.04, 2.99). Similarly, the multidisciplinary team were more likely to meet to discuss management of patients with aphasia (90% vs. 87%; aOR 1.50; 95% CI 1.28, 1.76), however there was a greater reliance solely on family involvement in these discussions (8% vs. 4%; aOR 2.26; 95% CI 1.87, 2.74). Patients with aphasia and/or their family were more likely to receive information about stroke management (61% vs. 58%; aOR 1.16; 95% CI 1.05, 1.28), and their carers were more likely to receive support needs assessments (68% vs. 65%; aOR 1.17; 95% CI 1.00, 1.38) and training (73% vs. 68%; aOR 1.33; 95% CI 1.13, 1.57).
Regression analyses of associations between aphasia status and processes of care relating to communication, support and discharge planning.
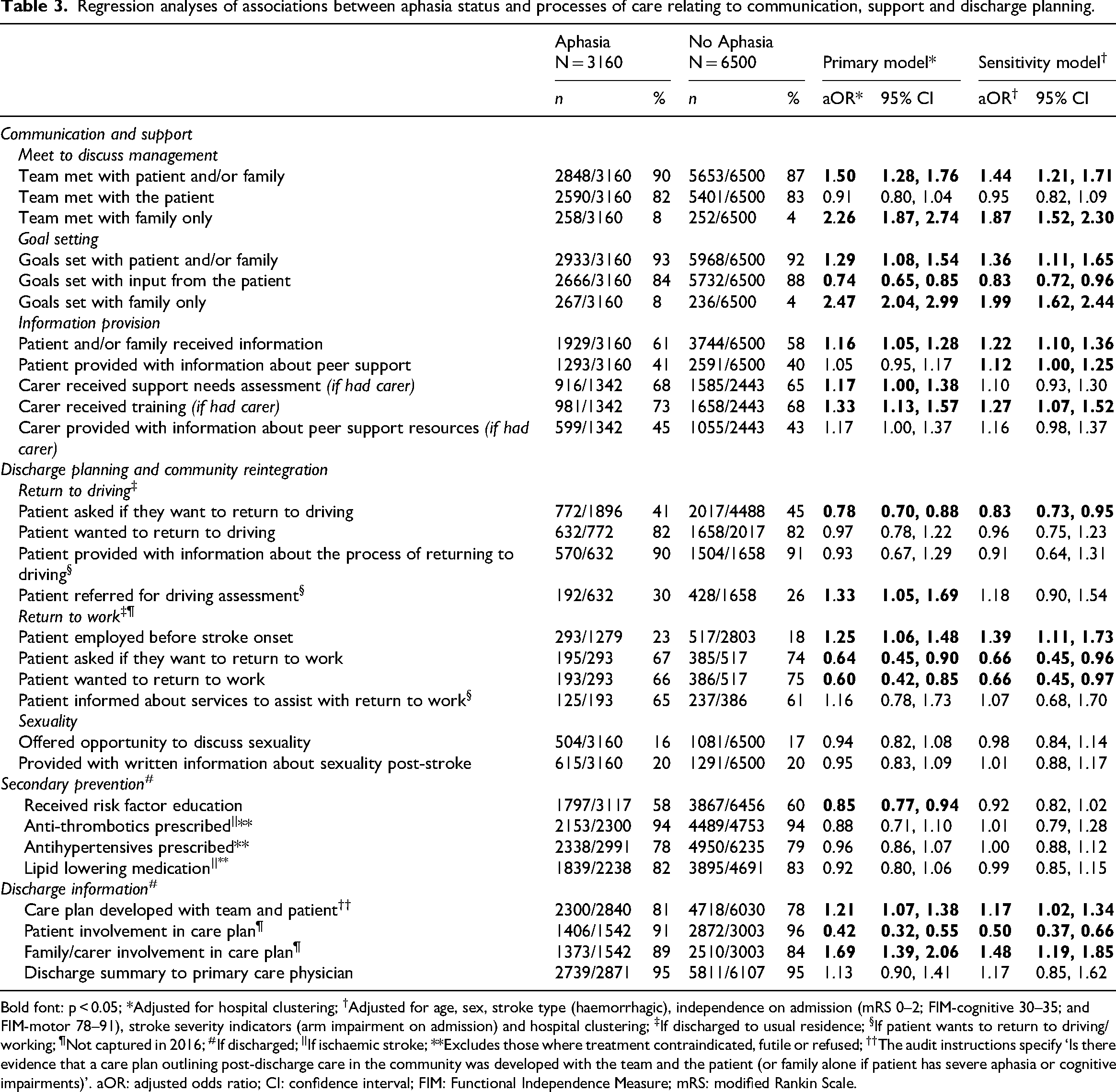
Bold font: p < 0.05; *Adjusted for hospital clustering; †Adjusted for age, sex, stroke type (haemorrhagic), independence on admission (mRS 0–2; FIM-cognitive 30–35; and FIM-motor 78–91), stroke severity indicators (arm impairment on admission) and hospital clustering; ‡If discharged to usual residence; §If patient wants to return to driving/working; ¶Not captured in 2016; #If discharged; ||If ischaemic stroke; **Excludes those where treatment contraindicated, futile or refused; ††The audit instructions specify ‘Is there evidence that a care plan outlining post-discharge care in the community was developed with the team and the patient (or family alone if patient has severe aphasia or cognitive impairments)’. aOR: adjusted odds ratio; CI: confidence interval; FIM: Functional Independence Measure; mRS: modified Rankin Scale.
With regard to discharge planning (Table 3), although more patients with aphasia were likely to work pre-stroke (23% vs. 18%; aOR 1.25; 95% CI 1.06, 1.48), they were less likely to be asked if they wanted to return to work (67% vs. 74%; aOR 0.64; 95% CI 0.45, 0.90) or driving (41% vs. 45%; aOR 0.78; 95% CI 0.70, 0.88), and were more likely to be referred for driving assessments (30% vs. 26%; aOR 1.33; 95% CI 1.05, 1.69) than those without aphasia. Patients with aphasia were less likely to receive behaviour change education (58% vs. 60%; aOR 0.85; 95% CI 0.77, 0.94). While they were more likely to have a care plan (81% vs. 78%; aOR 1.21; 95% CI 1.07, 1.38), patients with aphasia were less likely to be involved in their care plan development (91% vs. 96%; aOR 0.42; 95% CI 0.32, 0.55), with a greater reliance on family and carer involvement (89% vs. 84%; aOR 1.69; 95% CI 1.39, 2.06).
Differences in care following sensitivity analyses adjusting for patient and clinical factors are outlined in Tables 2 and 3 (see Sensitivity Model section). Of note, the differences between groups for psychological cognitive-behavioural interventions, anti-depressants, risk factor education and driving assessment were not statistically significant when adjusted for further patient and clinical factors. There was no change in statistical significance across processes of care when adjusted for audit year.
In-hospital outcomes
Some differences in in-hospital outcomes were identified for patients with and without aphasia (Table 4). Compared to patients without aphasia, those with aphasia had a longer length of stay. Although numbers were small, patients with aphasia had an increased likelihood of experiencing aspiration pneumonia (6% vs. 3%; aOR 1.46; 95% CI 1.17, 1.81), fever (10% vs. 7%; aOR 1.26; 95% CI 1.06, 1.49) and urinary tract infection (16% vs. 12%; aOR 1.23; 95% CI 1.07, 1.40) during their rehabilitation admission.
Regression analyses of associations between aphasia status and in-hospital outcomes.
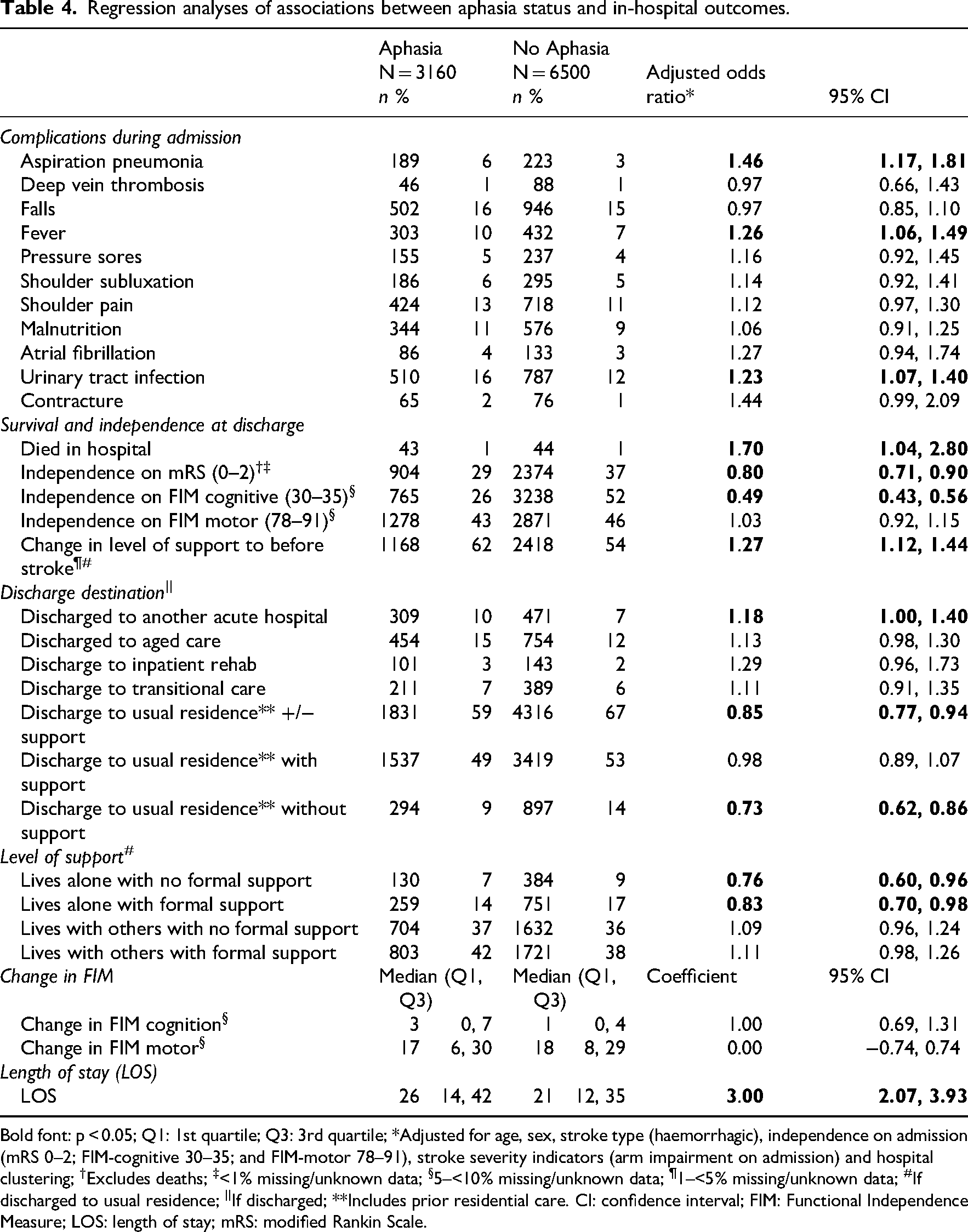
Bold font: p < 0.05; Q1: 1st quartile; Q3: 3rd quartile; *Adjusted for age, sex, stroke type (haemorrhagic), independence on admission (mRS 0–2; FIM-cognitive 30–35; and FIM-motor 78–91), stroke severity indicators (arm impairment on admission) and hospital clustering; †Excludes deaths; ‡<1% missing/unknown data; §5–<10% missing/unknown data; ¶1–<5% missing/unknown data; #If discharged to usual residence; ||If discharged; **Includes prior residential care. CI: confidence interval; FIM: Functional Independence Measure; LOS: length of stay; mRS: modified Rankin Scale.
On discharge, patients with aphasia were less likely to be independent on both the mRS (29% vs. 37%; aOR 0.80; 95% CI 0.71, 0.90) and the FIM cognitive (26% vs. 52%; aOR 0.49; 95% CI 0.43, 0.56). Patients with aphasia were more likely to have a change in level of support compared to pre-stroke (62% vs. 54%; aOR 1.27; 95% CI 1.12, 1.44). Patients with aphasia were more likely to be discharged to another acute hospital (10% vs. 7%; aOR 1.18; 95% CI 1.00, 1.40). They were less likely to be discharged to their usual residence, either with or without support (59% vs. 67%; aOR 0.85; 95% CI 0.77, 0.94), and were less likely to live alone post-discharge, either with (14% vs. 17%; aOR 0.83; 95% CI 0.70, 0.98) or without (7% vs. 9%; aOR 0.76; 95% CI 0.60, 0.96) formal support. There was no change in statistical significance of differences in outcomes when adjusted for audit year.
Discussion
For the first time, we compared inpatient rehabilitation care and outcomes for patients with and without aphasia in Australia and found important differences in stroke care. Most notably, differences were identified in person-centred care, mood management and community reintegration. Patients with aphasia also experienced different in-hospital outcomes, which, consistent with previous studies,7,12,13 included a longer length of stay, and a decreased likelihood of being independent, or discharged home. The longer length of stay may represent worse outcomes or could instead be reflective of increased intensive rehabilitation needs in the context of cognitive and communication impairment. Communication is fundamental to patient-centred 4 and effective rehabilitation. 1 Yet known communication challenges exist for patients with aphasia, and as such, identified differences in care require careful examination.
Rehabilitation should begin as soon as possible post-stroke. 31 In this study, patients with aphasia were admitted to inpatient rehabilitation one day later on average, potentially due to their increased impairments and complications. On a positive note, however, there was no difference in admission to dedicated stroke rehabilitation units for people with and without aphasia. This is important given stroke unit care has been associated with improved outcomes, 32 arguably due to structured processes 2 and specialist multidisciplinary care. We nevertheless found differences in rehabilitation processes of care received by patients with and without aphasia.
Person-centred care is foundational to rehabilitation 33 and synonymous with high-quality care. 34 Within a model of person-centred care, individuals are recognised as active partners in their care. 33 Patient participation in decision making and goal setting have been emphasised by stakeholders as key components of successful person-centred rehabilitation.33,35 Both of these processes can be impacted by patient–provider interactions, 10 and as such, person-centred care has been identified as a quality indicator for aphasia services. 36 In this study, patients with aphasia were less likely to be involved in goal setting or care plan development. Instead, there was greater reliance on family involvement in goal setting, care planning and management discussions, when compared to those without aphasia. The impact of aphasia type and severity on patient participation in these components of care is unknown, and person-centred care for patients with severe aphasia might appropriately include greater family involvement. Nevertheless, these findings align with previous research, which shows that patients with communication difficulties experience decreased involvement in decision making, and reduced participation in healthcare discussions.9,37 Furthermore, healthcare workers have described limiting or avoiding communication with patients with aphasia, 38 and greater reliance on their family members. 9 Similarly, while patients with aphasia and/or their family were more likely to receive information about stroke management, it is unknown to what extent patients with aphasia were involved in the information provision process. Patients with aphasia were less likely to receive education about behaviour change for modifiable risk factors; yet there were no differences in the prescription of stroke prevention medications for people with and without aphasia. These findings suggest that the presence of aphasia may have been a barrier to interventions that are dependent on patient participation, such as education. Reassuringly, carers of patients with aphasia were more likely to receive carer training. Finding ways to support the involvement of people with aphasia is critical. People with aphasia have expressed their desire for increased involvement in their healthcare.9,39 Furthermore, participation may influence the effectiveness of stroke rehabilitation 40 and contribute to better outcomes 41 ; and self-management is associated with improved self-efficacy and quality of life post-stroke. 42 Our findings support the need for staff training, education and support to improve patient–provider communication,10,40,43 and the promotion of communication-accessible environments to enhance person-centred care for patients with aphasia. 44
Successful rehabilitation and the delivery of high-quality stroke care is also dependent on a multidisciplinary team with specialist expertise.2,45 Encouragingly, in this study, patients with aphasia were more likely to be assessed by a speech pathologist, social worker and dietitian. However, patients with aphasia, either with or without an identified mood impairment, were less likely to be assessed by a psychologist during their rehabilitation admission. This is noteworthy, given the increased rates of mood impairment for patients with aphasia in this study, compared to those without. For those with a mood impairment, patients with aphasia were more likely to be managed with prescription of antidepressants and less likely to receive psychological cognitive-behavioural interventions compared to patients without aphasia. This could be related to the reduced psychologist input, and may suggest a lack of skill or confidence in working with patients with communication-support needs. Further research is needed to understand these differences, given evidence for cognitive-behavioural interventions 46 and the known prevalence of psychological distress and post-stroke depression experienced by people with aphasia. 47
The ultimate goal of rehabilitation is to optimise quality of life and social integration. 2 Thus, the focus of rehabilitation should extend to community reintegration, 15 including activities such as employment and driving. People with aphasia face barriers in returning to driving 48 and work, 49 and there is a lack of clinical practice guidance to support return to these activities. 50 Returning to driving has been identified as a top-ten research priority by people with aphasia, 51 however, in our study, patients with aphasia were less likely to be asked about returning to driving. For those who were asked, there was no difference in the provision of information about return to driving, and people with aphasia were more likely to be referred for driving assessments. This is promising, given those who receive advice and evaluations are more likely to return to driving post-stroke. 52 Patients with aphasia were also less likely to be asked about returning to work, and when asked, were less likely to want to return to work. This finding aligns with previous research in which people with aphasia did not identify return to work as a top research priority. 51 However, the presumption that this finding is related to people with aphasia already being at retirement age 51 is incongruent with the increased likelihood of people with aphasia working pre-stroke in this study, compared to those without aphasia.
This study has several strengths. To minimise reporting and selection bias, data collection for the Rehabilitation Audit followed a standardised data collection process. This included extracting data for the first 40 consecutive stroke admissions over a pre-defined period, and inter-rater reliability and data verification checks. 20 The Rehabilitation Audit is a comprehensive national dataset, and the processes of care investigated reflect Australian stroke rehabilitation guidelines. We acknowledge several limitations. Firstly, the retrospective and cross-sectional data provides a snapshot in time and may not be representative of all stroke presentations. The findings also reflect stroke care in the Australian hospital context, which may not be generalisable to other settings or countries. Although audit year was included as a sensitivity analysis, the characteristics of the participating hospitals and audited patients may differ between years. The identification of aphasia was based on Yes/No documentation in clinical notes, and the severity and type of aphasia were not captured. As such, the potential impact of aphasia characteristics on care provision and participation is unknown. Assessment to determine the presence and severity of aphasia has been identified as a quality indicator for post-stroke aphasia. 36 Collection of this information in future audits would support an understanding of the impact of aphasia severity on participation and quality of care. While the proportion of patients with aphasia reported in the study is consistent with prevalence reported in other rehabilitation studies, 7 the validity of aphasia diagnosis is unknown. As the Rehabilitation Audit does not include a stroke severity scale, impairments and independence on admission were used to adjust for stroke severity and case-mix, based on validated prognostic models. 29 However, we acknowledge that additional unmeasured confounders may have contributed to stroke outcomes, and we cannot definitively conclude that the identified differences in in-hospital outcomes can be fully attributed to the presence of aphasia. As such, the outcome findings should be interpreted cautiously. Similarly, additional clinical factors may have contributed to identified differences in quality of care for patients with and without aphasia, and further exploration of these factors is warranted. There was also the potential for reporting and documentation bias, whereby documentation practices may vary for patients with and without aphasia. Finally, while this study contributes to a wider body of work investigating quality of stroke care and outcomes for people with aphasia, it does not include processes of care specific to aphasia. Research is underway to address this gap, including the development 36 and pilot 53 of a minimum set of quality indicators for aphasia in hospital settings.
In summary, identified differences in care and outcomes for those with and without aphasia highlight areas for improvement to support equitable care and increased participation of people with aphasia in their rehabilitation.
Clinical messages
Differences in stroke rehabilitation care and outcomes exist for patients with aphasia, compared to those without aphasia.
Patients with aphasia were less likely to be involved in some aspects of their care, and there was a greater reliance on family involvement.
Further investigation and improvement in mood management, risk factor education, and return to work and driving is indicated.
Communication partner training, resources and support are needed to promote person-centred rehabilitation care for patients with aphasia.
Footnotes
Acknowledgements
We acknowledge the hospitals and clinicians that participated in the National Stroke Audit. The Stroke Foundation approved access to the National Stroke Audit data.
ORCID iDs
Ethical approval
Ethical approval was granted by Monash University Human Research Ethics Committee (Project ID 35037) and ratified by the University of Queensland Ethics Committee (Project No. 2023/HE000015).
Consent to participate
The stroke services of participating hospitals volunteered to participate in the Rehabilitation Audit.
Author contributions
MS was involved in writing – original draft, conceptualisation, methodology, analysis and writing – review and editing; SW in supervision, methodology and writing – review and editing; DCo in supervision, methodology and writing – review and editing; CB and TP in analysis and writing – review and editing; DCa and KH in writing – review and editing; and MK in supervision, conceptualisation, methodology, analysis and writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MS has been supported by an Australian Government Research Training Program scholarship. SW (1175821 and 2032983) and DAC (1154273) received Research Fellowship support from the NHMRC and MK (105737) receives a Future Leader Fellowship from the National Heart Foundation.
Declaration of conflicting interest
MK is the Deputy Chair of the Stroke Foundation Research Advisory Committee. KH is the Executive Director Stroke Programs, Research and Innovation at the Stroke Foundation. CB, TP, DACa and MK report funding from the Stroke Foundation, paid to the institution. DACa reports educational grants from Boehringer Ingelheim and Amazon Web Services, paid to Institution.
Data availability statement
On request from the Stroke Foundation (Australia).
