Abstract
Objective
This study estimated the prevalence of facial palsy after stroke and identified risk factors; examined recovery patterns; and evaluated the effect of hyperacute treatments on its recovery.
Design and setting
This was a secondary analysis of data prospectively collected in the first 7 days of stroke admission for the Bergen NORSTROKE study.
Participants
Of 5987 patients, a subgroup of 2293 with facial palsy were analysed for recovery, including 1954 with ischaemic stroke assessed for treatment effect.
Main measurements
Facial palsy was assessed repeatedly at irregular intervals from admission using the National Institute of Health Stroke Scale.
Analysis
Prevalence was summarised descriptively, and multifactorial logistic regression identified risk factors. The recovery was analysed with Kaplan–Meier curves and Cox models.
Results
Facial palsy was observed in 2578 patients (43%), including 2406 (40%) with minor or partial paralysis and 172 (3%) with complete paralysis. Facial palsy was more common in haemorrhagic (360, 54%) than ischaemic stroke (2218, 42%), reflecting greater motor impairment. Among patients without motor impairment, prevalence was lower in haemorrhagic strokes (15, 10%) than in ischaemic strokes (284, 14%). Risk factors included sex, age, and the degree of motor and sensory deficit. By day 2, 573 patients (25%) had fully recovered, while 1434 (63%) still had symptoms by day 7. Worse recovery was associated with more severe motor and sensory impairments. Intravenous thrombolysis was associated with better recovery.
Conclusions
Our study provides a comprehensive analysis of post-stroke facial palsy, highlighting recovery patterns and the need for ongoing monitoring and rehabilitation.
Introduction
Stroke remains one of the leading causes of disability worldwide, affecting approximately 12.2 million people annually. 1 Among the various symptoms of stroke, facial palsy represents a major challenge that impacts both functional outcomes and quality of life. Facial palsy, characterised by weakness or paralysis of facial muscles, can severely affect essential daily activities such as eating, drinking, and speaking while also having profound impact on patients’ psychological well-being, social interactions, and emotional health.2–4
While post-stroke facial palsy is frequently encountered in stroke clinical practice, its exact prevalence varies considerably in the literature, with reported prevalence ranging from 45% to 60%.5–8 Facial palsy occurs in both ischaemic and haemorrhagic strokes, and its prevalence and severity may vary between stroke subtypes. For example, in one rehabilitation cohort, 88% of cases were ischaemic and only 12% haemorrhagic, 4 while another study reported 11 ischaemic and 5 haemorrhagic cases with weakness of eye closure after post-stroke facial palsy. 9 These proportions are relatively straightforward to compare, but statistically robust analyses are limited by the small haemorrhagic sample sizes in most studies. Although modern stroke interventions such as intravenous thrombolysis and mechanical thrombectomy have revolutionised acute stroke care, their specific impact on facial palsy recovery has not been systematically evaluated.10,11 Despite its clinical importance, there is limited comprehensive data on the risk factors for facial palsy after stroke, what recovery occurs following stroke and the effect of any stroke interventions.4,12–14
Understanding the prevalence, risk factors, and recovery patterns of post-stroke facial palsy is important for several reasons. First, accurate prevalence data can inform resource allocation and rehabilitation planning in stroke units.6,15 In clinical practice, rehabilitation for post-stroke facial palsy is delivered mainly by speech and language therapists and physiotherapists, most often involving orofacial exercises and massage techniques. Assessment typically relies on clinical observation, with supplementary use of scales such as the National Institutes of Health Stroke Scale and the Sunnybrook Facial Grading System. However, management practices vary considerably, and robust evidence or consensus guidelines are lacking. 16 Second, the identification of risk factors could enable early intervention and potentially prevent severe cases. 15 Third, understanding the relationship between hyperacute treatments and facial palsy recovery could influence treatment decisions and prognosis.17,18
This study aims to address these knowledge gaps through a comprehensive analysis of data from the Bergen NORSTROKE registry, one of the largest stroke databases in Norway. Our objectives were threefold: (1) to establish the prevalence of post-stroke facial palsy and identify associated risk factors; (2) to examine facial palsy recovery within the first 7 days post-admission; and (3) to evaluate the potential effect of hyperacute treatments (intravenous thrombolysis and mechanical thrombectomy) on recovery outcomes of facial palsy. By utilising this extensive dataset from 2006 to 2021, we aimed to provide robust evidence to guide clinical practice and inform future research directions.
Methods
This was a secondary analysis of data prospectively collected for the Bergen NORSTROKE study. Participants were admitted to the Department of Neurology, Haukeland University Hospital, between February 2006 and February 2021. Data collection and analysis were approved by the hospital's institutional review board. This study was approved by the Regional Ethics Committee for Medical and Health Research Ethics in Western Norway (approval no. 2012/1483 Norstroke).
The National Institute of Health Stroke Scale was used to assess stroke severity, based on bedside neurological impairments and scored from 0 to 42. 19 Patients had up to 14 National Institute of Health Stroke Scale assessments carried out at irregular intervals within their first 7 days of hospital admission. The exact date and time of each assessment were recorded if available. Facial palsy measurements were extracted from the National Institute of Health Stroke Scale facial palsy item. The National Institute of Health Stroke Scale assesses facial palsy by observing facial symmetry during movements, scoring from 0 (normal) to 3 (complete paralysis).
Patients with missing National Institute of Health Stroke Scale on admission, transient ischaemic attack, missing date and time of admission National Institute of Health Stroke Scale assessment, or any measurement or recorded error in date and time of assessment were excluded from the study.
The point prevalence of post-stroke facial palsy on admission was calculated in two ways. First, we determined the proportion of patients with facial palsy on admission (facial palsy item > 0) relative to the total sample. Second, we summarised prevalence by severity levels at admission using the original ordinal scale. We also calculated admission prevalence across stroke subtypes, both with and without adjustment for motor impairment. Motor impairment was classified using the NIHSS motor domain (Table 1), which combined scores for arm and leg function, limb ataxia, and dysarthria. Dysarthria is included in this domain because the NIHSS conceptualises it as a motor speech function reflecting orofacial muscle control, and it is therefore analysed together with other motor components. Adjustment was necessary because the relationship between stroke subtype and facial palsy is likely confounded by overall stroke severity, particularly motor impairment.
Classified neurological symptoms based on National Institute of Health Stroke Scale.

To identify factors associated with facial palsy on admission, a multifactorial binary logistic regression model was applied. The facial palsy scale on admission was dichotomised into a binary response variable (=1 when facial palsy scale > 0). Explanatory variables available in the dataset included patient demographics (age and sex), the timing of stroke onset (e.g. unknown onset time, wake-up stroke) and stroke subtype (ischaemic stroke or intracerebral haemorrhage). Additional clinical variables included other neurological symptoms extracted from items of the National Institute of Health Stroke Scale on admission. We extracted and decomposed them into four domains, including consciousness, cognition, motor and sensory, in Table 1 based on the published categories. 20 The results were presented as estimated odds ratios with 95% confidence intervals.
The recovery of facial palsy post-admission was treated as a time-to-event outcome and analysed using Kaplan–Meier survival curves, which estimated recovery rates within 7 days post-admission. 21 Recovery time was defined as the interval from admission facial palsy assessment with score >0 to the first recorded disappearance of facial palsy (facial palsy scale = 0).
Patients who did not recover were censored at the last available assessment. Kaplan–Meier curves excluded data beyond the first treatment event to examine natural recovery rates unaffected by intravenous thrombolysis or mechanical thrombectomy.
To identify factors associated with recovery post-admission, a Cox proportional hazards model was applied, accounting for the same set of covariates including age, sex, stroke subtypes, symptom severity clusters as defined above and types of symptom onset time. 22 Here, we explored if the risk factors of interest were also associated with the recovery of facial palsy, not just the presence of the symptoms on admission. The results were presented as estimated hazard ratios with 95% confidence intervals.
The effect of hyperacute treatments on the recovery of facial palsy is dynamic since treatment administration depends on when patients are admitted to the hospital. Kaplan–Meier curves were applied to visually compare the difference in recovery trajectories with and without censoring after treatment. The treatment effect was then formally analysed using a Cox proportional hazards model with time-dependent covariates. 23 An extended Cox model with time-dependent covariates allows for the inclusion of variables that change their values over time during the observational period. In this study, two binary time-dependent indicators were created and combined to capture the association of treatments with recovery. Please see the details of the method in the Supplemental Material Part 1. We performed an unadjusted model only considering the time-dependent indicators and an adjusted model including calendar year, stroke severity (excluding facial palsy score) on admission, age and sex as confounders. Results were reported as hazard ratios with 95% confidence intervals.
All analyses were conducted in R Studio (Version 2023.12.1).
Results
The study followed a three-stage process, as shown in Figure 1, beginning with data cleaning, where 6786 patients were screened, and those with missing facial palsy scale (n = 226), or transient ischaemic attack (n = 573) were excluded. In Stage one, 5987 patients were included to estimate prevalence, with further exclusions for missing covariates (n = 44), leaving 5943 patients for risk factor analysis. Stage two focused on recovery analysis, including 2293 patients after excluding those with time measurement errors (n = 302), missing treatment information (n = 209), and patients without facial palsy on admission (n = 3139). Stage three analysed treatment effects in 1954 ischaemic stroke patients, excluding those with mechanical thrombectomy before intravenous thrombolysis (n = 6), treatment time errors (n = 6), and intracerebral haemorrhage patients (n = 327).
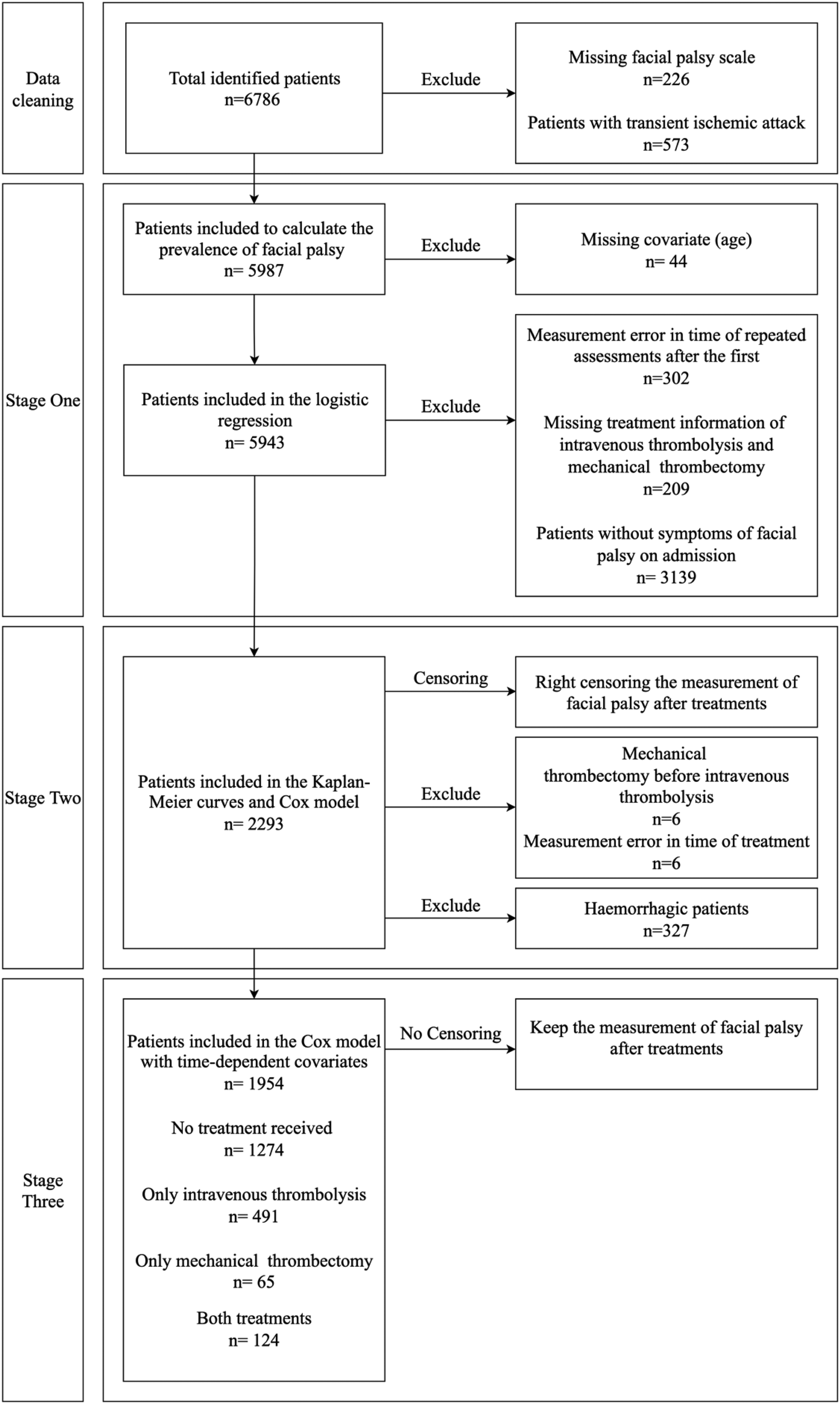
Flow chart of data cleaning and patient screening.
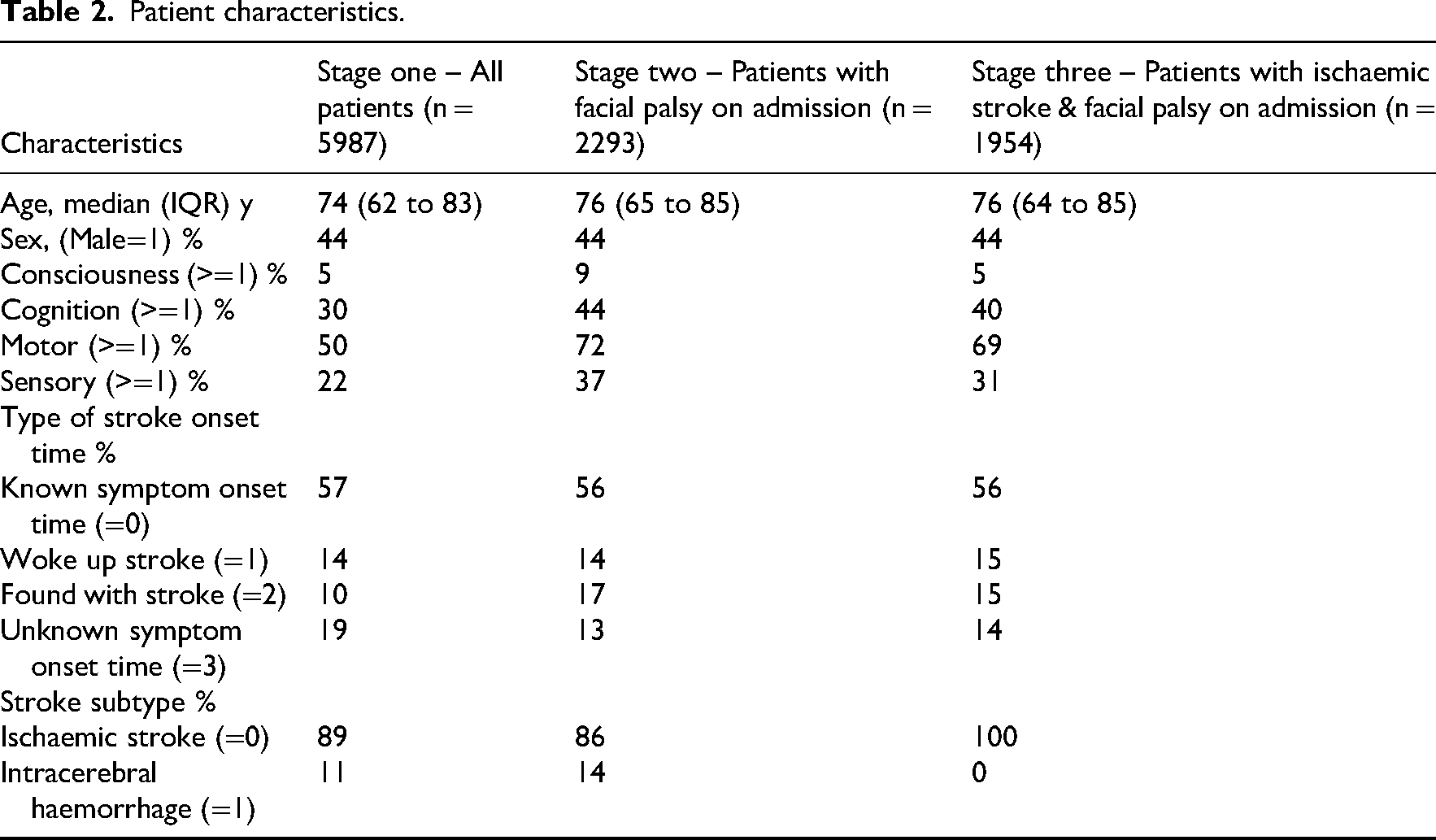
Patient characteristics are shown in Table 2. The median age of patients was 74 years (IQR 62–83) in stage one (all patients), 76 years (IQR 65–85) in stage two (patients with facial palsy on admission), and 76 years (IQR 64–85) in stage three (patients with ischaemic stroke and facial palsy on admission). The proportion of male patients remained consistent at 44% throughout different stages of analysis. Patients with facial palsy symptoms had more severe impairment in cognition, motor, and sensory functions compared with the overall patients. The majority had a known stroke onset time (∼57%), and ischaemic stroke was the predominant subtype.
Patient characteristics.
The prevalence of post-stroke facial palsy among 5987 patients admitted with stroke is presented in Table 3. Facial palsy affected 43% of the patients on admission, highlighting its relatively high prevalence as a stroke symptom. Most patients with facial palsy had either minor or partial paralysis (40% combined), while complete paralysis was relatively rare, affecting only 3% of patients. This is consistent with the neuroanatomy of facial innervation: most supranuclear strokes produce partial weakness sparing the forehead. Complete facial palsy, therefore, occurs rarely, since it would typically require either a brainstem lesion affecting the facial nucleus, a lower motor neuron lesion of cranial nerve VII, or, less commonly, bilateral cortical involvement. Facial palsy is more prevalent and severe among patients with intracerebral haemorrhage, with 54% experiencing symptoms and 11% experiencing severe symptoms (complete paralysis), compared with patients with ischaemic stroke, of whom 42% experienced symptoms and only 2% experienced complete paralysis.
The prevalence of post-stroke facial palsy on admission.
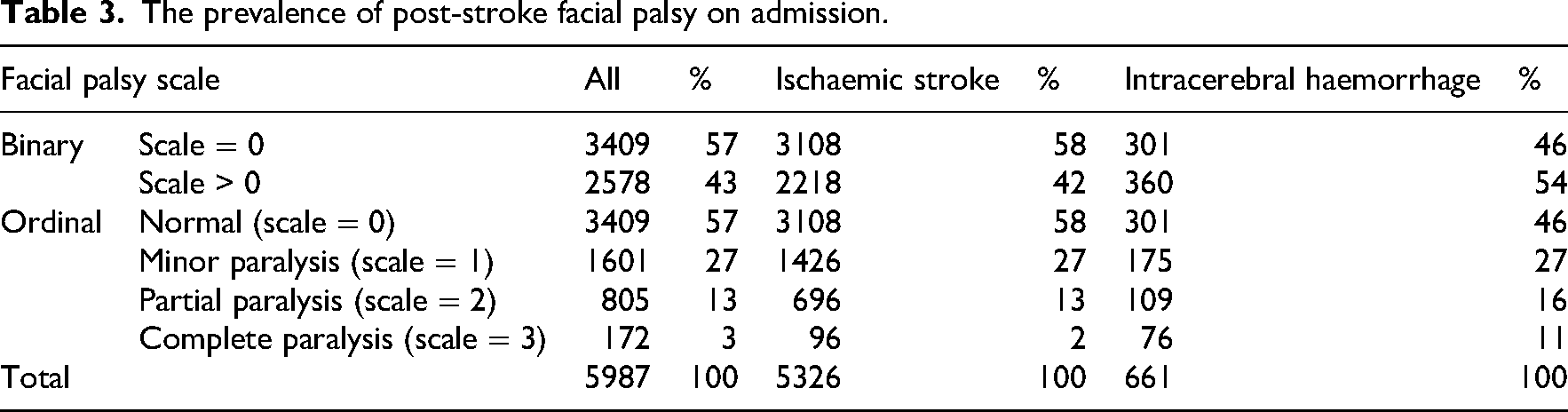
Table 4 presents odds ratios and 95% confidence intervals for risk factors associated with the presence of facial palsy on admission. Age, sex, motor and sensory function, and stroke subtype were significantly associated with the outcome. Specifically, each 10-year increase in age was associated with a 6% increase in the odds of facial palsy. Male patients had 17% lower odds of facial palsy compared to female patients. Greater severity in motor and sensory impairments was associated with 42% and 20% higher odds of facial palsy, respectively.
Estimated odds ratios for the risk factors of facial palsy on admission with 95% confidence intervals.
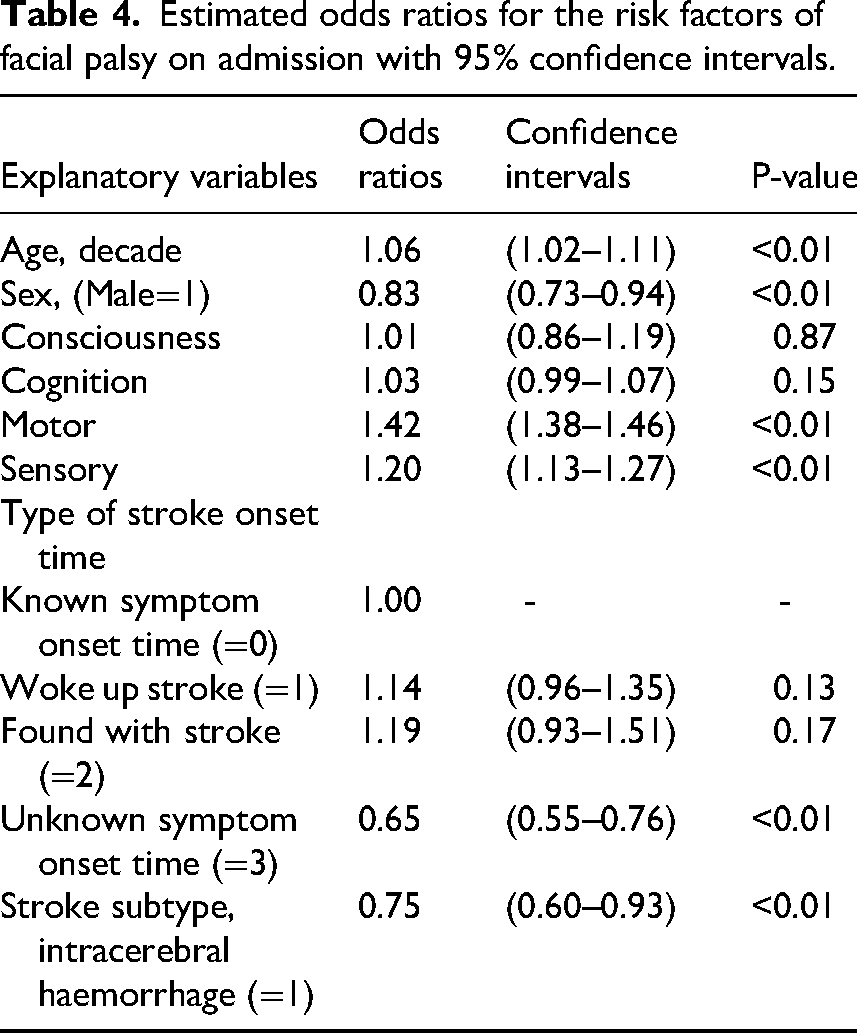
Interestingly, stroke subtype showed a reversed association with facial palsy in the adjusted model (Table 4), an example of Simpson's paradox – where the direction of an association changes once a confounding variable is adjusted in the model, in this case motor impairment. Full details, including a three-way cross-tabulation by subtype and motor function, are provided in Supplemental Material Part 3, Table 1.
The Kaplan–Meier curve illustrates the estimated recovery rate of post-stroke facial palsy during the first week after admission, with censoring applied beyond treatment events. In Figure 2, the recovery rate starts from 1 at time 0, indicating that all patients did not recover on admission. The first 25% recovered before day 2. Only 859 out of 2293 (37%) patients recovered within the first 7 days post-admission.
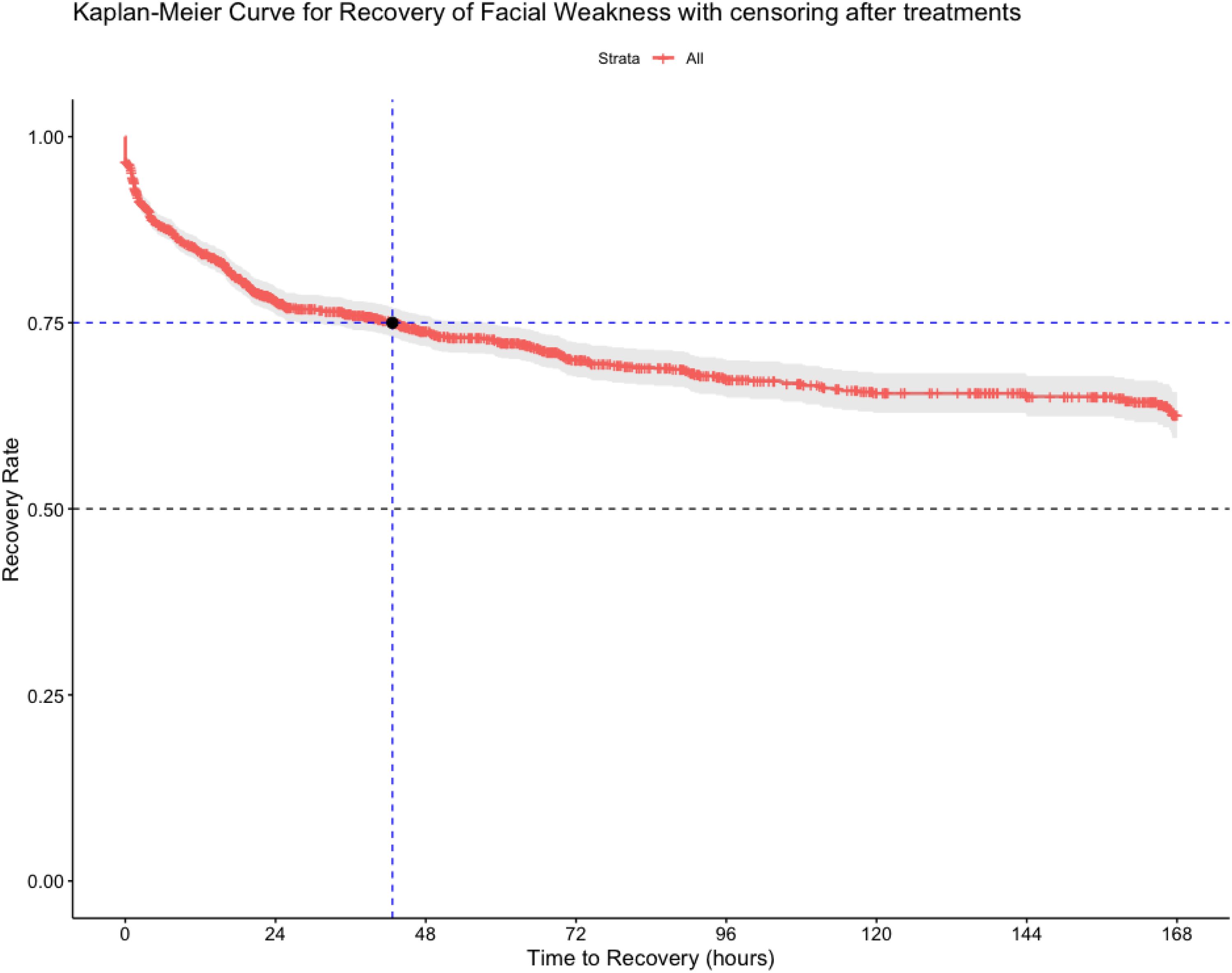
Estimated recovery rate of post-stroke facial palsy within a week after admission (censoring after treatments).
Motor function, sensory function, and different stroke types are potential risk factors for the recovery of facial palsy post-admission, as indicated by results from the Cox model in Table 5. Patients with greater impairments in motor and sensory function are also associated with 16% and 12% lower hazard of full recovery post-admission, respectively – that is, they recovered more slowly over time compared to those with fewer impairments. Patients with intracerebral haemorrhage had 46% higher hazard of full recovery, meaning their probability of achieving recovery at any given time point was greater than that of patients with ischaemic stroke.
Estimated hazard ratios for the risk factors of facial palsy recovery post-admission with 95% confidence intervals.

The Kaplan–Meier curves (Supplemental Material Part 2, Figure 2) suggest better facial palsy recovery with early reperfusion therapies. Results from the unadjusted extended Cox model in Table 6 indicate that only receiving intravenous thrombolysis is significantly associated with an increased odds of 25% of full recovery for facial palsy post-admission. The results of the adjusted extended Cox model show that receiving intravenous thrombolysis is still significantly associated with better recovery of facial palsy after adjusting for the known confounders.
Estimated hazard ratios with 95% confidence intervals in the unadjusted and adjusted extended Cox model with treatment as time-dependent covariate.

We adjusted for calendar year, age, sex, and admission stroke severity (excluding the facial palsy item) as confounders in the model.
Discussion
Our comprehensive analysis of post-stroke facial palsy in a large cohort of 5987 patients reveals several key findings that advance our understanding of this condition. Facial palsy affects 43% of stroke survivors on admission, predominantly consisting of minor to partial paralysis (40%), while complete paralysis was relatively rare (3%). Recovery patterns show that 2293 patients continued to have facial palsy that persisted beyond admission, with only 25% of patients achieving full recovery by day 2 and about 60% still experiencing symptoms at day 7. Potential risk factors identified included: being in the older age range, being a woman (who had a higher likelihood of facial palsy compared to men), and having more severe motor and sensory impairment on admission. Additionally, while intravenous thrombolysis showed a potential benefit in both unadjusted and adjusted analyses, whereas mechanical thrombectomy or combined treatments demonstrated no association with recovery. These findings represent a comprehensive analysis of post-stroke facial palsy and have important implications for clinical practice and prognosis.
The prevalence rate of 43% in our study contributes important insights to the existing literature, where reported rates have varied considerably due to differences in inclusion criteria, sample sizes, and assessment methods. Two smaller studies reported prevalence rates between 60% and 45%, but their limited sample sizes of 47 to 176 participants make these findings less reliable.5,7 In contrast, larger datasets like the Sentinel Stroke National Audit Programme reported prevalence rates of 54% (n = 88,974) 6 and 50% (n = 339,139), 8 which are likely to be more generalisable. While our prevalence rate is slightly lower than those reported in these studies, this difference may reflect variations in study design and patient populations. For example, some studies have focused on highly selected cohorts such as emergency department stroke admissions, 7 which may overrepresent patients with visible neurological signs, whereas others have been based on national audit registries that capture consecutive unselected stroke admissions.6,8 In addition, study populations differed in terms of stroke severity, with larger registry studies tending to include a greater proportion of patients with severe strokes, who are more likely to present with facial palsy, compared to smaller clinical cohorts with milder cases. These methodological and population differences are therefore likely to explain at least part of the variation in reported prevalence.
In addition to reporting the prevalence of facial palsy in the early days after stroke, this data has allowed us to explore facial palsy severity on admission. We found that most cases involved minor to partial paralysis (40%), while complete paralysis was rare (3%). This detailed severity distribution, which has not been well-documented in previous studies, offers valuable insights for rehabilitation planning and resource allocation in stroke units. While the National Institute of Health Stroke Scale was used to measure the presence and severity of facial palsy, its reliability in identifying severity compared to specialised facial movement assessments warrants further investigation.24,25
Our findings regarding stroke subtypes and facial palsy present an interesting contrast to previous literature,4,15 which often focused on ischaemic stroke and excluded patients with severe impairments. Our data indicates that although patients with ischaemic stroke account for the majority of facial palsy cases in absolute numbers (reflecting the higher overall prevalence of ischaemic strokes), patients with haemorrhagic stroke showed a higher frequency of facial palsy development than that patients with ischaemic stroke (54% vs 42%) and more severe presentations (complete paralysis in 11% vs 2%). This study examined the proportion of facial palsy within each stroke subtype, providing a more accurate view of individual risk. The higher rate of facial palsy in patients with haemorrhagic stroke is largely explained by more severe motor impairments; when motor function is considered, facial palsy is less prevalent in patients with haemorrhagic stroke who have milder or no motor deficits. Therefore, facial palsy should not be interpreted in isolation; assessment strategies should consider other neurological impairments, especially motor function, as stroke subtype alone can be misleading.
The identification of risk factors for post-stroke facial palsy in our study provides valuable insights. The higher risk of post-stroke facial palsy observed in female patients is consistent with other studies exploring the increased risk factors associated with sex.26–29 Symptom onset time appeared less relevant; patients with unknown onset times had lower odds of facial palsy, possibly due to delayed hospital admission or unrecognised symptoms allowing mild facial palsy to resolve before assessment. Greater motor and sensory impairments were associated with poorer recovery. Although patients with intracerebral haemorrhage appeared to recover better, this may reflect collider bias, a type of bias that can occur when analyses adjust for a variable (e.g. stroke severity) that is influenced by both the exposure and the outcome, potentially creating a spurious association. Further research is needed to clarify these associations. Identifying these specific risk factors may help clinicians recognise patients at higher risk of facial palsy and support more targeted monitoring and early intervention during the acute phase.
To the best of our knowledge, this is the first time that recovery of facial palsy has been explored in the acute post-stroke period, within the first 7 days of stroke. Around 25% of patients recover by day 2, with around 60% still affected at day 7. This is useful clinical information and should also be used to inform future intervention research. Intervention research has started to emphasise the need for targeted rehabilitation interventions on post-stroke facial palsy, and this study also highlights the need for extended monitoring and support after the initial week post-stroke.13,14
Early reperfusion therapies for stroke, such as intravenous thrombolysis and mechanical thrombectomy, are being used more frequently,17,30 and this study has allowed us to explore the effect of this on facial palsy recovery. Intravenous thrombolysis is significantly associated with improved recovery from facial palsy. However, this association may not reflect a true treatment effect. Patients eligible for intravenous thrombolysis typically had known and early symptom onset times, allowing for quicker hospital admission and earlier assessment. As a result, more transient or acute cases of facial palsy may have been captured in this group, with symptoms resolving over time regardless of treatment. In this case, we can't conclude that treatment alone improved recovery from the study, as patients who receive treatment may also be those more likely to recover. Further investigation is needed for this matter, considering other unmeasured confounders not included in this study.
The strengths of our study include a systematic assessment approach and analysis of early recovery patterns within the first week post-stroke. Using the Bergen NORSTROKE registry provided access to comprehensive clinical data with structured monitoring of outcomes, including frequent early assessments and specific follow-up time points. However, several limitations should be acknowledged. The study's focus on acute-phase recovery within the first 7 days means that long-term outcomes beyond the hospital stay remain to be explored. Except for the reperfusion therapies mentioned previously, all patients underwent standard medical care. We excluded patients with missing data on facial palsy at admission without applying imputation methods, which may affect the accuracy of prevalence estimates even if the missing rate is low. Although the sample size is relatively large, it was derived from a single stroke unit, limiting the generalisability of the findings to broader populations. Other stroke units may serve patient populations that differ in case-mix, for example, a higher proportion of minor versus severe strokes, different demographic profiles (e.g. age, sex distribution, ethnicity), or distinct referral and admission pathways. These differences could influence both the observed prevalence and recovery patterns of facial palsy. Finally, we used the time of the first facial palsy assessment as the starting point for recovery analysis rather than the time of symptom onset. This approach may introduce bias due to left truncation, where patients who already recovered before observation are not captured, or delayed entry, where patients are only included in the analysis after a variable time from stroke onset. In other words, patients who recovered quickly before their first assessment would be missed in the analysis, making the estimated recovery rate appear slower than it actually is.
In conclusion, our study provides a comprehensive analysis of post-stroke facial palsy, its risk factors, and recovery patterns. These findings have important implications for clinical practice and future research directions. The high prevalence and persistent nature of facial palsy suggest a need for routine screening and extended monitoring protocols. Although current rehabilitation strategies often include exercises and manual techniques, practice varies considerably and standardised guidelines remain lacking. Future research should focus on developing specialised rehabilitation protocols for post-stroke facial palsy and investigating the long-term outcomes beyond the first week of stroke since admission. This work sets the foundation for future research and improvements in clinical care for this common but challenging post-stroke complication.
Clinical messages
Post-stroke facial palsy is common, affecting over 40% of stroke patients at admission. It is more prevalent and severe in patients with more severe motor impairment. Most patients do not recover facial function within the first week; only 25% recover by day 2, and around 60% remain affected at day 7. Older age, female sex, and greater motor and sensory impairment at admission are potential risk factors for developing facial palsy after stroke. Early reperfusion therapies such as intravenous thrombolysis may benefit facial palsy recovery, but further research is needed to evaluate their effects.
Supplemental Material
sj-docx-1-cre-10.1177_02692155251391672 - Supplemental material for Post-stroke facial palsy: Prevalence, recovery patterns within the first 7 days, risk factors, and effect of hyperacute treatments
Supplemental material, sj-docx-1-cre-10.1177_02692155251391672 for Post-stroke facial palsy: Prevalence, recovery patterns within the first 7 days, risk factors, and effect of hyperacute treatments by Zewen Lu, Havva Sumeyye Eroglu, Halvor Naess, Matthew Gittins, Amit K Kishore, Craig J Smith, Andy Vail and Claire Mitchell in Clinical Rehabilitation
Footnotes
Correction (November 2025):
The article has been updated to correct few errors identified after publication. In the Abstract section, the number of patients who had fully recovered by day 2 has been corrected from 2573 to 573 and the patients having symptoms has been corrected from 1376 (60%) to 1434 (63%). Additionally, in the Results section, the percentage of patients recovered in the first 7 days of post-admission has been corrected from 38% to 37%. In the Discussion section, the phrase “persisted beyond the first 7 days of admission” has been updated to “persisted beyond admission.”
Ethical considerations
This study was approved by the Regional Ethics Committee for Medical and Health Research Ethics in Western Norway (approval no. 2012/1483 Norstroke).
Author contributions
ZL, HSE, AV, and CM conceived the study. MG and AV supervised the statistical analysis. CM, CJS, AKK, and HN contributed to the clinical knowledge. HN managed data acquisition and ethics. ZL performed the data analysis and interpreted the results. ZL and HSE wrote the first draft of the manuscript. CM, AV, MG, CJS, AKK, and HN reviewed and edited the manuscript. All authors read and approved the final manuscript. ZL and HSE contributed equally to the study and should be considered as joint first authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
All data produced in the present study are available upon reasonable request to the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
