Abstract
Objective:
To evaluate the cost effectiveness of a psychological adjustment group shown to be clinically effective in comparison with usual care for people with multiple sclerosis.
Design:
Randomized controlled trial with comparison of costs and calculation of incremental cost effectiveness ratio.
Setting:
Community.
Participants:
People with multiple sclerosis were screened on the General Health Questionnaire 12 and Hospital Anxiety and Depression Scale, and those with low mood were recruited.
Interventions:
Participants randomly allocated to the adjustment group received six group treatment sessions. The control group received usual care, which did not include psychological interventions.
Main measures:
Outcomes were assessed four and eight months after randomization, blind to group allocation. The costs were assessed from a service use questionnaire and information provided on medication. Quality of life was assessed using the EQ-5D.
Results:
Of the 311 patients identified, 221 (71%) met the criteria for having low mood. Of these, 72 were randomly allocated to receive treatment and 79 to usual care. Over eight months follow-up there was a decrease in the combined average costs of £378 per intervention respondent and an increase in the costs of £297 per patient in the control group, which was a significant difference (p=0.03). The incremental cost-effectiveness ratio indicated that the cost per point reduction on the Beck depression inventory–II was £118.
Conclusion:
In the short term, the adjustment group programme was cost effective when compared with usual care, for people with multiple sclerosis presenting with low mood. The longer-term costs need to be assessed.
Keywords
Introduction
A high proportion of those with multiple sclerosis have emotional problems and these adversely affect their quality of life.1,2 Psychological interventions have been shown to reduce depression and anxiety in people with multiple sclerosis.3,4 These interventions may be delivered in groups or individually. One potential advantage of group interventions, as compared with individual therapy, is that they may be more cost effective. Yet previous evaluations of the cost-effectiveness of similar treatments for elderly people with depression have indicated that while the treatments are effective they are also associated with an increased cost. 5 However, the cost effectiveness of psychological treatments for people with multiple sclerosis has not been examined. In a recent trial of a psychological adjustment group for people with multiple sclerosis and low mood, 3 results indicated that intervention improved mood and reduced the impact of multiple sclerosis in everyday life. These encouraging results suggested that the intervention should be provided as part of routine clinical practice. The aim of the present study was to determine the cost effectiveness of the intervention.
There were two health economic objectives. The first was to compare the average costs associated with the adjustment group programme, with a usual care control group. The second was to estimate the changes in the Beck depression inventory-II (BDI-II) 6 score as a point estimate to calculate the incremental cost-effectiveness ratio (ICER). The analysis was conducted from the perspective of the UK National Health Service (NHS).
Methods
Participants were recruited for a randomized trial of adjustment groups for people with multiple sclerosis and low mood between June 2008 and September 2009. The details of this study have been published elsewhere. 3 However, in summary, participants were included who scored >2 on the General Health Questionnaire 12, 7 a measure of psychological distress, or >7 on the anxiety and depression subscales of the Hospital Anxiety and Depression Scale, 8 thus confirming they had low mood.
Clusters of eight participants, who could attend groups on the same day, were randomly allocated, using a web-based randomization programme prepared in advance by the Clinical Trials Unit at Nottingham University, to an intervention or control group. Those allocated to intervention were invited to attend a psychological adjustment group for six sessions over 12 weeks. The groups were delivered by two Band 5 assistant psychologists, who were supervised by a Band 8a clinical psychologist, experienced in working with people with multiple sclerosis. The groups involved teaching people to recognise symptoms of distress and introduced them to strategies to improve their mood. Participants allocated to the control group received all other rehabilitation routinely provided and were offered the opportunity to attend groups once they had completed the study.
Four and eight months after random allocation, participants were sent a booklet of outcome questionnaires to complete and return by post. This booklet included the measures administered at baseline, the General Health Questionnaire 12 and Hospital Anxiety and Depression Scale, and in addition the BDI-II, 6 a measure of the severity of depression. The EQ-5D 9 was included as a generic quality of life measure. To ascertain costs, a questionnaire was used to assess the use of NHS services and prescribed medication in the previous three months. This included questions about the frequency of contact with community health services, including general practitioners, nurses and other health and social care professionals. The length of inpatient stays and number of outpatient appointments were recorded. The information was subdivided according to whether it was related to multiple sclerosis or for other reasons. Current medication and any changes in the previous three months were noted.
Those who were not able to complete the outcome measures, such as people with visual problems, were visited by a researcher who was unaware of their group allocation, who completed the questionnaires with them. If participants failed to return outcome questionnaires they were telephoned once to remind them.
Resource consumption was calculated using the UK Department of Health National Tariff 10 and from the Personal Social Services Research Unit (2009). 11 Medication costs were calculated using data from the World Health Organization (WHO) Collaborating Centre for Drug Statistics Methodology (2010) 12 and the British National Formulary (2009). 13 The costs used are shown in Table 1.
Unit costs of health and social care (£) (2009).
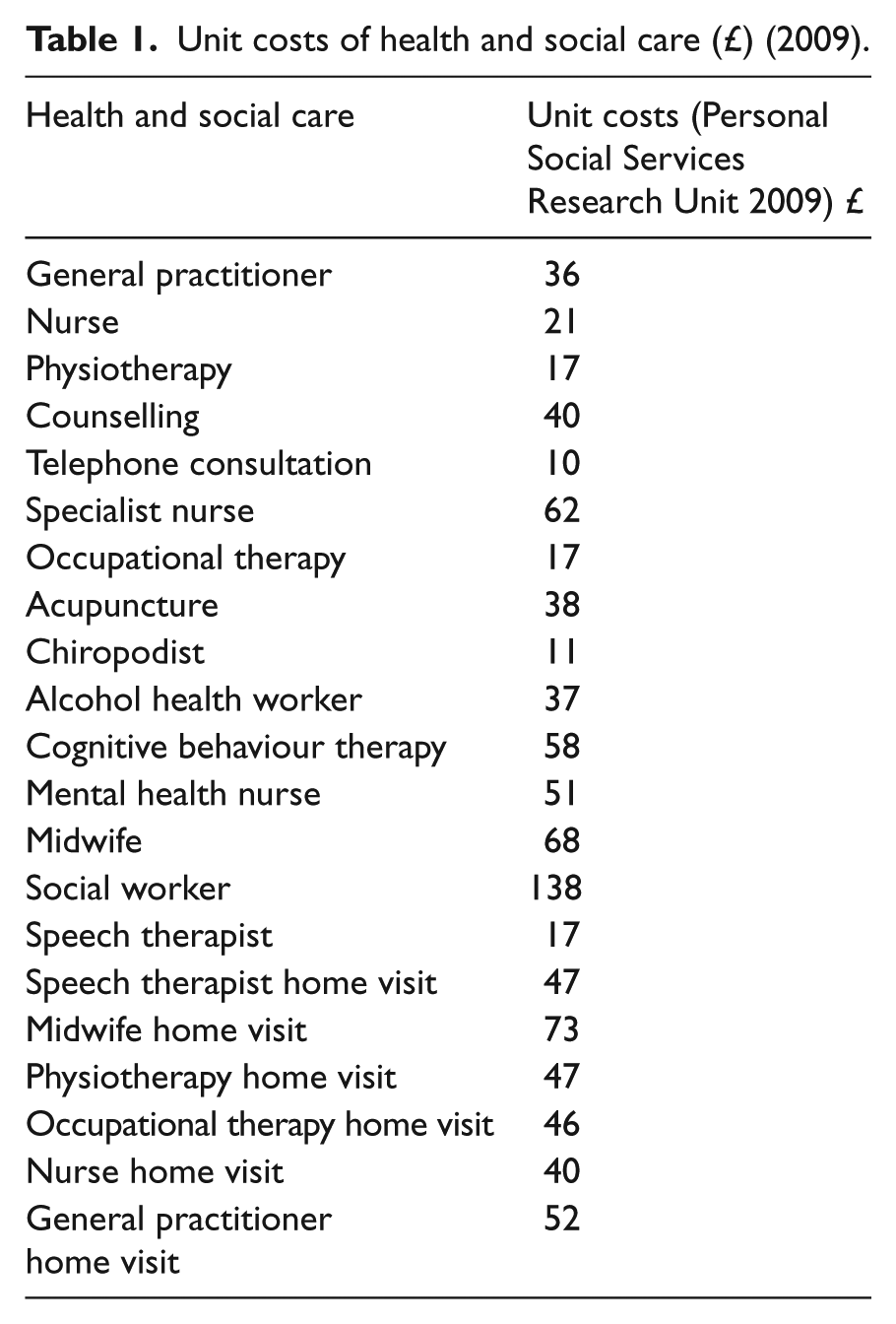
Patient-level costs were calculated by multiplying the frequency of contacts with health service providers with unit costs from the Personal Social Services Research Unit 2009. 11 Intervention costs were based on the number of sessions attended by each patient.
The main analysis evaluated the total costs of the intervention, resource use and medication at eight months after randomization, but costs at four months follow-up were also recorded. Baseline costs were examined for differences between groups, but were not used to adjust the results. A cost-effectiveness analysis was performed comparing the adjustment group with the usual care control group. Effectiveness was initially defined as the change in EQ-5D scores and a cost-effectiveness analysis was performed using this outcome measure. In addition, reductions in the BDI-II score from baseline to follow-up were analysed as an alternative measure of the cost effectiveness. The ICER, i.e. the difference in average costs divided by the difference in average effects between groups, was also calculated. In order to capture the uncertainty around the estimates, 1000 non-parametric bootstrap replications were generated from the sets of multiply imputed data, and mean cost and effect were plotted in a cost-effectiveness plane. As there are no published data on the threshold willingness to pay value for additional effectiveness associated with the group programme described, the probability that the programme will be considered cost effective was calculated for a range of threshold values and presented in a cost-effectiveness acceptability curve.
Results
There were 151 participants recruited to the trial; 72 randomly allocated to the treatment group and 79 to the control group. The CONSORT 14 diagram of the recruitment and follow-up of participants is shown in Figure 1.
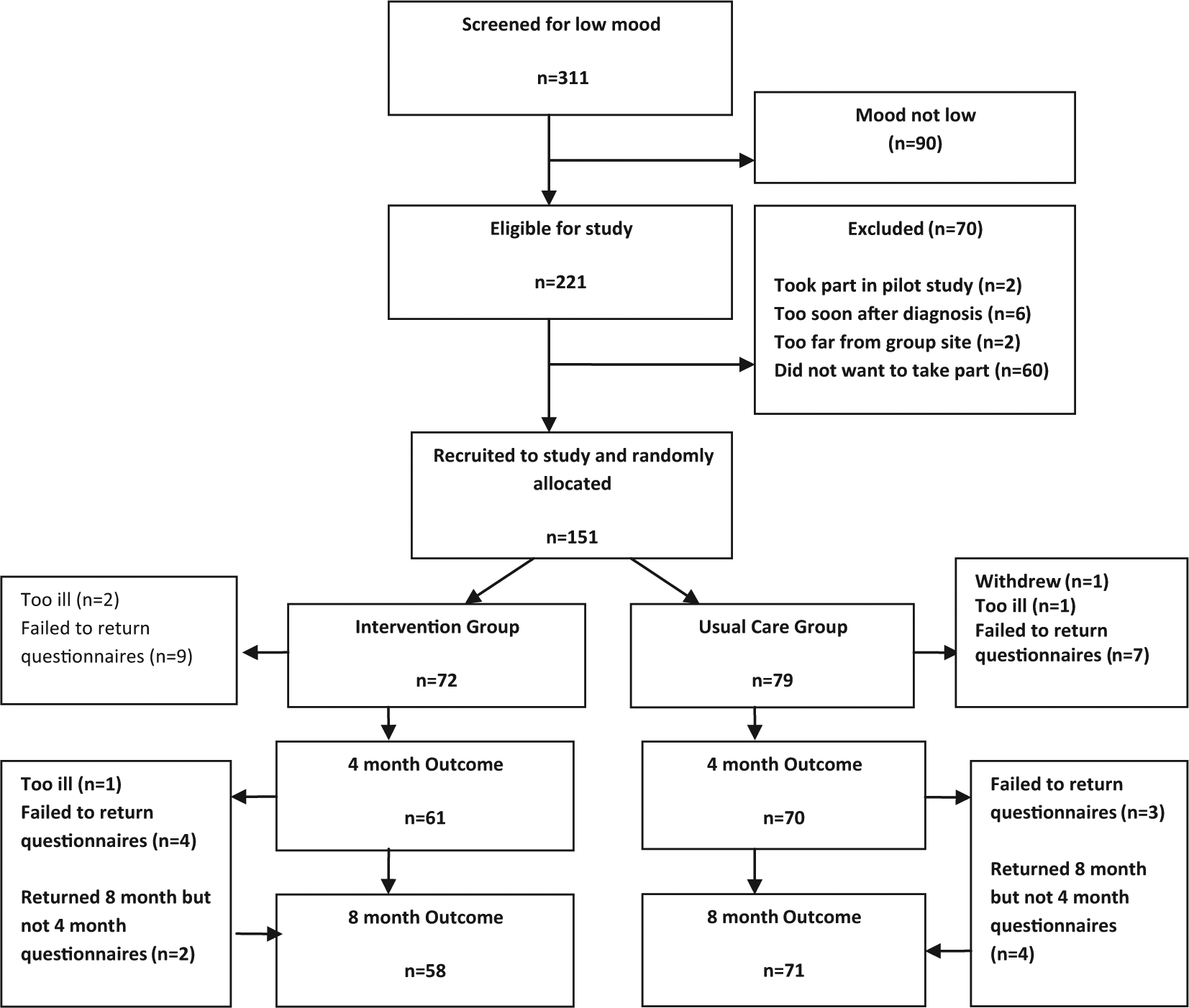
Recruitment and follow-up of participants in the trial.
The groups were well matched on all baseline variables. Participants’ characteristics are shown in Table 2.
Characteristics of participants.
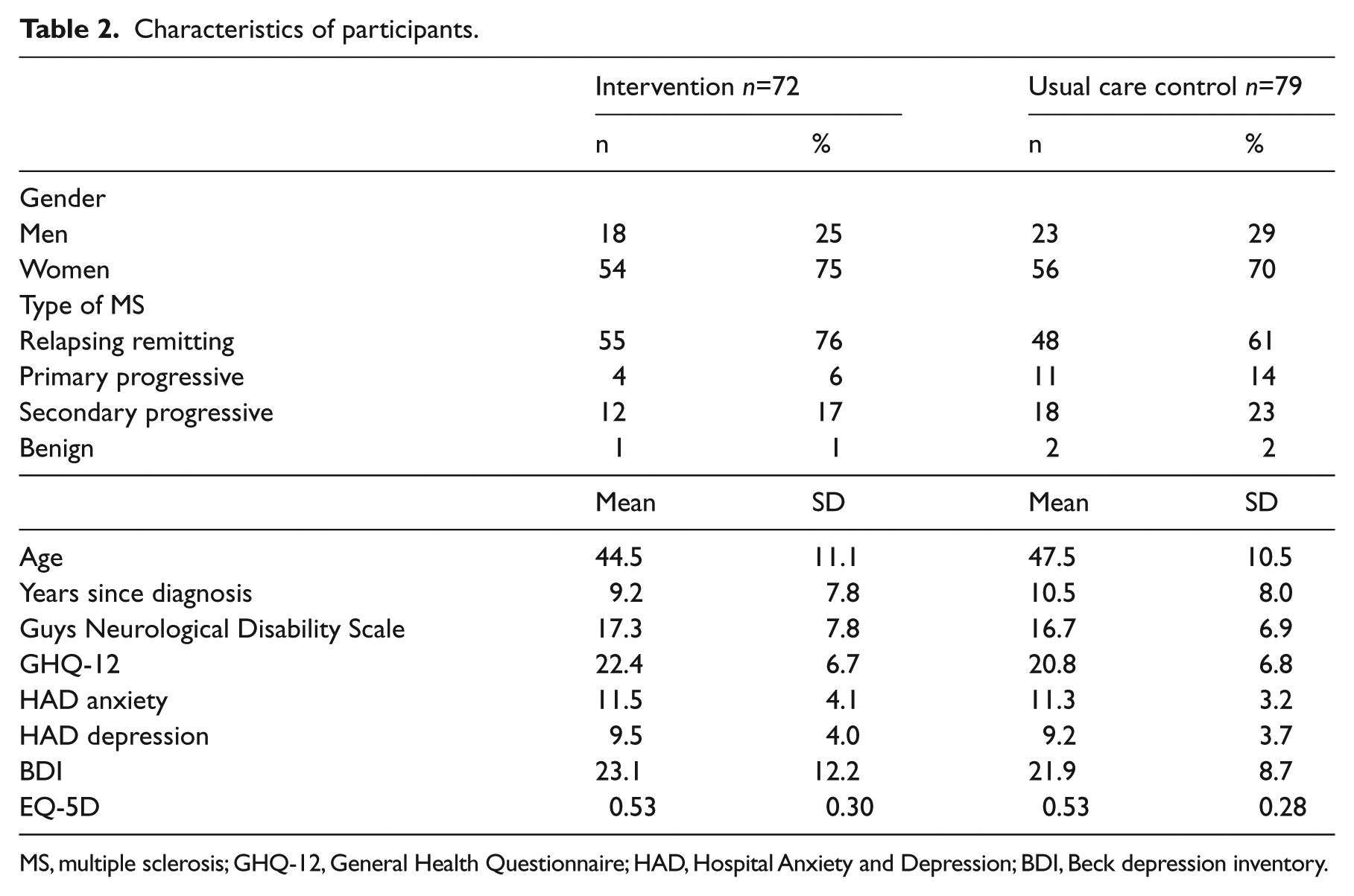
MS, multiple sclerosis; GHQ-12, General Health Questionnaire; HAD, Hospital Anxiety and Depression; BDI, Beck depression inventory.
The total running costs for the programme were £17,824. This was based on 54 three-hour sessions (including room hire and refreshments) and the salary of two NHS Band 5 assistant psychologists and one Band 8a clinical psychologist. 11 The average cost per person in the intervention group (n=72) was £248. This cost figure was added to the total baseline resource and medication cost for the intervention group.
The initial analysis was conducted on the service use questionnaire at four and eight months after randomization. The costs at each time point are shown in Table 3. In the intervention group, there was an overall reduction in resource use from baseline to eight months follow-up of £30,985 (mean per patient reduction of £429). In the control group, mean resource-use costs per patient at baseline were £536. There was an overall increase in resource use from baseline to eight months follow-up of £5,581 (mean per patient increase of £71). A Levene’s test for equality showed there were statistically significant group differences in both the four months resource costs (p=0.02) and the eight months resource costs (p=0.04). (See Table 4).
Level of resource utilisation across groups and at each time point.
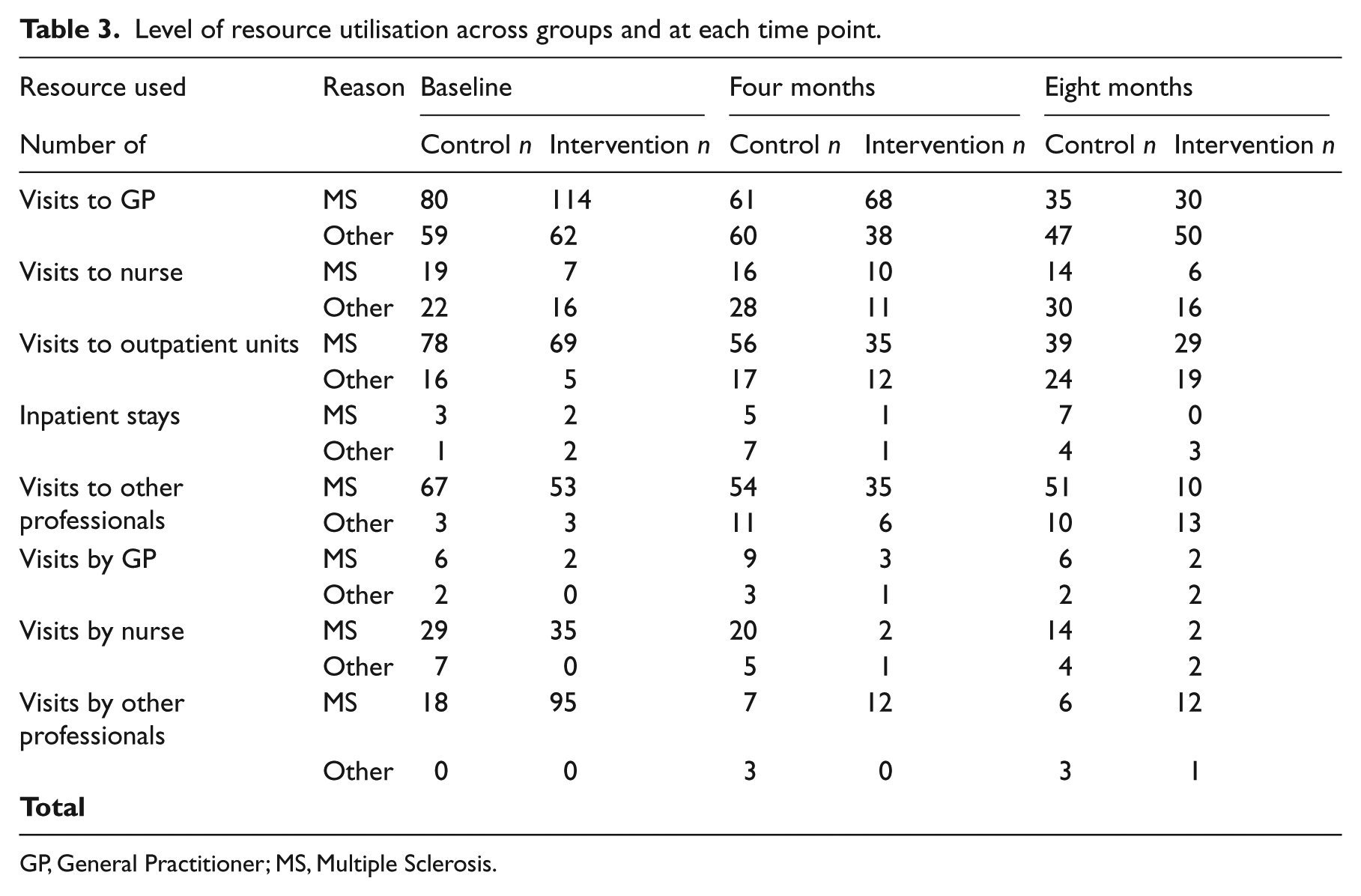
GP, General Practitioner; MS, Multiple Sclerosis.
Comparison of costs and outcome measures the eight months after randomization.
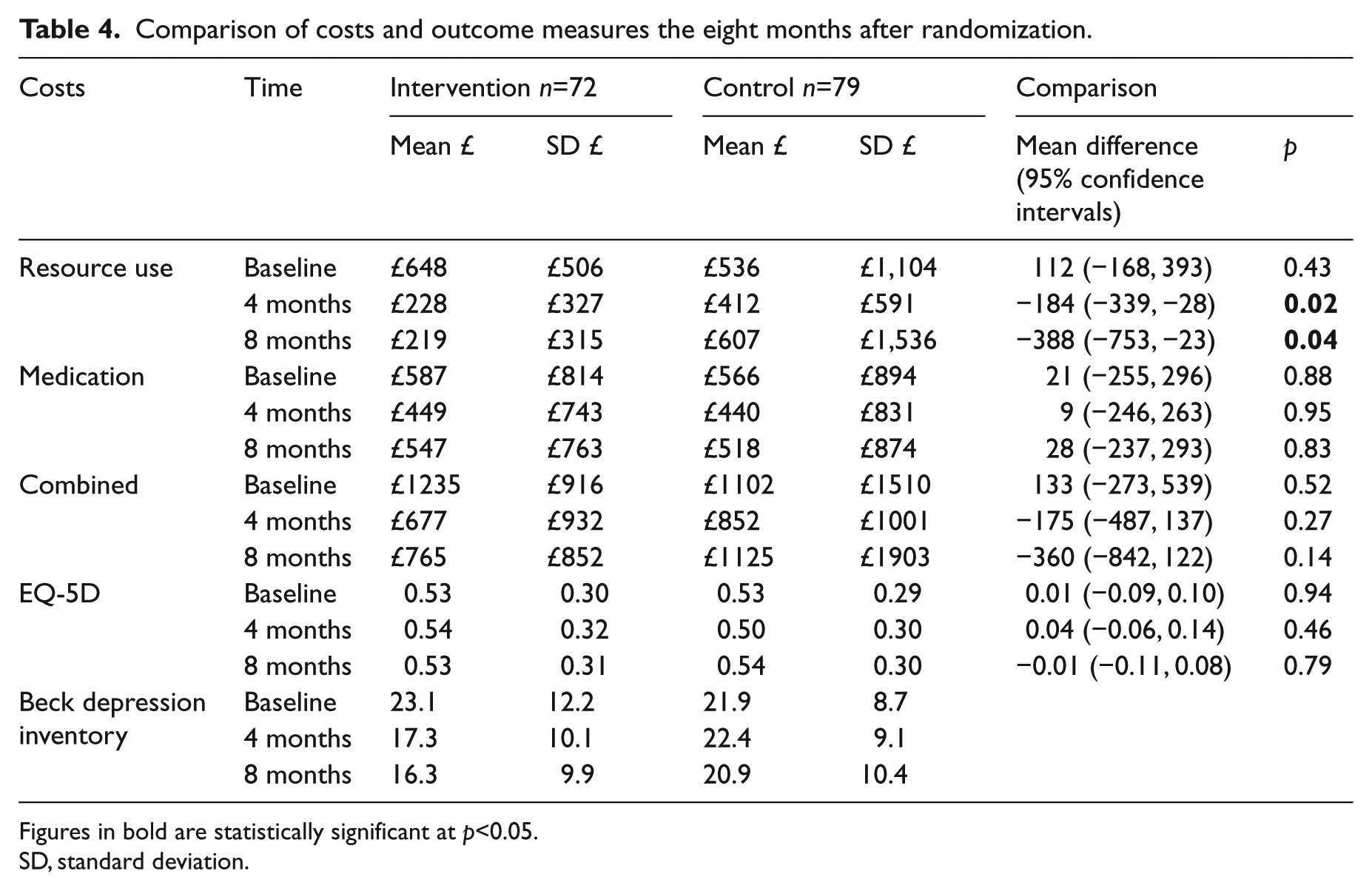
Figures in bold are statistically significant at p<0.05.
SD, standard deviation.
In the intervention group, there was an overall reduction in medication costs from baseline to eight months follow-up of £2888 (mean per patient reduction of £40). In the control group, there was an overall reduction in medication costs from baseline to eight months follow-up of £3782 (mean per patient reduction of £48). There were no significant differences between the groups (p>0.05).
The resource and medication costs were combined and the intervention cost added. In the intervention group, there was an overall reduction in combined resource use and medication costs from baseline to eight months follow-up of £33,873 (mean per patient reduction of £470). In the control group, there was an overall increase in combined resource use and medication costs from baseline to eight months follow-up of £1799 (mean per patient increase of £23).There were no significant differences between the groups in combined costs (p>0.05).
A Levene’s test for equality showed that the decrease in the intervention group mean resource-use costs compared with the increase in the control group at both four months and eight months follow-ups were statistically significant (four months mean difference −184,95% CI −339, −28, p=0.02 and eight months mean difference −388, 95% CI −753, −23, p=0.04). In the intervention group, 64 (89%) decreased in cost (−£36,722) compared with 8 (11%) increasing (£2881.5). In the control group, 53 (67%) decreased in cost (−£30,327), 2 (3%) stayed the same and 24 (30%) increased in cost (£32,128). The resource-use costs that reduced most were visits to general practitioners, visits to GP nurses and attendance at outpatient clinics (both for general and MS related problems).
There were 55 complete data sets (i.e. Baseline, four months and eight months) of EQ-5D data from the control group and 43 complete sets from the intervention group. Owing to the level of missing EQ-5D data at follow-up, a ‘last value carried forward’ imputation approach was adopted. 15 There was an overall mean reduction in the EQ-5D score from baseline to eight months follow-up of 0.008. In the control group, there was an overall mean increase in the EQ-5D score from baseline to eight months follow-up of 0.009. Results are shown in Table 2. The differences between the groups were not statistically significant at any time point (p>0.05).
The BDI-II was evaluated as an alternative indicator of the effectiveness of the intervention. A Levene’s test for equality showed a statistically significant difference (p=0.01) in the point reduction in the BDI between the intervention and control group over eight months (intervention mean −2.38 SD 4.72, control mean −0.67 SD 3.44, mean difference −1.70, 95% confidence interval −3, −0.4) . The incremental costs effectiveness ratio was calculated by dividing the mean reduction in costs between intervention and control (−£401) by the mean reduction in BDI score (−3.41). The adjustment group was associated with an incremental cost effectiveness ratio of £118 per additional point reduction in BDI score. Uncertainty in the incremental cost effectiveness ratio was estimated through bootstrapping, and by plotting the 1000 replications of mean cost and effect differences on a cost-effectiveness plane. Results are shown in Figure 2.
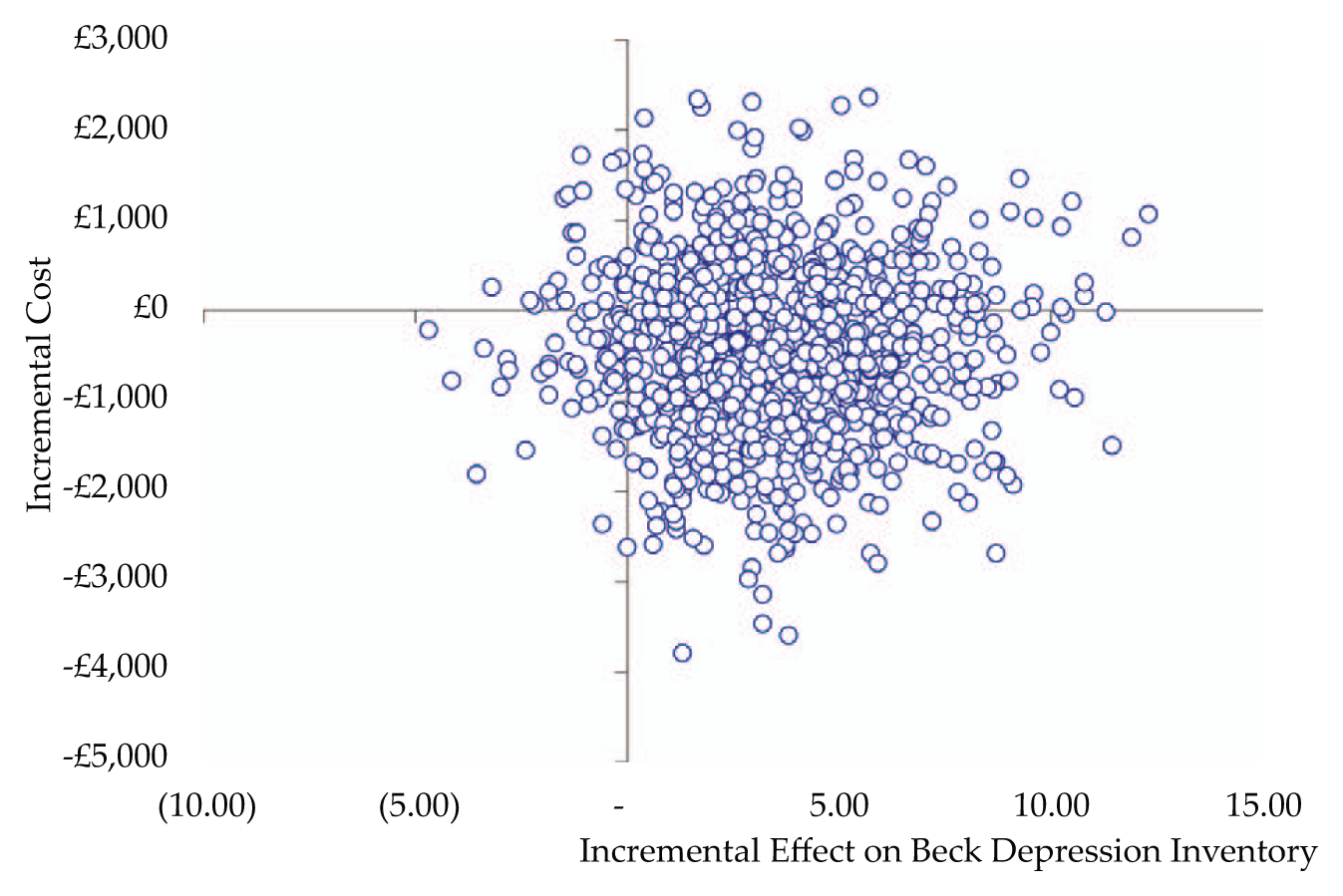
Incremental cost-effectiveness plane for the psychological adjustment group programme.
As 93% of re-samples fell within the bottom right-hand quadrant of the plane, there was a high likelihood of the adjustment group having lower costs and better outcomes, as measured by the BDI. A cost effectiveness acceptability curve, was plotted, which showed that the likelihood that the adjustment group was more cost-effective than usual care for different values placed on a unit improvement in the BDI. This showed a 93% probability that the adjustment group will be considered cost effective if purchasers are willing to pay up to £118 per point reduction in BDI score.
Discussion
The primary analysis was the cost effectiveness of an adjustment group, as compared with a usual care control group for people with multiple sclerosis who had low mood. Over eight months follow-up there was a decrease in the combined average costs of £378 per intervention respondent and an increase in the amount of combined resource and medication costs of £297 per patient in the control group, which was a significant difference (p=0.03). This indicates that not only did the intervention improve outcome, it also saved money. This provides justification for the provision of this intervention in routine clinical practice. The service in which the intervention was evaluated had no clinical psychology service specifically for people with multiple sclerosis. Our results suggest that the costs of providing such a service to allow the provision of treatment groups would pay for itself. However, elsewhere in the UK and in Europe, the provision of psychological services for people with multiple sclerosis is better than in our centre, it is possible that the benefits may be less marked. The main resource-use cost drivers included visits to general practitioners and outpatient clinics. The lack of difference in medication probably reflects that the medication cost drivers were high-cost disease-modifying drugs, such as Rebif, Avonex and Copaxone. These would not be affected by the mood of participants. In addition there were high levels of Beteferon and Gabapentin and also relatively inexpensive drugs (when compared with disease-modifying drugs), such as Amantadine and Baclofen. These all increased the costs but would not be affected by an improvement in mood. In addition, there were a few outliers with very high costs. At the four months follow-up there were two participants in the intervention group that generated costs in the £2000s using general inpatient services. Another participant in the intervention group generated costs in the £2000s by mostly using GP nursing services. One participant in the control group generated similar costs using general outpatient services. At the eight months follow-up, there were four intervention patients who were major outliers (£6000s). This was owing to long-term inpatient stays that were MS related.
There was a significant effect of the intervention on mood (Beck Depression Inventory) but not on quality of life. This is probably because the intervention was targeted at mood, and the EQ-5D assesses other aspects of quality of life that would not be affected by the intervention. The cost-effectiveness plane illustrates the ‘acceptable’ cost-effective ratio, i.e. the maximum threshold willingness to pay for a unit of effect. Our results indicate that the adjustment group programme was more likely to be cost effective compared with usual care for people with multiple sclerosis presenting with low mood, assuming a willingness to pay threshold of more than £118 per point reduction in BDI score. The cost-effectiveness of the group programme in this patient group depends on how much purchasers are willing to pay for reductions in depression scores. Further economic evaluations of psychological interventions studies should focus on costs that are expected to vary between groups as a result of an intervention. Such studies are needed given the lack of cost –effectiveness evaluations in relation to rehabilitation for people with multiple sclerosis. 16
The main limitation to the study is that it relied on self reports of services used and participants may not have been accurate in their recall of contacts with services. However, this should have occurred to the same extent in both intervention and control groups. The follow-up period was relatively short. A longer follow-up would indicate whether the costs savings were sustained over time. Yet these limitations do not detract from the key finding of this research that, in the short term, the adjustment group was a cost effective intervention.
Clinical messages
A psychological adjustment group for people with multiple sclerosis and low mood reduced the overall costs of clinical services in the short term.
There was little effect of the intervention on overall quality of life.
Every point reduction on the Beck Depression Inventory cost £118.
The provision of the psychological group intervention would pay for itself by saving overall costs.
Footnotes
Acknowledgements
We would like to thank Jessica Holmes and Karen Treece for running the treatment groups, Faye Yuill and Emma Ford for conducting the outcome assessments, the multiple sclerosis nurses for assistance with recruitment, and Sarah Armstrong and Cris Constantinescu for assistance with the trial. We also thank our multiple sclerosis partners who provided useful feedback from the service user perspective and the Multiple Sclerosis Society for financial support.
Conflict of interest
The author declares that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
