Abstract
Objective:
To assess the feasibility of conducting a randomized controlled trial of occupational therapy predischarge home visits for people after stroke.
Design:
Randomized controlled trial and cohort study. We randomized eligible patients for whom there was clinical uncertainty about the need to conduct a home visit to a randomized controlled trial; patients for whom a visit was judged ‘essential’ were enrolled into a cohort study.
Setting:
Stroke rehabilitation unit of teaching hospital.
Participants:
One hundred and twenty-six participants hospitalized following recent stroke.
Interventions:
Predischarge home visit or structured, hospital-based interview.
Main outcome measures:
The primary objective was to collect information on the feasibility of a randomized controlled trial, including eligibility, control intervention and outcome assessments. The primary outcome measure was the Nottingham Extended Activities of Daily Living Scale at one month after discharge from hospital. Secondary outcomes included mood, quality of life and costs at one week and one month following discharge.
Results:
Ninety-three people were allocated to the randomized controlled trial; 47 were randomized to intervention and 46 to control. Thirty-three were enrolled into the cohort study. More people were allocated to the randomized controlled trial as the study progressed. One hundred and thirteen people (90%) received the proposed intervention, although there was a need for stricter protocol adherence. Follow-up was good: at one month 114 (90%) were assessed. There were no significant differences between the groups in the randomized controlled trial for the primary outcome measure at one month. The average cost of a home visit was £208.
Conclusion:
A trial is feasible and warranted given the resource implications of predischarge occupational therapy home visits.
Introduction
People who have had a stroke and who are admitted to hospital may be offered a predischarge home visit by an occupational therapist. A home visit to evaluate patients in their usual environment is believed to increase the ability to cope at home and in the wider community 1 as well as enabling any issues about safety to be addressed. 2 In addition to assessing for potential problems, the visit provides the patient with the opportunity to practise the techniques they have learnt in hospital in their own home. This may be particularly important for people after stroke who may have hemiparesis, impaired cognition or aphasia. The 2006 National Sentinel Stroke Audit 3 reported that 73% of patients admitted to a stroke unit had a home visit before discharge, but there is no evidence on whether they actually improve patient outcome.
Performing home visits constitutes a significant element of practice for NHS occupational therapists; a recent survey of the total time spent on home visits (including preparation, travel, visit, administration) with patients following a stroke was just under 4 hours per visit. 4 However, there is little evidence to support the effectiveness of these visits. Barras 5 identified four randomized controlled trials of home visits, but these included both pre- and postdischarge assessments, focused on older people and concentrated particularly on falls prevention. The data were too limited and the follow-up periods were too short to reach any meaningful conclusions and both the paucity of quality and quantity of the studies reviewed were highlighted. No randomized controlled trials of home visits after stroke have been identified.
One of the reasons for the lack of evidence may be the concern that it would be unethical to withdraw such an established and accepted treatment. 6 A pilot study of home visits with older people in Australia 1 recruited only ten participants over three months despite admission records suggesting many more people should have been available. The authors believed that therapists were concerned about patients being allocated to the control group and consequently did not enrol them in the trial.
The aim of this study was to assess the feasibility of a randomized controlled trial in order to test out and revise the design for a definitive trial. In order to address the potential ethical and recruitment issues, we set and agreed criteria for a ‘home visit essential’ cohort study to include those patients whom the clinical therapists would be unwilling to randomize due to safety concerns. The objective was to establish whether participants could be recruited to a randomized controlled trial while acknowledging the clinical concerns of therapists.
Method
All patients were approached for informed, written consent. In those who lacked capacity, consent was obtained in keeping with the research provisions of the Mental Capacity Act of England, and as approved by the Research Ethics Committee (Berkshire Research Ethics Committee ref. 10/H0505/41). Participants were free to withdraw from the trial at any stage. Data were included in the analyses up to the point of withdrawal.
The design consisted of a randomized controlled trial and a cohort study. Eligible patients for randomization were those for whom the multidisciplinary team felt there was clinical uncertainty about whether or not a home visit was indicated. These patients were randomized to either an intervention (home visit) or control (no visit) group.
Patients included in the cohort study were those for whom ward clinicians believed a home visit was essential. The criteria were that these patients had new, significant functional impairment and/or environmental concerns which staff believed could not be assessed without a home visit (e.g. they were dependent in transferring or the home needed to be assessed for major equipment such as a hoist).
In conducting these studies, we were primarily interested in gathering and analysing information on eligibility criteria, consent procedures, intervention, collaboration with participating NHS staff and the completion rate of outcome assessments.
All patients transferred from the acute stroke unit to the stroke rehabilitation unit in Royal Derby Hospital between July 2010 and October 2011 with a confirmed diagnosis of stroke were considered. During their first 10 days on the stroke rehabilitation unit, the decision as to whether patients required a home visit (i.e. were eligible for cohort study) or were eligible for randomization was made by the multidisciplinary team.
Patients were excluded if they: did not speak English; would not normally be offered a home visit (e.g. those with existing comorbidities who needed to be transferred to other wards); were due for discharge out of the Derbyshire area; required an access visit only (a visit by the occupational therapist without the patient being present). Patients who were to be discharged to residential or nursing homes were eligible for inclusion. Ward occupational therapists made the initial approach to patients about participating in the trial. Patients who were agreeable were then seen by a member of the HOVIS (home visits for patients with a stroke) research team who provided full details of the study and who obtained informed consent. After getting consent, baseline data were collected on all participants in both the randomized controlled trial and cohort studies.
Patients recruited to the randomized controlled trial were registered using a web-based randomization program. This was managed by Nottingham Clinical Trials Unit who held a preprepared list in random varying block sizes. Allocation to either the control group (no visit) or the intervention group (home visit by occupational therapist) was immediate following completion of baseline assessments.
Those allocated to the intervention group were offered a predischarge home assessment visit with an occupational therapist. Patients were assessed in their own home and any potential problems were discussed and addressed in the home environment. The patient’s relative or carer(s) were invited to be present during the visit. Referrals were made to other agencies where required. On the visit, patients were offered advice, given practice in transfers and activities of daily living (ADLs), and offered equipment or adaptations, such as grab rails.
Those allocated to the control group received a predischarge home assessment structured interview with an occupational therapist in the hospital. The patient’s discharge and any potential problems were discussed in general terms. The patient’s relative or carer(s) were invited to participate in the interview. Referrals on to other agencies were made as required and patients were given the opportunity to practise using equipment in hospital, if necessary.
Patients in the cohort study received a home visit using the same protocol as those in the randomized controlled trial intervention group. A record was kept of the clinical team’s reasons, from the pre-agreed criteria, for deciding that a visit was essential, and the team was asked to record as much detail as possible on the form provided.
Patients in all groups were treated both by ward occupational therapists and by the HOVIS research occupational therapist who was based in the unit; visits were shared equally between all the therapists in order to control for the effect of individual therapists. The research occupational therapist was available to assist with providing patient information, consent and in monitoring home visit decisions about patients. She also delivered and assisted ward therapists to deliver the structured interview and the home visits. The research occupational therapist had previously worked on this unit clinically.
Baseline assessments were conducted on all consented patients by a member of the research team, or data were taken from patient notes. For participants in the cohort study this was prior to the visit and for those in the randomized controlled trial it was prior to randomization. Assessments included disability as measured by the Modified Rankin Scale 7 and the Barthel Index, 8 mood using the General Health Questionnaire-28 9 and the Stroke Aphasic Depression Questionnaire 10-item hospital version 10 for those with communication problems (completed by patient’s named nurse); cognition using Addenbrooke’s Cognitive Examination Revised 11 and EQ-5D 12 for the health-related quality of life.
The primary outcome measure was the Nottingham Extended Activities of Daily Living (NEADL) 13 at one month after discharge from hospital. Secondary outcomes were:
disability as measured by the Barthel Index 8 ;
mood using the General Health Questionnaire-28 9 and the Stroke Aphasic Depression Questionnaire – 10-item hospital version 10 ; those with communication problems had only the SADQ-H10 completed;
health-related quality of life, measured using the EQ-5D 12 questionnaire;
general mobility using the Rivermead Mobility Index 14 ;
number of falls and readmissions;
carer strain, if there was a main carer, using the Carer Strain Index 15 ;
resource use. For the home visit this included: costs of staff attending, travel time, time at home, administrative time associated with visit, cost of transport, cost of time taken to recommend actions, equipment and for referrals. For the interview this included: staff present, duration of interview, recommendations made, equipment tested, referrals made and any information supplied.
For the outcome assessments the researcher was masked to group allocation. All participants were followed up at one week and at one month following discharge from hospital. Assessments were conducted wherever the patient was at this time point – hospital, home or nursing home.
Data were analysed using SPSS version 16 (SPSS Inc., Chicago, IL, USA). The majority of the data were entered and analysed blind to group allocation; specific home visit data were entered last by an independent researcher. The analysis focused on determining feasibility, examining the primary outcome measure and on examining any differences between participants in the cohort and randomized controlled trial studies. Analyses were carried out on the basis of intention to treat.
For baseline and outcome measures where less than 10% of the total data were missing, mean values were imputed for individual missing items. Where 10% or more data were missing, the entire measure was coded as ‘missing’. The exceptions were the Addenbrooke’s Cognitive Examination Revised, 11 which was used only at baseline, and the EQ-5D 12 ; no values were imputed for these measures and missing values were coded as ‘0’.
Results
Participants
The flow of participants through the study is shown in Figure 1. Out of 297 patients admitted to the stroke rehabilitation unit, 216 met the eligibility criteria. Of the 81 not eligible, 36 were discharged before a decision about the need for a home visit was made, 10 were deemed to need access visits only, 9 were transferred (5 to other wards and 4 outside the catchment area), 7 died, 6 did not speak English, 5 were terminally ill, 4 were still on the ward when study was closed and 4 were ‘other’ reasons (e.g. diagnosis of stroke unconfirmed, readmission, planning not to return directly to own home and missed).
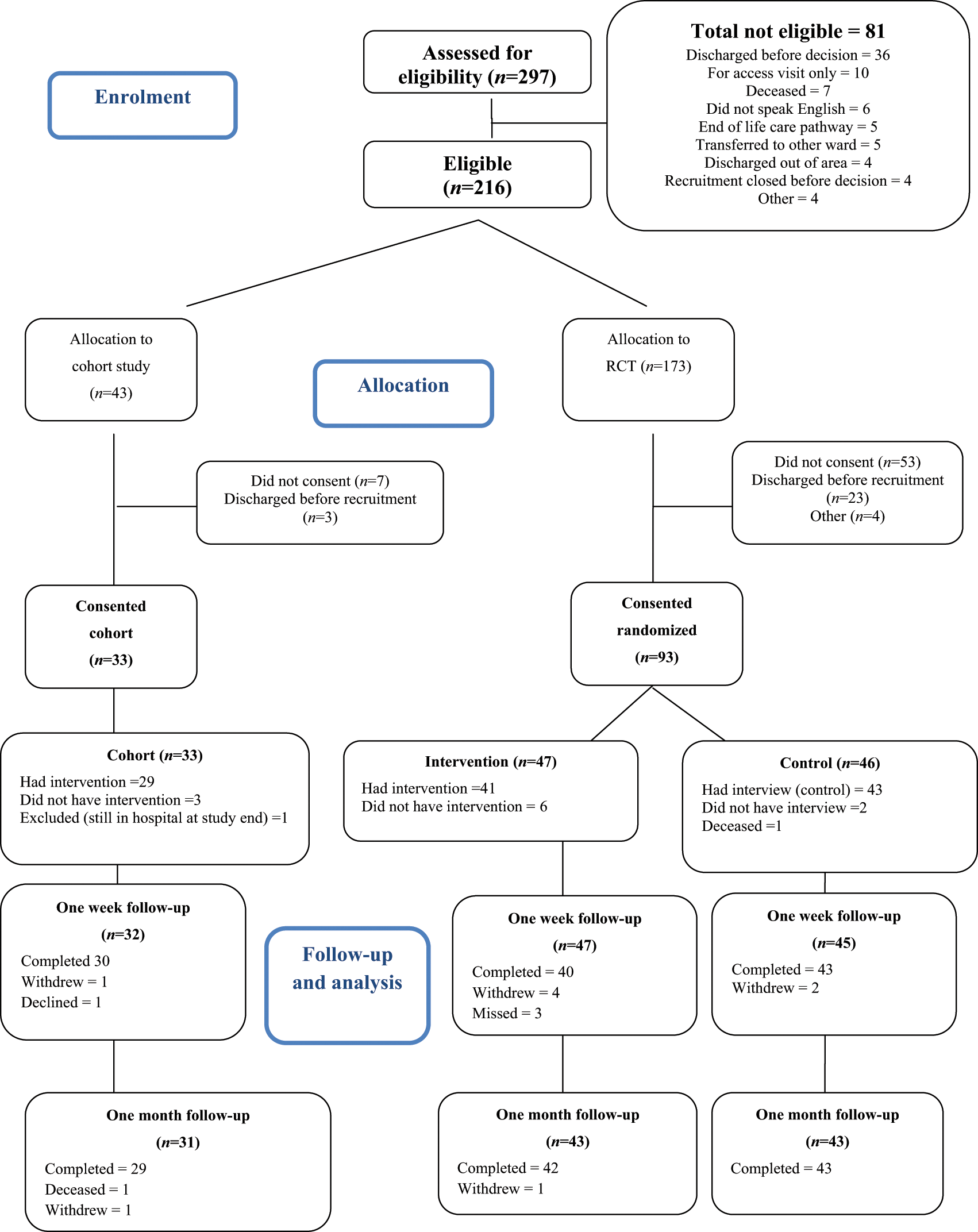
CONSORT diagram.
Of the 216, 173 were suitable for the randomized controlled trial and 93 were subsequently randomized (47 to the intervention; 46 to control). Thirty-three people were enrolled into the cohort study. Baseline characteristics are shown in Table 1. The mean age at randomization to the study was 72 years (SD 14.67, range 34–99). The randomized controlled trial groups were well balanced at baseline for demographic characteristics and baseline measures.
Baseline characteristics by group
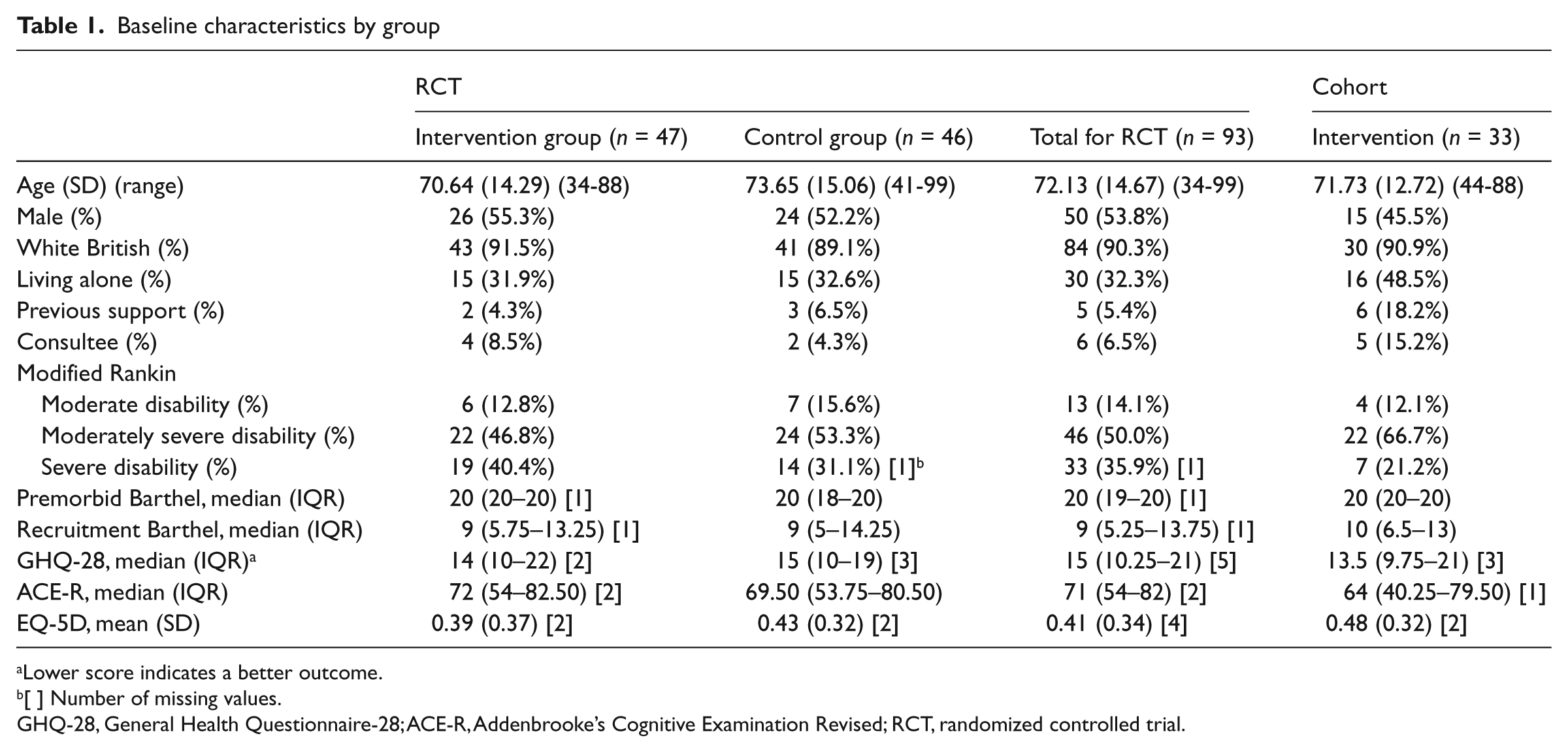
Lower score indicates a better outcome.
[ ] Number of missing values.
GHQ-28, General Health Questionnaire-28; ACE-R, Addenbrooke’s Cognitive Examination Revised; RCT, randomized controlled trial.
By comparison with participants in the randomized controlled trial, the participants in the cohort study were more likely to be female, to live alone and to have received a support package prior to admission. They were also more likely to have been consented by a consultee and have lower cognition scores as measured on the Addenbrooke’s Cognitive Examination Revised. 11 The most common reasons occupational therapists cited on the form for allocating patients to the cohort study were specific environmental concerns at home (e.g. stairs), cognitive issues including lack of insight and the patient living alone. Reasons were recorded for 24 (73%) of the cohort participants; for 23 of them, more than one reason was supplied. A total of 100 individual reasons were listed for meeting the criteria of needing a home assessment visit.
Outcome measures
There were no significant differences between the groups in the randomized controlled trial for any measure except mood (measured on the Stroke Aphasic Depression Questionnaire – 10-item hospital version
10
at one week) and readmissions to hospital at one month (Table 2). The former was in favour of the intervention group; the latter in favour of the control group. More participants in the intervention group (
Outcome measures
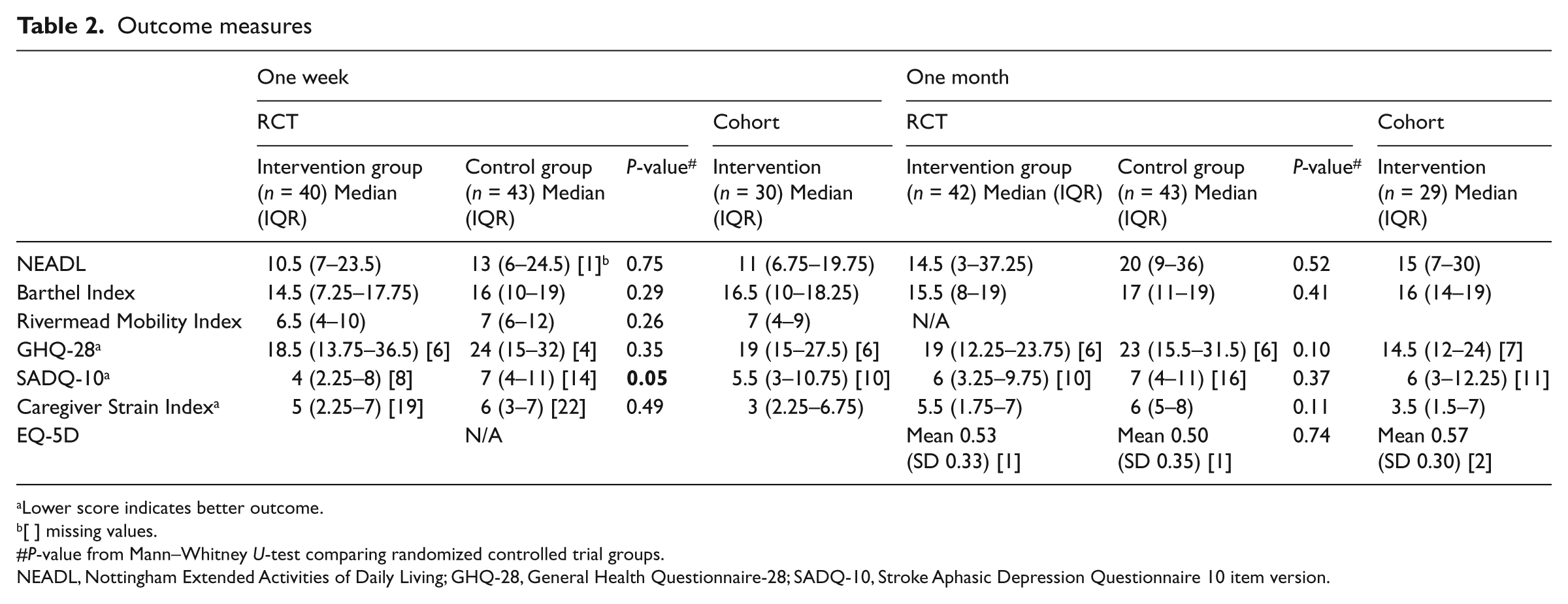
Lower score indicates better outcome.
[ ] missing values.
NEADL, Nottingham Extended Activities of Daily Living; GHQ-28, General Health Questionnaire-28; SADQ-10, Stroke Aphasic Depression Questionnaire 10 item version.
The main resource use associated with home visits related to the amount of staff time required, which was attached to NHS staff costs. 16 The mean (SD) time spent by the primary member of staff on a home visit (including organizing, completing and writing-up visits) was 180 (85) minutes for the randomized controlled trial participants and 203 (63) minutes for those in the cohort study. The mean (SD) total cost of staff time in the randomized controlled trial home visit was £158 (£75). The mean (SD) cost of staff time in the cohort study was £215 (£122). Other costs related to home visits included travel costs, parking fees and the provision of milk for kitchen assessments. The mean (SD) total cost of a home visit for randomized controlled trial participants was £183 (£81). For the cohort study group the mean (SD) cost was £243 (£130), giving a mean (SD) across all home visits of £208 (£107).
With regard to the randomized controlled trial control group, the mean (SD) time spent by the primary staff member on the hospital interview was 99 (53) minutes, and the mean (SD) total cost of a hospital interview was £75 (£40).
Feasibility
As the trial progressed, clinical staff allocated more patients to the randomized controlled trial; in the first four months of recruiting the majority of patients were considered to be ‘essential’ for a home visit and enrolled to the cohort study, whereas in the last four months the situation was reversed (Figure 2).
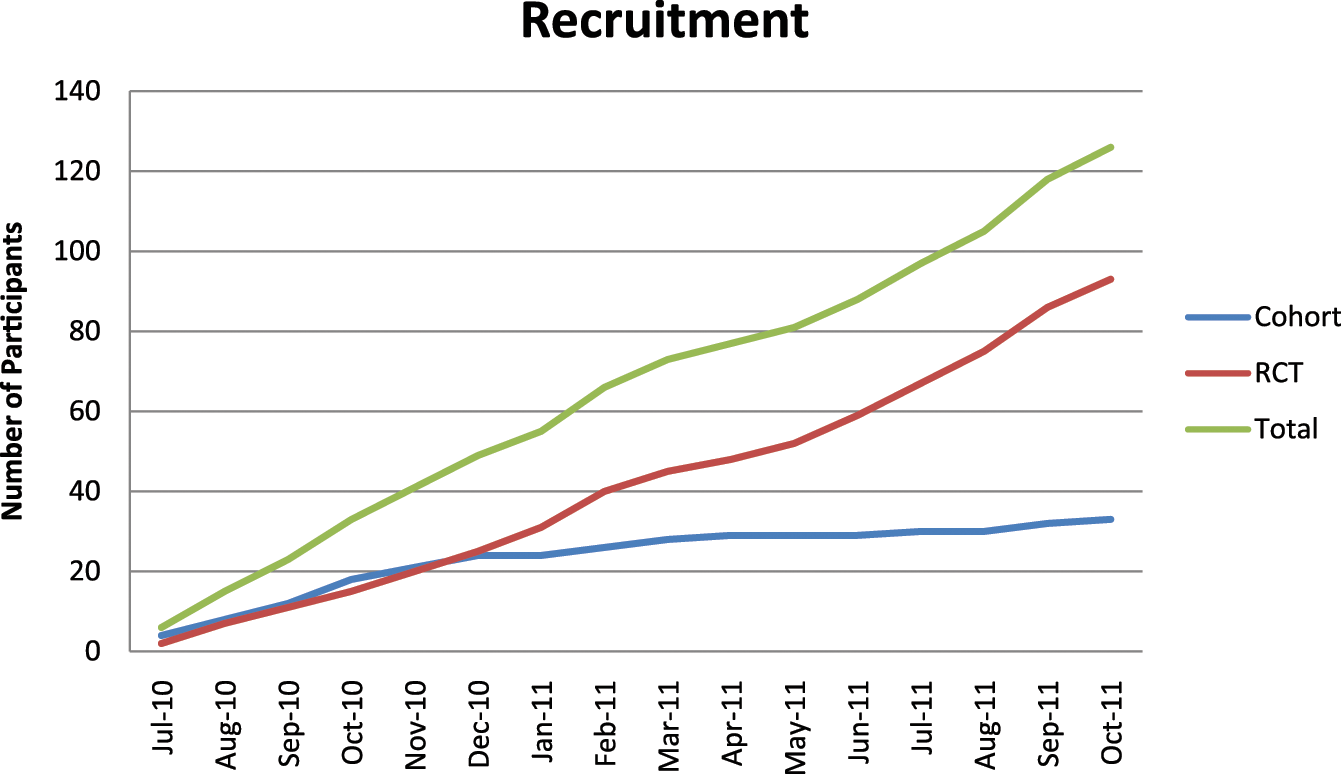
Recruitment during study.
With regard to consent, as Figure 1 shows, many people suitable for the randomized controlled trial declined to participate (
With reference to questionnaire completion, the majority of measures were fully completed. The main exception was the General Health Questionnaire-28, 9 which had the most incomplete responses (see Table 2), even with an assessor available to help. Several questions were systematically missed by patients who felt they were intrusive such as questions related to suicidal thoughts.
In delivering the home visit intervention, 29 participants in the cohort study received the intervention although 2 of these had two home visits and 1 was discharged on the visit. Of the 4 people who did not subsequently have a visit, 1 went into a nursing home, 1 was for re-housing, 1 was transferred prior to having a visit and 1 was still in hospital when the study closed. In the randomized controlled trial, 41 people had the intervention (1 person had two visits, 1 person had an access visit plus a home visit, 8 people were discharged on the visit and 3 people received the visit after discharge); 6 people did not (3 were transferred to other wards, 2 were discharged before intervention and 1 received the control interview). In the control group, 43 people had the hospital interview, however, of these 4 people also received an access visit and 2 people received a home visit (one of whom was discharged on the visit). Two people did not receive the intervention (1 was discharged and 1 withdrew from the trial).
Discussion
This was a feasibility trial undertaken in a clinical setting; the eligibility criteria were agreed beforehand and the use of a parallel cohort study meant that clinicians did not have to randomize patients for whom they felt a home visit was essential. We believe it was for this reason that we were able to recruit to this study – in contrast to the problems experienced by Australian colleagues. 1 We also think that giving control to the clinicians allowed them to be more confident in suggesting patients for randomization, which increased steadily over the study period. However balance is needed between research rigor and clinical concerns. Although we were successful in conducting this study, one of the limitations was that despite agreeing criteria for the essential group prior to commencing, there was a range of reasons given to justify visits. While these broadly met the agreed criteria of ‘new, significant functional impairment and/or environmental concerns’, these criteria would need clearly to be more explicitly defined in a future study.
Moreover, although we have demonstrated that such a trial is feasible in terms of recruitment and follow-up, we have identified some important issues. There were problems with patient completion of one of the measures (General Health Questionnaire-28 9 ), for which several questions were systematically not answered. This leads us to question the use of this measure in a subsequent trial. There was also a need for stricter protocol adherence in the randomized controlled trial; some people received a home visit or an access visit, when they should not, or received more than one visit or were discharged on the visit. This would clearly need to be managed more strictly in a definitive trial. With regard to safety, there were more falls and hospital readmissions in the randomized controlled trial intervention group. All adverse events were reviewed unblinded to intervention by a stroke physician and these events were not thought to be related to intervention, however this would also need to be addressed in a larger trial. It is possible that a home visit makes people more confident on discharge and thus the possibility that this may put them at more risk of falls or other adverse events needs to be investigated.
There were also key issues identified regarding the control group. It is likely that patients in this group received more intervention than is standard care in most centres; we know that in some hospitals patients are discharged from hospital following a stroke without any visit 4 and interviews are not routine practice. It may be that the control group received too much intervention – although it would have been difficult to give people nothing in a service where the majority of patients previously had a visit. It is also interesting, even allowing for the fact that the study was underpowered, that patients who had the interview seemed to have similar outcomes to those who had a home assessment. This may reflect that the in-depth ‘control’ intervention attenuated any outcome differences between the two groups, or may reflect a genuine lack of efficacy of home visits. This clearly needs further investigation. It is vital that the results of this study are not summarized inaccurately to suggest either that structured interviews can be used in place of home visits or that home visits are not effective. Moreover, this study did not investigate whether the collection of data on the home environment is important or not as both groups collected this data. Rather this study shows that a definitive trial occupational therapy home visits is both possible and warranted given the cost of visits and the lack of evidence to date of efficacy.
Clinical messages
This research shows that a trial of home visits is both feasible and warranted. It also provides important data on the costs of undertaking home assessment visits.
A careful balance must be struck between the involvement and concerns of clinicians and with research rigor.
Footnotes
Acknowledgements
We are grateful to the members of our steering committee and expert advisors: Dr Nicola Brain, Dr Boilang Guo, Professor Marilyn James, Dr Annie McCluskey, Mr Oswald Newell, Dr Cecily Palmer, Dr Ruth Parry, Dr Kate Radford, Dr Tracy Sach, Professor Cath Sackley, Dr Karen Stainer and Professor Marion Walker. We are grateful to all the staff on stroke wards at Derby, particularly the occupational therapists and the medical consultants for supporting this trial. We are also grateful to the patients and their carers for agreeing to participate in this research. We would also like to acknowledge the assistance of the Nottingham CTU, and in particular Daniel Simpkins.
Authors’ note
This paper presents independent research commissioned by the National Institute for Health Research (NIHR) as part of the Collaboration for Leadership in Applied Health Research and Care – Nottinghamshire, Derbyshire and Lincolnshire (CLAHRC-NDL). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Contributions of authors
AD contributed to the conception and design of the protocol, conducted follow ups, performed analysis, interpreted data and drafted the final paper. PW contributed to the design, patient recruitment, conducted follow ups, analysis, interpretation of data and drafted sections of the paper. KF contributed to the design, recruited patients, designed and conducted interventions, interpretation of data and commented on drafts of the paper. NS contributed to the design, interpretation of data and commented on drafts of the paper. CS led and conducted analysis and interpretation of health economic data and drafted sections of the paper. CE conducted data entry, analysis, interpretation of data and commented on drafts of the paper. NL contributed to the conception and design, interpretation of data and commented on drafts of the paper.
Conflict of interest
The authors declare no conflicts of interest in preparing this article.
Funding
This research received funding from the National Institute for Health Research (NIHR).
