Abstract
Background
Compression therapy (CT) is widely prescribed for chronic venous disease (CVD), lymphedema, and lipedema despite robust evidence supporting its effectiveness only in a minority of clinical scenarios.
Aim
Provide an overview of the methodological parameters essential for obtaining a high-quality scientific trial.
Methods
The International Compression Club (ICC) chairman prepared a preliminary list of the main methodological flaws in publications on CT, and a list of parameters to be included in high-quality randomized controlled trials (RCTs). This list was circulated among ICC board members, who provided their comments and suggestions. The document was then presented and discussed at an ICC consensus meeting with various professionals in May 2024 in London. Meeting minutes were distributed to ICC members, and the final document reflecting all additional comments and suggestions was compiled. In summary, the following recommendations represent the consensus of the ICC members.
Results
The ICC recommends that the following parameters be reported in randomized clinical trials (RCTs): appropriate patient selection, adequate sample size, detailed classification of venous and lymphatic diseases, detailed description of CT modality, adherence to CT, and reasons for non-adherence. Tailored recommendations for specific scenarios and the use of standardized quality-of-life (QoL) tools are emphasized. Despite their value, RCTs face limitations and do not reflect real-world clinical practice. Real-world evidence (RWE) can complement RCTs by providing insights into routine applications, safety, compliance, and cost-effectiveness across diverse patient populations by standardized methodologies, large cohorts, and reliable validated registries to ensure validity.
Conclusions
Combining data from RCTs and RWS allows for a comprehensive understanding of CT. RCTs provide evidence of effectiveness in a controlled scenario, while RWE offers real-world perspectives on compliance and economic outcomes in clinical practice. Together, these approaches enable a more holistic evaluation of CT, addressing current gaps in knowledge and guiding its optimal use.
Keywords
Introduction
Even though compression therapy (CT) applied by multiple modalities (medical compression stockings, elastic and inelastic bandages, wraps, pumps) is extensively prescribed in many clinical scenarios of chronic venous and lymphatic disease, the evidence of its effectiveness is limited to a few indications. In a recent publication reporting a literature review on CT, 1 it was proved effective in venous insufficiency clinical classes C3 to C6 [venous edema, lipodermatosclerosis, healed venous ulcer to prevent recurrences, venous leg ulcers (VLU)], and in deep vein thrombosis (DVT) and lymphedema. However, both in VLU and DVT, which kind of CT [if elastic compression stockings (ECS) or inelastic materials (bandages or wraps)] is more effective is not yet clear. In many potential indications for CT (venous symptoms control, venous disease progression, CT after venous procedures, thromboprophylaxis, prevention and treatment of post-thrombotic syndrome, during or after sports activities), scientific studies report no or weak evidence for CT effectiveness. In other indications (for instance, CT after venous procedures), there are conflicting data due to studies reporting CT efficacy and other studies denying any effectiveness. However, too many studies on CT are burdened by significant methodological flaws. As a practical consequence, it is necessary to find out when missing, weak, or contrasting data on CT effectiveness are due to CT ineffectiveness or to the methodological flaws of the studies, which cannot provide solid data in favor or against CT. The gold standard to investigate the efficacy and safety of any therapeutic intervention is represented by RCTs and Meta-analysis.2,3 Although the parameters necessary for high-quality RCTs should be clear to all the researchers, several methodological mistakes and bias burdening many studies encouraged the International Compression Club (ICC) board members to provide all researchers interested in publishing on CT with minimal items required for a good study on CT.
This work aims to provide a consensus on the data that must be included in high-quality RCTs investigating the effectiveness of CT, including their limits and possible solutions to overcome them.
Methods
Methodological flaws in RCTs and their consequences.
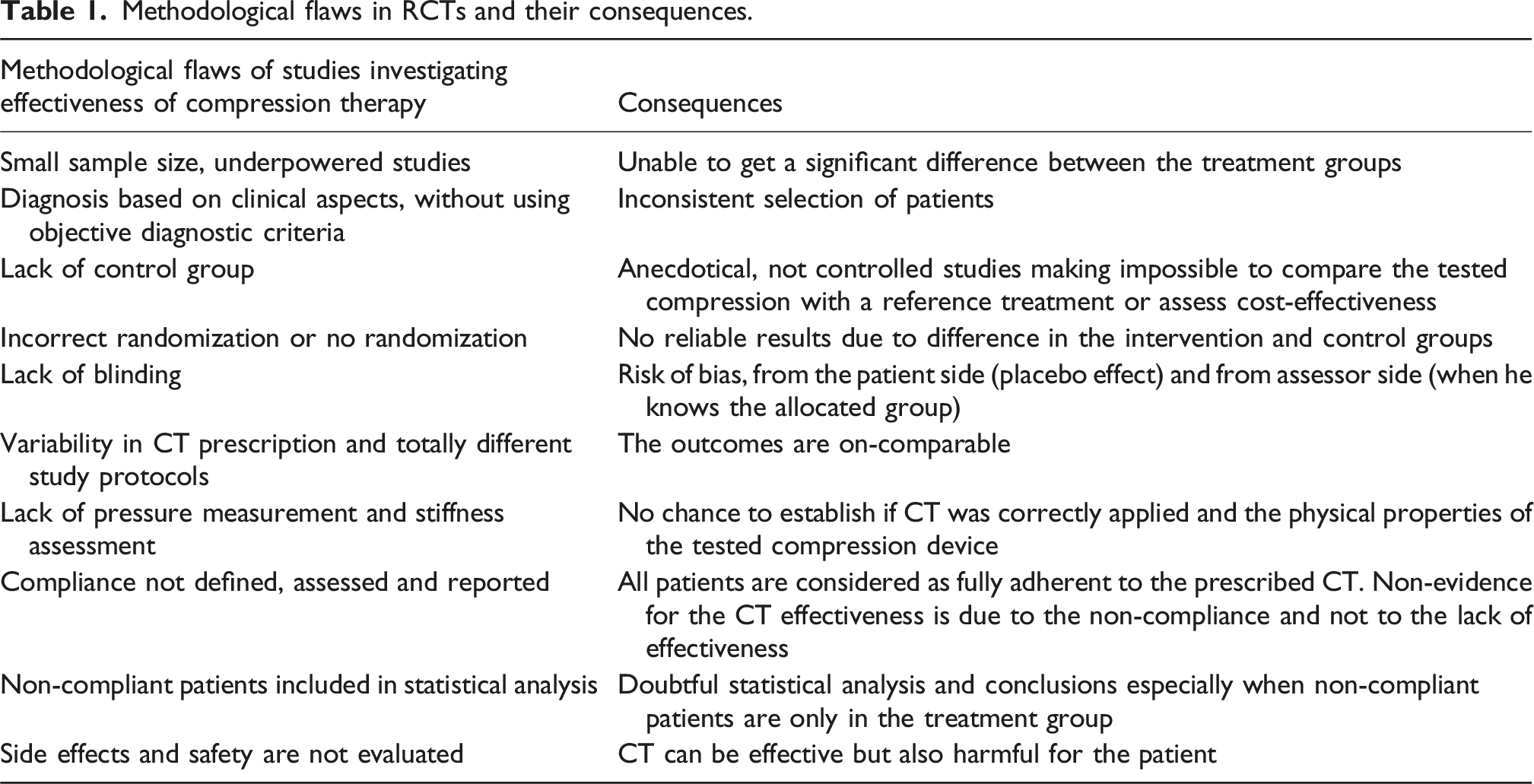
Results
The ICC members first requested that data from the so-called “real world” be considered in addition to adequately designed and powered randomized controlled trials (RCTs). Consequently, though the consensus focused on mandatory requirements for publications on RCTs, it was decided to highlight their limitations and define how RCTs data can be integrated with data from real-world studies.
The ICC members agree that the protocol of a high-quality RCT about the effectiveness of CT must meet the following requirements:
Sample size and statistical power
The sample size must be adequately powered to detect a significant effect on the study’s primary outcome. It must also be statistically calculated based on the expected effect sizes and variability. Proper sample size calculation is crucial to ensure that the study can provide reliable and valid conclusions.
Patient selection criteria and baseline characteristics
The patients must be carefully selected. Inclusion and exclusion criteria have to be well defined. Diagnostic tools, such as ultrasound, are necessary to prove the presence of venous disease or its absence when enrolling normal volunteers. The patient’s clinical class must be reported, which is usually the case. However, the etiology, anatomy, and pathophysiology of the patient’s venous disease, according to the updated CEAP classification, 4 are also mandatory information. Weather including or excluding patients with a history and signs of previous chronic venous insufficiency (CVI), must be defined in the protocol. The pre-existing primary diseases must be described if patients with previous CVI are included in the study.
The same precise details must be added relatively to a possible simultaneous arterial disease: distal pulses assessment, ankle-brachial pressure index, and, possibly, the pain-free walking distance to assess the clinical grade of arterial impairment. For lymphedema patients, lymphoscintigraphic or indocyanine green findings are helpful for the diagnosis. Their clinical stage has to be defined following the International Society of Lymphology Consensus. 5 The baseline characteristics of the study population, including all potentially relevant parameters that impact outcomes must be described: individual and disease characteristics that can act as risk factors for lymphedema, potential confounding factors, known prediction factors or prognostic factors.
Control groups and randomization
A control group is mandatory to ensure the study’s validity. Patients in the control group should receive a comparator or a placebo device or no compression. Attention should be paid to placebo compression that does not exist. Every compression pressure, even light, produces some effects, as clearly shown in a paper where a stocking exerting less than 10 mmHg effectively reduced leg edema. 6 Therefore, a placebo device should not exert any compression. The method section must clearly outline the randomization method used to assign the patients to the control or treatment group to have well-balanced groups. Reporting the characteristics of the treatment and control group is requested to demonstrate that the randomization worked appropriately, minimizing the risk of bias resulting in population differences. Very often, information on the specific characteristics of study populations is minimal, raising doubts about whether the study arms are correctly balanced.
Blinding strategies to minimize bias
Double blindness should be considered to minimize bias risks. ICC members agree that, although recommended, double blindness is almost always impossible when dealing with compression devices; however, a single blinding (the assessor’s blinding), in addition to the statistician’s blinding, is highly recommended.
Standardization of compression therapy protocols
Compression therapy prescription and study protocol should be well described and consistent. In particular, the type of compression used in the specific study population must be accurately described: what kind of medical compression stockings, the composition of multicomponent bandages, what kind of wraps. Wearing time and timing for compression change must be defined and reported. This recommendation will avoid meta-analysis or systematic review, including studies with different compression modalities and protocols, leading to the impossibility for the researchers to come to any conclusion (a typical example is the effectiveness of compression therapy after endovenous ablation). 7
Compression dosage and pressure measurement
The dosage of compression therapy, represented by the compression pressure, must be reported. Assuming that other essential compression characteristics (such as a correct application with an even pressure distribution to avoid strangulation) are respected, CT, like any other therapy, depends on its dosage. Compression pressure must be measured at least at application time. If there is an interest in assessing the pressure behavior over time for different compression devices, additional measurements must be performed at removal time or intervals defined in the protocol.
Static stiffness index (SSI) assessment
The SSI must be assessed to determine whether the compression device is stiff or non-stiff. This data is critical, especially in multicomponent bandages, where the stiffness of the final bandage can be different from the stiffness of every component. 8 Some ICC members argued that compression pressure measurement and SSI assessment are not standard clinical practice and may influence the use and application of the compression device. However, most members agreed that these assessments are mandatory in RCTs on CT.
Compliance monitoring and reporting
Compliance must be assessed. The ICC members agreed that compliance requires wearing all compression modalities for 7 days a week and 24 h a day when applied for ulcer treatment, DVT in the acute phase, and lymphedema in the treatment phase when decongestion is necessary. In other venous indications and the lymphedema maintenance phase, MCS or wraps must be worn at least 6 days a week and can be removed overnight. The wearing time must be reported in the study. Patients who significantly fail to adhere to the wearing time outlined in the protocol must be considered as non-compliant. Reasons for non-compliance must be listed.
Handling of non-compliance in the analysis
Including or excluding non-compliant patients from the final analysis was a very debated topic. Including non-compliant patients, according to the intention-to-treat (ITT) analysis, is more scientifically correct as it prevents potential bias due to patients’ exclusion. 2 However, this distinction is particularly relevant in compression therapy studies, where patient adherence significantly impacts outcomes and might introduce bias if improperly analyzed. Some members raised their concern about situations such as non-compliant patients being only in the treatment group, as patients in the control group do not wear any compression or placebo stockings, which is the usual case when dealing with studies on CT. In this case, the possible beneficial effects of the treatment cannot be demonstrated. On this point, a consensus was not achieved. However, most ICC members favored excluding non-compliant patients according to the per-protocol (PP) analysis and reporting all the causes for non-compliance. Some solutions were proposed during the debate: one solution was to select the patients during their first visit, before enrollment, based on their availability to wear compression according to the study protocol. Another solution was to include only patients who had already worn some compression in the past and were already compliant with compression. In this case, non-compliance should be kept at a shallow level.
Specific recommendations for specific studies
Specific recommendations should be added for specific studies. Some examples are ulcer characteristics, including size, age, location, wound bed conditions, infection, and so on, for studies on compression for ulcers; vein diameter, location (if in the thigh, leg, or both), concomitant tributaries, and so on, for compression after vein procedures. These parameters should be defined according to the study hypothesis.
Innovative tools: Sensors for compliance monitoring
Utilizing sensors in compression devices is an innovative approach. The ICC members agree that integrating sensors into the compression device would be an excellent solution to assess patient compliance. It could also be very important if the same sensor recorded the patient’s physical activity, which is well-known to increase CT effectiveness. However, concerns about the sensor’s availability and cost have been raised. If they are available and reasonably priced, the ICC members agree that it is a significant advantage to integrate them in the compression device.
Quality of life (QoL) Assessments in compression therapy studies
QoL scores deriving from specific questionnaire compression- and disease-related must be reported. It must be specified which QoL tool was used, when it was completed, and who completed the questionnaire. The ICC has developed a specific questionnaire to evaluate the quality of life (QoL) related to compression therapy. 9 This questionnaire assesses the effects of different compression materials and systems and their acceptance and experience by patients and care providers.
Side effects and safety
All side effects, unwanted effects, and all other signs concerning compression safety and risks for the participants must be reported.
Discussion
There are several methods for determining consensus, but there is still uncertainty about the optimal method. 10 We chose the reported method to involve a big number of ICC members after explaining them the reasons for this consensus and discussing together every single parameter useful to increase the scientific level of clinical trials. This extensive work could not always be possible by using other methods. During the ICC consensus meeting, the participants agreed that RCTs are necessary to investigate the effectiveness of CT and agreed on the minimal requirements for such RCTs. Safety and risks must also be evaluated in RCTs, even if ideal, controlled conditions in a selected population of patients result in limited value of these data.
Randomized control trials must be planned based on predefined protocols, including a proper patient number. The correct sample calculation is mandatory; if the sample size is too small, the study will not achieve statistically significant outcomes; if it is too large, an unacceptable number of patients will be submitted to a placebo treatment with risk for their health status.
The participants must be selected through an accurate and objective diagnostic procedure and by considering well-determined inclusion and exclusion criteria.
Patients must be randomized into a treatment and control group. Studies without a control group may simply report the effectiveness of a compression modality. However, this kind of study cannot report whether the tested compression system is more or less effective than a reference compression modality. Cost-effectiveness can also not be assessed; consequently, these studies have severe limitations.
Compression pressure must be measured and SSI assessed. Compression pressure represents not only the dosage of compression that must be declared as it happens for any medical treatment. ICC members highlight that compression pressure is also essential to understand if the compression device was correctly applied. If one compression device must exert a pressure of 50 mmHg when applied according to the indications provided by manufacturers, previous studies, or ex-vivo measurement, 11 every participant in the study must be sure to apply this pressure level. When compression pressure is not measured, it is impossible to know if the compression device was applied according to the protocol requirement. Especially when using multicomponent bandages, the stretch and the bandage superimposition can vary for each component from one application to another and among different healthcare providers. Although marks on the bandage can help achieve a consistent application, doubts about application appropriateness can be raised, and study conclusions can be argued. Stiffness assessment is essential, especially when testing multicomponent compression devices where the stiffness of the final device can be different from the stiffness of every single component. For instance, the so-called four-layer bandage has been considered elastic because it comprises four elastic bandages. However, when its stiffness was assessed, it resulted in the range of stiff materials. 8 Consequently, all the studies comparing the “elastic” four-layer with an inelastic bandage actually compared two inelastic bandages, making the conclusions of these studies hard to accept.
The wearing time of compression and patients’ compliance with the protocol must be reported. Without these data, it is impossible to know if the protocol was respected, making the outcomes, once more, doubtful.
Whether to include or exclude non-compliant patients from the final analysis was the only very debated issue. Most members favored excluding non-compliant patients, according to the per-protocol analysis, even if trying to adopt some criteria during the inclusion process to keep non-compliance at a minimum.
Sensors assessing compliance and patients’ physical activity should be used if cheap and widely available.
Specific details for specific studies must be reported.
A dedicated QoL questionnaire must be used.
Additional parameters are necessary in RCTs. Even if not discussed during the consensus meeting, they are universally accepted, though sometimes neglected. They include a precise formulation of the study hypothesis, the validity of measurement instruments, the consideration of variables, and proper statistical analysis. In addition, the conclusion should concisely but precisely report the data concerning the kind of compression applied and the specific study population. This would avoid misinterpreting the study and extrapolating its results to other types of compression and other patient populations.
During the consensus meeting, participants raised the issue of the limitations of RCTs. Although RCTs represent the second highest level of the evidence pyramid 12 and are required for regulatory approval, their limitations must be considered. The strict inclusion and exclusion criteria result in a homogeneous yet small and highly selected sample that does not reflect the general population with various comorbidities and treatments. Additionally, the controlled conditions and the necessity to adhere to all protocol requirements do not mirror real life. The selected population may bias the safety assessment, as the safety results cannot be generalized to the broader population with comorbidities. Bias can also arise from including or excluding non-compliant patients. Furthermore, RCTs are often very expensive, particularly when a large patient enrolment is needed to achieve a statistically significant difference from the control group. Finally, insurance for all the enrolled patients is requested.
These limitations are why ICC members advocate for considering and integrating data from real-world studies (RWSs).13–16 These studies provide real-world evidence (RWE) by analyzing data from routine clinical practice. This is crucial for understanding how compression therapies perform outside the controlled environment of RCTs. Real-world data can reveal variations in treatment effectiveness across different patient populations and settings, offering a more comprehensive view of clinical outcomes. They can be conducted in various formats, such as prospective (Observational Studies) or retrospective research (Database Research or site-specific Medical Records Evaluations, maybe artificial intelligence-assisted), without all the restrictions of RCTs. Utilizing existing data makes these studies more cost-effective. This allows for the exploration of multiple research questions without the need for additional data collection, thereby accelerating the pace of research. Finally, these studies can track patient outcomes over extended periods, providing insights into compression therapies’ long-term effectiveness and safety. This is particularly important for chronic conditions like venous leg ulcers and lymphoedema, where long-term management is crucial and impossible to report in clinical trials.
However, observational studies have limited value regarding treatment effectiveness. A recent editorial 15 highlights that the AMA Manual of Style instructs that, contrarily with randomized trials where terms such as effect and causal relationship can be used, “observational studies cannot lead to causal inferences and should thus be described in terms of association or correlation, avoiding cause-and-effect language.” Despite these limitations, real-world evidence from medical claims, electronic health records databases and wearable devices must be used to deliver data on compression performance, safety outcomes, compliance, and reasons for noncompliance in real-world conditions. While the CT application is standardized in RCTs with guided pressure levels (pressure measurements), this application does not represent the device’s real-world use. As product application techniques, capabilities, and approaches greatly vary for compression care, real-world evidence should be used to determine outcomes across care providers and various patient profiles, including mobility or limb shapes with potential implications for bandage slippage and wear time. Economic outcomes (required for budget impact assessments) such as frequency of care/interventions, material use, and complications may be highly variable in the real-world setting, not subject to rigid care protocol defining schedules or interventions. Moreover, economic care parameters are health care system-specific and patient pathway-specific and require considering country particularities. They can best be determined only in RWE. These are the reasons why RWEs represent a significant integration to the RCTs and are also expected by the Notified Bodies to obtain a CE Mark renewal. However, the RWSs are also burdened with some limitations. They rely on existing data, which may be incomplete or of variable quality. Missing data, inconsistent documentation, and coding errors can introduce bias and affect the accuracy and precision of data, resulting in misleading conclusions.
Consequently, even real-world studies on CT need some minimal requirements:
Registries
When these studies are based on medical records, it is crucial that Registry Systems follow specific regulations and features to make this kind of research trustworthy.
Patients
A large cohort of an unselected population (hundreds of patients) is necessary, which can enhance the statistical power of studies. This is particularly beneficial for identifying rare adverse events or outcomes that smaller RCTs may not capture. Large sample sizes also allow for more robust subgroup analyses, helping to tailor compression care to specific patient needs.
Data analysis
Intention to treat analysis must be used even when the data are noncomparative.
Indications
The definition of all indications that the medical device is covering must be reported.
Even if not mandatory, adequate compression application and high patient compliance are desirable for trustable outcomes.
Adequate application mainly refers to compression by bandages. Bandages are very often not adequately applied. Even expert healthcare professionals apply inelastic bandages with about the same compression pressure as an elastic stocking and lower than a hosiery kit. 17 However, when correctly applied, an inelastic bandage should exert much higher pressure than an elastic stocking or hosiery. When comparing elastic material and inelastic bandages, the results are hard to accept if we are not sure that the inelastic bandages have been correctly applied.
Compliance is a critical issue, too. Compression therapy is often compared with no compression or a very light compression. Non-compliant patients obviously are only in the compression group; if the compliance is low, the result will be against CT effectiveness, but this result is due to non-compliance and not to lack of CT effectiveness. All the systems that maintain noncompliance at a minimum should be considered as this increases the value of these data.
Conclusions
Compression therapy is one of the most effective treatments for the conservative management of chronic venous insufficiency (CVI) lymphedema and lipedema, but its effectiveness is proven only in a minority of clinical scenarios. This is too often due to the poor quality of CT studies. With this consensus document, ICC members want to emphasize the mandatory parameters in RCTs on CT, their potential limitations, and the need to integrate their results with data from real-world studies. In particular, RCTs respecting all parameters listed above are mandatory to prove CT’s effectiveness in treating the different clinical scenarios of chronic venous insufficiency, lymphedema and lipedema and in studies comparing different compression modalities. The outcomes of RCTs should be integrated with data from real-world studies assessing safety and risk in real-world patients and long-term outcomes in compliant patients even when compression is applied by healthcare providers with different levels of experience, which is the case in the real world. Real-world studies are also very important to provide us with data on compliance, real clinical performance (including reasons for non-compliance and modalities to improve compliance), and cost-effectiveness in the general population under different conditions and in different countries.
Footnotes
Author contributions
GM, SB and CB wrote the first draft. All co-authors contributed suggestions and comments. GM created a pre-final version and sent it to all the co-authors, who provided their final suggestions, which were incorporated into the final version. All co-authors approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor
GM.
