Abstract
Objective
The aim of this study is to present our results with an innovative extracorporeal thermal therapy using High Intensity Focused Ultrasound (HIFU) to treat Superficial Venous Insufficiency (SVI).
Design
Observational retrospective study with prospective data collection.
Methods
A total of 102 consecutive patients were included. All types of incompetent veins that met the HIFU-device criteria were treated. The primary endpoint of our analysis was vein occlusion rate. As secondary endpoints, shrinkage, absence of reflux, clinical improvement and complications related to the treatment were analyzed. Clinical and Ultrasound (US) follow-up was planned at 15 days, 3 months, 6 months, and 12 months. For clinical assessment, simplify CEAP classification and Venous Clinical Severity Score (VCSS) were registered.
Results
A total of 164 veins were treated: 92 saphenous trunks [65 Great Saphenous Veins (GSV), 15 Short Saphenous Veins (SSV), 12 Anterior Accessory Saphenous Veins (AASV)], 48 perforator veins and 24 other veins (15 tributaries, 4 Giacomini veins, 5 neovascularizations). For all types of veins, occlusion rates were 85% to 96% at early follow-up and from 85% to 90% twelve months after the treatment. Regarding reflux-free rate, it ranged from 85% to 96% 2 weeks after treatment, and from 85% to 93% at twelve months. Finally, shrinkage rates ranged from 96% to 97% 2 weeks after the treatment, and from 94% to 97% at twelve months. All patients showed statistically significant clinical improvement in VCSS and CEAP classification trough the different follow-ups. No major adverse events were observed.
Conclusion
Treatment of varicose veins with the HIFU therapy seems to be competitive with the state-of-the-art techniques and has shown to be effective and safe in our initial 102 patients’ data analysis. Nevertheless, further studies are needed for stronger evidence.
Keywords
Introduction
Chronic Venous Disease (CVD) of the lower limbs is a common medical condition with a prevalence of up to 30% of the adult population. Its prevalence and morbidity increase with age, suggesting the earliest possible care. According to the National Institute for Health and Care Excellence (NICE), superficial venous insufficiency (SVI) of lower limbs is the most common form of CVD in symptomatic patients. The first-line recommended treatment of SVI and truncal reflux involves endothermal ablation, 1 as endorsed by both NICE and the European Society for Vascular Surgery guidelines. 2 According to the American Venous Forum, all endovenous ablation techniques are favored as the first choice of treatment for SVI. 3
Endovenous thermal procedures involve introducing a catheter or a laser fiber into the incompetent vein and heating the vein wall. These catheter-guided thermo-ablation techniques have been demonstrated to be safe and effective methods to treat incompetent truncal veins.4,5 However, inserting and navigating a catheter into pathologic vein can be difficult. The tortuosity increases the risks of adverse events and can be challenging for the physician. Certain types of veins such as perforators or superficial tributaries are not suited for this type of approach.6,7
High-Intensity Focused Ultrasound (HIFU) is a treatment method whose development and applications have been demonstrated in various medical fields over the last 80 years. Its feasibility and safety have been proven in treating various pathologies such as thyroid nodules, hepatic tumors, or intracerebral lesions.8–10
In comparison to endovenous methods, Extra-Corporeal HIFU Venous Ablation technique (ECHVA) is an appealing non-invasive thermo-ablative alternative for treating SVI. The efficacy and safety of HIFU as a non-invasive thermal method for treating SVI has already been demonstrated in some clinical studies.11–13 This paper presents our results at different follow-up intervals using HIFU therapy to treat all incompetent superficial veins that meet the indications for its use.
Methods
A total of 102 consecutive patients were included in the study. Data were prospectively collected and analyzed retrospectively. All patients received treatment from the same operator at the same center [Clínica Vascular Marbella, International Vascular and Endovascular Institute (IVEI), Marbella, Spain] between March 2022 and December 2024.
The inclusion criteria were: adult population (>18-year-old), life expectancy >2 years, presence of venous insufficiency class C2s or higher according to the Clinical, Etiology, Anatomy, Pathophysiology (CEAP) classification and veins characteristics meeting the device criteria for treating veins (vein depth between 9 and 24 mm when the vein is collapsed). During the first visit, a full clinical and ultrasound assessment was performed. During duplex ultrasound examination of their affected limbs, vein diameters and depths was measured to evaluate the applicability of HIFU treatment.
The Sonovein S Hifu system
All procedures were performed utilizing the CE-marked SONOVEIN® S HIFU device manufactured and provided by Theraclion® (Malakoff, France). This HIFU system is equipped with various subsystems, including a motorized treatment head (Figure 1). Within the treatment head, both a therapy transducer and an ultrasound imaging probe are integrated, with the imaging probe positioned centrally within the therapy transducer for real-time imaging, precise targeting, and treatment monitoring. Working alongside a single-use disposable kit, which includes a pre-filled pouch containing degassed fluid, a balloon, and connecting tubes, the system guarantees optimal cooling at 10°C throughout the entire treatment procedure. This cooling mechanism, facilitated by the circulation of fluid from the pouch to the balloon and back, efficiently removes excess heat from the patient’s skin. The Sonovein S during a treatment. 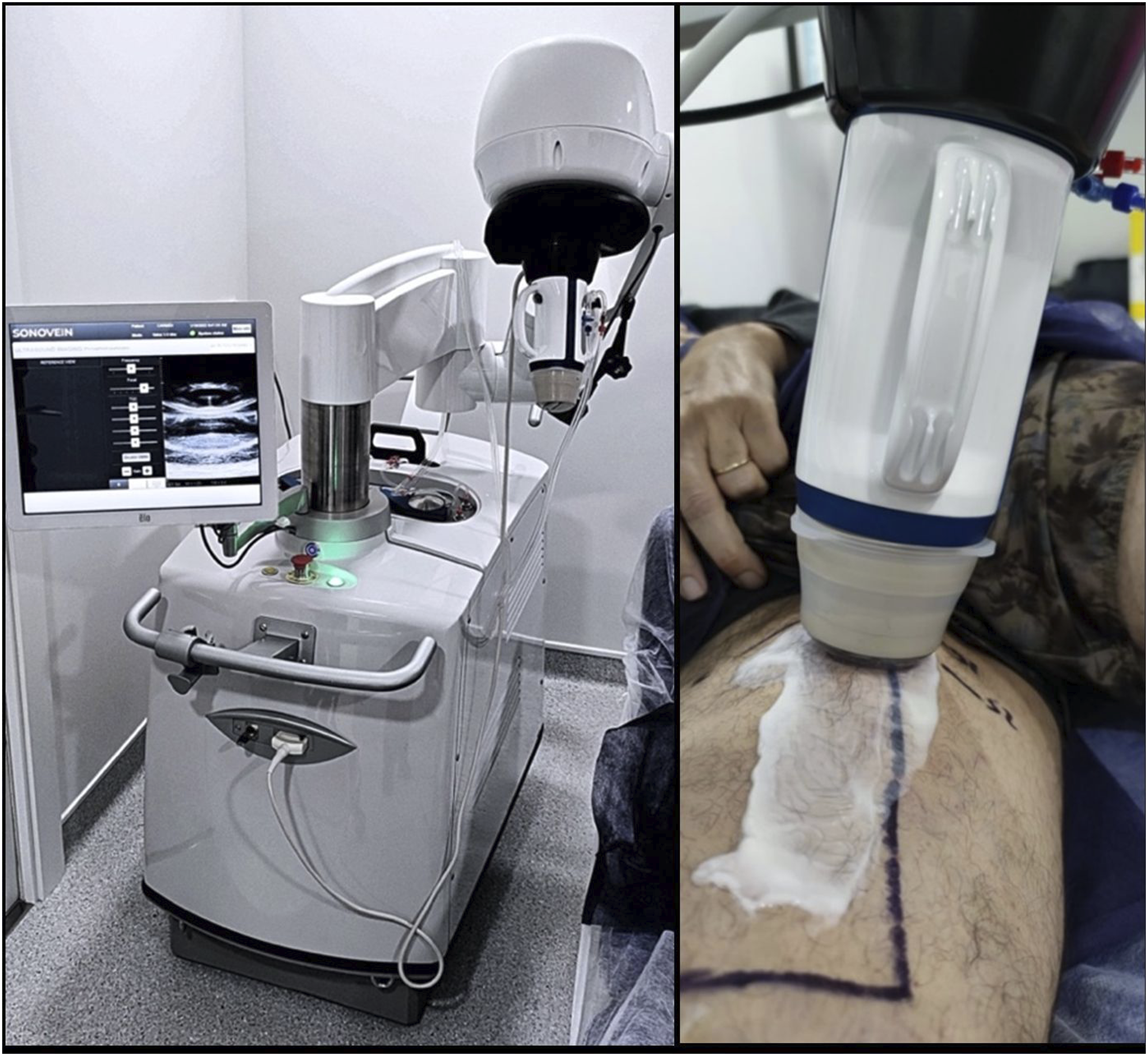
When applied to tissue, the focusing of high-power acoustic beams enables the induction of high temperatures locally at the focal point, producing localized tissue ablation. Based on previous studies,14,15 the dominant effects leading to vascular occlusion after HIFU application include vein wall thermal coagulation, HIFU-induced thrombogenesis and permanent fibrosis.
Treatment procedure
All procedures were conducted in an outpatient setting. With the patient in standing position, an accurate mapping of the target veins was performed over the skin. After placing the patients in the supine position, the International Vascular & Endovascular Institute (IVEI) anesthesia protocol, specifically implemented in our institution for ECHVA, was carried out. This involves a low volume peri-venous, ultrasound guided anesthesia protocol using lidocaine 2% to prevent pain during treatment (Figure 2). The IVEI anesthesia protocol entails small injections of 2 mL, performed with 27 G/4 cm needle punctures every 3 cm along the entire length of the target vein, to achieve a homogeneous anesthetic effect. IVEI anesthesia protocol. Low volume lidocaine 2%. Red: saphenous compartment. Blue: perivenous anesthesia. Green: venous wall.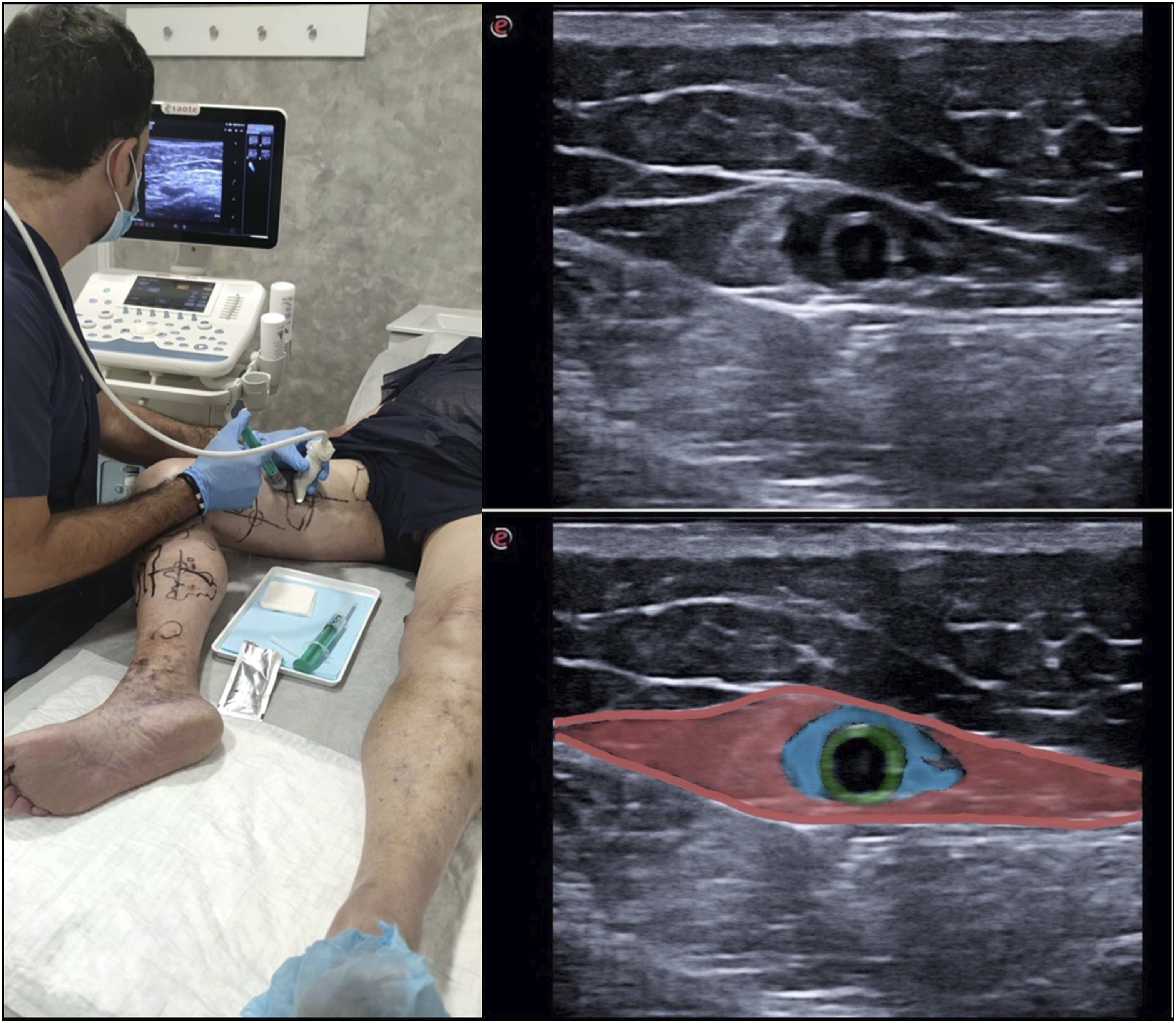
Once the patient was positioned for treatment, ultrasonic gel was applied to the skin over the vein to ensure appropriate coupling. The HIFU device head was first positioned manually in the region of interest and then adjusted robotically to focus on the targeted vein. Before each sonication, the vein was mechanically compressed with a balloon until no blood flow was detected to maximize thermal damage to the vein wall. HIFU pulses of either 0.5 or 1 seconds were then delivered on the vein wall, depending on vein depth. The acoustic power to be delivered is set by the operator during the treatment based on the occurrence of hyper-echoic mark, resulting from tissue heating. When treating primary incompetent saphenous trunks, the starting position for ECHVA has been between 2 and 5 cm from the saphenofemoral and/or popliteal junctions. During and immediately after the treatment, procedural pain was evaluated using a Visual Analog Scale (VAS).
Technical success was defined as the occlusion and shrinkage of the targeted vein after the procedure. Vein occlusion was defined as the absence of any flow along the treated vein, while shrinkage was defined as a reduction in vein diameter of 50% or more immediately after the procedure.
Ultrasound Guided Foam Sclerotherapy (UGFS) was the concomitant treatment used for tributary varicose veins in all cases. UGFS was applied in a different session for all Saphenous trunks, spaced at least 1 month apart from the initial ECHVA. In cases of Perforators, neovascularizations or tributaries treatment, UGFS was applied synergically with HIFU from the first treatment session.
Patients at risk of venous thrombosis or previous history of phlebitis were advised to wear compressive stockings for 3 weeks. All patients returned immediately to their normal activities after the treatment session.
Follow-up
The follow-up protocol was planned at 15 days, 3 months, 6 months, and 12 months. Each follow-up visit consisted of a duplex ultrasound examination of the treated leg. Occlusion, shrinkage, and presence of reflux were assessed for all treated veins. Simplify CEAP clinical classification and VCSS were recorded.
Primary and secondary endpoints
The primary endpoint of our analysis was vein occlusion rate immediately after the procedure and during follow-up visits. As secondary endpoints, we also evaluated shrinkage, absence of reflux, clinical improvement and complications related to the treatment.
Statistical analysis
Initially, a descriptive analysis of the characteristics of the study population was conducted, including both sociodemographic variables and those describing the patient’s situation at the time of diagnosis, preoperative evaluation, and treatment procedure. The description of these variables was carried out using measures of central tendency and dispersion for quantitative variables (mean and standard deviation), and frequency distribution for categorical variables.
The proportion of patients with continued vein occlusion was calculated using Kaplan-Meier method, separating saphenous trunks, perforator veins, tributaries, and others. The log-rank test was also carried out to compare the survival distributions of the three vein types. The paired student’s t test was used to compare the VCSS evolution throughout the different follow-up visits.
The statistical analysis was performed using the Python programming language, specifically with Spyder (Scientific Python Development EnviRonment) as the development environment.
Results
A total of 164 veins were treated in 106 legs of 102 patients: 92 saphenous trunks [64 Great Saphenous Veins (GSV), 15 Small Saphenous Veins (SSV), 12 Anterior Accessory Saphenous Veins (AASV)], 48 perforator veins and 24 other veins (15 tributaries, 4 Giacomini veins, 5 neovascularizations).
Demographics and baseline characteristics
Patient population characteristics.
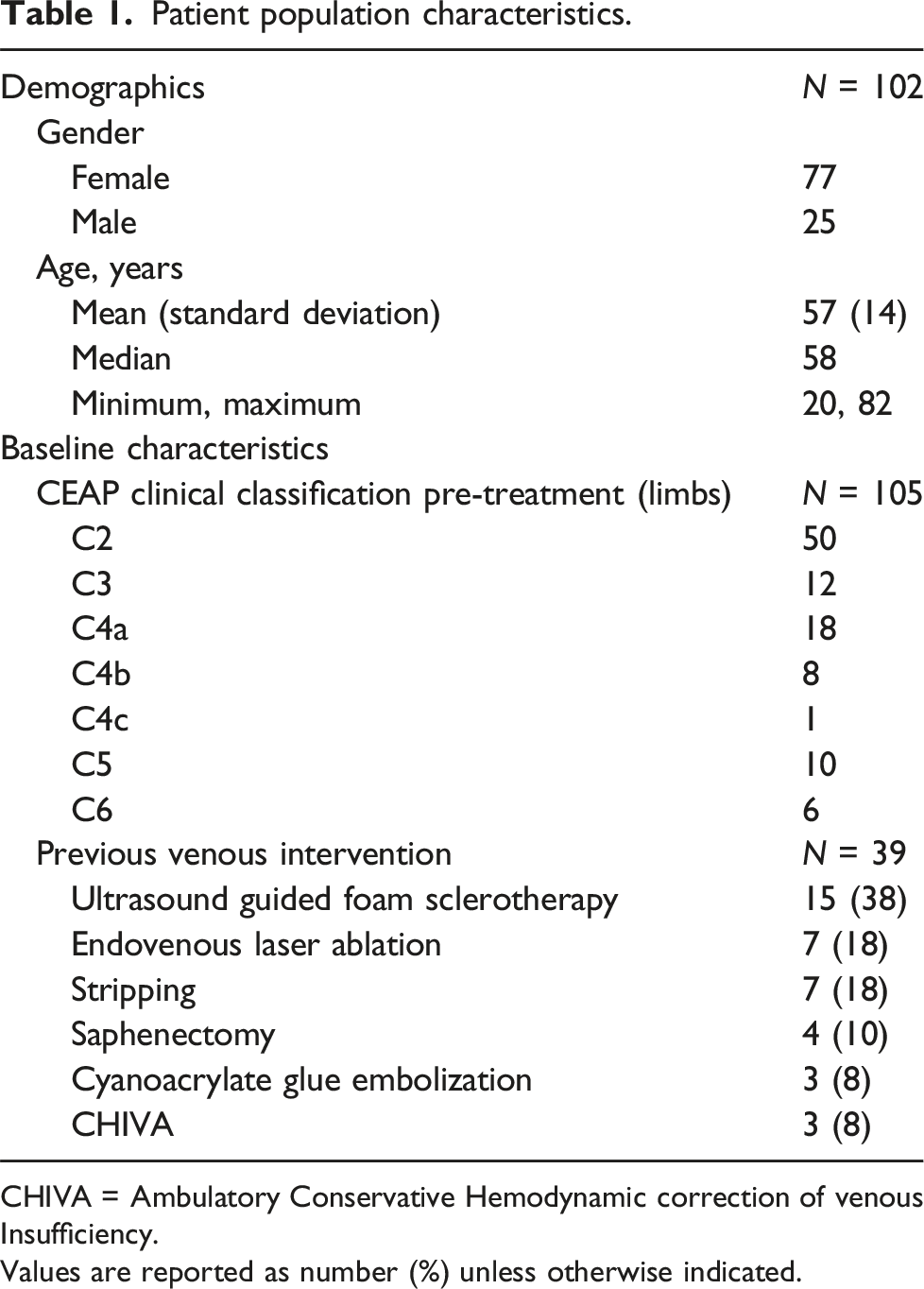
CHIVA = Ambulatory Conservative Hemodynamic correction of venous Insufficiency.
Values are reported as number (%) unless otherwise indicated.
Procedural characteristics
Pre-treatment vein diameters measured in the supine position averaged 7.2 ± 1.7 mm (mean ± SD) for the saphenous trunks (7.6 ± 1.7 mm for GSVs, 7.1 ± 1.4 mm for SSVs and 5.5 ± 1.1 mm for AASVs), 5.6 ± 1.6 mm for the perforator veins and 6.3 ± 1.4 mm for other veins (5.6 ± 1.5 mm for tributaries, 6.5 ± 1.3 mm for Giacomini veins, 10.0 ± 2.8 mm for neovascularizations). The volume of anesthetic injected before treatment was of 9.1 ± 2.9 mL for saphenous trunks, 3.5 ± 0.8 mL for perforator veins & 4.2 ± 1.6 mL for other veins. UGFS was performed as complementary treatment for tributary varicose veins at the same time for 30% of the patients. During treatment, patients reported a mean VAS pain score of 3.4 ± 1.7, and immediately after treatment, the mean VAS score was 0.4 ± 0.5.
Endpoints results
Clinical results for saphenous trunks, perforator veins, tributaries and others during follow-up visits.
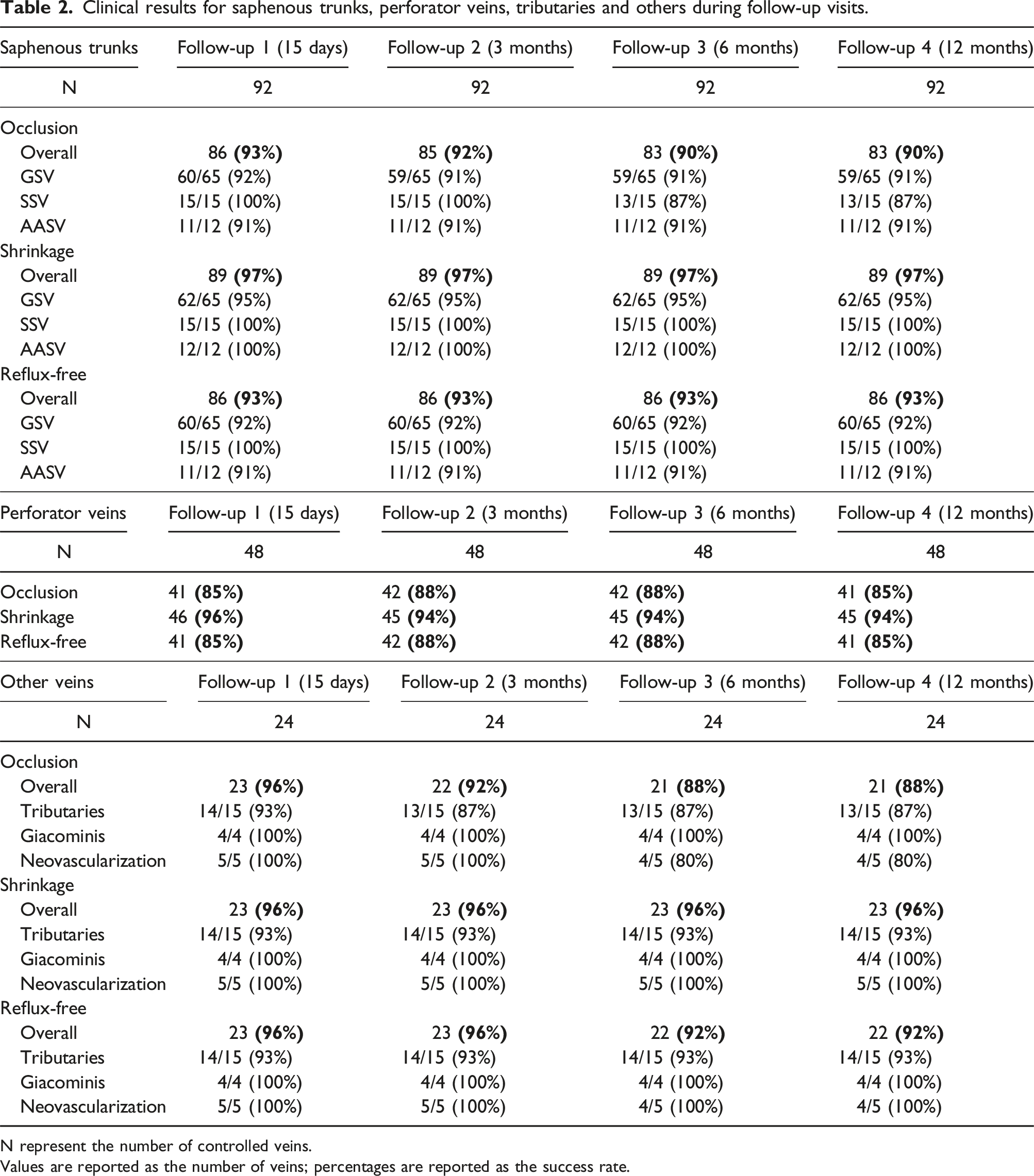
N represent the number of controlled veins.
Values are reported as the number of veins; percentages are reported as the success rate.
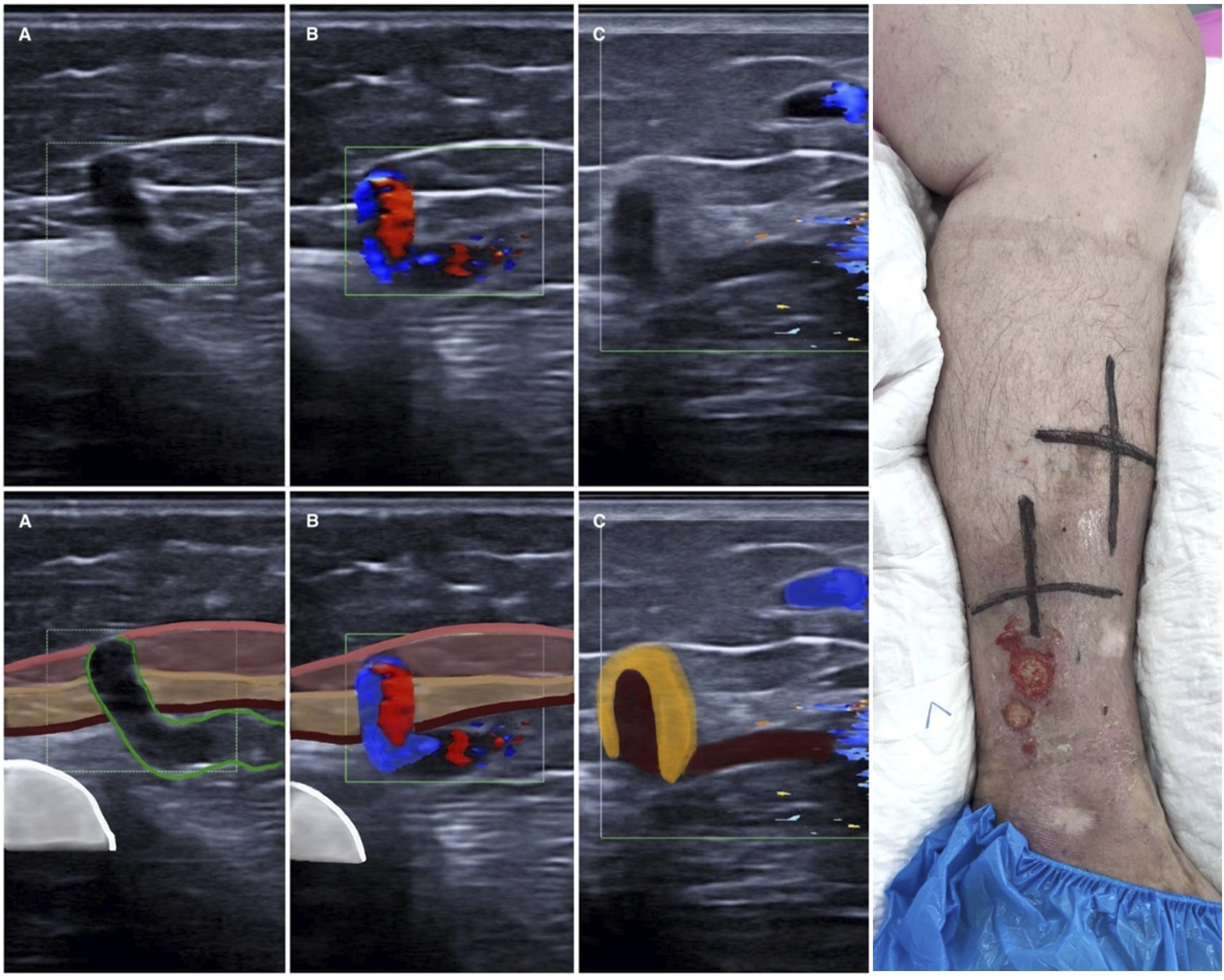
Some illustrations showing GSV (Figure 3) and perforator veins (Figure 4) under ultrasound examination before and after HIFU treatment are presented in this paper. GSV under duplex ultrasound examination before and after HIFU treatment A) GSV 8 mm target vessel before HIFU. B) 1-month post-treatment: GSV fully occluded C) 3-month post-treatment. Significant shrinkage: GSV 4 mm D) 6-month post-treatment. GSV is almost invisible: complete fibrosis process. Perforator under duplex ultrasound examination before (a) and (b) and after (c) HIFU treatment (a) Green: venous wall. (a) and (b) Pink: superficial subcutaneous fascia. Camel: deep subcutaneous fascia. Brown: muscle fascia. White: warm (b) Red and blue: reflux and flow. (c) Yellow: perivenous inflammatory tissue. Brown: occluded perforator. Blue: patent varicose veins.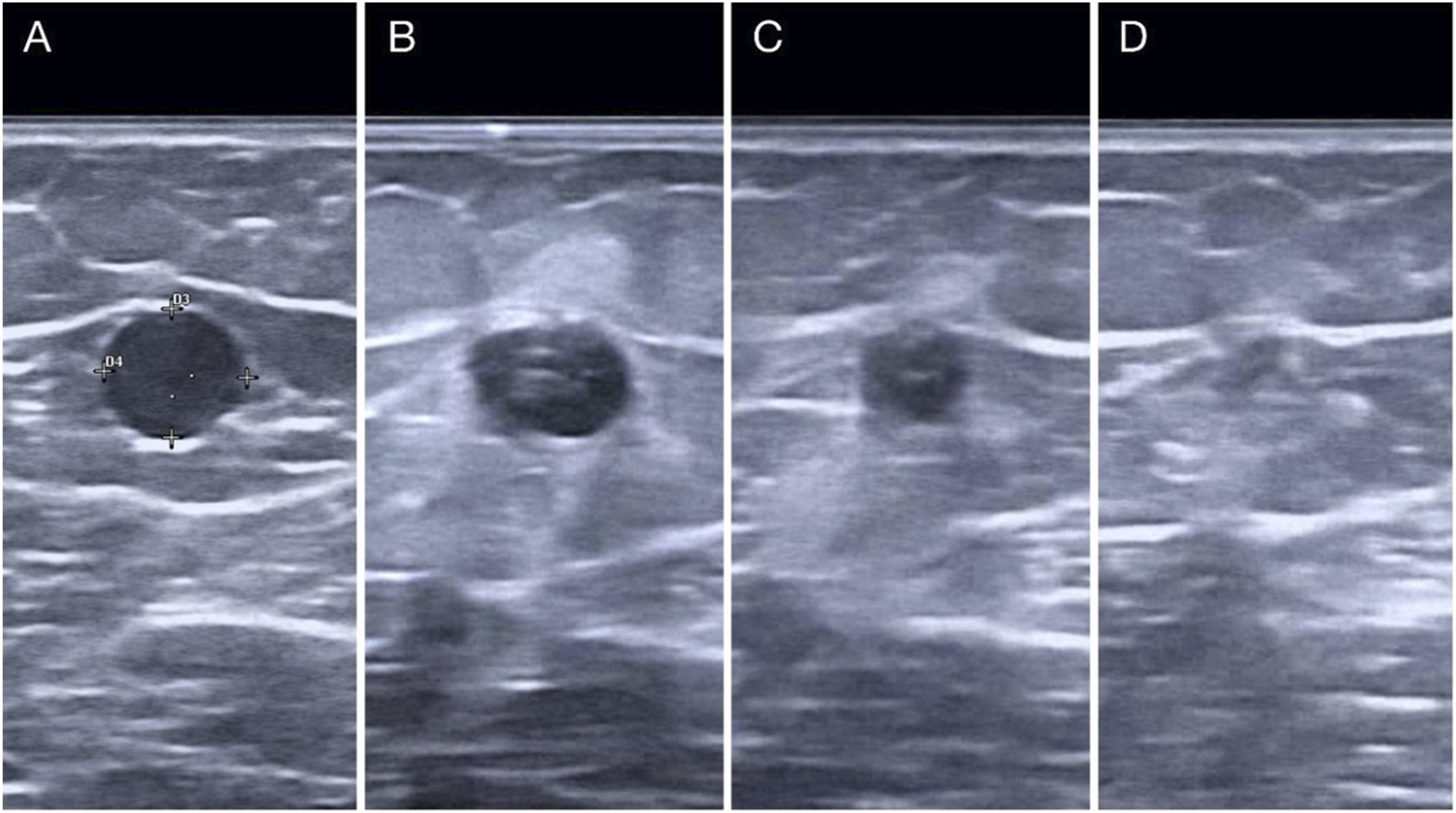
Kaplan-Meier analysis was performed to assess the survival rate free from reopening (Figure 5). This includes all patients whose initial vein occlusion was confirmed. Saphenous trunks, perforator veins, tributaries and others were assessed separately in three samples. One failure appeared at 3 months for tributaries and others, one failure at 6 months for perforators and two failures, in the same patient, at 6 months for saphenous trunks. No additional failures were reported through 12 months. Thus, we observed survival rate of 0.91 for other veins, 0.97 for saphenous trunks and 0.98 for perforator veins for. This indicates a high level of success persisting over time. Moreover, the log-rank test revealed that the three survival distributions are statistically equivalent (p = .75). Kaplan Meier analysis comparing survival rate free from reopening after ECHVA for saphenous trunks, perforator veins, other veins.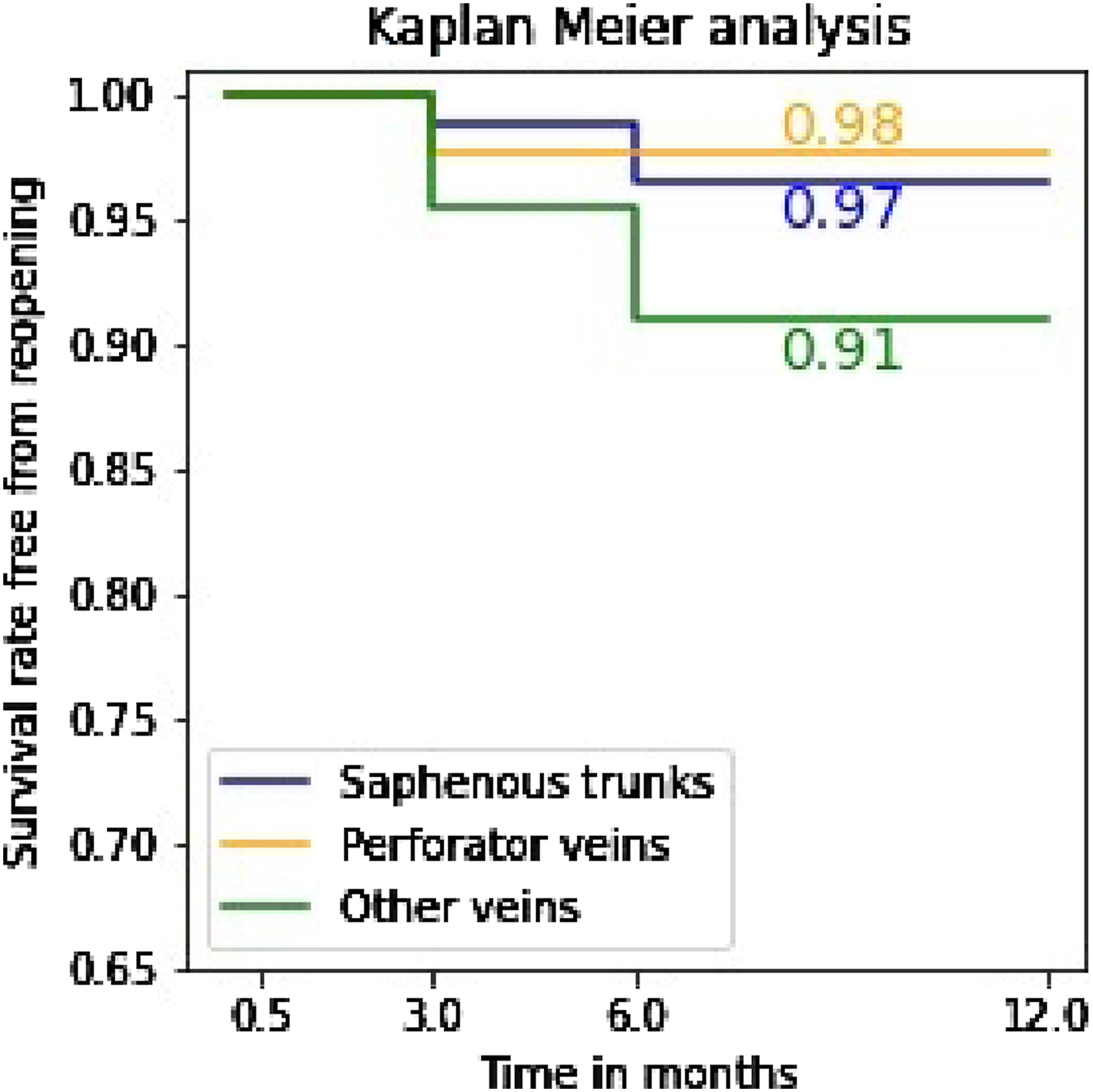
CEAP and VCSS improvement
The CEAP clinical classification at 15 days, 3 months, 6 months, and 12 months demonstrated a reduction in disease severity compared to pre-treatment (Figure 6). There is a significant clinical improvement 15 days after treatment, continuing during subsequent follow-up visits. Indeed, the number of limbs classified as ≤ C2 was as follows: 50 (pre-treatment), 67 (2 weeks), 72 (3 months), 74 (6 months) and 75 (12 months). Concomitantly, C3 class, C4 class decreased throughout the follow-up visits: 39 (pre-treatment), 22 (2 weeks), 17 (3 months), 15 (6 months) and 14 (12 months).). We also noticed a reduction in C6 class during the different follow-up visits, with 6/6 ulcers healed at 3 months of follow-up. Clinical, Etiology, Anatomy, and Pathophysiology (CEAP) clinical classification before treatment and at different follow-up visits.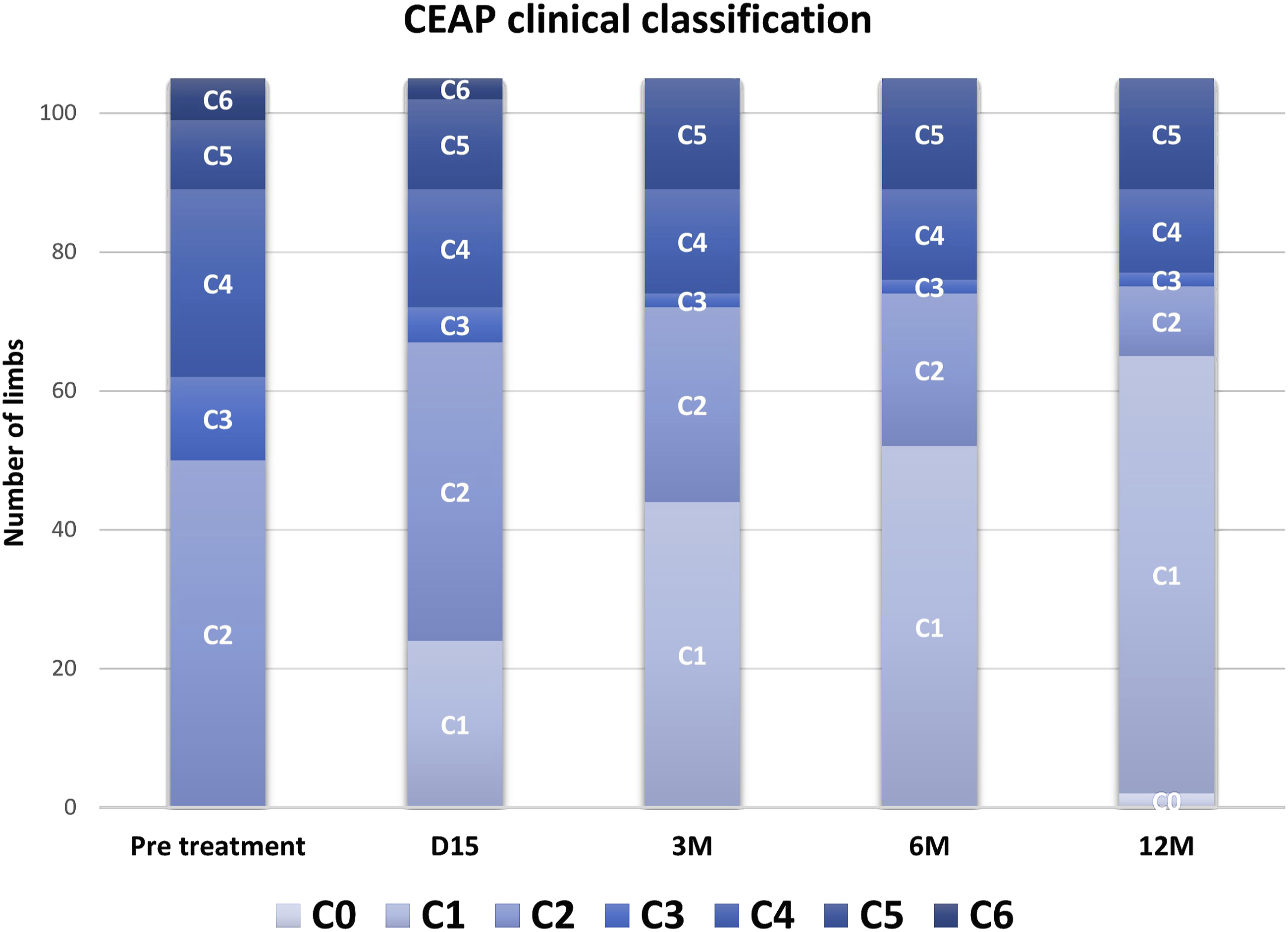
To complement the CEAP clinical classification and further assess the disease severity, the VCSS was also evaluated for the 58 patients. The mean VCSS before treatment was 8.6 ± 4.2 ranging from 2 to 20 (Figure 7). Statistically significant VCSS improvement 15 days after treatment was observed (p < .001), as well as between each subsequent follow-up visit (p < .05) until 12 months. Venous Clinical Severity Score (VCSS) during 12-month follow-up. Values represent mean ± standard error.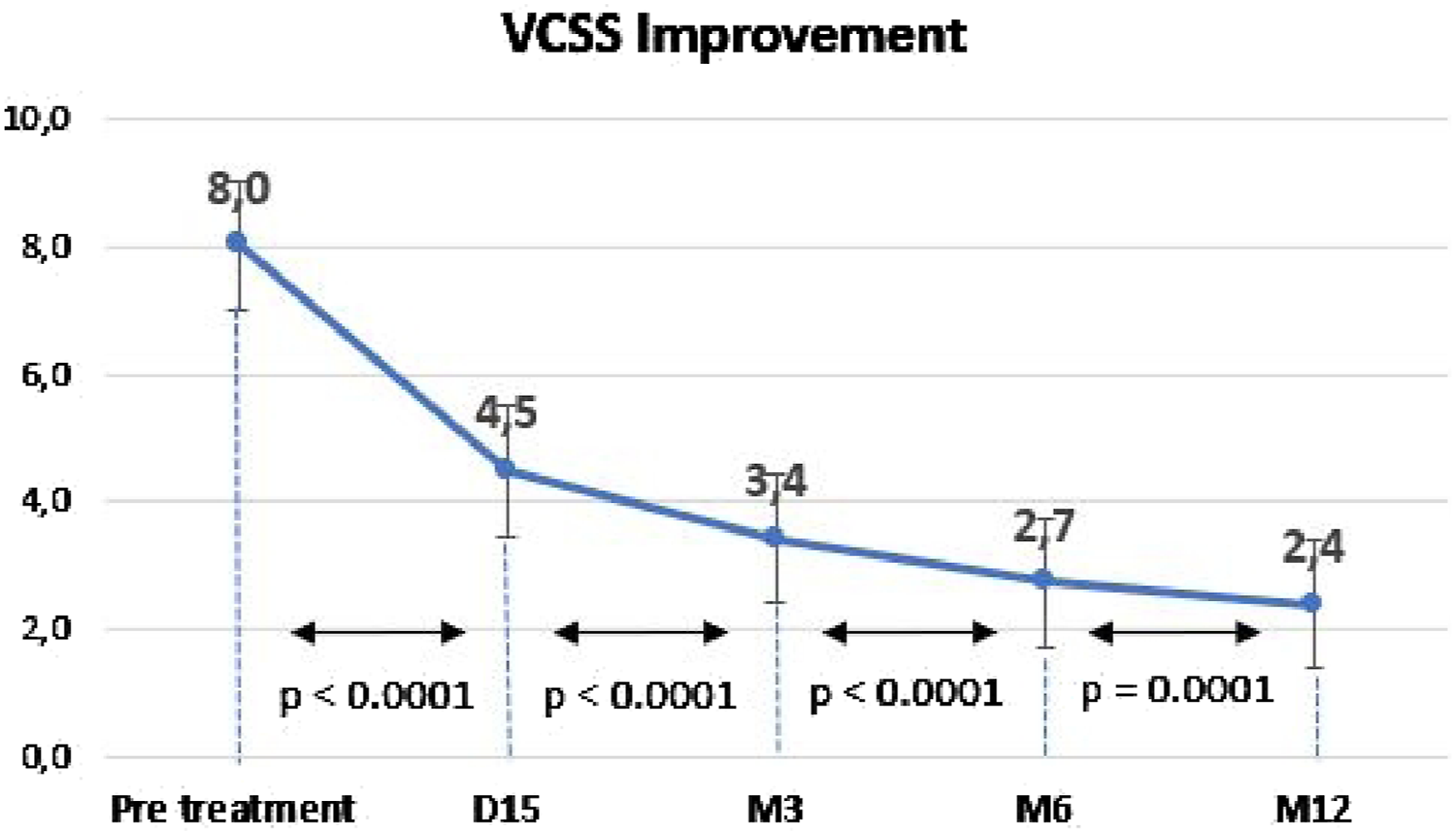
Safety analysis
No major adverse events were observed during treatment or follow-up. No deep-venous thrombosis or pulmonary embolism occurred. Among the 164 treated veins, minor adverse events included 4.4% superficial skin burns (less than 1 cm2), 2.5% dysesthesia and 0.6% superficial vein thrombosis distal to the treated area.
Discussion
Endovenous catheter-guided, thermal ablation is now the gold standard treatment for SVI, with the best occlusion and clinical rates in long-term follow-up studies. It is also the first choice of treatment recommended in the most recent international guidelines.1–3 Given this context, extracorporeal venous treatment could represent the next evolutionary step in the quest for non-invasive varicose vein treatments. HIFU therapy which allows for extracorporeal thermal ablation for varicose veins (ECHVA), combines the advantages of current thermal ablation techniques with a non-invasive approach.
Endovenous thermal ablation techniques requires tumescent anesthesia, which involves injecting much larger volumes, generally 300 mL to 500 mL. EVLA (Endovenous Laser Ablation) or RFA (Radiofrequency ablation) procedures under tumescent anesthesia are often associated with significant stress and discomfort for the patient. 16 ECHVA treatment with our low volume anesthesia protocol simplifies the procedure for both the patient and physician. On average, only 9.1 mL of volume was used to treat saphenous trunks.
The HIFU therapy is a very precise technique, effectively heating a 3 mm area with each shot. This precise targeting enables ECHVA to treat practically all types of veins, including tortuous veins, (Image 5) which present significant challenges for endovenous techniques. Elias & al. 17 reported that while other devices can attempt to treat tortuous veins, the inability to insert the fiber into the vein necessitates the use of a steerable guidewire, increasing procedural complexity.
In this paper, we present our first series of 102 consecutive patients and 164 veins treated with ECHVA, with prospectively collected data. Our retrospective analysis aimed to encompass a diverse range of patients and vein types to demonstrate the versatility and broad applicability of HIFU therapy. The primary endpoint was vein occlusion rates exclusively, evaluated as an independent outcome to ensure external validity for comparison with other thermal and non-thermal existing therapies, in contrast to the approach by Casoni et al. published in April 2024. 18
Results indicate consistent and persistent occlusion rates across all vein types. These rates are competitive with other endovenous thermal ablation techniques, showing occlusion rates from 93% to 90% (15 days to 12 months follow-up) for saphenous trunks and from 96% to 88% for other types of veins. Rasmussen & al. 19 reported failure rates up to 1-year post-treatment for EVLA, RFA, sclerotherapy and stripping at 7%, 6%, 20% and 4% respectively. Concerning perforator veins, current state of literature shows lower closure rates compared to saphenous trunks, with a technical success rate of 60% to 80%. 20 Our study presents occlusion rates of 85% for perforator veins, both at 15 days and at 12 months follow-up, showing better results than other techniques. This improvement may be influenced by the synergistic effect of sclerotherapy with HIFU, enhancing long-term occlusion rates for perforators. Kaplan-Meier distribution and log-rank test confirmed that vein occlusion persisted throughout follow-up, including all vein types. Considering all these results, ECHVA seems to be as effective as endovenous thermal ablation.
Our treatment strategy was based in an ablative approach akin to endovenous techniques. However, as we gained experience with the HIFU therapy, we realized that it allows for a more conservative strategy and could facilitated hemodynamic vein care. The ability to treat any vein at any site makes a hemodynamic approach suitable for maximizing HIFU features and benefits. Even proximal stumps, cavernomas, secondary varicose veins or new incompetent perforators are good targets for ECHVA (Figure 8). HIFU treatment: mapping of various vein types (a) Long GSV: 27 cm length. (b) Short GSV: 10 cm length. Short AASV: 7 cm length (c) 3 perforators after recurrencies. (d) Aberrant Accessory Saphenous Vein: 23 cm length.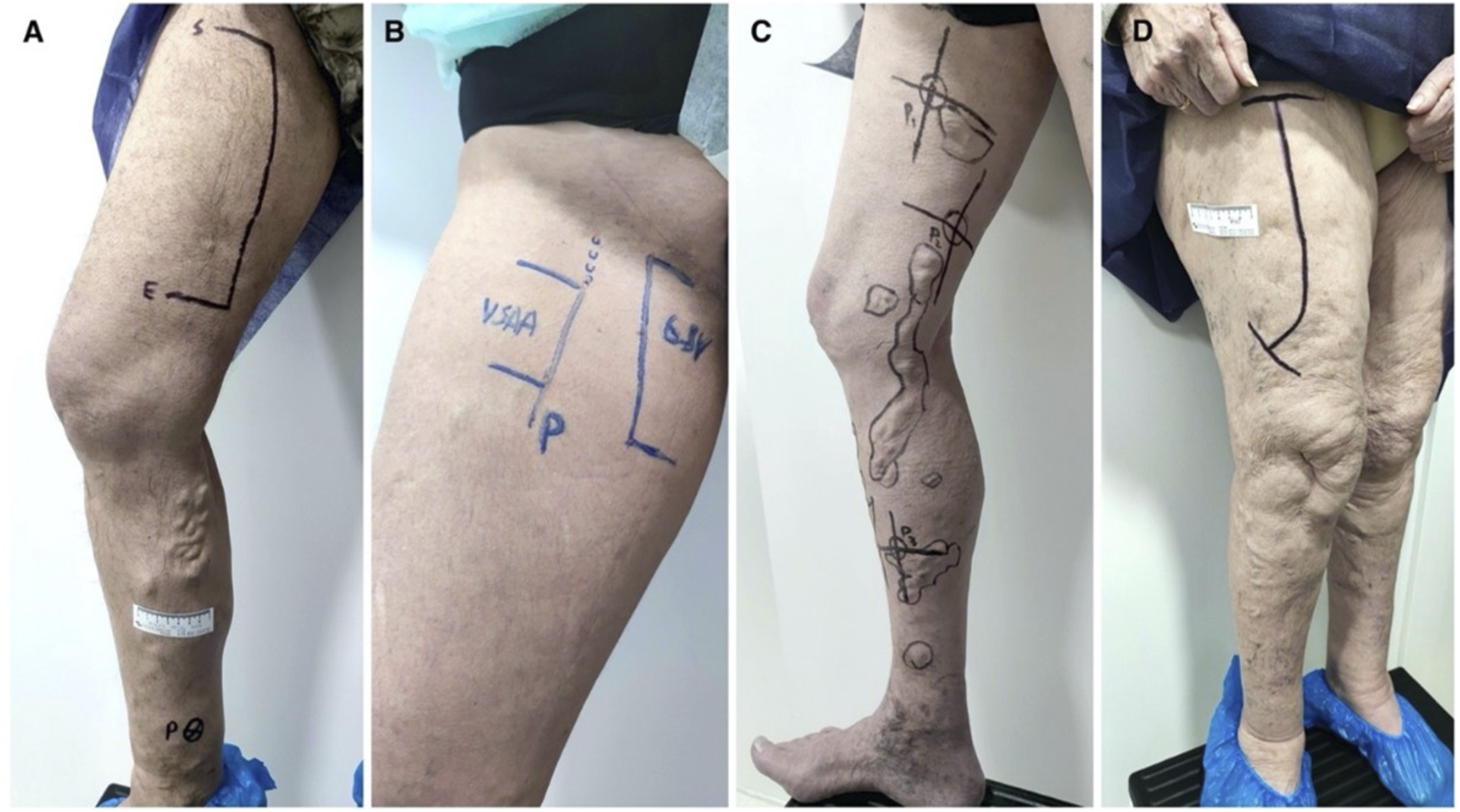
Regarding clinical findings, ECHVA demonstrated excellent improvement rates in CEAP class and VCSS variation pre- and post-treatment, which were statistically significant. The VCSS improved from a mean score of 8.6 before treatment to 2.9 after 12 months. Rajendran & al 21 reported a baseline VCSS of 9.3 in the EVLA group and 9.2 in the UAVS (Ultrasound Assisted Vascular Surgery) group, both decreasing to 1.3 after 12 months, aligning with our findings.
In terms of complications, no major events were detected in any patient during treatment or follow-up. Minor events occurred in 13 out of 102 patients, with 7 experiencing skin injuries. All of these were superficial lesions that healed without issues within 2 weeks.
ECHVA enables patients to return to normal activities immediately after treatment. Lawson & al. 22 reported that the median time for return to work was 1 day, with the maximum time being 14 days after EVLA or RFA treatments. Rasmussen & al. 19 in a study comparing the results of EVLA, RFA sclerotherapy and surgical stripping, showed that the median time to resume work was 3.6 days, 2.9 days, 2.9 days and 4.3 days, respectively.
Conclusions
In conclusion, the treatment of varicose veins with the HIFU therapy seems to be competitive with the state-of-the-art techniques and has proven to be effective and safe in our initial 102 patients’ data analysis. ECHVA has successfully integrated into our current practice and is now routinely employed in our superficial venous insufficiency treatment protocol. Nevertheless, further studies are necessary to refine patient selection criteria and to gather stronger evidence. This preliminary report opens new possibilities in varicose veins care, offering an extracorporeal approach for the first time.
Footnotes
Acknowledgements
To the Theraclion® team for their support and involvement in this project, especially to Raphael Gourgue, Jeremie Anquez, Nesrine Barnat, Marie Berne, Michel Nuta and Martin Deterre.
Author Contributions
RC.R is an HIFU pioneer practitioner and the main author and writer of this paper. The rest of authors have contributed to the paper, all of them, with data collection and writing reviewing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
