Abstract
Purpose
To evaluate longer term outcomes of the Zilver Vena Venous Stent in patients undergoing venous stenting.
Materials and Methods
Patients with iliofemoral obstructive venous disease and treated with venous stents were retrospectively enrolled in a physician-led real-world data collection effort. Results were analyzed by etiologies: post-thrombotic syndrome (PTS), non-thrombotic iliac vein lesion (NIVL), and iliocaval acute deep vein thrombosis (aDVT). Patency outcomes (primary, assisted-primary, secondary), reinterventions (in-stent and all), adverse events, and venous clinical outcome measures (VCSS, CEAP, Villalta Score) were reported using Kaplan-Meier estimates and summary statistics.
Results
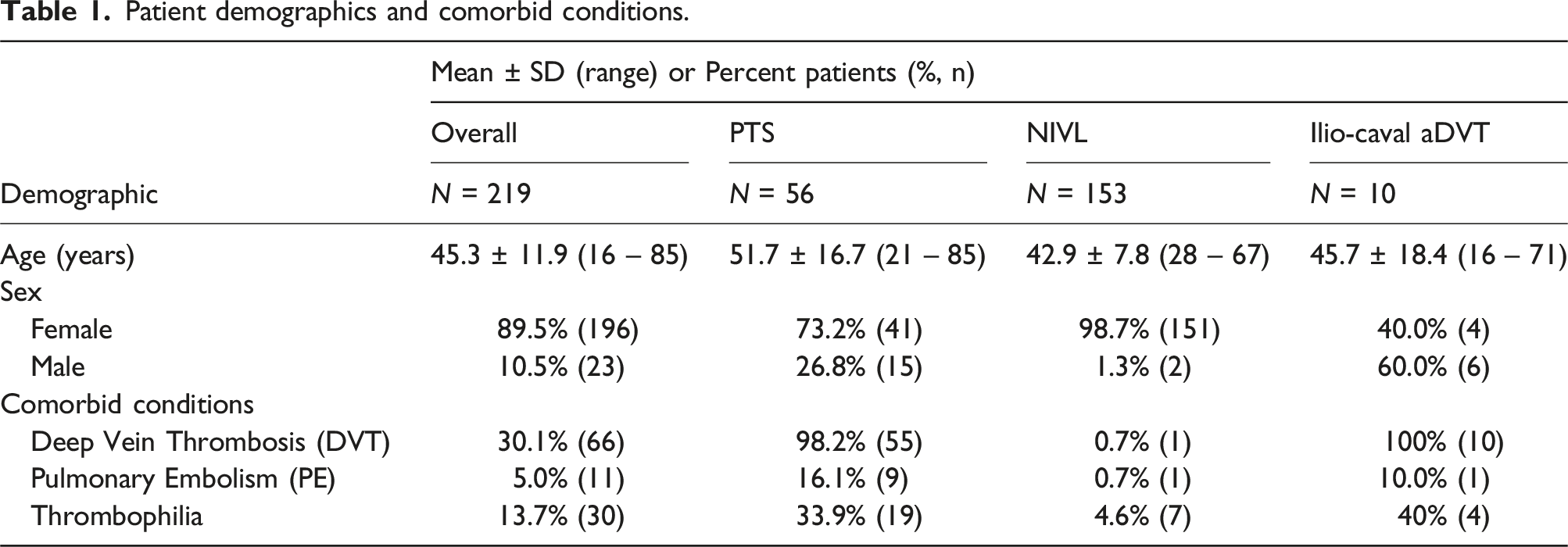
A total of 219 patients (89.5% women, mean age 45.3 ± 11.9 years) were identified: 56 PTS, 153 NIVL, and 10 aDVT patients. Devices were placed across the inguinal ligament in 80.4%, 0%, and 70.0% of patients in the PTS, NIVL, and aDVT groups, respectively. Through 1-year, Kaplan-Meier estimated primary patency rate was 96.4% for PTS, 99.3% for NIVL, and 100% for aDVT patients, respectively, and followed the same trend through 3 years. Freedom from (FF) all and within stent reinterventions estimated by Kaplan-Meier was 87.9% and 90.1% for PTS, 98.2% and 98.2% for NIVL through 3 years. No reinterventions were reported for the aDVT group. Improved venous clinical outcome measures were seen in all groups at the last follow-up visit. Stent fractures occurred in 2 PTS patients without related reintervention or clinical sequelae. No stent migrations were reported.
Conclusion
Real-world use showed high patency rates and improved venous clinical outcome measures (VCSS, Villalta, and CEAP) after venous stent placement.
Introduction
Venous disease (VD) symptomatology varies depending on duration and severity, making treatment challenging. Broadly, acute VD is defined as symptom presentation within 30 days; chronic venous disease (CVD) symptoms persist beyond 30 days. 1 CVD prevalence may be as high as 60% in adults 2 and can result in leg ulcerations when untreated.3,4
VD treatment requires identification of iliofemoral venous outflow obstruction and the presence of acute or chronic pathologies such as thrombosis. Post-thrombotic syndrome (PTS) is the most common long-term complication of deep vein thrombosis (DVT). Up to 50% of patients with iliac DVT develop moderate to severe PTS.5,6 Non-thrombotic iliac vein lesions (NIVL) result from anatomical variants 7 and external compression, most commonly May-Thurner Syndrome (MTS). NIVL often results in intrinsic vein stenosis with wall fibrosis and intraluminal webs/spurs.8,9
Newer diagnostic tools reveal a high percentage of proximal pathologies in the CVD population. 10 Conservative treatments can improve symptoms 11 and prevent secondary complications and disease progression. 12 When conservative measures are unsuccessful, alternatives, such as venous stent placement or surgical intervention, should be considered. 12 These alternative treatments have resulted in improved CVD symptoms and quality of life (QOL). 13
This real-world data collection evaluated venous stent patency, reinterventions, device integrity, and clinical outcomes to assess the performance and safety of the Zilver Vena Venous Stent for the treatment of iliofemoral obstructive VD (OVD).
Methods
Study design
This observational, retrospective analysis of prospectively collected data was conducted between 2012 and 2021 to evaluate stent patency post endovascular treatment of iliofemoral OVD, in accordance with the Declaration of Helsinki. Data elements were prospectively defined. Informed consent was not required for retrospective data review. The local ethics committee (University Hospital Jiménez Díaz Foundation) approved the data collection (EC approval number E0190-19). Three physicians from sites in Spain participated.
The primary objective was evaluation of stent patency (i.e., primary, assisted-primary, secondary) for PTS, NIVL, and iliocaval acute deep vein thrombosis (aDVT) patients through 3-year follow-up. Secondary objectives included evaluation of reinterventions and device integrity (fracture, migration) through 3-year follow-up. Clinical, Etiological, Anatomical, and Pathophysiological (CEAP) “C” classification, Venous Clinical Severity Score (VCSS), and Villalta Score changes were evaluated at last follow-up. Periprocedural complications and adverse event rates were reported throughout the entire reporting period.
Device description
Detailed device information for the Zilver Vena® Venous Self Expanding Stent (Cook Ireland Ltd, Limerick, Ireland, henceforth referred to as Zilver Vena Stent) has been published previously. 14
Patient eligibility
Consecutive patients treated for iliofemoral OVD with the Zilver Vena Stent at the index procedure were included. All patients treated from 2012 to 2021 were included, there was no pre-defined sample size.
Patient history and clinical measures were obtained prior to the procedure; grouping was based on symptoms and lesion characteristics assessed via imaging. PTS was characterized by a thickened noncompliant venous wall, with valvular reflux and often mechanical obstruction and venous hypertension. PTS patients had symptoms for over 21 days and were treated 3 to 6 months after the initial event. NIVL was characterized by extrinsic compression of the left common iliac vein (CIV) between the right common iliac artery and the spine (sacral promontory or L5). The NIVL group comprised patients with lower limb symptoms, CEAP “C” scores ≥3, VCSS scores ≥3, or venous claudication or patients with pelvic symptoms, including those with pelvic venous disorders secondary to NIVL. All NIVL patients were treated with endovascular stent placement if they had ≥70% stenosis, identified using transabdominal ultrasound and confirmed with multiplanar venography. aDVT was characterized with symptom onset within 2 weeks of presentation; patients were treated with thrombolysis 1-21 days from the initial event.
Implant procedure
Stents were implanted in accordance with the Instructions for Use. Procedures were performed under general anesthesia with patients in the supine position with the left leg placed in either the “frog leg” position (femoral or common femoral vein (CFV) access) or prone position (popliteal vein (PV) access). Access was achieved with ultrasound guidance using a 10 cm, 9 French sheath. Non-fractionated heparin was given to prevent coagulation.
For PTS patients, a stiff 0.035-inch (0.889 mm × 260 cm) guidewire was advanced with a multipurpose A curve angiographic 5 French (1.67 mm × 65 cm) catheter and a chronic total occlusion catheter with a long sheath available for extra support as needed. PTS patients required use of postero-anterior, lateral, and oblique venography projections to ensure proper positioning after crossing the lesion. Progressive predilatation with standard percutaneous transluminal angioplasty (PTA) was performed followed by postdilatation with a high-pressure balloon.
For NIVL patients, a stiff 0.035-inch (0.889 mm × 260 cm) guidewire was advanced with a C2 (Cobra) 5 French (1.67 mm × 65 cm) catheter into the contralateral external iliac vein (EIV). With the C2 catheter positioned within the EIV, angiography was performed with subtraction using 15 mL of iodine contrast solution injected at 15 mL/second by the sheath and manually by the catheter to obtain a bilateral image. Signs of NIVL, such as bullseye, compression (defined as a visualization of left common iliac vein compression during the venogram as a small central ellipse with no contrast within a dilated vein with contrast around the ellipse), collateral redistribution, or contralateral hypogastric vein fast filling (before the ipsilateral CIV) were assessed using multiplanar venography and transabdominal ultrasound. Predilatation, using a high-pressure balloon, was performed with the ultrasound adjusted to the exact position of the balloon with no more than 1.5 to 2 cm above the maximum compression point. Predilatation of the CIV and EIV was performed, as necessary.
To decrease thrombus burden and potential valvular damage with residual clot in patients with aDVT, pharmacomechanical (Angiojet™, Boston Scientific), mechanical thrombectomy (Aspirex™, Becton Dickinson), or catheter-directed thrombolysis was performed. Iliofemoral stent placement was then performed in the same manner used for PTS patients.
Stent deployment was performed using fluoroscopy and transabdominal ultrasound. The stent was partially deployed (1 cm) followed by a wait time of 60 seconds. Full stent deployment was completed using a slow pullback maneuver. For PTS lesions, the stent coverage was from healthy vessel to healthy vessel, typically requiring placement of multiple stents to avoid overlap across the inguinal ligament. For NIVL, measurements were done with transabdominal ultrasound in the distal CIV or proximal EIV, ensuring 1 mm oversizing, with the balloon adjusted to the diameter of the healthy vein. Full stent postdilatation was performed after all deployments with a high-pressure balloon. A final venogram and transabdominal ultrasound ensured correct stent position and absence of bullseye or collateral circulation.
For PTS and aDVT patients, systemic anticoagulation was given based on procedural activated clotting time (250–300 seconds) and a full dose of low molecular weight heparin (LMWH) was given after the procedure. Discharge anticoagulation for PTS and aDVT patients was most commonly low molecular weight heparin and followed the 2021 European Society for Vascular Surgery (ESVS) Clinical Practice Guidelines on the Management of Venous Thrombosis. 15 For NIVL patients, systemic anticoagulation was given based on activated clotting time (250–300 seconds) during the procedure. A prophylactic dose of LMWH was given 4 hours after the procedure and 12 months of antiplatelet medication (aspirin 100 mg or clopidogrel 75 mg) was prescribed.
Patient follow-up
Patients were evaluated post-procedure, and at 1 week, 1 month, 6 months, 1 year, and annually thereafter. Echo-doppler ultrasound (to evaluate stent patency) and physical examinations were obtained at each follow-up visit. Clinical outcome measures were recorded at the last follow-up visit (e.g., a patient with follow-up through 3 years would have clinical outcome measure data available at baseline and 3 years). Abdominal X-ray in 2 projections was performed at 1 month and annually thereafter to evaluate stent integrity.
Study definitions
Primary patency was defined as uninterrupted venous patency following initial stent deployment. Assisted-primary patency was defined as patency after a procedure or intervention was performed on the target lesion to prevent occlusion in cases of symptomatic restenosis. Finally, secondary patency was defined as patency of the target lesion after treatment of reocclusion.
Data analysis
Data were collected and recorded in a pseudo-anonymized database. Statistical Analysis Software (SAS) for Windows (release 9.4 or higher; SAS Institute, Cary, NC) was used to perform statistical analyses. Continuous variables were described as means and standard deviations. Categorical variables were described as counts and percentages. Structured follow-up periods were not defined for data entry. Analysis of primary outcomes used complete case analysis. Patients were at various follow-up stages. Mean patient follow-up was 1231 days post-procedure; therefore, Kaplan-Meier FF loss of primary, assisted-primary, and secondary patency, as well as FF all-cause and within-stent reinterventions are reported through 1, 2, and 3-year follow-up. FF stent fracture was reported through 3-year follow-up. Kaplan-Meier estimates were used to reduce the risk of bias as patients with missing data were censored.
Results
A total of 241 patients underwent stent placement. Two patients did not have a stent placed at the index procedure and were excluded from data analysis. Additionally, 20 patients who had the stent placed for indications other than iliofemoral OVD were excluded. A total of 219 patients were included (Figure 1): 69.9% NIVL (153/219), 25.6% PTS (56/219), and 4.6% aDVT (10/219). Mean patient age was 45.3 ± 11.9 years; 89.5% of patients treated were female. Common comorbidities are listed in Table 1. Patient inclusion/exclusion flow chart. Patient demographics and comorbid conditions.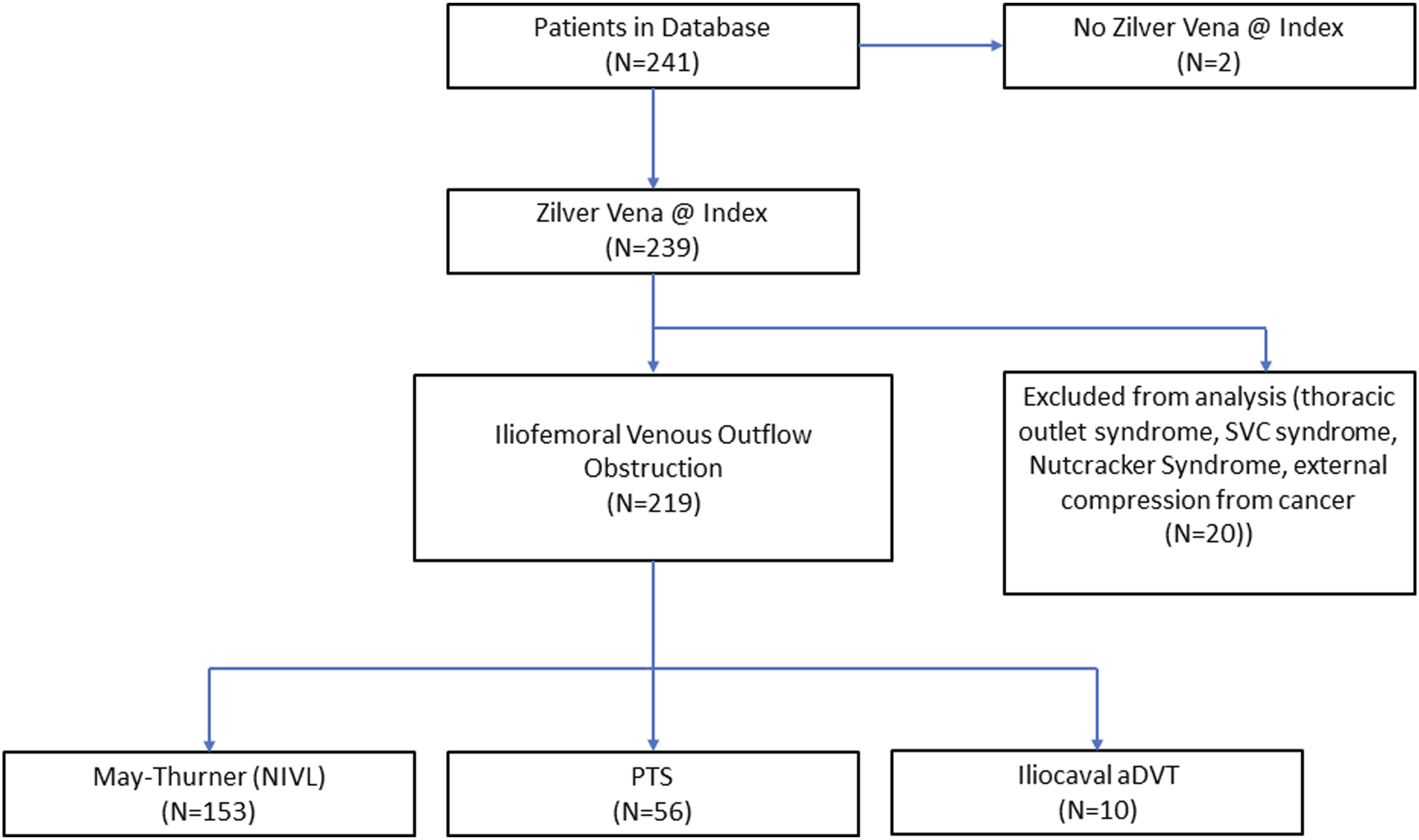
Procedural information
Baseline lesion characteristics and procedural details.
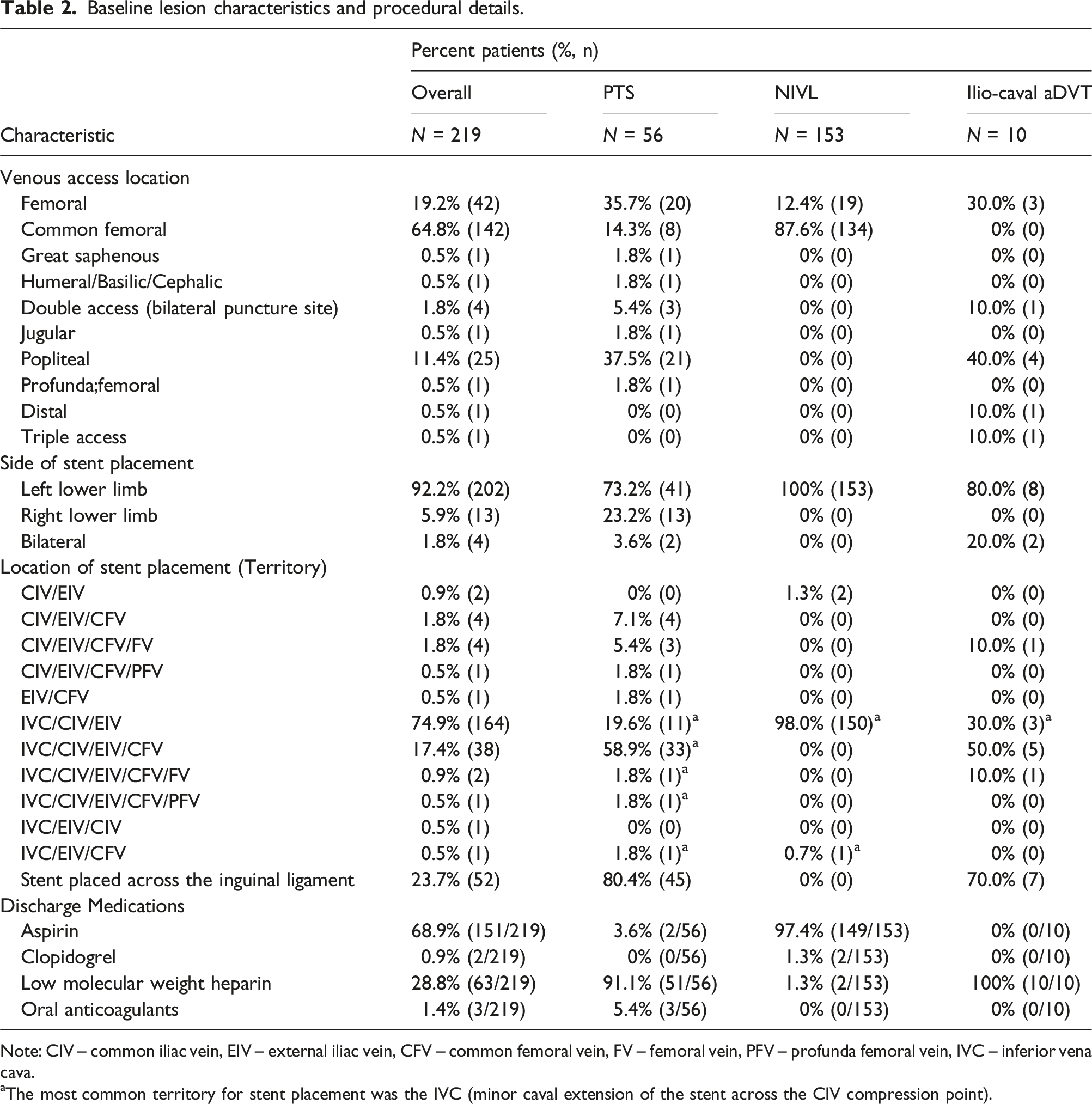
Note: CIV – common iliac vein, EIV – external iliac vein, CFV – common femoral vein, FV – femoral vein, PFV – profunda femoral vein, IVC – inferior vena cava.
aThe most common territory for stent placement was the IVC (minor caval extension of the stent across the CIV compression point).
Clinical outcome measures.
Results through last follow-up
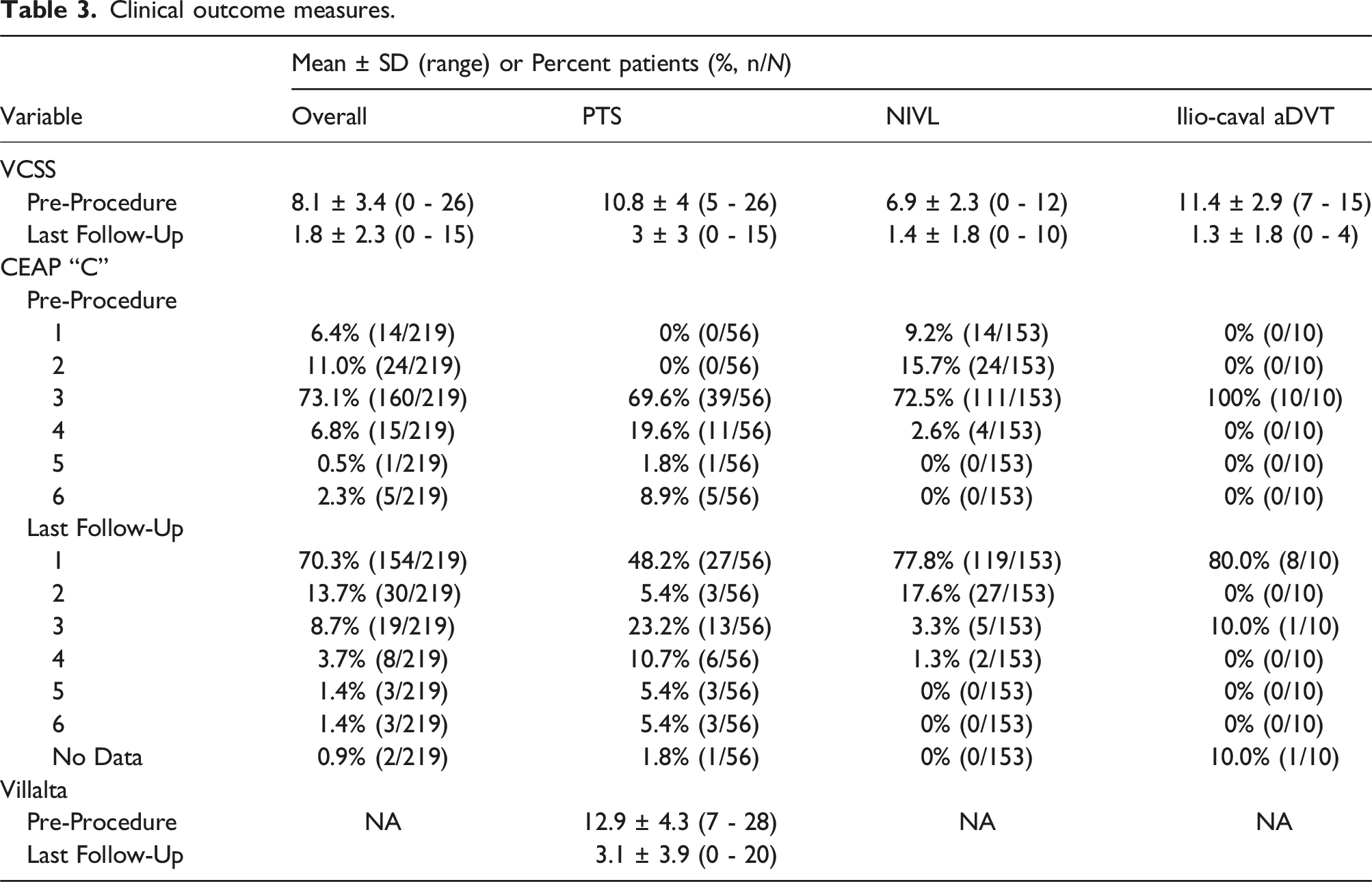
Mean follow-up was 1230.9 ± 672.4 days (range: 1–3315 days) for all patients; 1448.6 ± 748.8 days for PTS (range: 1–2707 days), 1169.3 ± 607 days for NIVL (range: 30–2738 days), and 955.2 ± 926.6 days for aDVT (range: 61–3315 days). Loss-to-follow-up was noted in 16% (35/219) of patients.
Clinical outcome measures from the last follow-up visit are shown in Figure 2 and Table 3. Mean baseline VCSS for the overall cohort was 8.1 ± 3.4, which improved to 1.8 ± 2.3 at the last follow-up. Most patients’ CEAP classification improved from C3 (73.1% [160/219]) at baseline to C1 70.3% (154/219) at last follow-up. Improvement in Villalta scores were also observed in PTS patients (mean Villalta score at baseline: 12.9 [N = 55], vs mean Villalta score at last follow-up: 3.1 [N = 54]). Overall, patients demonstrated clinical improvement after stent placement. Venous clinical outcome measures, including Venous Clinical Severity Score (VCSS, a), Villalta scores (b) and CEAP (c-f), were all improved following stent placement.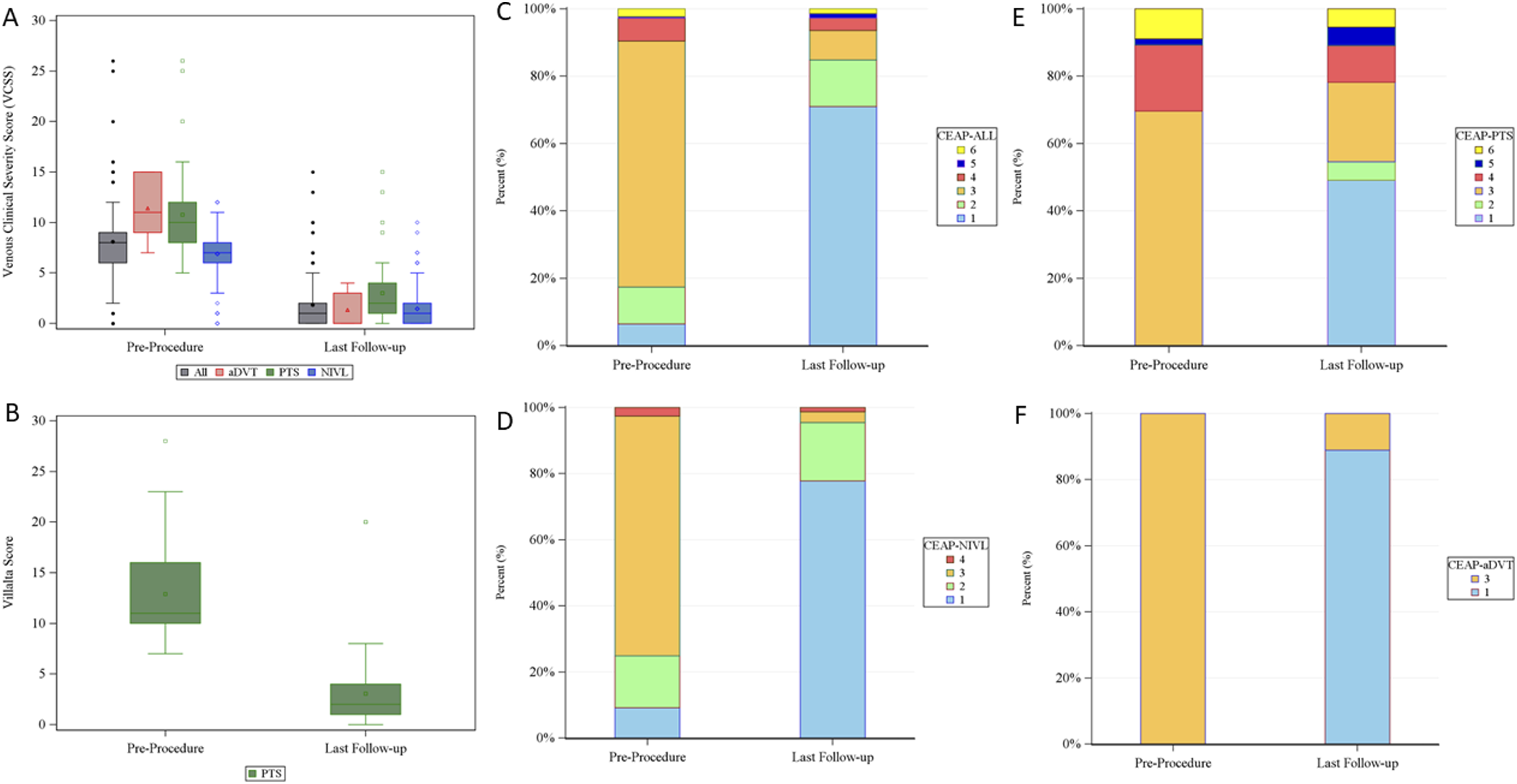
Index periprocedural complications and adverse events (entire reporting period).
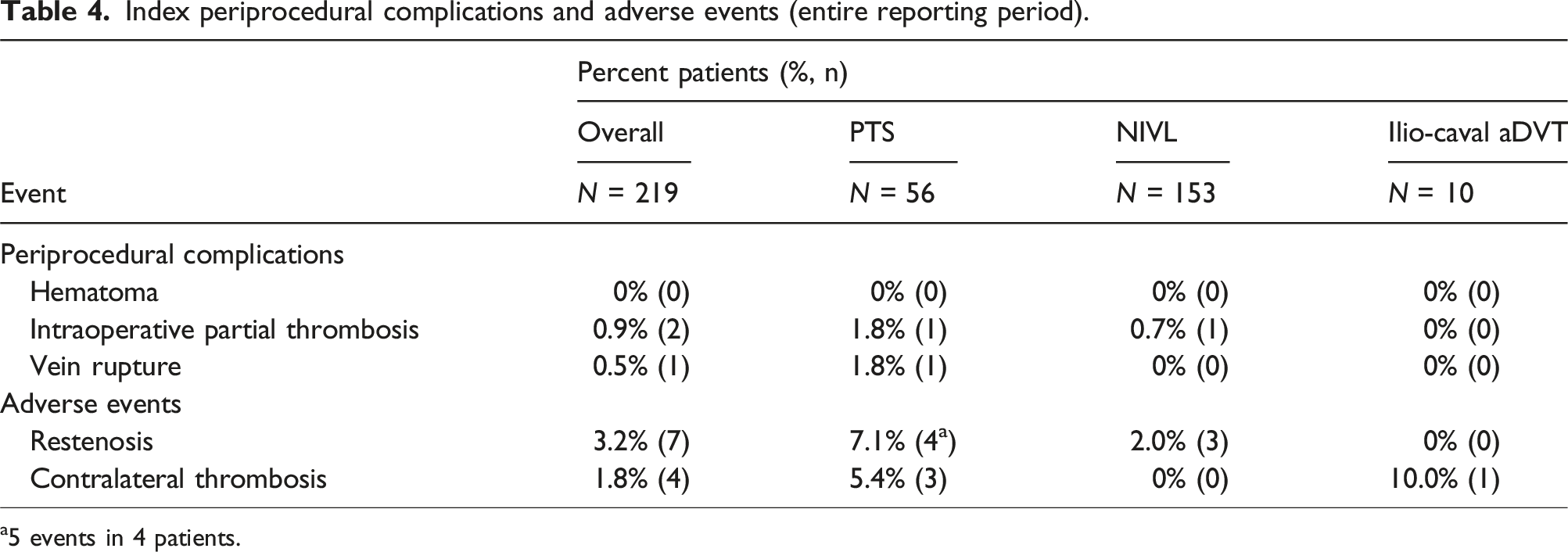
a5 events in 4 patients.
FF loss of primary patency was 98.6% ± 0.8% (1 year) and 96.1% ± 1.6% (3 years). Among PTS patients, FF loss of primary patency was 96.4% ± 2.5% (1 year) and 90.1% ± 4.5% (3 years); failed primary patency occurred in 5 patients requiring reinterventions. Loss of patency for one patient was related to non-compliance with anticoagulation at discharge, requiring fibrinolysis treatment 2 days post-procedure. Another patient experienced intraoperative partial thrombosis distally requiring an additional stent placement 22 days post-procedure. The third patient experienced asymptomatic restenosis resulting in PTA treatment 440 days post-procedure. The fourth patient experienced within-stent symptomatic partial thrombosis requiring fibrinolysis treatment 739 days post-procedure. The final patient required proximal stent extension (additional Zilver Vena Stent placement) 918 days post-procedure due to incomplete coverage of the lesion during the index procedure.
In the NIVL group, FF loss of primary patency was 99.3% ± 0.7% (1 year) and 98.2% ± 1.4% (3 years). FF loss of assisted-primary and secondary patency was 100% in the NIVL group through 3 years. Symptomatic restenosis requiring reintervention occurred in 2 patients, with PTA performed in one patient 107 days post-procedure and additional stent placement performed in the other patient 1011 days post-procedure.
FF loss of primary, assisted-primary, and secondary patency was 100% through 3 years in aDVT patients. Kaplan-Meier analysis of primary and assisted primary patency in PTS and NIVL patients through 3 years are shown in Figure 3. Kaplan-Meier primary (a) and primary-assisted (b) patency through 3-year follow-up. Primary and assisted-primary patency in the iliocaval aDVT group (not shown) was maintained in 100% of patients through 3-year follow-up. Secondary patency (not included above) was maintained in all patients (100%) through 3-year follow-up.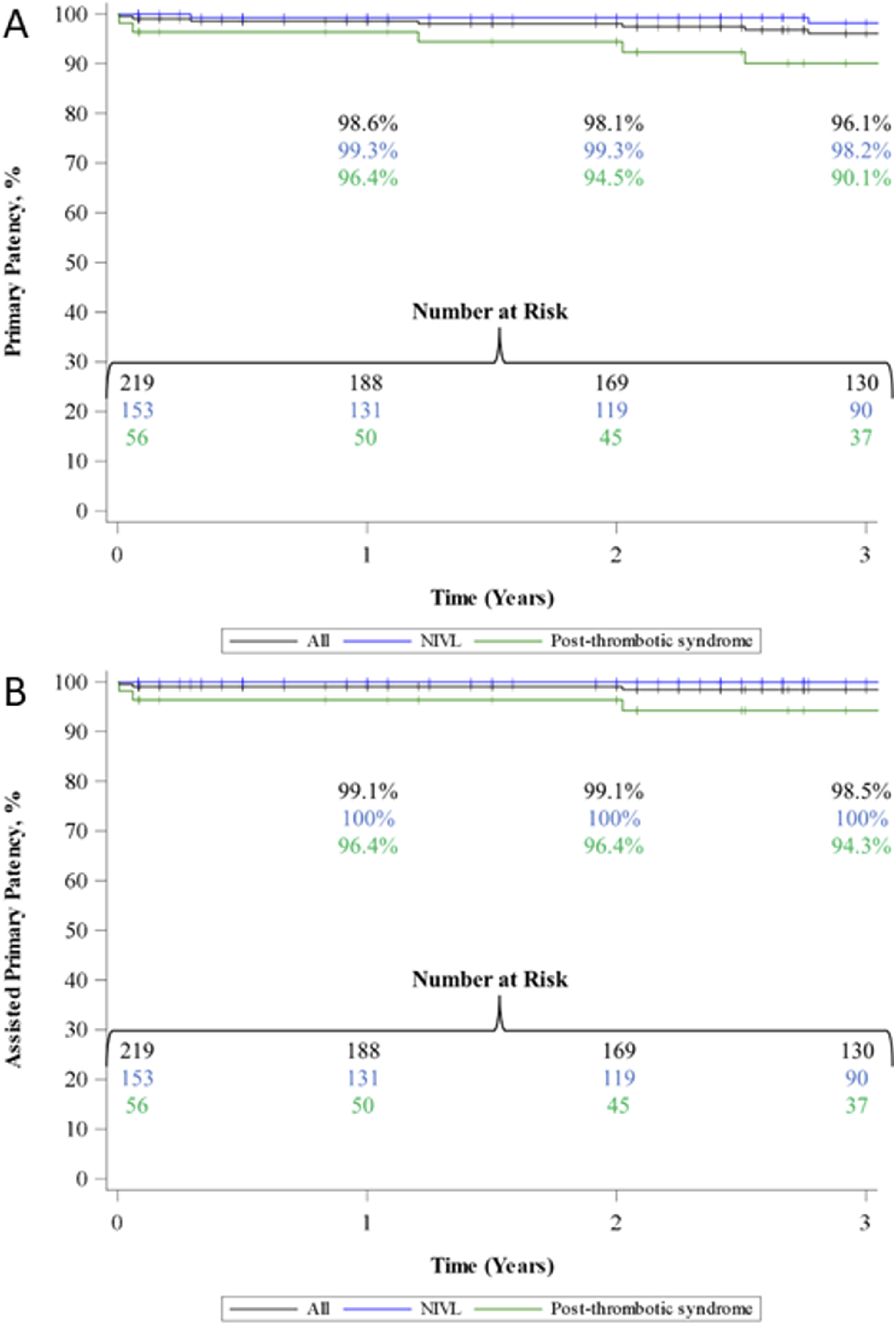
Overall FF all reinterventions was 98.6% ± 0.8% (1 year) and 95.5% ± 1.8% (3 years). FF reinterventions within the stent was 98.6% ± 0.8% (1 year) and 96.1% ± 1.6% (3 years). The PTS group had a higher rate of reinterventions compared to the NIVL and aDVT groups (Figure 4). Kaplan-Meier freedom from all reinterventions (a) and reinterventions within the stent (b) through 3-year follow-up. Freedom from reintervention was maintained in 100% of patients in the ilio-caval aDVT group (not shown). Note: 2 patients in the PTS group had all-cause reinterventions on the same day (day 918);3 patients had reinterventions beyond the 3-year period of the Kaplan-Meier analysis (PTS group, 1 all-cause reintervention day 2707; NIVL group, 1 in-stent reintervention day 1510; iliocaval aDVT group, 1 all-cause reintervention day 1232).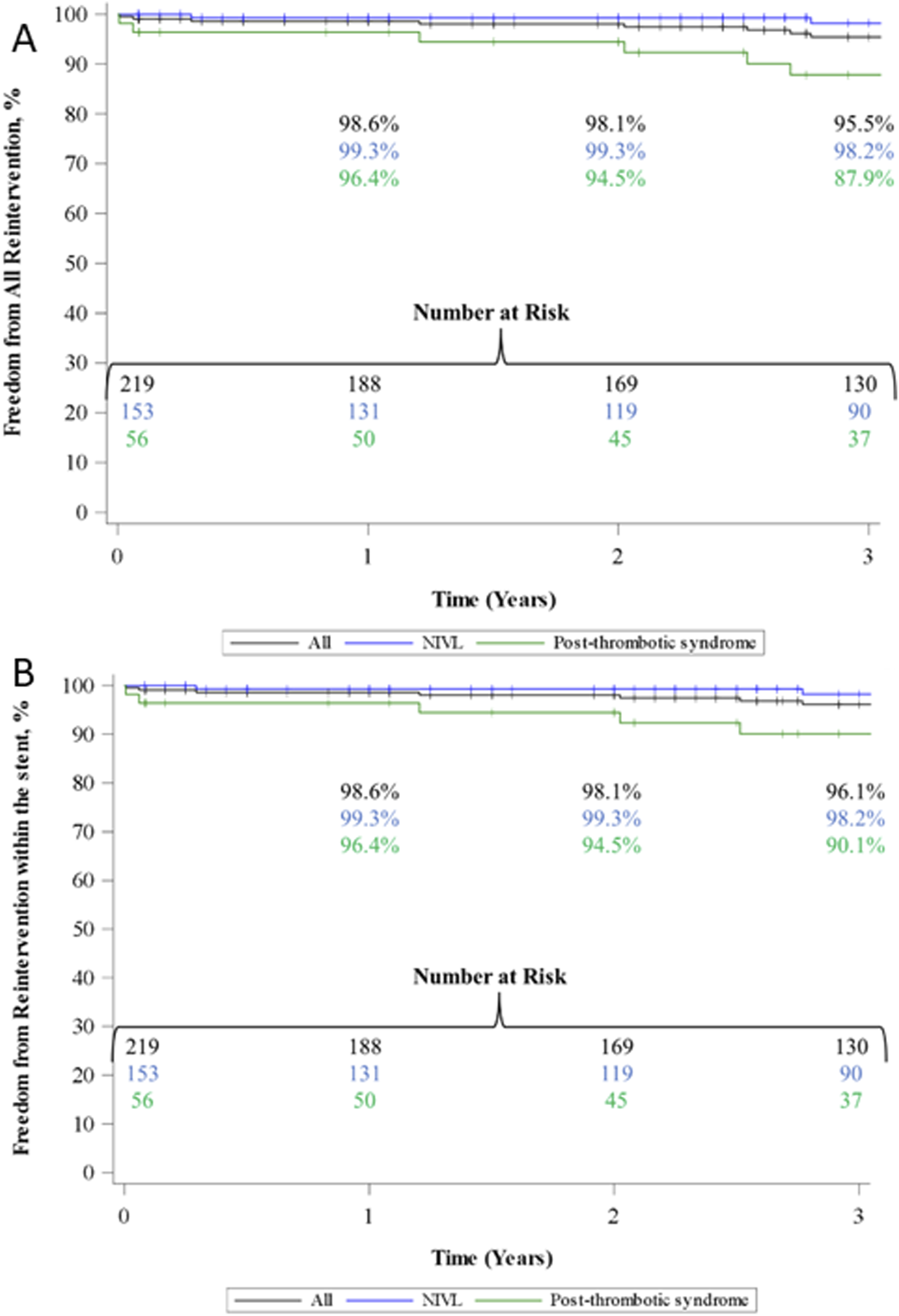
A total of 11 (5.0%, 11/219) patients required an all-cause reintervention, including reinterventions outside of the stented region or on the contralateral side, throughout the entire reporting period (7 PTS, 3 NIVL, 1 aDVT). Eight reinterventions occurred within 3 years (Figure 4(a)), and 3 reinterventions (1 PTS, 1 NIVL, and 1 aDVT) occurred beyond 3 years. Of the all-cause reinterventions, 8 patients (5 PTS, 3 NIVL) required reinterventions within the stent (3.7%, 8/219, Figure 4(b)).
FF stent fracture was 98.8% ± 0.8% through 3-year follow-up. Two patients in the PTS group experienced fractures, reported at 617 and 825 days (3.6%, 2/56). Importantly, no symptoms were associated with these fractures, and no reinterventions were required. No migrations were identified throughout the data collection.
Discussion
While no global standard of care has been established for venous stent placement, these data demonstrate results from real-world use for clinical improvement, patency, and durability through 3 years. Most patients (69.9%) presented with NIVL obstructions associated with MTS etiology or iliac vein compression, while the remaining patients had PTS (25.6%) or aDVT (4.6%). The aDVT group presented with more extensive lesions than the typical focal lesions associated with acute thrombosis within iliac veins, as reflected by stent placement across the inguinal ligament for most aDVT patients (70%) to protect the inflow.
Consistent with referral patterns, 89.5% of the patients were female; this may limit generalizability. Patients were referred for treatment from outpatient offices and specialties, including gynecologists treating symptomatic women with pelvic venous disorders (PeVD). PeVD may cause a variety of symptoms and signs emanating from the veins of the pelvis and their drainage pathways. 16 PeVD can have underlying anatomic lesions consistent with NIVL and present with venous claudication or chronic pelvic pain without typical venous symptoms in the lower limbs. In this data collection, patients treated for less severe peripheral disease were treated for pelvic symptomatology.
Improvement in VCSS scores after stent placement is consistent with previous reports through 1,14,17–19 2, 19 and 3 years. 20 This change in VCSS scores reflects improvement from moderate/severe disease to absent disease. 21 Improvement in Villalta scores aligns with observations from other studies following stent placement.18,22–24 Finally, improvement in CEAP “C” classification, aligns with other reports through 3 20 and 6 years, 25 with results corresponding to improvement in patients’ clinical condition.
Consistent with previous reports, overall high patency rates were observed through both 1 year7,14,18,20,23,24,26–29 and 3 years. 29 Meta-analyses and clinical studies have shown higher patency rates associated with treatment of NIVL compared to thrombotic etiologies.7,22,24,30 This trend was observed in the current study, with lower primary and assisted-primary patency rates in the PTS group compared to the NIVL group through 3 years. The high patency rates observed in the PTS group may be due to extending the stent below the inguinal ligament into a healthy vein segment.
FF all-cause and within stent reintervention rates tended to be higher than rates reported elsewhere through both 1-14,23,24,26 and 3-22,30 years. This may be related to the high proportion of patients with non-thrombotic indications. FF all-cause and within-stent reinterventions through 3 years was higher in the NIVL group compared to the PTS group. Both patency and FF reintervention, (all-cause and within stent) were 100% for aDVT group, potentially influenced by the small number of patients in the group and the even smaller number of patients who completed 3-year follow-up. Overall, the lower FF reintervention aligns with the lower primary and assisted-primary patency rates in the PTS group compared to the NIVL and aDVT groups.
Adverse events were low, with intraoperative partial thrombosis and vein rupture being the only perioperative events reported. One case of contralateral thrombosis was attributed to contralateral jailing. Two reports of stent fractures for unknown causes were identified at days 617 and 825 in stents placed across the inguinal ligament. In the PTS group, over 80% of stents were placed across the inguinal ligament. Information regarding patency and safety outcomes of stents placed across the inguinal ligament is contradictory. 31 Despite insufficient evidence for or against the practice, current expert opinion supports stent placement in this location. 31
This data collection provides insights from a real-world setting with limitations for both study design and patient population. All patients were treated for iliofemoral OVD with the Zilver Vena Stent with no exclusion criteria applied or restrictions on reinterventions. Time periods followed standard-of-care for clinical outcomes measures; thus, measures only reflect the baseline and last follow-up visits. QOL assessments, such as CIVIQ-20 32 and Veines-QOL, 33 have been useful to assess patient improvement; however, these assessments were not yet in widespread use in Spain at the conception of this data collection. Still, dedicated venous stents have been shown to improve both QOL and VCSS compared to conservative treatment strategies. 34 As the data collection reports device use and outcomes from centers in Spain, data may not represent global standard-of-care.
Conclusions
This data collection provides real-world data supporting the safety and performance of the Zilver Vena Stent for the treatment of iliofemoral OVD. Patency and FF reintervention were higher in the aDVT and NIVL groups compared to the PTS group. Adverse event rates were low. Improvements in clinical measures (VCSS, Villalta, and CEAP “C” scores) demonstrated clinical improvement and a decrease in the symptoms of VD after stent placement for all patient groups.
Footnotes
Author notes
Presented at Leipzig Interventional Course (LINC) 2023, June 6-9, 2023.
Acknowledgements
The authors thank Sara Sherman, M.S., and Nicholas Dey, PhD, full-time employees of Cook Research Incorporated, a Cook Group Company, for their contributions to summarizing and editing the information presented in this manuscript.
Author contributions
MRO was responsible for the conception and design of the study. RMO, EPM, EMB and OTT performed data collection. ES performed data analysis and interpretation, and stastical analysis. AG performed data analysis and interpretation and writing of the manuscript. MRO, EPM, EMB, OTT, ES, and AG provided critical revision, provided approval of the manuscript, and agree to be accountable for the manuscript.
Declarations of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MRO is a consultant to Cook Medical, Medtronic, BD, and Balt. EMB is a consultant to Balt and Medtronic, ES and AG are full-time employees of Cook Research Incorporated. OTT and EPM have no competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Guarantor
MRO.
