Abstract
Background
The term Anterior Accessory of the Great Saphenous Vein suggest this is a branch tributary vein despite this vessel’s anatomic features of a truncal vein. A multisocietal group suggested to designate this the Anterior Saphenous Vein (ASV). This study was aimed to evaluate its ultrasound anatomy in normal and varicose limbs.
Methods
The clinical anatomy of the ASV was evaluated by narrative review of the literature. Additionally, the course of the ASV was evaluated in 62 limbs with no evidence of venous disease and 62 limbs with varicosities.
Results
The ASV length, patterns of origin and termination are reported in both normal and patients with varicose veins. Discussion of the patterns is supported by the narrative review of the literature.
Conclusions
The ASV must be considered a truncal vein and its treatment modalities should be the same that for the great and small saphenous veins rather than a tributary vein.
Keywords
Introduction
In recent years, endovascular ablation has emerged as the best technique to treat truncal reflux whereas phlebectomy or sclerotherapy are the best option for varicose tributaries. Accordingly, the distinction between a truncal and a tributary vein is increasingly important in the treatment of superficial venous disease. This distinction was not well defined until the late 1990s. All superficial veins in the lower limbs were considered as surrounded by the amorphous subcutaneous fat until 1997 when it was demonstrated that the Great Saphenous Vein (GSV) courses for its entire length in a proper fascial compartment, deeply located in the subcutaneous tissue, known as the so-called “Saphenous Compartment” (SC).
1
This compartment is delineated deeply by the muscular fascia and superficially by a portion of the Membranous Layer of the Subcutaneous Tissue that was called “saphenous fascia” (SF) (Figure 1).
1
In 2001 the same planar anatomy and fascial relationships were demonstrated for the Small Saphenous Vein (SSV).
2
The compartment of the GSV continues on the dorsum of the foot along the medial marginal vein, the dorsal arcuate vein and the lateral marginal vein to connect itself to the compartment of the SSV.
3
The saphenous compartment. D: dermis. MF: muscular fascia. SF: saphenous fascia. GSV: great saphenous vein. ASV: anterior saphenous vein.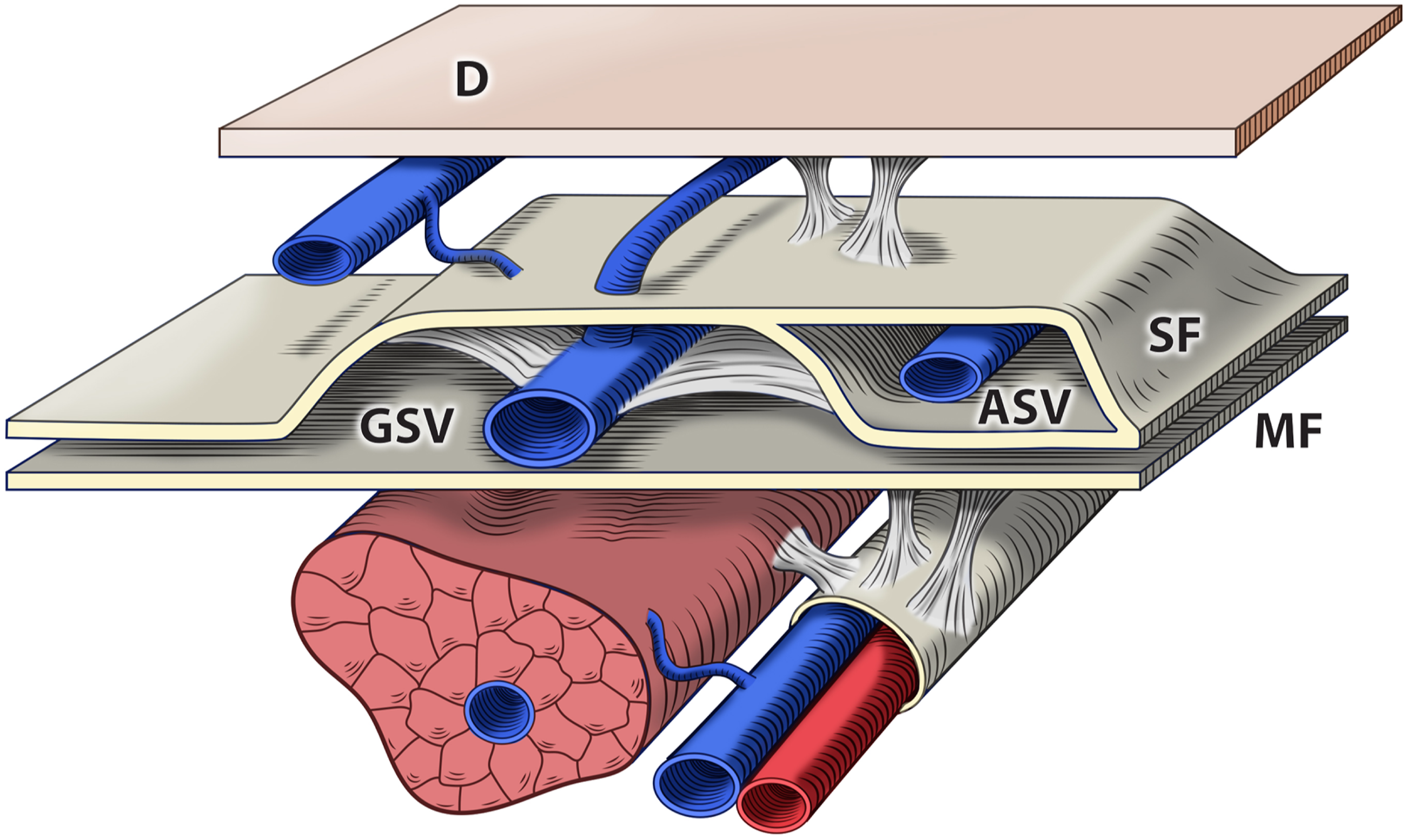
In 2001, an international interdisciplinary committee which included the International Union of Phlebology, the International Federation of Anatomical Associations and the Federative International Committee on Anatomical Terminology introduced a clear discrimination between the GSV and the SSV from the other “superficial” veins because of their deep and interfascial path. 4 The fascial compartment characterized the GSV and SSV as proper truncal veins in contrast to other superficial veins in the legs which were characterized as tributary veins. The latter are smaller and course close to the skin, in a real superficial plane of the subcutaneous layer. 5 This was designated as the “epifascial plane” because it is superficial with respect of both the muscular and the saphenous fasciae. 6 An exception to this general rule was also acknowledged, i.e., that at the thigh a vein courses deep in the subcutaneous layer (SCL) in a proper interfascial compartment parallel but more anterior to that of the GSV, referred to at the time as the Anterior Accessory of the GSV (AAGSV). 6 This anatomical consensus statement did not address whether this vein was by definition truncal, due to its fascial envelopment, or a simple tributary of the GSV (Figure 1). In fact, by definition, the superficial truncal veins should be distinguished from other veins in the subcutaneous tissue (tela subcutanea), because they are enclosed in the “saphenous fascia” or more appropriately, contained within the “compartimentum saphenum”. 7
The ASV was not mentioned in the 1995 and 2004 versions of the CEAP8,9 as well as in the original Venous Segmental Severity Score (2000). 10 After its description in 2005, 6 the ASV appeared in the UIP Consensus documents on Duplex investigation of venous disease (2006) 11 and on Duplex investigation after treatment for varicose veins (2011). 12 Finally, the last revision of the CEAP classification introduced the Anterior (Accessory) Saphenous Vein and clearly discriminated it from other superficial veins. (Nonsaphenous Vein, NSV). 13
Multiple studies have demonstrated a relevant role of the ASV in the pathogenesis, pathophysiology, and treatment of primary and recurrent varicose disease of the lower limbs.13–15 There are also unique natural history considerations that make ASV reflux similar to that in the GSV and SSV. 15 The lack of clarity and consensus has resulted in a limited access to care for some patients when treating superficial truncal reflux in this distribution.
To help alleviate potential confusion from the adjective “accessory” that can suggest this vein is a simple tributary of the GSV, it has been proposed that the term AAGSV be abandoned in favor of the term Anterior Saphenous Vein (ASV) to provide more anatomic clarity to guide optimal clinical treatment options and coverage guidelines. 16
This manuscript is part 2 of a 4 part series supporting changing the terminology to ASV. This recommendation is supplemented by three accompanying parts to this series: Part 1: The Anterior Saphenous Vein. A position statement endorsed by the American Vein and Lymphatic Society (AVLS), the American Venous Forum (AVF) and the International Union of Phlebology (UIP). Part 3: Systematic review of the literature and payor coverage policies and Part 4: a review of the clinical and technical considerations when treating patients with ASV reflux. The purpose of this article is to outline the clinical anatomy of the ASV by a narrative review of the literature supported by data obtained in both normal volunteers and in patients with primary varicose veins of the anteromedial thigh.
Methods
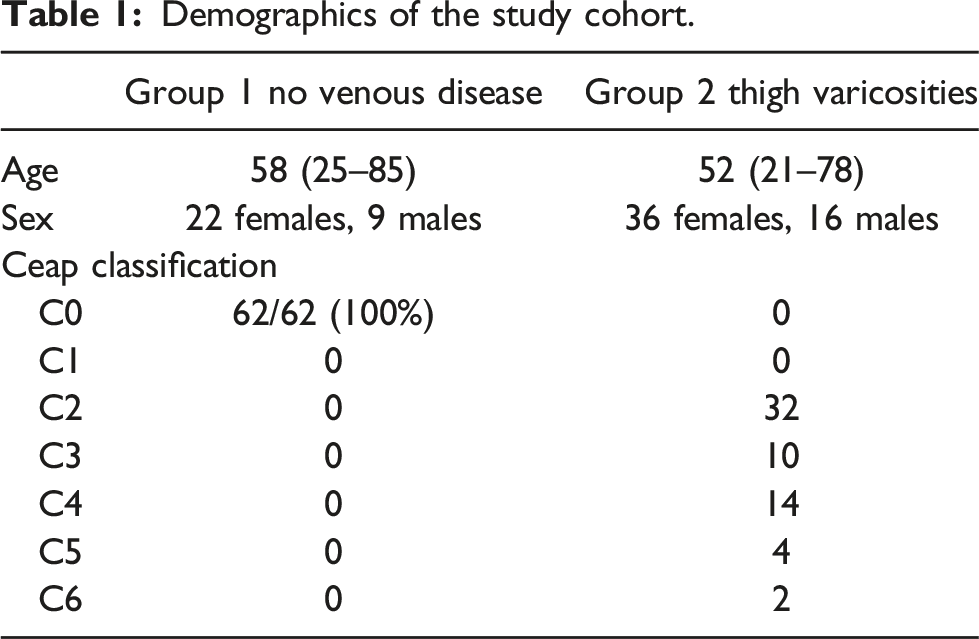
Demographics of the study cohort.
All ultrasound examinations were performed in the standing position using a Sonoscape S9 echograph and a linear 8–14 MHz transducer. Real-time B mode images were used to follow the course of the superficial veins in the thigh from the sapheno-femoral junction (SFJ) caudally. The caliber of the GSV and of the ASV were calculated by digital calibers at the upper third of the thigh. Reflux was determined by pulsed Doppler in response to the release of manual calf compression and defined as retrograde flow >0.5 s in duration. The ASV was defined as the vertical vein that courses deep in the subcutaneous layer of the antero-medial thigh, parallel but more anterior to the GSV, between the saphenous and the muscular fasciae. To evaluate the extension of the interfascial path of the ASV, the thigh was divided into the groin area (femoral triangle), upper, mid and lower third. The ultrasound findings in this study were compared to those previously reported in the literature. Publications concerning the anatomy of the superficial veins of the thigh were reviewed after a Pubmed and Google Scholars review. No previous studies were identified specifically evaluating the anatomy of the ASV in normal limbs.
Results
The path and fascial relationships of the ASV
In all patients in both Group 1 and Group 2 the ASV courses in a deep plane of the SCL of the antero-medial thigh, above the sartorius and vastus medialis (Figure 2(a)). Its course is parallel but more anterior to that of the GSV. In the proximal thigh it runs parallel to the femoral artery and vein to reach the fossa ovalis, crossing the lateral margin of the femoral triangle (Figure 2(b)). At the groin, the ASV courses between several lymph nodes and receives most of the veins draining them. In both groups it was noted that these connections form a lympho-venous network, often associated with primary and recurrent varicose veins of the thigh as reported separately (Figure 3).
17
As the GSV and SSV, the ASV courses in a defined compartment delimited deeply by the muscular fascia (MF) and superficially by a duplication of the Saphenous Fascia (SF) (Figures 1 and 2(b)). (a) Muscle relationships of the ASV. (b) The relationships of the ASV with the femoral vessels (FV) and the saphenous fascia (arrowheads). GSV: great saphenous vein. (a) The relationships of the ASV (arrow) with inguinal nodes (N). (b) ASV ectasia in primary varicose veins. (c) recurrent varicose veins originated by dilated veins of the inguinal nodes (*). FA: femoral artery. FV: femoral vein.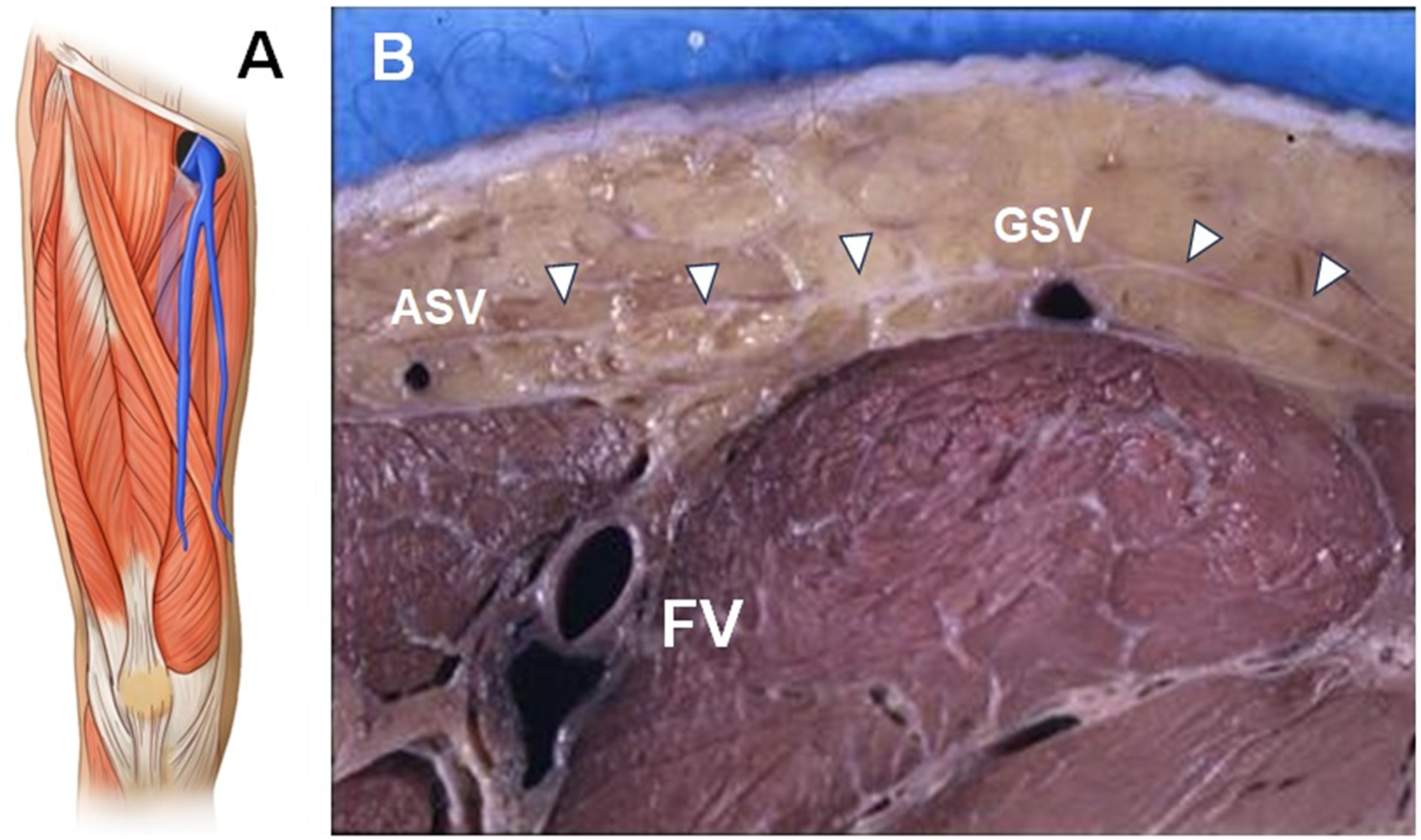
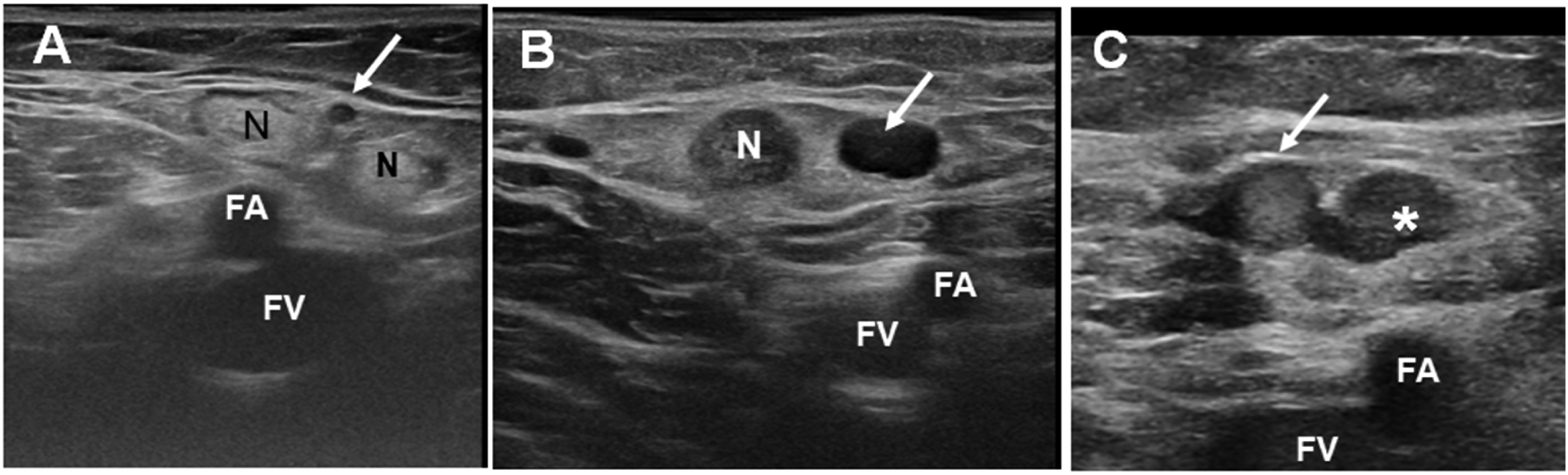
Ultrasound anatomy of the ASV
The deep and interfascial path (the so-called “Aegyptian eye sign”) (Figure 4(a)) of the ASV allows to it to be easily differentiated from other superficial veins in the anterior and medial thigh. However, this sign does not allow discrimination of the ASV from the GSV, both appearing as the pupil of the respective eyes. The ASV and GSV are, however, discriminated by the “alignment sign”. The ASV courses immediately above the superficial projection of the femoral vessels, whereas the GSV courses along a more medial path (Figures 2(b), 3(a)–(c), 4(b)). Ultrasonography of the ASV. (a) The double saphenous eye. (b) The ASV (white arrow) courses between the saphenous fascia (SF) and the muscular fascia (MF), aligned above the femoral artery (FA) and vein (FV). GSV: great saphenous vein.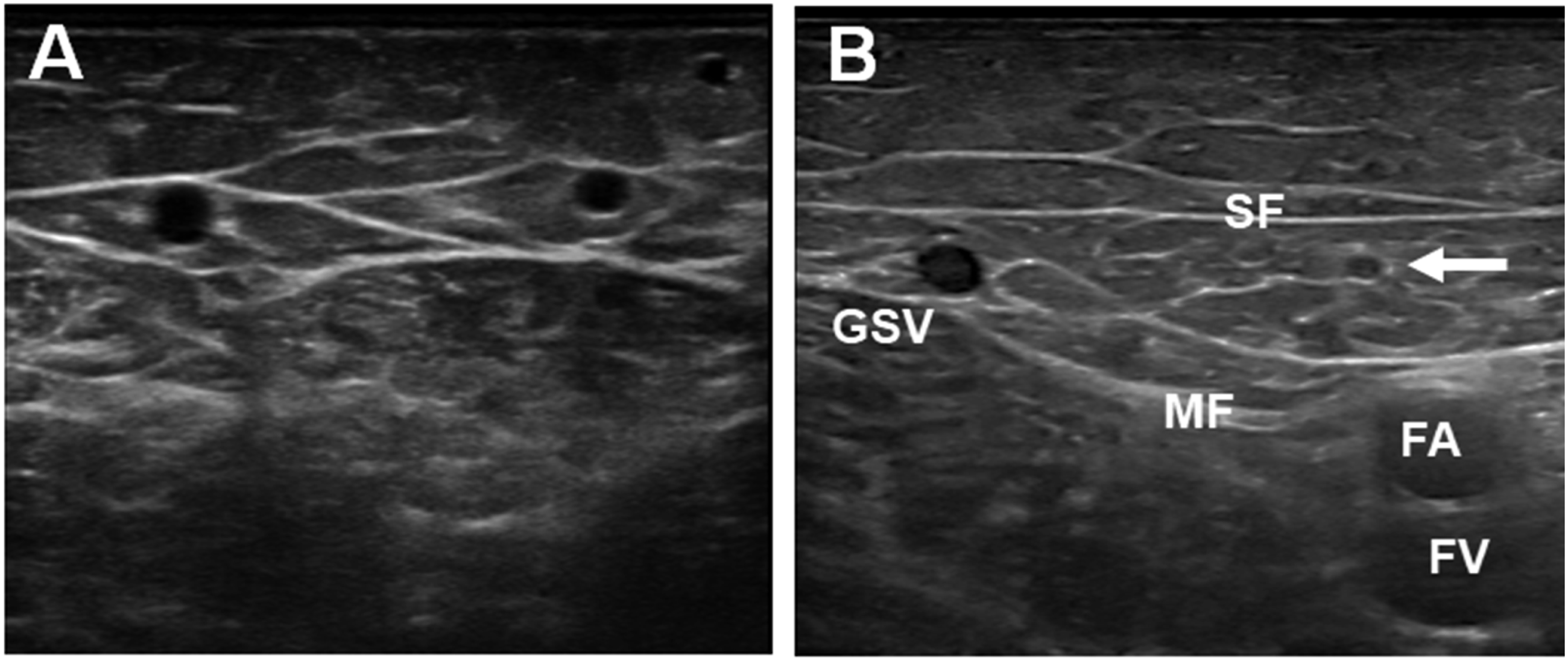
The length of the ASV
As reported by other studies, we confirmed in both groups that the distinctive anatomic characteristic of the ASV is its relatively short interfascial path (5 to 20 cm from the SFJ), generally not extending below the knee. 18
In 12/62 (19%) group 1 limbs, the ASV was visible by US only at the groin and consisted of a small vein from the inguinal nodes joining the SFJ (Figures 3 and 5(a)). In 18/62 (29%) limbs, the ASV was visible only at the upper third (Figure 5(b)) and in 26/62 (42%) also at the mid third (Figure 5(c)). In only 6/62 (9.6%) limbs, was the ASV also visible at the lower third of the thigh (Figure 5(d)) (Table 2). The extension of the ASV. (a) the ASV is visible only at the groin. (b) the ASV is visible at the upper third of the thigh. (c) the ASV is visible also at mid third and, (d) at the lower third. The length of the ASV.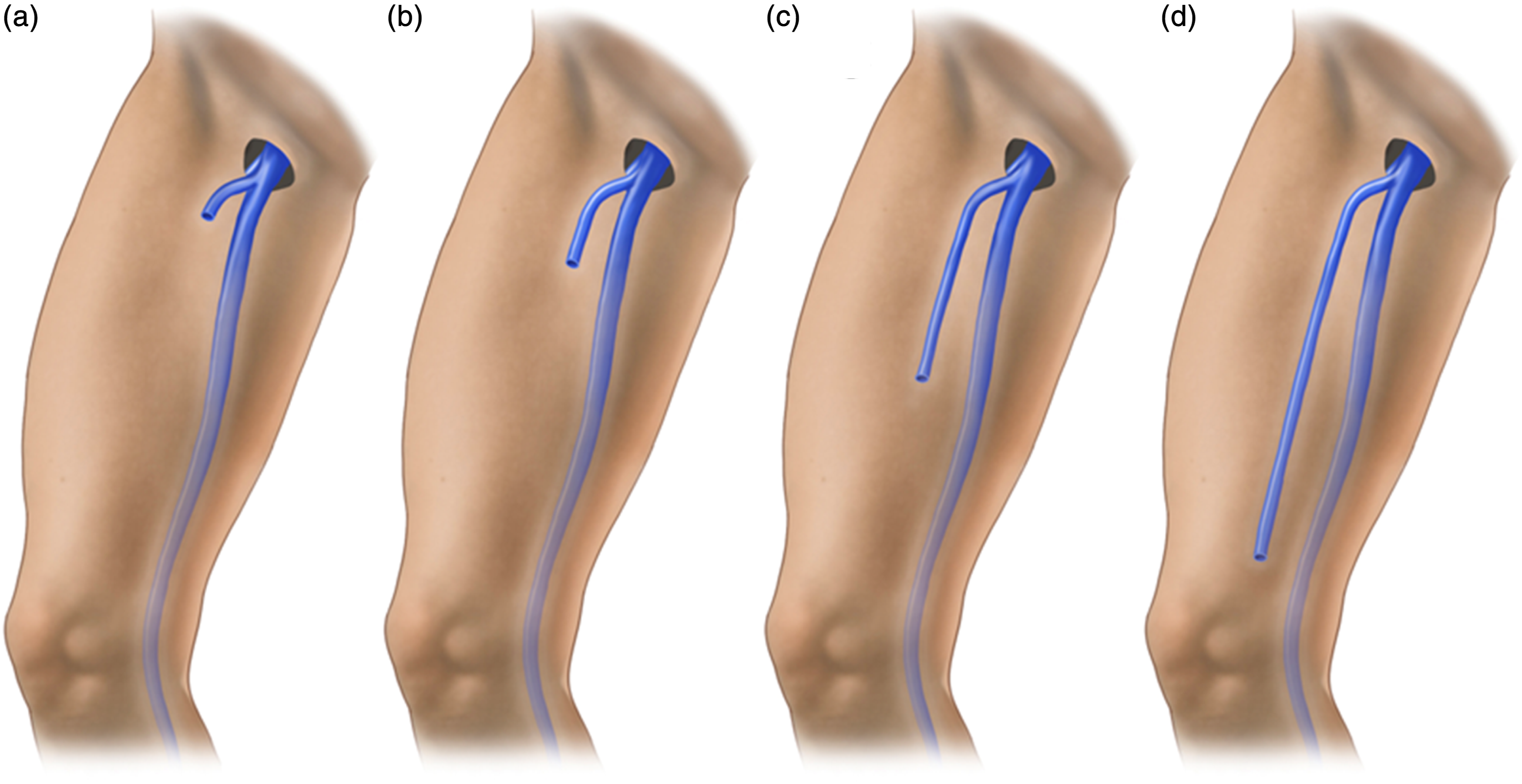
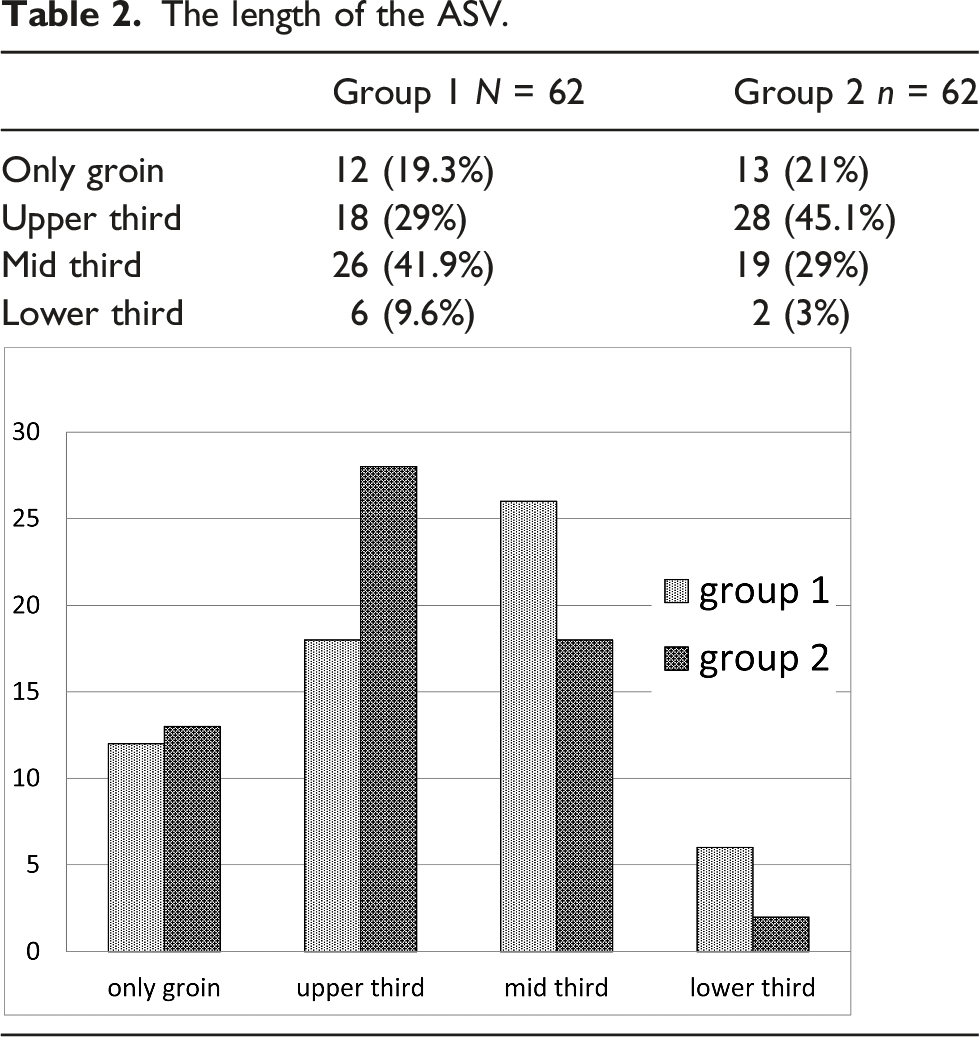
In group 2, the ASV was not visible in the upper, mid or lower third of the thigh in 13/62 limbs (21%). In the remaining 49 limbs (79%) the ASV was visible at least at the upper third of the thigh (Table 2).
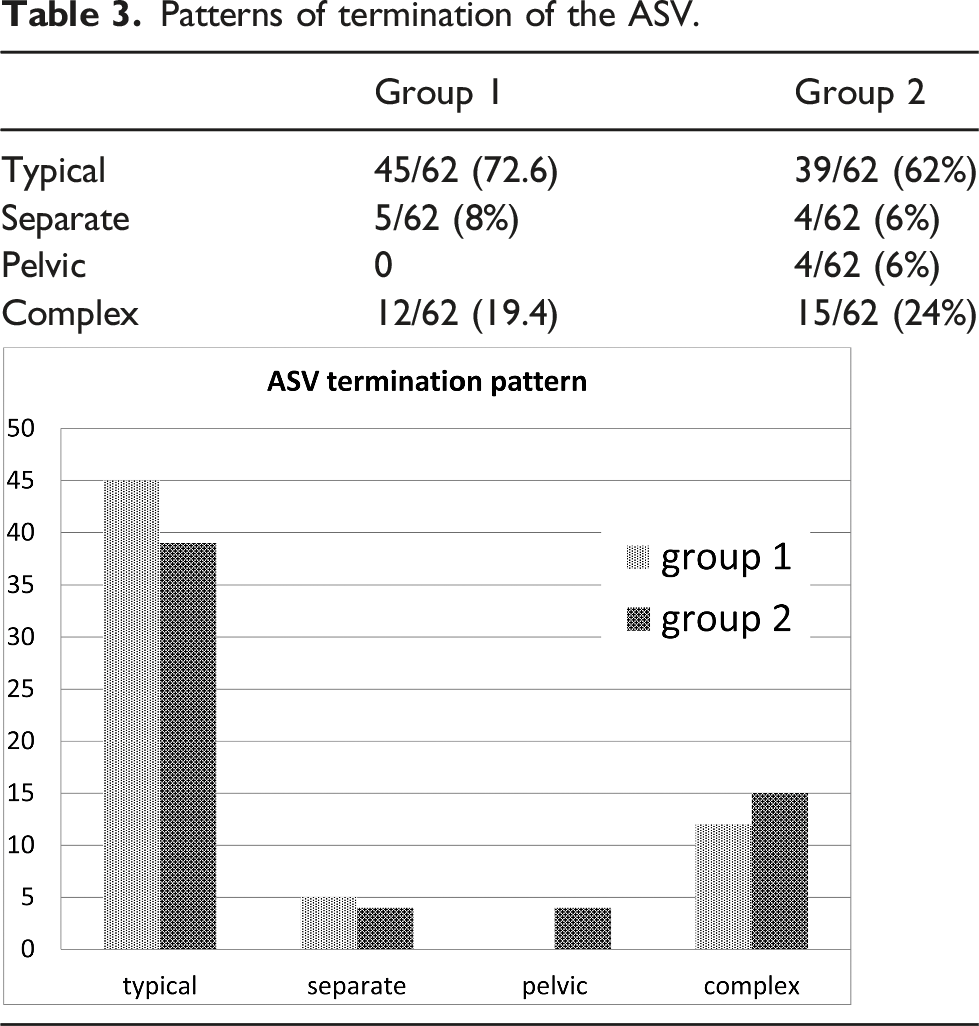
The ASV termination
It has been well characterized in prior studies that the ASV generally ends in the GSV at the SFJ above the Preterminal Valve (PTV), either directly or joining one of the other SFJ tributaries (Figure 6(a)).
18
Less frequently, the ASV joins the GSV below the PTV (Figure 6(b)) or finally, the Common Femoral Vein with a separate junction (Figure 6(c)). It is reported that occasionally it continues as a superficial vein of the anterior abdominal wall (inferior epigastric vein) (Figure 6(d)).
18
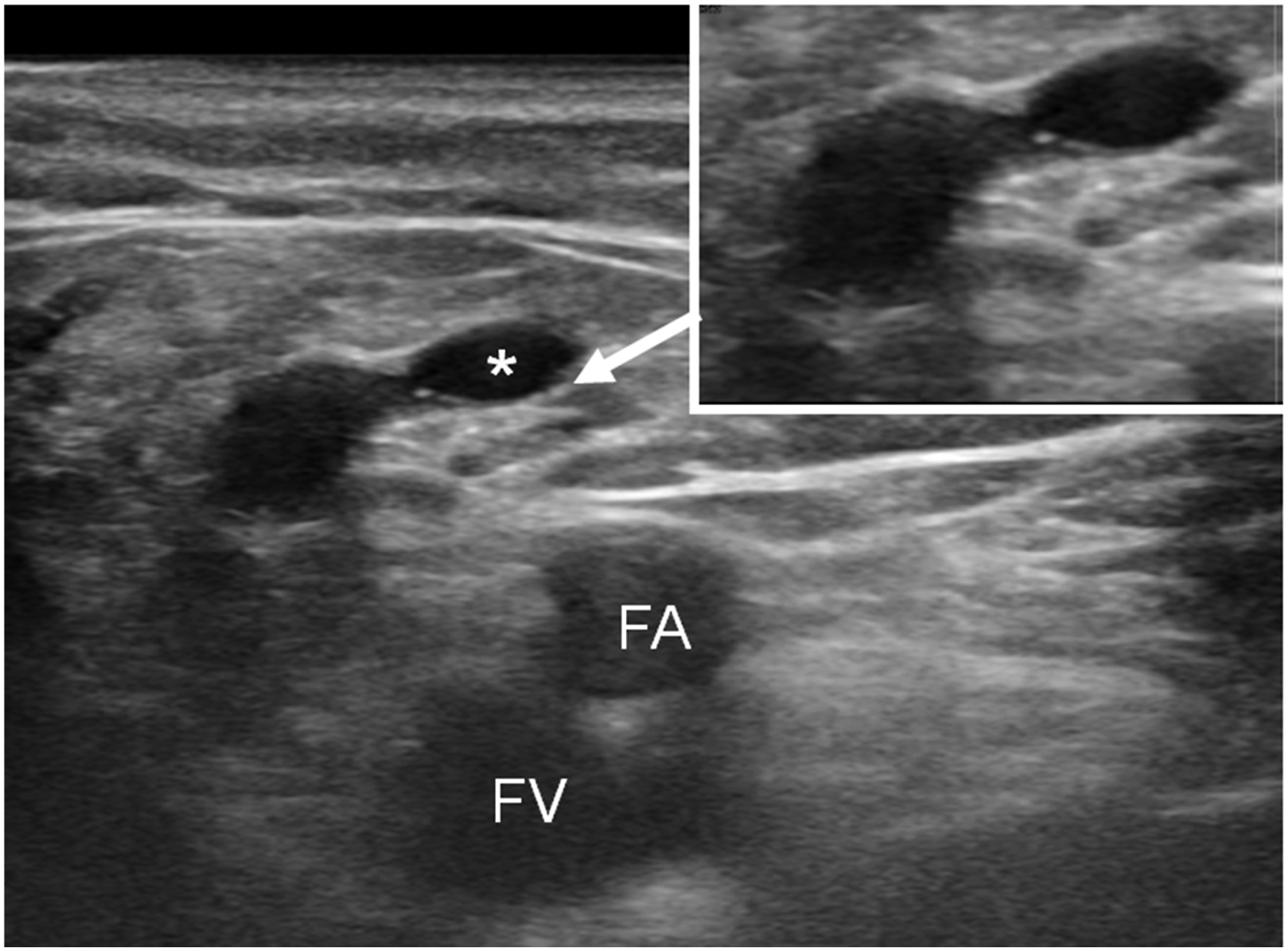
A “terminal valve” is located just below the ASV termination (Figure 7). The patterns of termination of the ASV. (a) Between the preterminal and terminal valves of the GSV. (b) Below the preterminal valve of the GSV. (c) Separate termination into the femoral vein. (d) Connection with the veins of the pelvic diaphragm. The terminal valve of the ASV (*). FA: femoral artery. FV: femoral vein.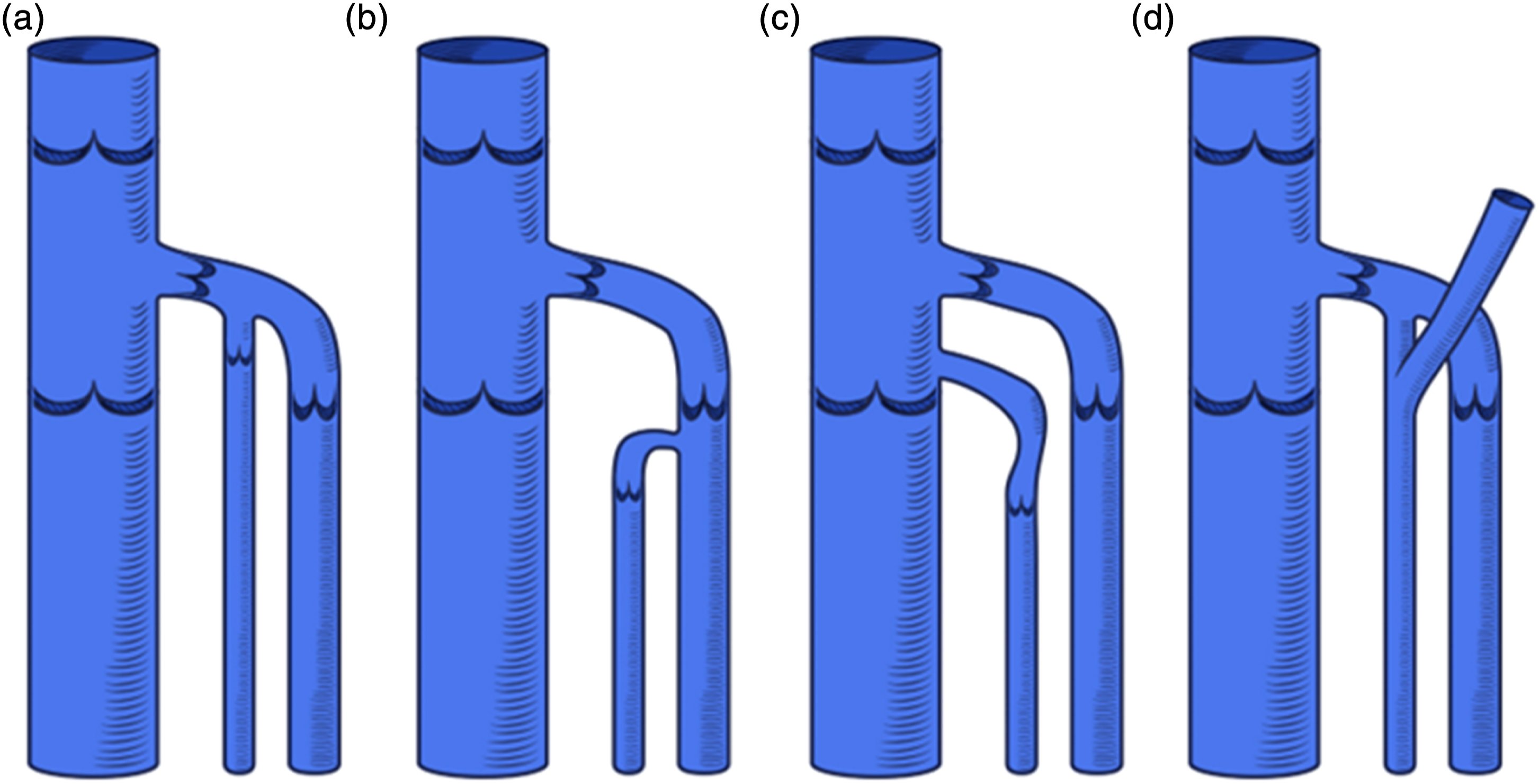
Patterns of termination of the ASV.
The origin of the ASV in normal limbs
There can be a variety of origins of the ASV. The interfascial path of the ASV may arise from the Anterior Thigh Circumflex Vein (ATCV) (Figure 8(a)), from the trunk of the GSV (Figure 8(b)) or, finally, from epifascial veins of the anteromedial thigh (Figure 8(c)). The ATCV is a vessel that originates from the veins of the Lateral Plexus and courses obliquely in the epifascial plane of the SCL of the anterior thigh. This vein may terminate at the SFJ independently from the ASV and can be mistaken for the ASV. The ATCV can be distinguished from ASV by its course closer to the skin and lack of any fascial ensheathment. The origin of the ASV. (a) From the anterior thigh circumflex vein which drains the Lateral Plexus. (b) From the GSV trunk. (c) From a net of unnamed veins of the anteromedial thigh. (d) The anterior thigh circumflex vein may course more vertically in the epifascial plane to drain directly into the sapheno-femoral junction.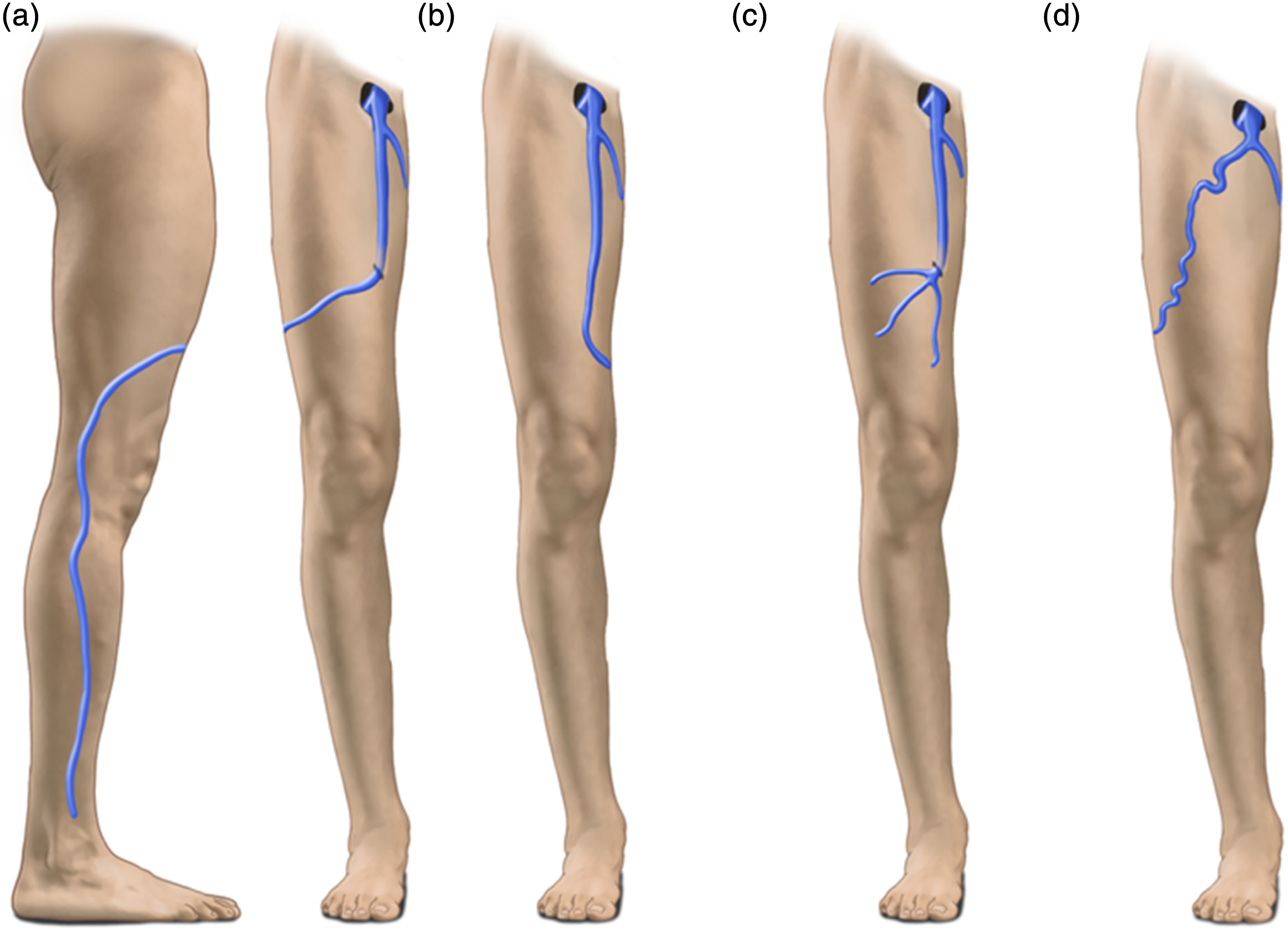
Alternatively, the ASV may originate from the trunk of the GSV at the mid or less frequently the lower thigh, resembling a saphenous duplication. 19 This arrangement has been designated “the embryonic pattern of the saphenous system” 20 and appears as two large vertical veins in the subcutaneous layer of the anteromedial thigh. In some cases, the segment of the GSV cranial to the origin of the ASV may be markedly reduced in caliber (GSV hypoplasia or aplasia) and the main superficial vein of the thigh is the ASV. 21 This is particularly evident when the ASV is incompetent. 14
In contrast to the subjects with venous reflux (Group 2) where the veins are larger, in subjects with normal limbs (Group 1) it was not always possible to establish the precise origin of the ASV or to define all its connections. The origin of the ASV could be evaluated in only 50 out of 62 limbs of the group 1 (81%) in which it was visible at the upper third of the thigh. In 13/50 (26%) limbs, the ASV appears to be the upward continuation of a vertical epifascial vein which goes deeply below the saphenous fascia. In 4/50 (8%), the predominant root of the ASV is the ATCV. In 11/50 (22%) limbs the ASV arises from the GSV trunk in the thigh. In 6 of these limbs (12%), the GSV cranial to the origin of the ASV was markedly reduced in caliber or absent. In the remaining 22 limbs (44%), the interfascial segments of the ASV arises from a network of epifascial veins of the anterior thigh, with no evident predominance of any vein.
The size of the ASV in normal limbs
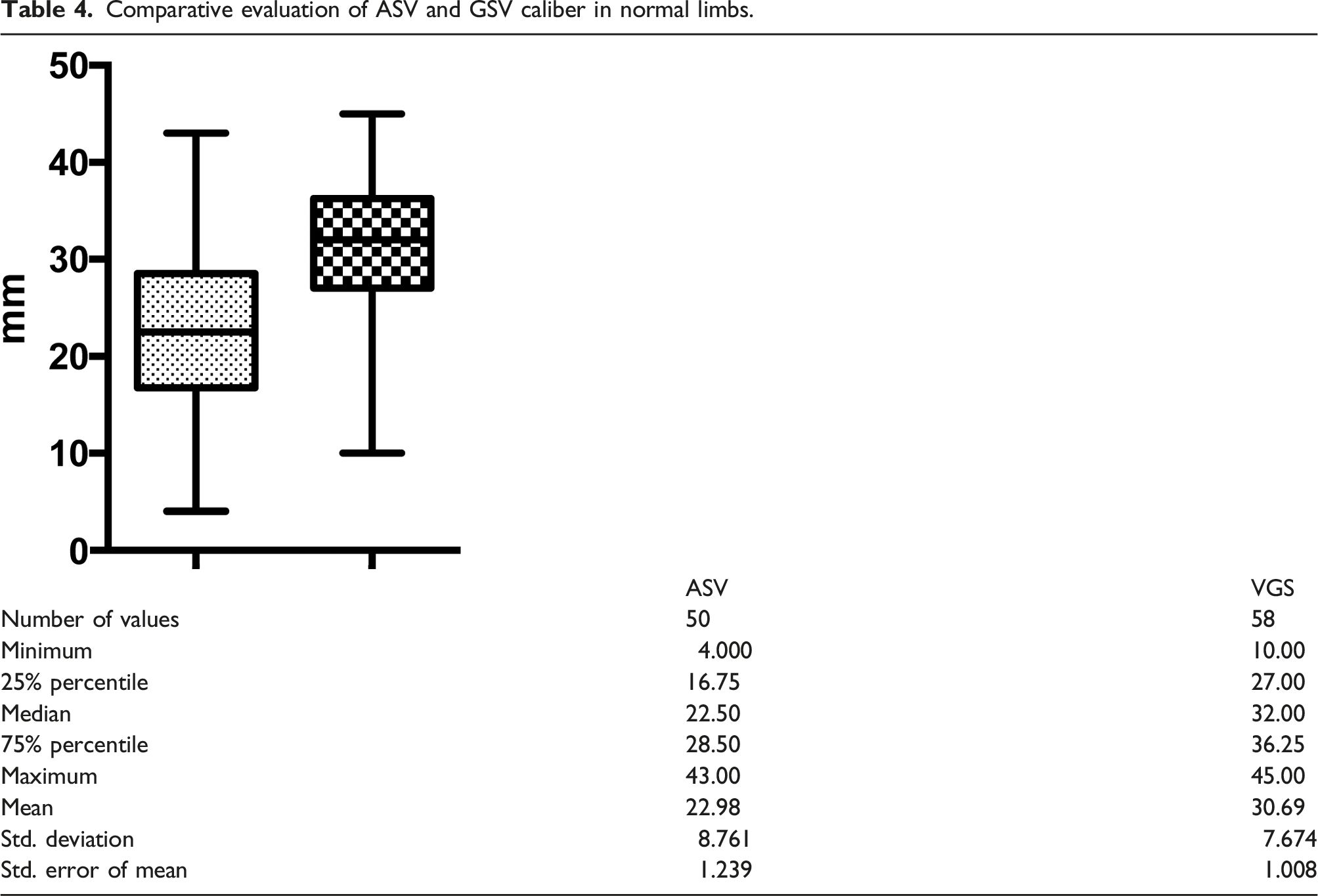
The caliber of the ASV was evaluated only in the 50 limbs of the group 1 in which it was visible at the upper third of the thigh. The caliber of the ASV was greater than that of the GSV in 13/62 (21%). In 32/62 (51.6%) the caliber of the GSV was greater than that of the ASV (Figure 9) (Table 4). The comparison was not possible in 5/62 (8%) legs because of aplasia of the GSV at that level. The mean caliber of the ASV was 2.35 mm, whereas that of the GSV at the same level was 3.07 mm (Table 4). The caliber of the ASV. (a) The caliber of the GSV (on the left) is greater than that of the ASV (on the right). (b) The GSV and the ASV show a similar caliber at the upper thigh. Comparative evaluation of ASV and GSV caliber in normal limbs.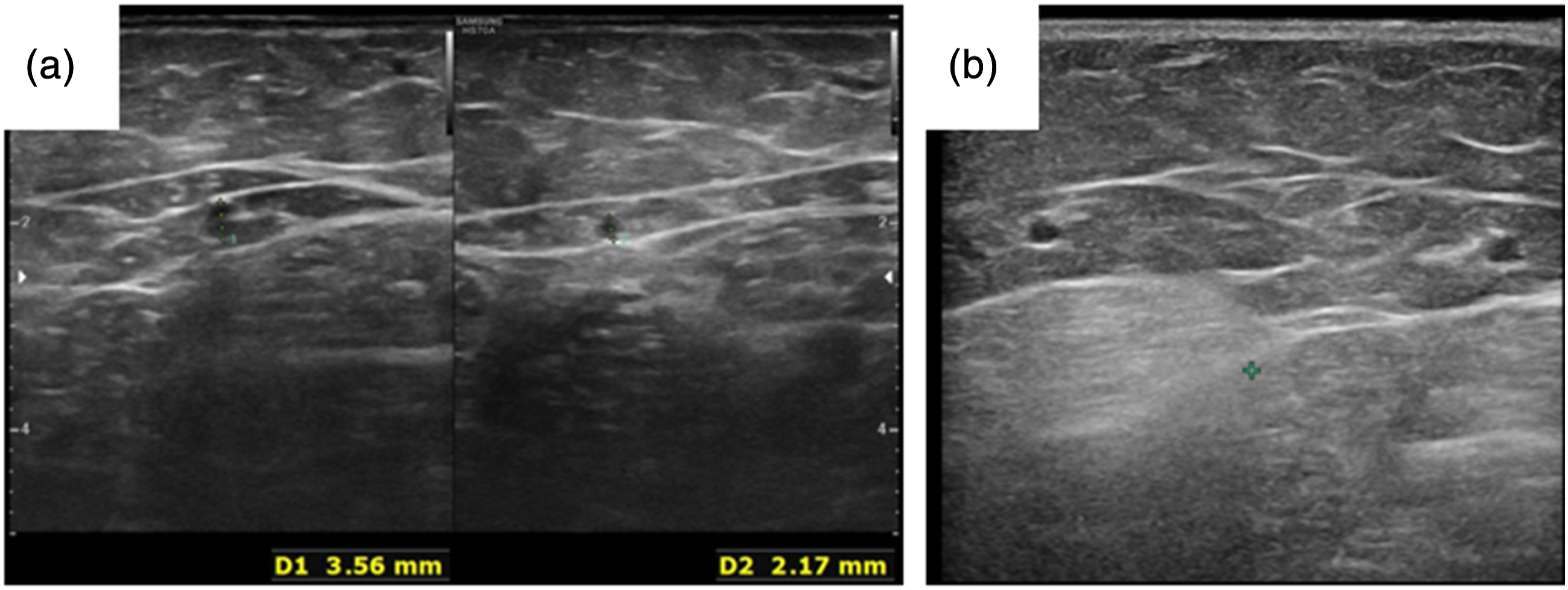
The origin of the reflux in the ASV
In the vast majority of cases, the reflux in the ASV is associated with the Common Femoral Vein, according to the following patterns:22,23 - Sapheno-femoral reflux occurred through the incompetent GSV terminal valve but is shunted into the ASV because of a competent preterminal valve (Figure 10(a)). - ASV reflux occurs in the absence of a GSV after previous GSV surgery (flush ligation + stripping, endoluminal ablation) (Figure 10(b)).
24
- The reflux is associated with neoangiogenic vessels following SFJ ligation and division. (Figure 10(c)) - The reflux is directly transmitted from the CFV to the ASV in the case of its separate termination (Figure 10(d)). The origin of the reflux in the ASV. (a) From the femoral vein, with an incompetent GSV terminal valve and a competent preterminal valve. (b) From the femoral vein, following SFJ interruption. (c) From neoangiogenetic vessels after crossectomy. (d) From the femoral vein, in the case of ASV separate termination. (e) From veins of the inguinal nodes. (f) From veins of the pelvic floor.
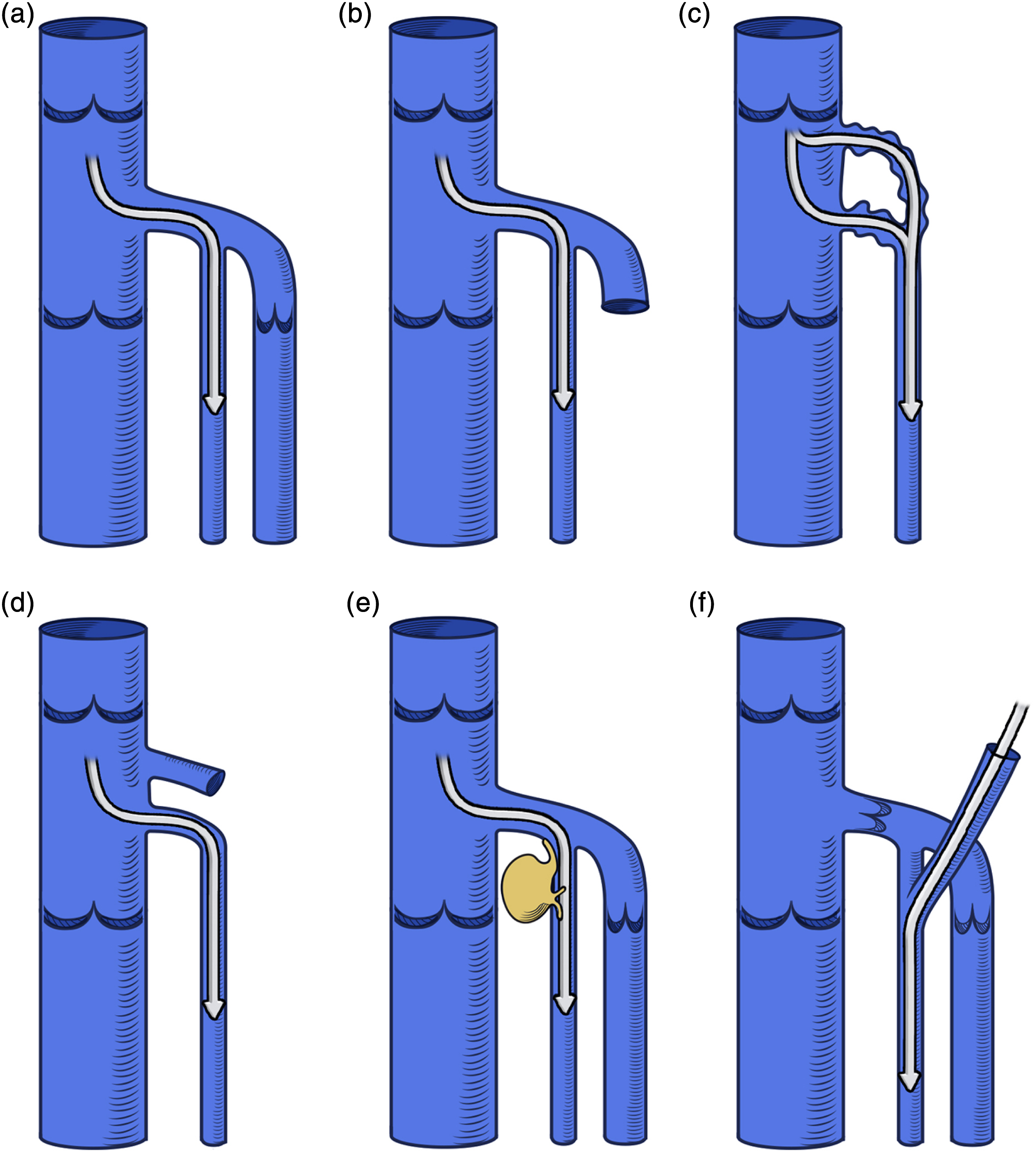
The reflux is associated with the veins of the inguinal nodes
17
(Figure 10(e)). - The reflux is associated with veins of the pelvic floor and is transmitted to the ASV via the external pudendal vein or via the veins of the round ligament through the inguinal point. (Figure 10(f)). Such an occurrence must be discriminated from the transmission of the reflux to the Posterior Accessory of the GSV. This vein courses devoid of any fascial covering superficially in the subcutaneous layer, parallel but more posterior to the GSV.
5
In the 62 limbs of group 2 with primary varicose veins of the anteromedial thigh, the reflux was only in the ASV in 12 whereas in 2 legs both the ASV and GSV were incompetent for a combined prevalence of 22.5%. In the remaining 48 limbs, the reflux was only in the GSV.
These findings agree with previous studies that reported the prevalence of ASV reflux in patients who have not had prior vein interventions: the ASV alone had reflux in about 11% of limbs with varicose veins of the antero-medial thigh. 18 Additionally, about 11% of patients had ASV reflux with concurrent GSV reflux for a combined prevalence of approximately 22%. 25
The fate of the reflux in the ASV
Venous reflux leading to varicose veins of the anteromedial thigh can extend caudally along four different paths that include along the GSV, along the ASV with a normally sized GSV, along the ASV with a hypoplastic GSV and, finally, along both the GSV and ASV. In most limbs with ASV incompetence, the reflux is distally transmitted to the ATCV (Figure 11(a)). The fate of the ASV reflux. The reflux is transmitted to the ATCV (a), to the GSV (b) or finally to unnamed veins of the anteromedial thigh (c).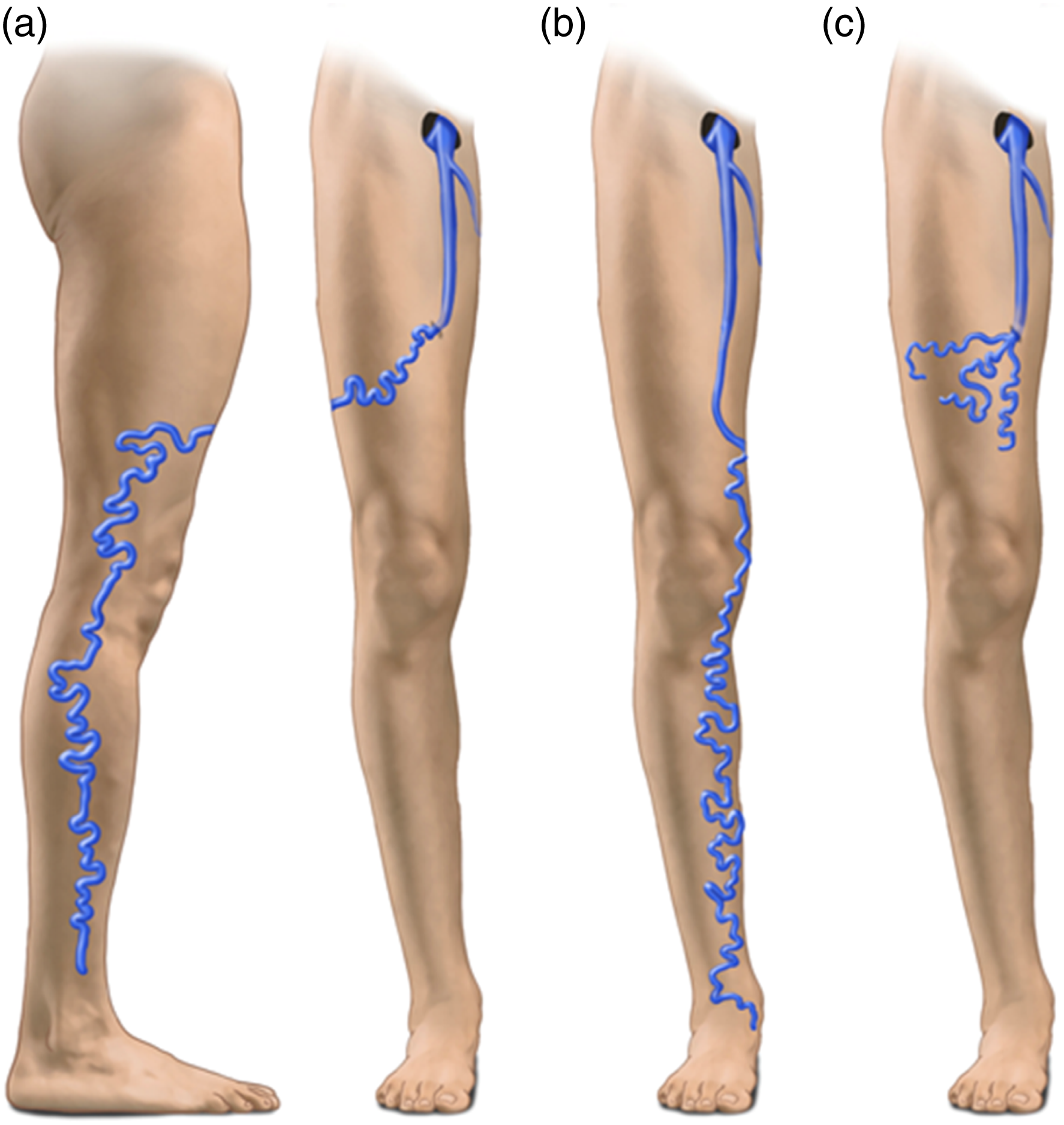
The refluxing ATCV may greatly enlarge with a typical serpiginous path and a cluster of large varicose veins may descend obliquely from the anteromedial thigh to the lateral knee and leg. Here the ATCV is connected to the veins of the Lateral Plexus with evident varicose veins of the lateral thigh and leg (Figures 11(a) and 12(a)). In other cases, the ASV reflux connects to the more distal GSV and saphenous tributaries in the calf (Figures 11(b), 12(b) and 13). Accordingly, the GSV is incompetent below the ASV entry point but competent above the entry point. This pattern is frequently associated with a marked reduction in the GSV caliber at the groin or at the upper thigh. Less frequently, the ASV exits the fascia and tracks medially in the upper thigh and around the patella and upper calf without contributing to GSV reflux. Finally, the ASV reflux can be distributed to unnamed veins of the anteromedial thigh (Figures 11(c) and 12(c)). The fate of the ASV reflux. (a) the reflux is shunted to the lateral plexus via the ATCV (arrow). (b) The ASV reflux is shunted to the GSV at mid-thigh. The GSV is competent above the entry point of the ASV. (c): the reflux descending along the ASV is shunted to unnamed veins of the anteromedial thigh. An incompetent ASV (*) coexists with a normally sized GSV (white arrows in b and c). The ASV is covered by the saphenous fascia for its entire length, down to the lower thigh where the reflux is transmitted to the GSV (b, c).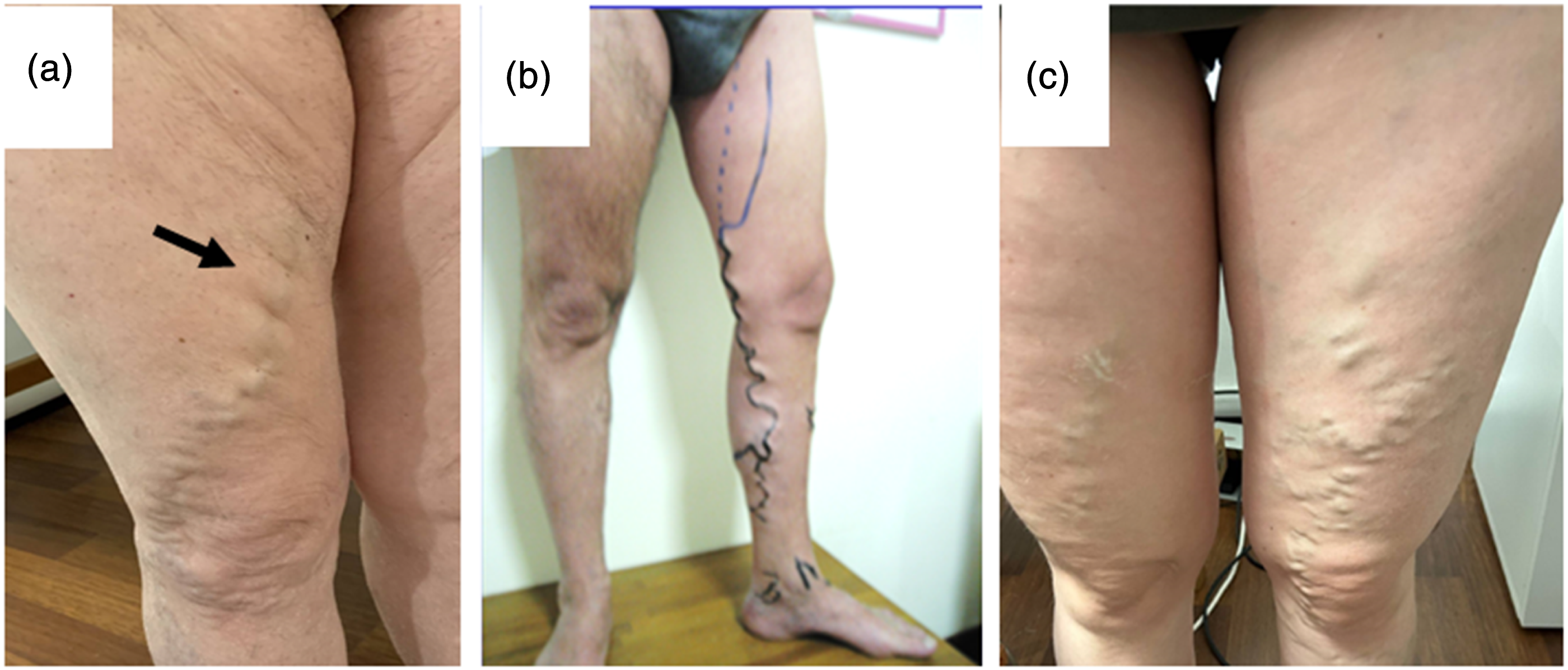
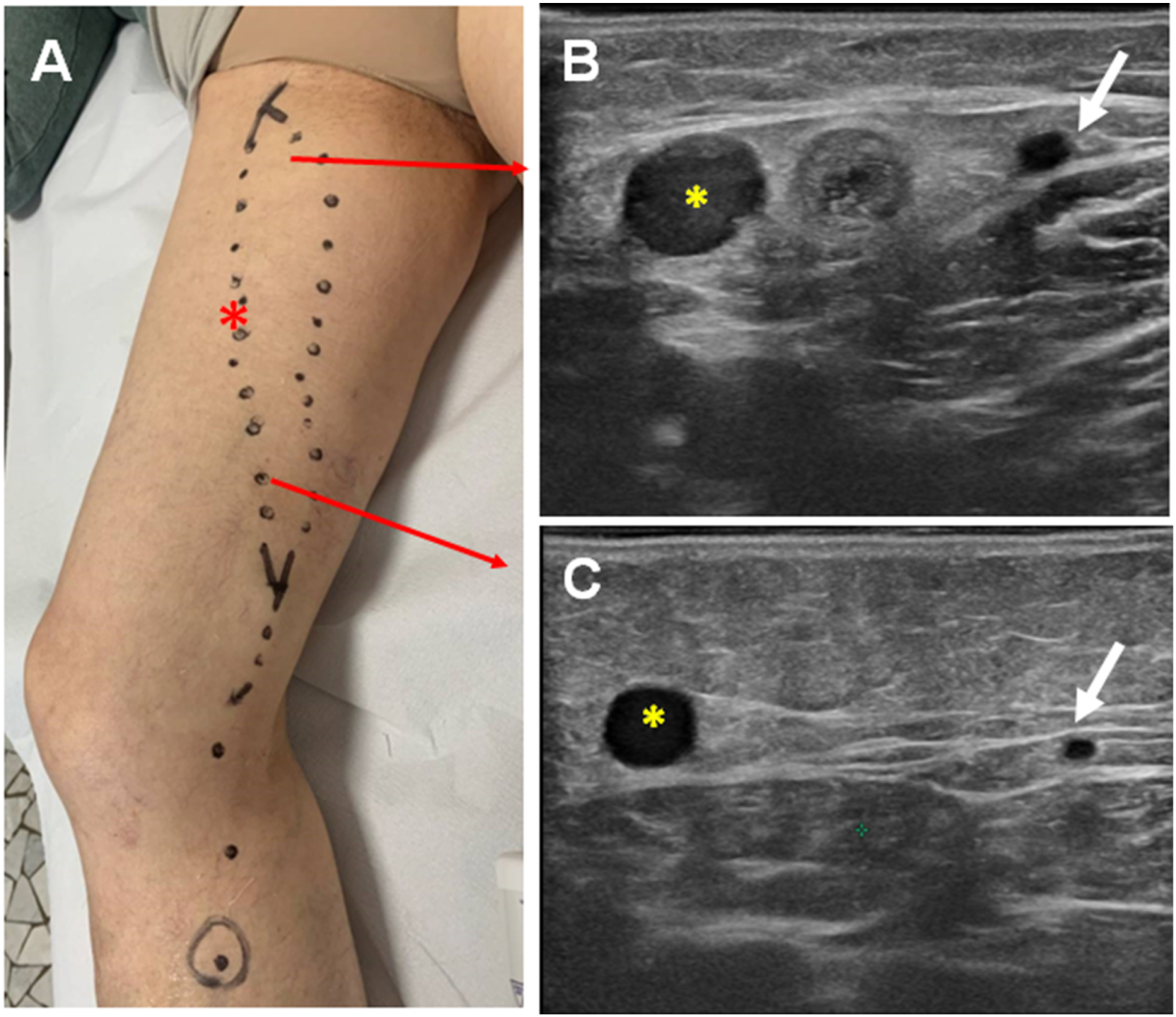
In 6 out of the 13 limbs of the group 2 with a primitive varicose ASV, the reflux was distally transmitted to the ATCV, in 2 to the GSV trunk and finally into anonymous epifascial veins of the anteromedial thigh in the remaining 5 cases.
Discussion
A truncal vein refers to a major subcutaneous vein that forms the primary pathway for blood return in a specific region of the body. In normal limbs, it drains the epifascial tributaries. In limbs with varicose veins, the reflux from truncal veins is connected to tributaries which are devoid of any fascial protection against dilation. 5
The anatomy of the GSV and of the SSV have been previously evaluated in normal and varicose limbs with reference to their course, size, fascial relationships, and connections. This has led to their designation as truncal veins. As they most often follow a straight course and are deeply situated within a fascial envelope, they are currently best treated with catheter-based techniques such as thermal ablation, proprietary foam, or cyanoacrylate. In contrast, superficial tributaries are often tortuous, situated more superficially and lack a fascial envelope – they are accordingly best treated with phlebectomy or foam sclerotherapy. Treatment algorithms for the ASV are similar to other truncal veins and the literature strongly suggest beneficial patient outcomes that are equivalent to GSV ablation by a variety of techniques. 15
Although the ASV clearly shares anatomic features with the other truncal veins, this has been less well described in the literature. While prior studies have detailed the characteristics of the ASV in patients with venous reflux, this is the first study to systematically evaluate its anatomy in normal limbs with the aim to describe the “normal” anatomy of the ASV in addition to that in limbs with venous reflux. The main difficulty in evaluating the US anatomy of the superficial veins in the absence of reflux (group 1) is the small caliber of the ASV and of its tributaries, especially in young subjects. Consequently, this study took particular care to prevent their compression under the pressure of the probe on the skin surface. With such careful technique, a macroscopic ASV was detected by US in 50/62 (81%) normal limbs. The length of the interfascial path of the ASV was variable, originating at different levels of the anteromedial thigh. In normal limbs there is more than one pattern of ASV termination. When present, its caliber was about 70% of that of the GSV.
Thus, these findings are consistent with prior studies where the anatomy of the ASV in varicose limbs is distinct in location but similar to that of the GSV in terms of path and fascial relationships in the proximal thigh.14,15,22,23
The recognition of the ASV as a proper truncal vein has important implications in treatment planning and for authorities that define treatment coverage. Improved patient symptoms, long term closure, and minimizing the incidence of recurrence can be achieved by following the principles and techniques of other truncal veins such as the GSV and SSV. Simply ablating the GSV has been shown to be inadequate in treating ASV derived varicosities resulting into higher recurrence rates. 15
As clinicians examine limbs with US, recognizing the unique anatomic attributes of the ASV as a truncal vein is critical to developing an appropriate treatment plan. This requires a keen understanding of normal ASV anatomy and its variants as well as the hemodynamic patterns of ASV reflux in limbs with thigh varicose veins. Finally, ensuring that the ASV is appropriately recognized and covered as a truncal vein by insurance carriers is critical to good patient care and outcomes.
Conclusions
Because available treatments and increasingly payer coverage policies make important distinctions between truncal and tributary treatments, venous specialists must carefully evaluate both truncal veins and their tributaries to provide appropriate and effective treatment for superficial venous disease. Moreover, it is imperative for clinicians to correctly and distinctly identify the GSV and ASV and discriminate their respective hemodynamic contribution to a patient’s symptoms to develop a correct, patient centered plan for treatment.
Both the anatomic characteristics of the ASV and the response to appropriate treatment supports recognition of the ASV as a truncal vein rather than a simple tributary of the GSV. The ASV is enclosed in the saphenous compartment as are the GSV and SSV, and the results of successful ablation are equivalent to the other truncal veins. The most recent update to anatomic nomenclature clearly states that the accessory saphenous veins (venae saphenae accessoriae) are located superficial to this compartment. 7 The term “Anterior Accessory of the Great Saphenous Vein” is therefore anatomically incorrect, which has led to many of the misunderstandings regarding importance. The international, multidisciplinary panel of the AVLS, AVF, and UIP recommends that the term “Anterior Accessory of the Great Saphenous Vein” be abandoned and substituted with the more appropriate “Anterior Saphenous Vein”. As a rule, it is important to distinguish terminology, or specialized terms for one scientific branch, from nomenclature, which require more detailed anatomic oversight using standardized terms according to defined classification principles.
Footnotes
Author’s note
Meissner M (MM); Boyle EM; (EB) Labropoulos N (NL); Caggiati A (AC); Drgastin R (RD); Doganc S (SD); Gasparis A (AG).
Acknowledgements
The authors acknowledge Michael Thompson for helping organize the group to develop and complete the project and Mesa Schumacher for her artistic contributions.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RD, EB, AC, SD and MM reports no conflicts. NL is a Consultant/Speaker with Philips, Medtronic, BD Bard, Boston Scientific; AG is a Consultant/Speaker: Medtronic, BD Bard, Boston Scientific.
Funding
The open access publication fees for this article was financially supported by Boston Scientific Corporation. No other funding was provided.
Guarantor
Mark Meissner, MD.
Contributorship
MM, AG, NL and SD conceived of the project. MM, AG, NL, EB, RD, AC and SD all contributed to developing the articles and contributing to the analysis and conclusions. EB, RD and AC wrote the drafts of the articles and all parties approved and agree to be accountable for the final manuscripts.
