Abstract
Objective
To perform a preliminary usability evaluation of a novel, compact pneumatic compression device in patients with lymphoedema.
Methods
This open-label, single-arm trial had two phases: the first focused on the fitting of the pneumatic compression device (Aria FreeTM, Aria Health, San Diego CA, USA) and the second focused on evaluating the comfort of the entire system during a 45-min usage period. Both phases were conducted in a monitored clinical environment. Patients aged ≥18 years with a diagnosis of lower limb lymphoedema who had used a pneumatic compression device for ≥3 months were eligible. Patients rated subjective fit, comfort and usability on an 11-point Likert scale (where higher scores indicate better fit/comfort/usability). The truncated cone method was used to infer limb volume before and after therapy in phase 2.
Results
Twenty-four patients were screened, and 15 were enrolled (80% female; mean age 62 years); all completed both study phases. Patients rated the garment as easy to set up and fit (median score 6.5), and all reported that the therapy was comfortable (median score 10; p < 0.001 vs. reference score of 6). There was a 1.85% reduction in limb volume after device use for 45 min (p = 0.018 vs. before therapy). No safety issues were identified.
Conclusions
The new pneumatic compression device fitted well, was easy to use and reduced leg oedema.
Keywords
Introduction
Lymphoedema is a chronic condition characterized by the accumulation of protein-rich fluid in the interstitial compartment as a result of impaired lymphatic drainage. 1 There are two main forms of lymphoedema: primary (resulting from innate abnormalities in the lymphatic system) and secondary (resulting from impairment of lymphatic vessels due to an acquired condition such as trauma, tumour, surgery, infection or obesity).1,2 Overall, between 90 and 250 million people worldwide are affected by lymphoedema,3,4 and secondary lymphoedema is more prevalent than the primary form. 1
Left untreated lymphoedema progresses over time, more rapidly in the lower extremities. 1 Regardless of the underlying cause, lymphoedema is characterized clinically by chronic swelling, localized pain, skin atrophy and increased vulnerability to bacterial infection. 2 The effects of lymphoedema have a significant negative impact on health-related quality of life, 5 and patients with lymphoedema have high levels of healthcare utilization, and both direct and indirect costs.6,7
The goal of treatment in patients with lymphoedema is to reduce morbidity and improve overall quality of life. This can be achieved by increasing lymph transport from the limb, thus reducing swelling and restoring functionality of the affected limb, which in turn helps to prevent complications. 8 The mainstay of noninvasive treatment is complete decongestive therapy (CDT), which includes two key stages.1,8 The first includes manual lymphatic drainage (MLD), compression bandaging, exercise, skin care and increasingly the use of pneumatic compression devices (PCDs).1,8 The second (maintenance) stage requires daily self-management by the patient, such as self-lymphatic drainage, compression therapy, skin care, exercise and regular home use of PCDs.1,8
PCDs surround the limb with a garment containing air-filled chambers. Traditionally, PCDs utilized a treatment algorithm where chambers are gradually filled with air to provide active sequential compression from distal to proximal. The aim was to address venous disorders and therefore the treatment was intended to mobilize fluid in the limb vasculature. 9 These devices have been shown to move tissue fluid proximally in lower limb lymphoedema. 10 Recently, newer generation PCDs have been designed to specifically impact the lymphatic system, utilizing lower pressure profiles and treatment sequences intended to simulate manual lymphatic treatment techniques. 8 Although there is still debate about the optimal strategy for acute and chronic management of lymphoedema, PCDs are typically seen as being more useful in the chronic, self-management phase. 11
Intermittent pneumatic compression delivered via a PCD has been shown to reduce symptoms, decrease limb circumference and the rate of complications, improve quality of life and reduce healthcare expenditure.12–15 However, currently available PCD systems have a number of limitations, especially with respect to usability, because they are large, bulky and difficult to use.16,17 Although it has not yet been formally evaluated, anecdotal evidence suggests that these issues can often lead to a lack of engagement, poor usage and/or termination of therapy. One small study reported that 70% of patients had <75% adherence to PCD therapy at 1 year after prescription, where adherence was defined as the usage frequency recommended by their lymphoedema clinician. 18 There is therefore a need for research on new-generation PCD devices. 19
This preliminary study was designed to evaluate the usability of a novel compact PCD in patients with lower limb lymphoedema.
Methods
Study design
This single-center, open-label, single-arm, non-randomized study was conducted between September 2019 and January 2020. The trial was a usability study that included two phases: the first focused on the fitting of the device during a single 60-min clinic visit and the second focused on evaluating the comfort of the entire system during a single clinic visit lasting 90 min. Both phases were conducted in a monitored clinical environment. The study protocol was approved by an independent ethics committee. All patients provided written informed consent prior to enrolment, and the study was conducted in accordance with ISO 14155:2011, Good Clinical Practice Guidelines and Australian Therapeutic Goods Administration requirements.
Study population
Eligible patients were aged ≥18 years, had lower limb lymphoedema, had been using a pneumatic compression device for ≥3 months, were able to understand and follow instructions from study personnel and were able to provide written and informed consent. Patients with active lower limb wounds, undergoing cancer treatment, with a history of pulmonary oedema or decompensated congestive heart failure, with any condition in which increased venous and lymphatic return was undesirable, with an implanted electrical stimulator device, and women who were pregnant or trying to become pregnant were excluded. Beyond the inclusion and exclusion criteria, etiologies of the patients’ lymphoedema were not recorded for this usability study.
Treatment
The pneumatic compression device used (AirWear Investigational System) was a prototype of the subsequently released product, Aria FreeTM (Aria Health, San Diego CA, USA), indicated for patients with primary or secondary lymphoedema. The system consists of an air pump, a foot-to-thigh pneumatic garment with seven chambers, and a connector tube (Figure 1). A set of optional self-fastening extension straps was also provided. The system is applied by wrapping the garment around the limb and connecting the tube to the garment and the pump. Pneumatic compression device components.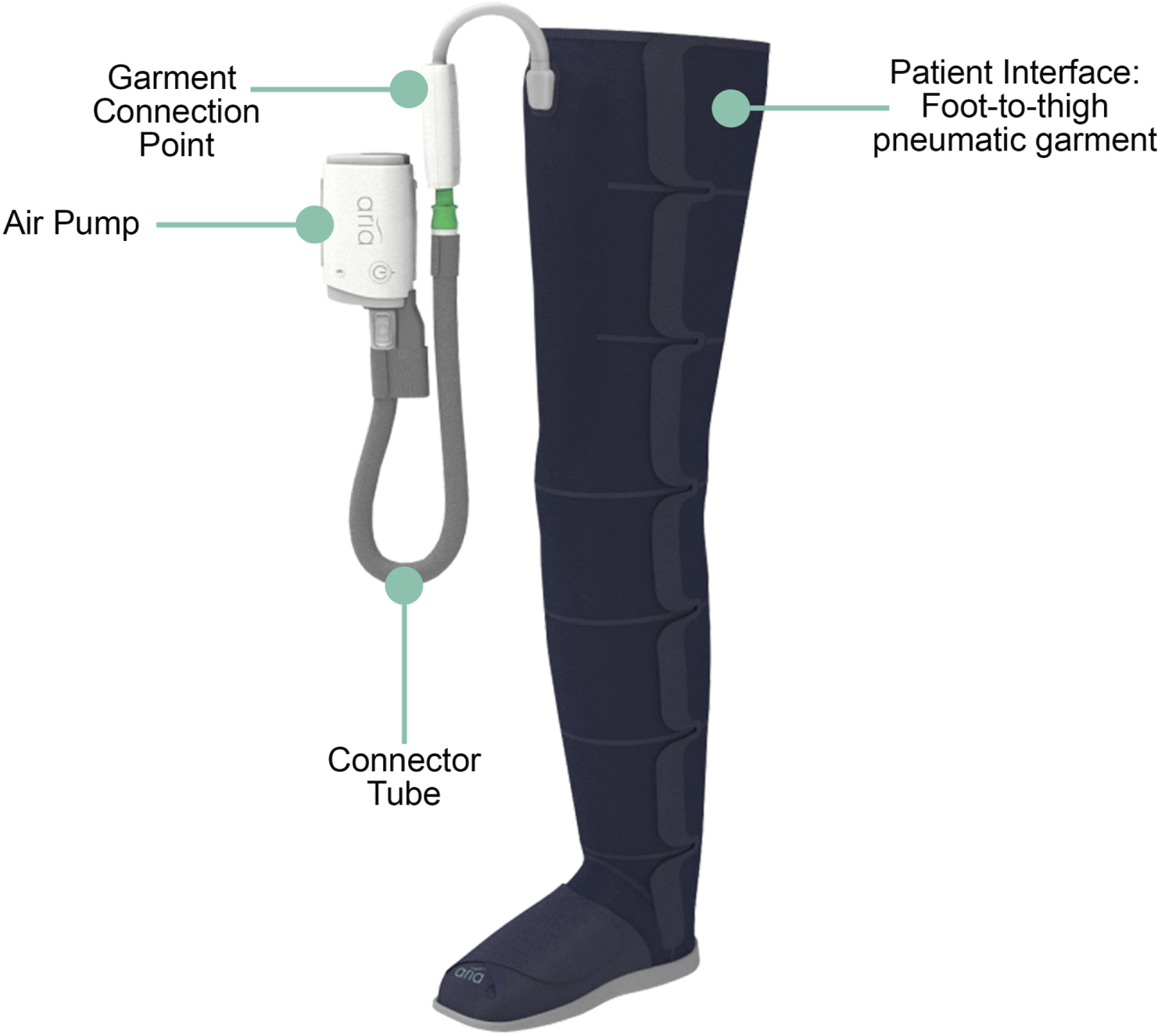
For this study, the therapy parameters were configured to deliver a 45-min session of therapy where the chambers were inflated to a pressure of 35 mmHg progressing from the distal to proximal end of the limb.
During each of the two study visits, participants were shown the study equipment and given the device in its packaged state. They were then asked to take the device out, fit it to their lower limb, then remove it. If lymphoedema was present in both lower limbs, the participant was given the choice of which leg to apply the garment. In phase 1, removal was immediate, whereas during phase 2 removal took place after 45 min of therapy.
Assessments
Prior to device usage in both study phases, non-identifying photos of the participant’s lower limbs were taken 1 m away from the participant to display the geometry of the lower limb, and limb size was determined by tape measure circumferential measurements at defined intervals from the distal medial malleoli, apex and base of patella and calculated using the truncated cone formula (Figure S1). 20 After the patient had fitted the device, lower limb photos were repeated, then patients removed the device and completed a questionnaire (Table S1 [phase 1] & Table S2 [phase 2]). In phase 2, patients also completed a pre-study questionnaire (Table S3), and turned the device on and left it on for 45 min prior to removal. At the end of both phases, participants underwent a post-study qualitative interview with the principal investigator (M.D.), with or without a design engineer, about their experience with fitting (phase 1) or fitting and using (phase 2) the pneumatic compression device (see online supplement for details of questions asked).
Within all questionnaires, device fit, usability and comfort were determined using an 11-point Likert scale, from 0 (worst score/unfavorable) to 10 (best score/favorable).
Adverse events, including those related to the device, were recorded during the study. An event was defined as related to the pneumatic compression device if it resulted from insufficiencies or inadequacies in the instructions for use, deployment, implantation/installation or operation, or any malfunction of the investigational medical device (this included any event resulting from usage error or intentional misuse).
Objectives
The primary objective of phase 1 was to evaluate the subjective fit of the pneumatic compression device; subjective ease of use and comfort were secondary endpoints. The primary objective of phase 2 was to assess the subjective comfort of the pneumatic compression device over 1 h of use; subjective ease of use and fit were secondary endpoints.
Statistical analysis
For this pilot usability study, the goal was to enroll 10 patients. The anticipated number of dropouts was zero, but it was determined that 90% of usability issues could still be identified if up to 2 participants dropped out of the study. 21 Likert scale scores were compared against a reference score of 6, a method used in previous medical device usability studies, where a score ≥6 is considered ‘acceptable’, as a score of 5 is the midpoint of the scale.22,23 The null hypothesis was that the score for the new device would be 6; if scores were >6 or <6 the product was better or worse, respectively, than the defined criterion. The rating for each assessment item was evaluated using the Wilcoxon Signed Rank Test. ‘Pass’ was defined as a significant p-value and a positive direction of change or p-value not significant, and ‘Fail’ was defined as a significant p-value and a negative direction of change.
Results
Study population
Participant demographic and clinical characteristics at baseline.
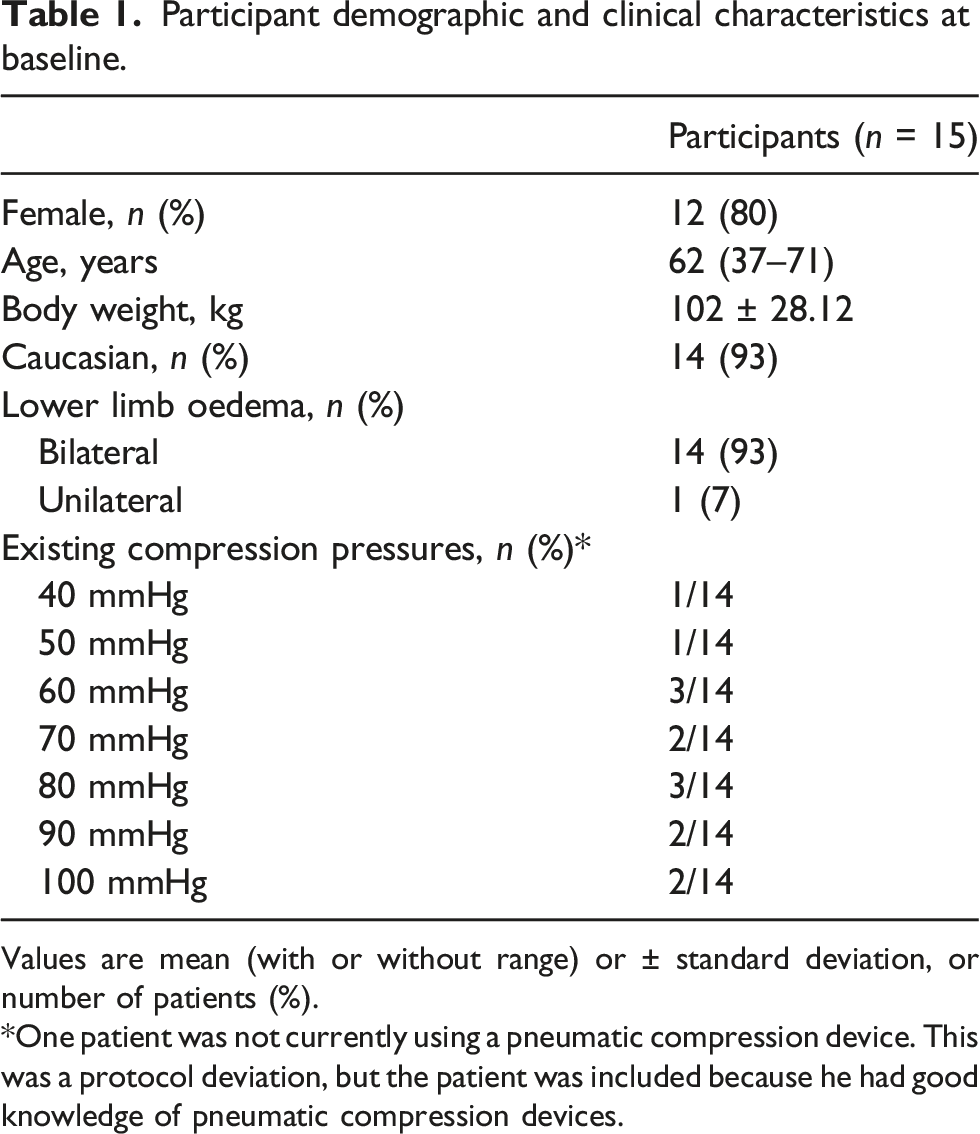
Values are mean (with or without range) or ± standard deviation, or number of patients (%).
*One patient was not currently using a pneumatic compression device. This was a protocol deviation, but the patient was included because he had good knowledge of pneumatic compression devices.
Phase 1
Likert scale scores for fit (phase 1) and comfort (phase 2) of the new pneumatic compression device system (on an 11-point scale from 0 [worst score/unfavorable] to 10 [best score/favorable]).
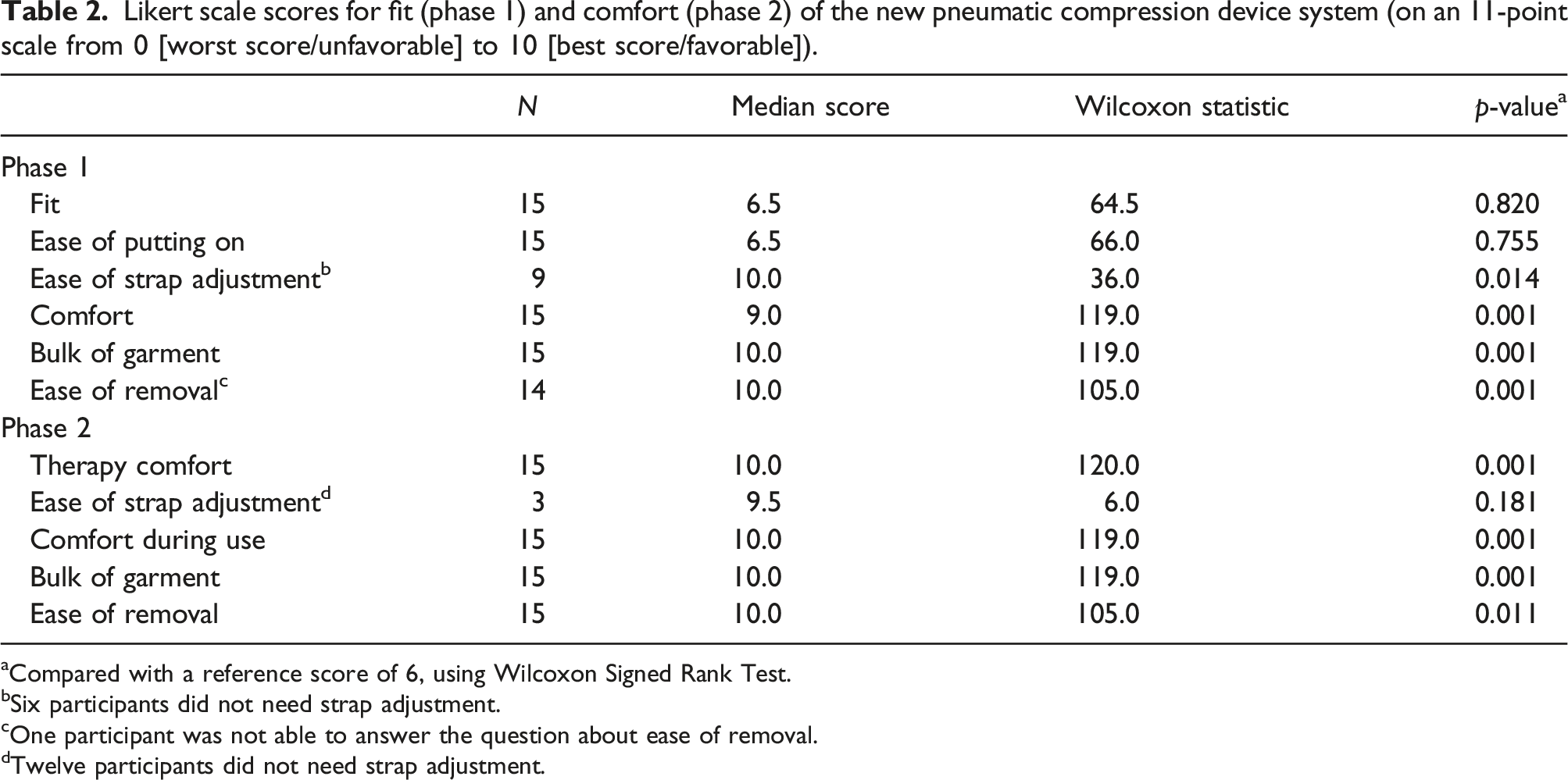
aCompared with a reference score of 6, using Wilcoxon Signed Rank Test.
bSix participants did not need strap adjustment.
cOne participant was not able to answer the question about ease of removal.
dTwelve participants did not need strap adjustment.
Phase 2
All participants rated the pneumatic compression device with a score of 10 for comfort (p = 0.001 vs. reference score of 6) (Table 2). Ease of use, comfort, bulk of garment and ease of removal scores were also significantly greater than the reference (Table 2). No patients reported any discomfort during device usage for 45 min. Mean percentage Change from Baseline of limb volume was −1.85% (p = 0.018) after 45 min of therapy. Positive comments about the new system related to the comfort of the fabric used in the garment, the small size of the air pump and system portability. Suggested improvements related to sizing, configuration of the foot piece, and ability to adjust more features (e.g. pressure). Five (5) participants volunteered feedback that they would purchase the new PCD if it was available.
There were no adverse events during the study.
Four (4) device deficiencies occurred: air pump would not turn on (remedied by correcting misalignment of the connector, allowing therapy to continue); 1 air chamber not fully inflating (therapy was able to be continued); disconnection at garment end (device replaced); and material separated around the lateral malleolus region (area reinforced with an extension strap). All of these were considered to be minor.
Discussion
This preliminary usability study showed that patients with lower limb lymphoedema found the new PCD easy to use and comfortable, and that the system was safe. All patients stated that they were generally happy with the design of the new device and, unprompted, one-third of patients also said they would be happy to use the device.
This study was designed to evaluate the usability of the pneumatic compression device system, not the therapeutic benefits associated with its use. Therefore, additional studies are required to evaluate the effects of ongoing therapy with the device on lower limb volume, other objective efficacy parameters, and patient-reported outcomes such as quality of life in patients with lymphoedema of the lower limbs. Nevertheless, the reduction in mean limb volume of 1.85% seen after a single 45-min treatment session seen in the current study is promising. A systematic review found limited evidence for conclusions regarding the minimal clinically important difference (MCID) in lymphoedema management and none of the included studies examined lower limb volume reduction from a single therapy session. 20 A Distribution-based MCID for scaled standard error of measurement (SEM) of 1% is recommended for the lower limb volume method used in this study but this is not necessarily clinically meaningful. 24 A leg volume increase of greater than 5% when measured by a perometer has been used as the threshold for referral for evaluation of possible lymphoedema flare-up. 25
Physical device attributes of the new pneumatic compression device system and existing recognized devices in the US market. a
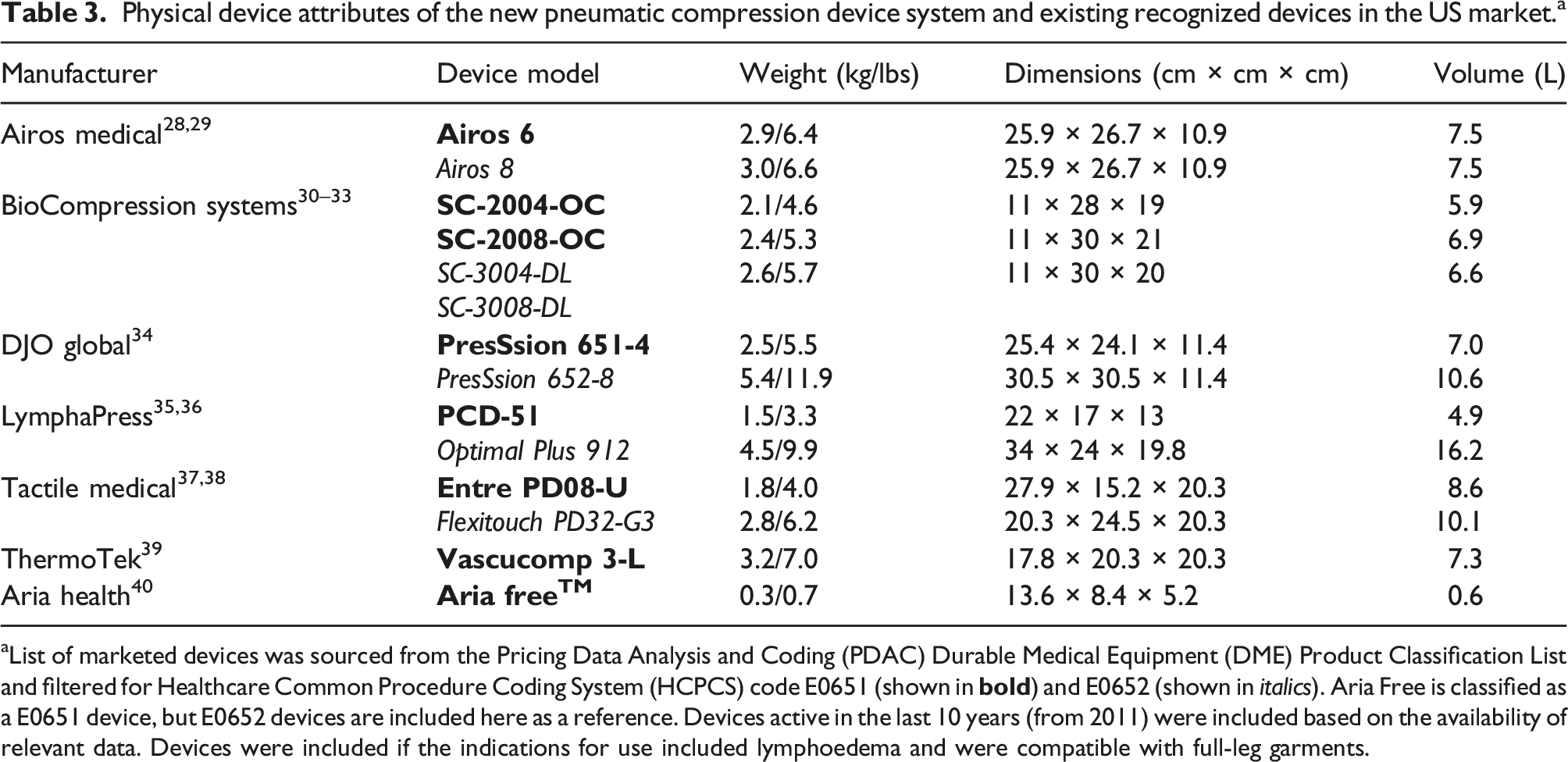
aList of marketed devices was sourced from the Pricing Data Analysis and Coding (PDAC) Durable Medical Equipment (DME) Product Classification List and filtered for Healthcare Common Procedure Coding System (HCPCS) code E0651 (shown in
Overall, there is a lack of robust data for the use of PCDs in lymphoedema and the quality of existing studies is low. 19 As PCD therapy can be self-administered in the home it is important that the individual patient experience is assessed.8,26 Two previous studies examined usability of PCD for lymphoedema: one included lower limb therapy and the other, treatment of the head and neck.25,27 Both utilized a rating scale to determine device usability. This was also the approach taken in our study, which found that the use of the new PCD system was both comfortable and feasible for a single session.
A key strength of the current study is that it is the first to investigate use of a novel compact PCD system in patients with lower limb lymphoedema. However, there are several limitations that need to be taken into account when interpreting the current findings. The number of patients studied is small, and the trial was conducted in a controlled clinical setting (rather than at home, which is where the device would mostly be used). In addition, this preliminary study had an uncontrolled design and used qualitative endpoints, meaning that the findings may be subject to bias. Furthermore, the study population included patients who had previous experience with PCDs, meaning that the results cannot be generalized to patients with lymphoedema who have not previously used PCDs.
Conclusions
This clinical evaluation supports the usability of the novel compact pneumatic compression garment with respect to both subjective fit and comfort during use. The features of this PCD system have the potential to address the limitations of currently available devices. Additional studies are needed to determine the effectiveness of the new PCD device, but preliminary data support its feasibility, acceptability and safety for potential use as part of the multidisciplinary management of lower limb lymphoedema.
Supplemental Material
Supplemental Material - Usability evaluation of a novel compact pneumatic compression device for the treatment of lymphoedema: Pilot study
Supplemental material for Usability evaluation of a novel compact pneumatic compression device for the treatment of lymphoedema: Pilot study by Martin Devitt, Dinesh Ramanan, Jeff Armitstead, Xueling Zhu and Helen Mackie in Phlebology.
Footnotes
Acknowledgements
Medical writing assistance was provided by Nicola Ryan, independent medical writer, funded by Aria Health.
Author contributions
M.D., D.R. and J.A. researched literature and conceived the study.
M.D., D.R. and X.Z. were involved in protocol development, gaining ethical approval, patient recruitment and data analysis.
H.M. provided clinical advice and was involved in patient recruitment.
M.D. wrote the first draft of the manuscript with assistance from a medical writer.
All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.D. is a paid independent consultant to ResMed Ltd, the parent company of Aria Health. D.R. is an employee of Aria Health. J.A. and X.Z. are employees of ResMed Ltd, the parent company of Aria Health.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by ResMed Ltd, the parent company of Aria Health.
Ethical approval
The study protocol was approved by an independent ethics committee (Bellberry Limited; ResMed Objective ID: A4778495). Human Research Ethics Committee Approval Application Number 2019-02-132.
Guarantor
M.D. takes full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
