Abstract
Objectives
This survey study evaluates current management strategies for venous ulceration and the impacts of the EVRA trial results.
Methods
An online survey was disseminated to approximately 15000 clinicians, through 12 vascular societies in 2018. Survey themes included: referral times, treatment times and strategies, knowledge of the EVRA trial and service barriers to managing venous ulceration. Data analysis was performed using Microsoft Excel and SPSS.
Results
664 responses were received from 78 countries. Respondents were predominantly European (55%) and North American (23%) vascular surgeons (74%). Responses varied between different countries. The median vascular clinic referral time was 6 weeks and time to be seen in clinic was 2 weeks. This was significantly higher in the UK (p ≤ 0.02). 77% of respondents performed surgical/endovenous interventions prior to ulcer healing, the median time to intervention was 4 weeks. 31% of participants changed their practice following EVRA. Frequently encountered barriers to implementing change were a lack of operating space/time (18%).
Conclusion
Venous ulcers are not managed as quickly as they should be. An evaluation of local resource requirements should be performed to improve service provision for venous ulceration. When interpreting the results of this survey consideration should be given to the response rate.
Background
Venous leg ulceration (VLU) affects an estimated 1% 1 of the population internationally. It costs approximately 2% of healthcare budgets in western societies 2 and has a significant impact on patient morbidity.3,4 There are no national guidelines for the treatment of VLU in England, however, the National Institute for Health and Care Excellence (NICE) guidance for varicose veins 5 suggest that patients with ulceration that persists for more than two weeks should be referred to a specialist vascular unit. Patients referred to vascular units often receive a duplex scan and, if indicated, surgical/endovenous ablation of superficial veins. The ESCHAR 6 study suggested that there was reduced ulcer recurrence in patients who received compression with surgery as opposed to compression alone. Consequently, surgery was frequently performed once ulcers had healed to prevent recurrence. This is further reflected in European and American guidelines.7–9 The Early Venous Reflux Ablation (EVRA) ulcer trial 10 identified that patients who underwent early endovenous ablation had improved ulcer healing rates and ulcer free time. The implementation of both the NICE guidelines on referrals and the EVRA study results are likely to be challenging; indeed it has been shown that patients with ulcers are not referred to specialist care within the 2 week limit in the UK.11,12
This survey study aims to determine the standards of the global management of patients with VLU, four to six months after the release of the EVRA trial results.
Methods
This descriptive, cross-sectional study utilised an online survey to collate VLU practices and perspectives. The initial survey was designed using the Qualtrics management platform (Qualtrics, Utah, USA) following discussion of important themes in a focus group with three vascular clinicians. Themes included: time from VLU development to secondary care referral and clinic visit, understanding of the NICE guidelines, knowledge of the EVRA study trial results and the impact these have had on practice. Additional information on clinician demographics was collected. The survey underwent five rounds of revision following review and feedback by a panel of vascular surgeons. The final online survey (Appendix, Supplementary material), consisting of 11 questions, was piloted on an additional five vascular surgeons prior to dissemination. The survey was circulated to approximately 15,000 participants through 12 national and international vascular societies. Reponses were collected over a four-month period between September 2018 and December 2018.
Outcomes of interest included referral time, time to vascular clinic review, aimed and actual time to surgical/endovenous intervention, whether interventions were performed before or after ulcer healing, whether EVRA trial results had changed practice, what the anticipated and actual barriers to implementing change were and views on the clinical and cost effectiveness of the EVRA trial findings.
Referral time was defined as the time between a patient’s first presentation to primary care for a venous ulcer to the date that they were referred to a vascular service. Time to vascular clinic review was defined as the time between the vascular clinic referral and first being seen in vascular clinic. Surgical/endovenous interventions encompassed all methods of superficial venous ablation. Time to intervention was defined as the time from the clinical decision to proceed with intervention to the patient undergoing this.
Descriptive statistics and normality were calculated using Microsoft Excel to analyse the results. All evaluated outcomes were not normally distributed and summarised using medians and interquartile ranges (IQRs). Categorical variables were summarised using frequencies and percentages. Free text in survey responses were categorised by common themes for the ease of interpretation. A Mann-Whitney U test in SPSS was used to compare differences between the UK and global responses.
Results
664 responses were received from 78 countries giving an approximate response rate of 4.4%. Respondent characteristics are summarised in Table 1. Respondents were predominantly vascular surgeons (74.2%). Most clinicians worked in Europe (54.5%) or North America (23%). Of 659 respondents, 454 (69%) had heard of the EVRA trial and 415 (63%) were familiar with the results. Of 627 respondents, 82% believed early surgical/endovenous intervention would reduce recurrence rates, 0.3% thought it would increase them, 13% thought there would be no effect, 3.7% thought it would depend on other factors such as the age of the ulcer, or extent of deep disease.
Baseline characteristics of respondents.

*Algeria (n = 1), Albania (n = 2), Argentina (n = 10), Australia (n = 19), Austria (n = 7), Bahrain (n = 1), Bangladesh (n = 1), Belarus (n = 1), Belgium (n = 12), Bolivia (n = 1), Bosnia (n = 1), Brazil(n = 40), Bulgaria (n = 7), Canada (n = 6), Chile (n = 1), China (n = 1), Colombia (n = 4), Costa Rica (n = 1), Croatia (n = 3), Cuba (n = 1), Czech Republic (n = 4), Denmark (n = 2), Ecuador (n = 2), Egypt (n = 5), El Salvador (n = 1), Finland (n = 4), France (n = 5), Georgia (n = 1), Germany (n = 22), Greece (n = 12), Honduras (n = 1), Hong Kong (n = 1), Hungary (n = 3), India (n = 11),Indonesia (n = 2), Iraq (n = 1), Ireland (n = 6), Israel (n = 4), Italy (n = 37), Japan (n = 2), Kenya (n = 1), Kosovo (n = 1),Kuwait (n = 1), Latvia (n = 2), Lithuania (n = 3), Malaysia (n = 1), Mexico (n = 19), Monaco (n = 1), Montenegro (n = 1), Netherlands (n = 8), New Zealand (n = 5), Norway (n = 4), Pakistan (n = 2), Palestine (n = 1), Paraguay (n = 1), Peru (n = 1), Poland (n = 8), Portugal (n = 23), Romania (n = 3), Russia (n = 9), Serbia (n = 3), Slovakia (n = 5), Slovenia (n = 6), South Africa (n = 8), South Korea (n = 1), Spain (n = 20), Sri Lanka (n = 2), Sweden (n = 10), Switzerland (n = 5), Taiwan (n = 1), Thailand (n = 9), Turkey (n = 6), Ukraine (n = 3), United Kingdom (n = 108), USA (n = 127),Uruguay (n = 1), Missing (n = 3)
Current intervention
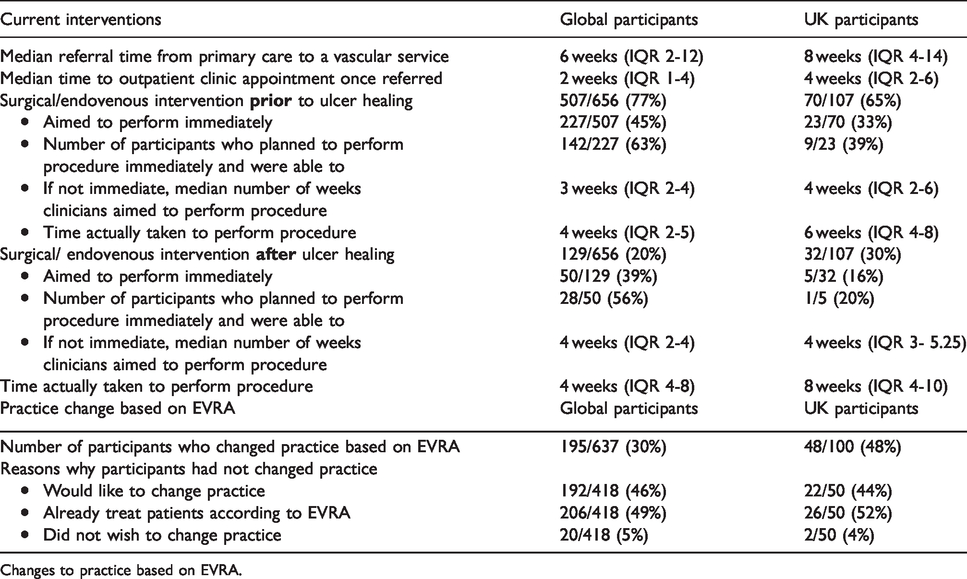
Globally, the median referral time from primary care to a vascular service was six weeks (IQR 2-12 weeks); this was longer in the UK where the median time was 8 weeks (IQR 4-14 weeks), p = 0.02. The median time to outpatient clinic appointment once referred was 2 weeks (IQR 1-4 weeks); increasing to 4 weeks in the UK (IQR 2 - 6), p < 0.01.
Of 656 global respondents, 507 (77%) reportedly performed surgical/endovenous intervention prior to ulcer healing, 129 (20%) after ulcer healing and 20 (3%) did not perform surgical/endovenous intervention for ulceration. Of the 507 global respondents who treated prior to ulcer healing, 227 (45%) aimed to perform the intervention immediately, of these, 142 (63%) were able to do this. 280 (55%) of global respondents did not aim to immediately perform intervention and instead aimed-to-treat at a median time of 3 weeks (IQR 2 to 4). The median time to actually treating patients was 4 weeks (IQR 2 to 5).
Of the 129 global respondents who treated after ulcer healing, 50 (39%) of participants would aim to perform the intervention immediately after healing, 28 (56%) of these participants were able to. For those who did not aim-to-treat immediately, the median aim-to-treat time was 4 weeks (IQR 2 to 4) after ulcer healing. The median time from healing to actual intervention was 4 weeks (IQR 4 to 8).
Of 107 UK respondents, 70 (65%) usually performed surgical/endovenous intervention prior to ulcer healing, 32 (30%) after and 5 (5%) did not perform intervention. Of the 70 respondents who treated prior to ulcer healing, 23 (33%) would aim to perform intervention immediately. However, only 9 (39%) of these were able to do so. For those who did not aim to treat immediately, the median aim-to-treat-time was 4 weeks (IQR 2 to 6). The median time to treatment was 6 weeks (IQR 4 to 8).
Of the 32 UK respondents who treated after ulcer healing, only 5 (16%) would aim to treat immediately after ulcer healing. Only one respondent was able perform the intervention immediately. For those who did not aim-to-treat immediately, the median recorded aim-to-treat time was 4 weeks (IQR 3 to 5.25) after ulcer healing. The median time from healing to treatment was 8 weeks (IQR 4 to 10).
Changing practice
Clinical practice before and after EVRA are described in Table 2. 195 (30%) of global respondents (n = 637) reported they had changed practice with respect to the timing of intervention based on the results of the EVRA study, 418 (66%) did not change practice and 24 (4%) did not answer the question. Of the 418 global participants who did not change practice, 192 (46%) stated that they would like to, 206 (49%) stated that they already treat prior to ulcer healing and 20 (5%) participants did not wish to change.
Current practices for surgical/endovenous interventions for venous ulcers.
Changes to practice based on EVRA.
In the UK, of 100 respondents, 48% stated that they changed their practice with respect to the timing of intervention based on the EVRA results, 50% did not and 2% did not answer. Of the 50 respondents who did not change following EVRA, 22 (44%) indicated that they would like to, 26 (52%) stated they already treat prior to ulcer healing and 2 (4%) participants did not wish to change.
Barriers to changing practice
195 (31%) global respondents stated they had changed practice with respect to the timing of intervention based on the results of EVRA study. The barriers faced by the respondents in changing practice are summarised in Table 3. Respondents could select more than one barrier and therefore 347 barriers were recorded in total. The most frequently anticipated and encountered barriers to implementing EVRA were a lack of operating space/time (18%) and a lack of theatre space (15%). A fifth of participants felt there were no barriers to changing practice.
Clinician perspectives on the barriers of implementing EVRA in normal practice.

Clinicians who have already implemented EVRA listed the actual barriers they experienced doing this. Clinicians who have not yet implemented EVRA listed the barriers that they anticipated would arise.
192 (43%) global respondents stated they had not changed practice with respect to the timing of intervention based on the results of the EVRA study but would like to. These respondents listed their anticipated barriers (Table 3).
62% of respondents stated that the cost effectiveness results would alter how they made clinical decisions and 30% said they would not have any impact. 4% stated that this would depend and 4% said the decisions are made by someone else such as Clinical Commissioning Groups (CCGs) in the UK.
Discussion
The survey responses generated from this study provide helpful insights into the global management of venous leg ulceration post the EVRA trial. 10
Globally, the median time to referral was 6 weeks, increasing to 8 weeks in the UK. This is longer than the recommendations issued by NICE. 5 Reasons for this could include education in primary care, 13 ease of referral, access to secondary care services and patient preference. Recent evidence suggests that most CCGs commission this service in the UK 14 and measures are being taken to improve referral access from primary to secondary care. 15
This survey has additionally shown that there are some perceived constraints in secondary care with approximately only 60% of participants feeling they are able to perform intervention in the time frame in which they hope to. This is reflected when evaluating perceived barriers to implementing EVRA as many respondents cited a lack of theatre space or time as barriers to care.
A minority of clinicians chose to perform surgical/endovenous intervention after ulcer healing. Although evidence from randomised control trials indicates that surgical/endovenous intervention can help promote venous ulcer healing, 10 this is not reflected in the UK, 5 European 9 or American guidelines. 8 The results of this survey suggest that most clinicians would align their practice with the EVRA trial results. The EVRA trial results should be reflected in national and international guidelines to better guide clinical practice.
This survey suggests that the UK comparatively has significantly longer referral times, longer waiting times to secondary care and longer times to intervention. This is supported elsewhere where it is suggested that the UK is possibly undertreating patients with chronic venous disease. 16 This may relate to relative constraints of the National Health Service compared to other privatised health care systems globally.
The survey was predominantly completed by vascular surgeons working in academic units in Europe; this selection bias could lead to inadequate representation of the care administered in other types of units. Although the number of respondents was high, there was a low overall response rate; this again could contribute to a selection bias. The survey was only performed 6 months after the EVRA trial results were published; the impact of the EVRA trial may become more pronounced with time. It is also important to note that this survey evaluates subjective clinician perspectives on how venous ulceration is currently managed; further work evaluating additional objective measures should be performed.
Conclusion
Evaluating clinician perspectives has provided helpful insights into the current management of venous ulceration and what impacts EVRA may have had on this. This survey has identified that although many clinicians are aware of EVRA, there may be a number of barriers in implementing its findings to clinical practice. The healthcare structures in each geographical region may vary significantly. Given the low response rate, a more in-depth evaluation of the barriers to achieving and delivering best practice care should be performed in each local region to advance service provision.
Supplemental Material
sj-pdf-1-phl-10.1177_0268355520966893 - Supplemental material for The management of venous leg ulceration post the EVRA (early venous reflux ablation) ulcer trial: Management of venous ulceration post EVRA
Supplemental material, sj-pdf-1-phl-10.1177_0268355520966893 for The management of venous leg ulceration post the EVRA (early venous reflux ablation) ulcer trial: Management of venous ulceration post EVRA by Safa Salim, Francine Heatley, Layla Bolton, Amulya Khatri, Sarah Onida and Alun H Davies in Phlebology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Infrastructure support for this work was provided by the NIHR Imperial Biomedical Research Centre.
Ethical approval
No ethical approval or patient consent was required for this article.
Guarantor
AHD.
Contributorship
FH and AHD conceived the research idea. FH and AK designed the questionnaire. FH performed the data collection and data analysis via Excel. SS performed the data analysis via SPSS. SS wrote the manuscript, supported by FH, SO and AHD. All authors commented on the manuscript.
Acknowledgements
The authors would like to thank the following societies for enabling dissemination of the survey: American Venous Forum (AVF), American College of Phlebology (ACP), Veith Symposium, Venous-lymphatics World International Network foundation, ONLUS, Vascular Society of Great Britain & Ireland (VSGBI), Venous Forum UK, Venous news / Charing Cross Symposium, The Australasian College of Phlebology (ACP), International Union of Phlebology (UIP), Venous Association of India (VAI), European Venous Forum (EVF), European Society of Vascular Surgery (ESVS), Turkish / Mexican / Baltic mailing lists. The authors would like to additionally thank Mr Colin Bicknell, Mr Manjit Gohel, Mr Joseph Shalhoub, Ms Marina Kafeza and Mr Roshan Bootun. The authors also acknowledges the support of Department of Vascular Surgery, Imperial College Healthcare Trust.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
