Abstract
Introduction
No existing patient-reported outcome instrument focuses solely on assessment of varicose veins symptoms that are bothersome to patients.
Methods
The VVSymQ® instrument is a five-item patient-reported outcome that assesses symptoms most important to patients with varicose veins (heaviness, achiness, swelling, throbbing and itching). This paper describes how the VVSymQ® instrument was incorporated into an electronic daily diary to monitor key outcomes over time and capture treatment benefit in two randomized, controlled, phase 3 clinical trials.
Results
Patients were highly compliant in completing the electronic daily diary, and the VVSymQ® instrument demonstrated ability to detect overall change and ability to detect change that is meaningful to patients.
Conclusion
The VVSymQ® instrument is a reliable, valid instrument responsive to measuring change in the patient experience of varicose vein symptoms pre- and post-intervention, and is uniquely focused on patient-reported symptoms compared with other widely used questionnaires completed by clinicians.
Introduction
Varicose veins are extremely common, affecting up to 73% of women and up to 56% of men. 1 While varicose veins can be unsightly, patients who suffer from this condition, especially as it progresses over time, are not only concerned about their appearance but also report significant symptoms stemming from varicose veins. 2 Interviews of patients with varicose veins confirm that these symptoms, typically including heaviness, achiness, swelling, throbbing and itching, are experienced on a regular basis. 2 Further, symptoms often lead to a reduction in health-related quality of life (HRQoL).3,4 Not surprisingly, as venous disease increases in severity, there is a concomitant increase in symptoms and an increasing impact on HRQoL.3,4 Despite the importance of symptoms and their impact on HRQoL, there has not been a systematic focus on assessing symptoms of varicose veins in clinical research. In the development program for polidocanol endovenous microfoam (Varithena®; Provensis Ltd, a BTG International group company), the need arose for a patient-reported outcome (PRO) instrument for evaluating symptoms that followed best practices for instrument development and validation and could be used in clinical trials. Historically, clinicians have characterized varicose veins primarily by signs and/or the degree of venous disease. Measures of venous disease severity include the Clinical classification portion of the Clinical, Etiology, Anatomy and Pathophysiology (CEAP) Classification of Venous Disorders and the Venous Clinical Severity Score (VCSS).5–8 These assessment tools are administered and scored by clinicians following an examination of the patient and do not systematically evaluate symptoms from the patient's perspective.
In order to directly evaluate the patient's experience, a few PRO instruments that are completed solely by the patient have been developed, including the Aberdeen Varicose Veins Questionnaire (AVVQ), the Venous Insufficiency Epidemiological and Economic Study instrument (VEINES-QOL/Sym), the Specific Quality of Life and Outcome Response – Venous questionnaire (SQOR-V), and the Chronic Venous Insufficiency Quality of Life Questionnaire (CIVIQ).9–12 Each of these evaluates several aspects of the impact of venous disease on patients, and do not focus solely on the key symptoms of varicose veins for which they seek treatment. For instance, the AVVQ measures various concepts including symptoms, pain medication use, concern about appearance and the impact of varicose veins on clothing choice and HRQoL. The SQOR-V questionnaire measures symptom severity; pain intensity at different times of the day; disease progression in the last year; and impact of vein problems on appearance of legs, clothing choice, and emotional, physical, role, sleep, sexual and social function. The VEINES-QOL/Sym instrument is used to assess a broad range of chronic venous disorders, symptoms, and HRQoL domains and is, therefore, not specific enough to measure change in symptoms for patients with varicose veins. 4 Finally, the CIVIQ-20 instrument measures a single symptom (ankle/leg pain severity), and remaining items assess the impact of leg problems on usual activities, physical, role, social, sleep, and emotional function.
In addition, most of the existing PROs for varicose veins rely on retrospective recall of symptoms experienced in the “past 2 weeks” (AVVQ) or “past 4 weeks” (VEINES-QOL/Sym, CIVIQ-20), which may not be an ideal data collection method to assess symptoms that can change in the course of a day or a week (i.e. retrospective symptom questionnaires may be subject to poor recall).
Further, most of the existing PRO instruments have not followed best practices for instrument development and validation; one key source for guidance on such best practices is the United States Food and Drug Administration's (FDA) guidance on PRO development. 13 For example, a current central best practice in instrument development is establishing content validity through interviewing patients about the condition, and then testing the instrument for comprehension and relevance in patients. Of these historic PROs, only the CIVIQ-20 instrument appears to have had development activities in alignment with FDA-recommended best practices for PRO instrument development, 13 but the CIVIQ-20 is not a direct measure of all key symptoms found to be important and relevant to patients with varicose veins. 2
The VVSymQ® instrument is a novel five-item PRO instrument specifically developed in accordance with the FDA Guidance for PROs, to evaluate varicose vein symptoms from the patient's perspective in clinical trials. 13 The VVSymQ® instrument focuses on symptoms most relevant to patients with varicose veins, and it was constructed by following instrument development best practices including direct patient input.2,13 The VVSymQ® instrument was evaluated in three studies of patients with varicose veins: a VVSymQ® instrument validation study and two phase 3 clinical trials.2,14,15 In the validation study, the VVSymQ® instrument was found to be reliable and valid. 2 This paper summarizes a number of findings regarding how the VVSymQ® instrument performed when it was used in two phase 3 clinical trials that evaluated a novel treatment, and its potential usefulness to clinical researchers and clinicians.
Methods
The relevant institutional review boards approved the protocols, and all patients gave written informed consent for their participation in the study.14,15
Procedure
The VVSymQ® score was used to assess the primary efficacy endpoint in two randomized, controlled phase 3 clinical studies (VANISH-1 & VANISH-2) evaluating microfoam ablation with polidocanol endovenous microfoam in patients with superficial venous insufficiency (for detailed presentation of the methods and efficacy results, see King et al. 15 and Todd et al. 14 ). The studies had virtually identical study designs and compared a vehicle placebo with varying dose concentrations of polidocanol endovenous microfoam (0.125% control, 0.5%, 1.0%, and 2.0% in VANISH-1, and all but the 2.0% dose in VANISH-2). Because the primary efficacy analysis was conducted at Week 8 post-treatment, the results presented here will focus on Baseline and Week 8.
Questions, responses, and method of scoring in the VVSymQ® questionnaire.

Additional assessment tools were administered at screening and/or Baseline and Week 8 that are relevant for the evaluation of patients with varicose veins, including the central Independent Photography Review by clinicians (IPR-V3); the Patient Self-Assessment of Varicose Veins (PA-V3); duplex ultrasound response; the physician-assessed VCSS, a measure of disease severity; and the VEINES-QOL, the quality-of-life component of the Venous Insufficiency Epidemiological and Economic Study instrument (VEINES-QOL/Sym), which focuses on the impact of varicose veins. The IPR-V3 and the PA-V3 instruments were validated in the development program for polidocanol endovenous microfoam and shown to be reliable. 16 Finally, the Patient Global Impression of Change (PGIC) questionnaire, administered at Week 8, was used to evaluate clinically meaningful change in the VVSymQ® score. The PGIC used was modelled after the widely used PGIC for pain intensity. 17
Patients
In both the VANISH-1 and VANISH-2 studies, patients were required to have symptomatic and visible varicose veins; a minimum score of ≥7 out of a possible 25 on the sum of the five symptoms that constitute the VVSymQ® score (reported on a paper version of the VVSymQ® for the previous week); and at least moderately noticeable (PA-V3) and moderately severe (IPR-V3) scores for the appearance of their varicose veins (a score of at least 2 out of 4). They were also required to have reflux >0.5 s (as measured by duplex ultrasound at the saphenofemoral junction) and incompetence of the great saphenous vein or other major accessory vein. The other inclusion and exclusion criteria were identical in the two studies.
VVSymQ® score analyses
In order to simplify presentation of the results, the patient samples from both the VANISH-1 and VANISH-2 studies were combined, and three of the polidocanol endovenous microfoam arms were pooled: 0.5%, 1.0%, and 2.0%. (The 0.125% arm that was used for control was not included in the current analyses.) The specific analyses conducted on the VVSymQ® instrument were aimed at examining the following key questions:
Did patients complete the daily e-diary each evening, per protocol? Did the VVSymQ® score change following treatment? Was it sensitive to treatment? Was the change that patients experienced on the VVSymQ® score clinically meaningful to them? If so, what is the clinically meaningful level of change in the VVSymQ® score? How does the VVSymQ® score relate to other measures of varicose veins?
Compliance rates for the daily e-diary were calculated to evaluate compliance with the protocol requirements for the Baseline and Week 8 periods in this study.
Sensitivity to treatment was computed by determining whether change occurred between Baseline and Week 8 using mean change and effect size. While mean change demonstrates the absolute magnitude of change, the stronger metric of sensitivity to change is effect size. Cohen effect size is a normalized measure of change relative to an instrument's variability and is calculated by dividing the mean change by the standard deviation of the Baseline value for that instrument. 18 This provides a standardized metric of change that can be compared across studies, independent of sample size. The suggested reference values for the magnitude of effect size are: small, ≥ 0.2; medium, ≥ 0.5; and ≥ 0.8 large. 19
Clinically meaningful change was evaluated using an anchor-based approach. 20 For the anchor-based approach, mean change in VVSymQ® score from Baseline to Week 8 was computed for each of the seven levels of the PGIC instrument, a single-question PRO with a 7-point scale on which patients rated their impression of whether they believed they improved, worsened, or remained the same following treatment. The ratings on the PGIC are “much improved” (score +3), “moderately improved,” “a little improved,” “no change,” “a little worse,” “moderately worse,” and “much worse” (score –3). The typical threshold for clinically meaningful change using the PGIC is “moderately improved”. 20
Finally, the correlation between the VVSymQ® score and other measures, including the two appearance measures (IPR-V3 and PA-V3), the VCSS, VEINES-QOL, and elimination of reflux on duplex ultrasound response (non-responders were assigned a value of 0, and responders assigned a value of 1) was assessed to evaluate degree of overlap.
Results
Patient demographics
Patient demographics and screening/Baseline characteristics.
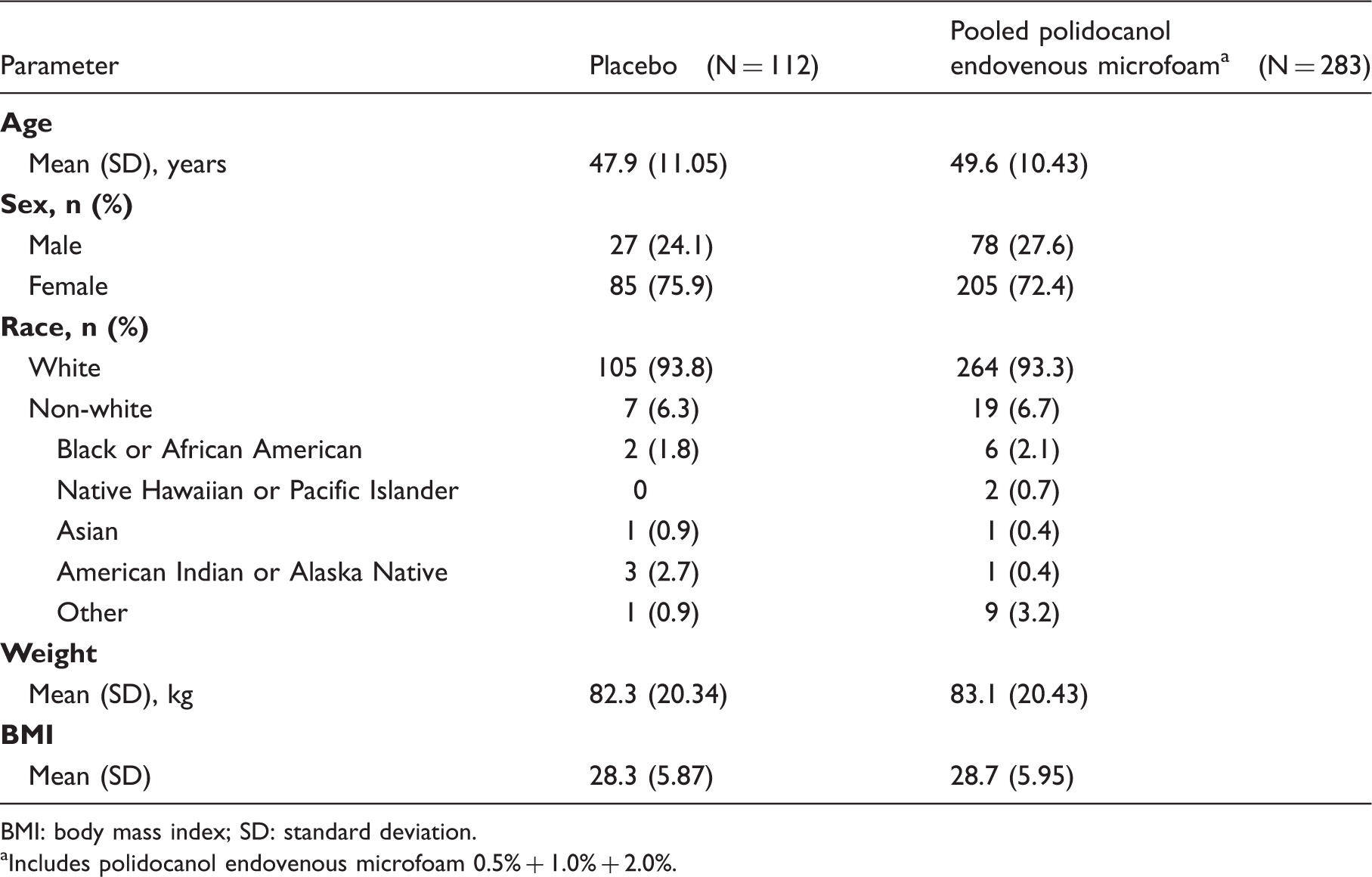
BMI: body mass index; SD: standard deviation.
Includes polidocanol endovenous microfoam 0.5% + 1.0% + 2.0%.
Compliance with the daily e-diary
Patient compliance with completing the daily e-diary in accordance with the protocol schedule was high. The mean compliance rate for the patient sample was 96.2% (n = 509, SD = 9.05) for the Baseline Week and 86.1% (n = 505, SD = 20.13) for Week 8.
VVSymQ® score sensitivity to treatment
Change from Baseline to Week 8 in the 7-day average VVSymQ® daily diary overall score and individual component symptom scores.
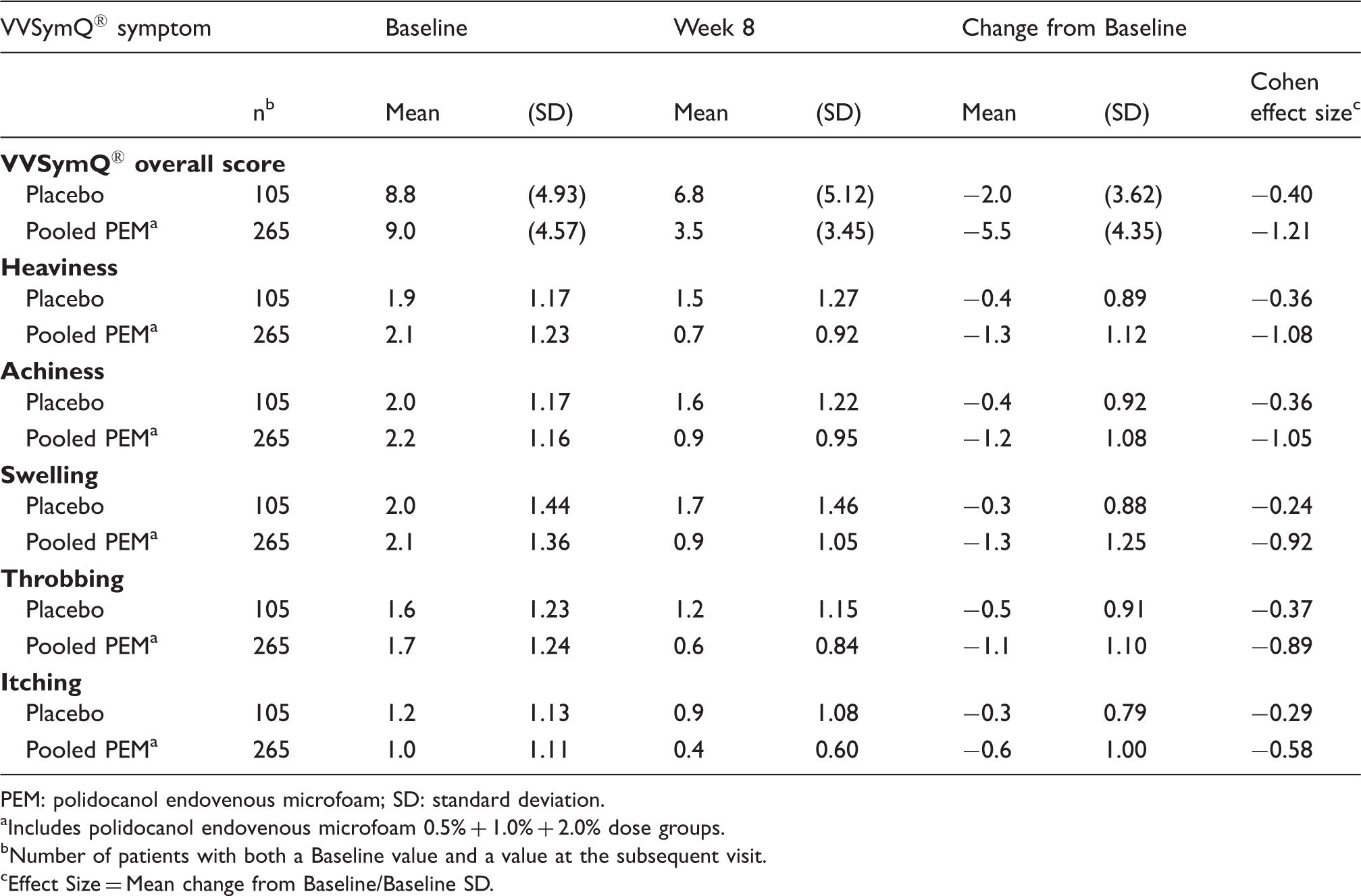
PEM: polidocanol endovenous microfoam; SD: standard deviation.
Includes polidocanol endovenous microfoam 0.5% + 1.0% + 2.0% dose groups.
Number of patients with both a Baseline value and a value at the subsequent visit.
Effect Size = Mean change from Baseline/Baseline SD.
The sensitivity of the VVSymQ® score to detect change after treatment was evaluated via Cohen effect size (Table 3). Following Cohen's guidelines for judging magnitude of effect size, the effect size was small to medium in the placebo group (−0.24 to −0.37) but large in the pooled polidocanol endovenous microfoam group (−0.58 to −1.08) for all of the individual symptom items. A similar trend was obtained for the VVSymQ® overall score, with the Cohen effect size −0.40 for placebo compared with −1.21 for the pooled polidocanol endovenous microfoam group. These findings are consistent with what would be expected for an instrument that was sensitive to change attributable to treatment.
Clinically meaningful change
Change from baseline to Week 8 in VVSymQ® scores across various levels of PGIC.
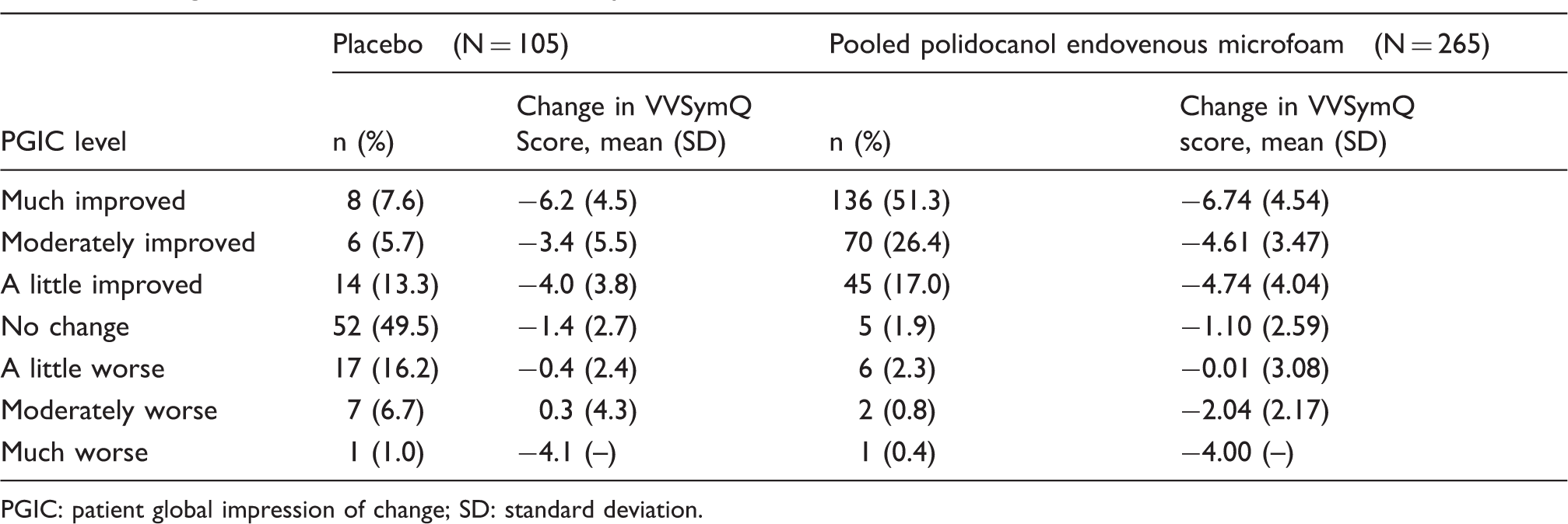
PGIC: patient global impression of change; SD: standard deviation.
Central to understanding the performance of the VVSymQ® score is the concept that changes in the clinical condition are reflected in instrument scores when patients have experienced an improvement. Table 4 demonstrates that the greatest level of Baseline-to-Week 8 change in the VVSymQ® score occurred when patients reported being “much improved” on the PGIC. Patients also showed notable changes from Baseline on the VVSymQ® when they were “moderately improved” on the PGIC. When patients reported “no change” or being “a little worse” on the PGIC, only small changes were observed in the VVSymQ® score (Table 4). The sample sizes for “moderately worse” and “much worse” on the PGIC were very small; therefore, the results are difficult to interpret.
VVSymQ® and other measures
Pearson correlations between VVSymQ® score and other measures.
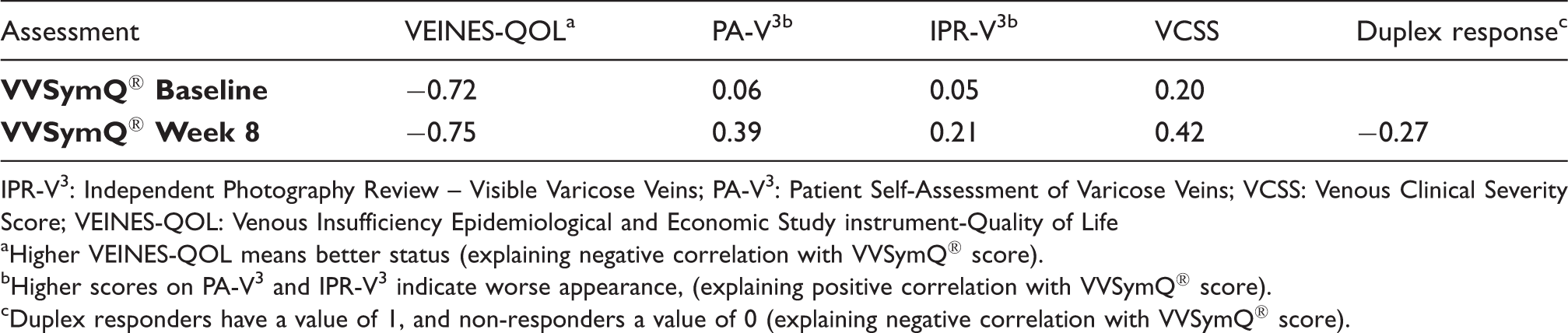
IPR-V3: Independent Photography Review – Visible Varicose Veins; PA-V3: Patient Self-Assessment of Varicose Veins; VCSS: Venous Clinical Severity Score; VEINES-QOL: Venous Insufficiency Epidemiological and Economic Study instrument-Quality of Life
Higher VEINES-QOL means better status (explaining negative correlation with VVSymQ® score).
Higher scores on PA-V3 and IPR-V3 indicate worse appearance, (explaining positive correlation with VVSymQ® score).
Duplex responders have a value of 1, and non-responders a value of 0 (explaining negative correlation with VVSymQ® score).
Discussion
The VVSymQ® instrument is the first PRO instrument developed in accordance with FDA guidelines to specifically evaluate varicose vein symptoms from the patient's perspective. Results from two randomized, controlled phase 3 clinical studies using the VVSymQ® instrument demonstrated high rates of compliance with the instrument and excellent sensitivity of the instrument to detect change, and indicated that the reflected change was clinically meaningful to patients (i.e. “moderately improved” or “much improved” on PGIC).
When developing a PRO instrument, an important consideration is whether patients will be willing and able to complete the instrument. Electronic daily diaries capture day-to-day fluctuations in symptoms, reducing the risk of recall bias and facilitating overall data quality. The rates of compliance clearly indicate that patients were able to easily use and comply with the protocol requirements for the electronic diary. The use of electronic versions of questionnaires has been shown to improve compliance, and electronic versions of instruments have correlated highly with paper versions (0.90 as the average weighted correlation), as described in a meta-analytic review performed by Gwaltney et al. 21
In clinical studies, as well as in the clinical setting, the effectiveness of treatment needs to be measured by an instrument designed to measure what it is intended to measure. If a treatment is successful, this will be demonstrated by changes in scores on that instrument. The VVSymQ® instrument was able to detect change in symptoms from Baseline to Week 8 in both VANISH studies, for the overall score as well as for the five individual symptoms (heaviness, achiness, swelling, throbbing and itching). These findings provide evidence that the VVSymQ® instrument is an effective tool for measuring pre-treatment symptoms and demonstrating treatment success.
Whether or not a treatment is successful in the eyes of a clinician or researcher, the ultimate goal is to treat patients in a way that is meaningful to them. To this end, in the VANISH-1 and VANISH-2 studies, the PGIC was used as a yardstick for determining whether or not the VVSymQ® score measured clinically meaningful change following treatment. The threshold for clinically meaningful change using the PGIC is “moderately improved”. 20 More patients reported having a meaningful change in the active treatment group than in the placebo group. Patients who responded that they were “moderately” or “much” improved on the PGIC had greater reductions in VVSymQ® scores than did patients who did not improve on the PGIC, indicating that the VVSymQ® score changed as expected in relation to patients' reporting of changes in their clinical condition. The VVSymQ® instrument detects clinically meaningful change.
The correlation of the VVSymQ® score to commonly used clinical measures shows the highest correlation to HRQoL, as expected. As the patients' symptoms improved, the impact of these symptoms on HRQoL, as measured by the VEINES-QOL instrument, also improved. The lowest correlations were observed between the VVSymQ® score and both the clinician rating of appearance and the duplex ultrasound response. The comparatively low correlations between these measures indicate that clinician rating and duplex ultrasound findings do not predict patient symptom response. The relative weakness of the duplex ultrasound response correlation might arise from the simplistic binary nature of the ultrasound findings (duplex responder or not), whereas the VVSymQ® score reflects symptom improvement on a graded scale. Similarly, the low to moderate correlations observed between the VVSymQ® and VCSS scores suggest that VCSS scores may characterize disease severity but do not predict patient symptom response.
Taken together, these findings suggest that the VVSymQ® instrument, with a focus on symptoms, is evaluating an aspect of varicose veins that is different from prior traditional measures of appearance or disease severity. In other words, to more completely understand patients with varicose veins, it may be best to evaluate symptoms and the impact of symptoms on patient HRQoL, in addition to disease severity and appearance. This recommendation marks a significant shift from historic measures that focused primarily on appearance scores, disease severity scores, and duplex ultrasound findings.
The VVSymQ® can be used for either unilateral or bilateral symptoms. When assessing bilateral symptoms, the patient completes one VVSymQ® questionnaire for one leg and a second questionnaire for the other leg. The scores from the two legs are evaluated independently, i.e. they are not summed.
The VVSymQ® instrument has been developed following the most up-to-date guidelines and fulfills an unmet need for an assessment of varicose veins based on patient reports of their symptom experience that can be used as part of an electronic daily diary to monitor symptoms and capture treatment benefit for individual patients.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The development of the VVSymQ® instrument was sponsored by BTG International Inc. JP, DTB, and CAE are current or former employees of ERT, which was contracted by BTG to develop the VVSymQ® instrument. DW is an employee of BTG International Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by BTG International Inc. Please direct inquiries about obtaining the VVSymQ® instrument to BTG International Inc., West Conshohocken, PA, USA.
