Abstract
Introduction
Due to anticoagulation during Extracorporeal Membrane Oxygenation (ECMO), patients face a higher risk of bleeding. This study aimed to assess the correlation between anticoagulation assays and hemorrhage in ECMO patients.
Methods
We reviewed records from patients in our critical care units, comparing anticoagulation parameters with clinical outcomes. Statistical analyses included the Wilcoxon rank sum test for continuous variables and chi-square or Fisher’s exact tests for categorical variables. We used Kaplan-Meier curves to depict hemorrhage incidence and logistic regression to identify factors associated with bleeding and mortality. Linear regression assessed coagulation factors linked to length of stay (LOS).
Results
Of 76 patients, 32 (42.1%) experienced hemorrhage; 15 (46.9%) of these survived. Survivors had a higher proportion without bleeding (68.2%). Hemorrhage patients had more circuit changes (18.8% vs 2.3%, p = .01). The 10-, 20-, and 30-days cumulative hemorrhage incidences were 36.9%, 58.8%, and 72.5%, respectively. Multivariable analysis suggested that bleeding patients had a higher aPTT prior to the bleeding event compared with the median aPTT in non-bleeding patients (p = 0.04), and ACT and PT levels showed a trend towards a significant association (p = 0.06). Longer ECMO runs and circuit changes increased hemorrhage risk (OR = 1.004 and OR = 12.85, respectively).
Conclusion
Our study found that higher aPTT and longer ECMO duration are independent hemorrhage risk factors, underscoring the need for timely ECMO decannulation.
Keywords
Introduction
Extracorporeal membrane oxygenation (ECMO) is a life-saving therapy employed in different disease processes. While ECMO has shown improved survival amongst patients with potentially poor prognosis, there is an increased risk for complications, including thrombosis and hemorrhage. 1 The etiology stems from various factors, ranging from intrinsic and extrinsic factors, the patient’s underlying medical condition, and the physiologic response to achieving oxygenation with a mechanical device. 2 Thus, anticoagulation is necessary to prevent thrombosis but must be balanced with the risk of bleeding. Children are at a higher risk of bleeding while on ECMO due to multiple factors.3–9 Several monitoring methods of anticoagulation have been employed, including a “clotting time” approach (i.e., Activated Clotting Time, ACT), Activated Partial Thromboplastin Time, aPTT), a “chromogenic” approach (i.e., Anti Activated Factor X, anti-Xa) or combination of both, along with vescoelastic assays (Thromboelastography (TEG) or Rotational thromboelastometry (ROTEM)). The Extracorporeal Life Support Organization (ELSO) has published guidelines for the management of anticoagulation on ECMO that detail the organization’s recommendations. 7 Our objective is to correlate different anticoagulation assays with hemorrhagic events of patients who received ECMO at our center and assess whether that correlation varies with the mode of ECMO support. Since we have a combined unit and continue to care for adults with congenital heart disease, those patients were also included in our analysis. We hypothesized that higher levels of aPTT, ACT, and anti-Xa were associated with increased bleeding risk in pediatric and adult patients receiving ECMO at our center.
Materials and methods
Study design
This is a retrospective chart review of patients admitted to the Neonatal and Pediatric Intensive Care Units (NICU and PICU) at the CHRISTUS Children’s Hospital of San Antonio who required VV or VA-ECMO. Anticoagulation parameters: Activated Clotting time (ACT), anti-Xa levels, Prothrombin Time (PT), International Normalized Ratio (INR), activated Partial Thromboplastin time (aPTT), were assessed and compared against clinical outcomes. Although TEG is performed on all patients, its influence on decision-making is minimal unless results are clearly abnormal, which is why it was not included in the analysis. The primary outcome is hemorrhagic complications, defined by ELSO as a bleeding event with clinically overt bleeding recorded in the medical and nursing charts associated with either administration of 2 or more RBC units in 24 h or a drop in hemoglobin greater than 2 g/L over 24 h, and if there was a hemothorax, central nervous system or retroperitoneal bleeding, or lastly if bleeding required an intervention. 7 A unit in pediatrics is defined as 10 ml/kg of PRBCs. If there were consecutive days with the same primary source of bleeding as preceding days, these were considered the same bleeding event. When a patient had more than one bleeding source on the same day, this was also recorded.
At our institution, we use ECMO circuits from Spectrum Medical®, including the Pedi Quadrox-iD® oxygenator for patients under 10 kg, with a ¼ inch circuit, which requires 2 units of blood and 100 ml of Fresh Frozen Plasma (FFP). For patients over 10 kg, we use the same oxygenator with a ⅜ inch circuit, requiring 3 units of blood and 150 ml of FFP. Our ECMO system incorporates the Sorin (now Liva Nova) Revolution® centrifugal pump, featuring a 94.5 g open impeller design with a 57 ml priming volume, a maximum operational speed of 3500 rpm, and a flow rate of up to 8 L/min.
Prior to cannulation, we draw CBC, PT, aPTT, fibrinogen, and antithrombin III (ATIII). ACT is measured every hour for the first 24 h post-cannulation, with subsequent adjustments based on clinical need. For the PICU, ACT is monitored every 3 h after the initial 24 h, and every 6 h as needed. In the NICU, ACT is monitored every 3 h for the first 24 h, then every 6 h as needed. Coagulation labs are drawn every 6 h, and TEG is performed every 24 h. Anti-Xa and D-dimer levels are checked every 6 h for the first 24 h, and plasma-free hemoglobin and antithrombin are checked every 24 h for 3 days.
Twenty four hours after cannulation, heparin titration is based on the anti-Xa levels, aiming for a range of 0.3-0.6 IU/ml. We administer a bolus of 10u/kg of heparin prior to giving cryoprecipitate and platelets. The heparin infusion is started at 30u/kg/hr immediately upon cannulation and titrated according to the anticoagulation protocol. Heparin doses range from 25 to 100u/kg prior to cannulation, and the heparin drip used is a 100/ml solution (25,000u/250 ml) in D5W. In the NICU, the heparin infusion is titrated using the anti-Xa heparin titration table for the first 24 h post-cannulation or after a circuit change. The NEO oxygenator is used in the NICU, while the Pedi oxygenator is used in the PICU.
We prime the ECMO circuit with RBCs, plasma, albumin (25%), heparin, NaHCO3, and CaCl2. Initially, we prime with plasmalyte, followed by blood, and add medications like NaHCO3, calcium, and heparin. For patients under 10 kg, blood priming is always used. Since the inception of the standardized ECMO program, ECMO runs, including transfusion thresholds, are protocolized and the decisions to transfuse are to a specific cutoff and will not be affected by individual decisions. The institution follows standardized transfusion targets for patients supported on ECMO to optimize hemostasis and minimize bleeding risk. Platelet counts are maintained above 100 × 103/µL, with a lowered threshold of >80 × 103/µL in children older than 6 years who are not requiring significant blood product support; platelets are administered at 10 mL/kg. Red blood cell transfusions are guided by a hematocrit goal of >30%, with higher targets (>35%) applied in neonates, postoperative cardiac patients, and those with cyanotic heart disease; RBCs are dosed at 10 mL/kg. Cryoprecipitate is used to maintain fibrinogen levels above 200 mg/dL, with an adjusted goal of >150 mg/dL in patients with minimal blood product utilization, administered as one unit per 10 kg body weight. Antithrombin III replacement is generally considered when levels fall below 50%, although Thrombate use is reserved for cases with high heparin requirements or suspected heparin resistance, and routine use of FFP for low antithrombin levels is not standard. While the NICU applies a similar 50% cutoff, they tend to be more proactive in administering Thrombate.
The study was approved by the Institutional Review Board for Human Subject Research for Baylor College of Medicine and Affiliated Hospitals (BCM IRB-H-51,520).
Study population
Patients admitted to the PICU and NICU at CHRISTUS Children’s of San Antonio from 2014 to 2023 who were with VV or VA-ECMO. Our PICU is a combined unit that also cares for adult congenital heart disease patients, some of whom require ECMO treatment. The patient population is heterogeneous, with wide age ranges and different pathologies.
Inclusion criteria
(1) PICU and NICU admissions at CHRISTUS Children’s of San Antonio. (2) VV or VA-ECMO initiation. (3) Anticoagulation using heparin.
Exclusion criteria
(1) Patients with a previously identified history of bleeding disorder. (2) Patients with bleeding episodes that happened before the initiation of ECMO and discovered within 24 h of ECMO initiation. (3) Patients receiving direct thrombin antagonists (DTA) for anticoagulation. These patients are monitored using aPTT only, and the use of DTAs is infrequent at our institution.
Statistical analysis
Descriptive data are reported as frequencies and proportions for categorical variables and median (interquartile range, IQR) for continuous variables. Differences between groups were determined by the Wilcoxon rank sum test for the continuous variables and chi-square or Fisher’s exact tests for the categorical variables. The cumulative incidence of hemorrhagic events was depicted by the Kaplan-Meier (KM) curves. Levels of select coagulation parameters were presented by the box plots. Logistic regression was used to determine coagulation factors associated with hemorrhagic events and mortality. Linear regression was used to determine the coagulation factors associated with the length of stay (LOS). Because of the heterogeneity of the cohort and the possibility of confounders, two multivariable models were constructed for each regression analysis, each with a different anticoagulant measurement used for those who experienced a hemorrhagic event. The median anticoagulant parameter was used in one model, while the other used the anticoagulant measurement taken within 6 h of the hemorrhagic event. Variable selections for the multivariable models were conducted using the least absolute shrinkage and selection operator (Lasso) method with the cross-validation (CV) selection option or clinical importance.10,11 Briefly, the Lasso program was run with all variables included in the univariable analysis and suggested good initial models, which included the variables with a high probability of being risk factors. Covariates were also reviewed by clinicians for clinical relevance. The best model was selected based on the smallest Bayesian Information Criterion (BIC). All analyses were performed on Stata version 18.5 (StataCorp LLC, College Station, TX, USA). 10 A p-value of <0.05 was considered statistically significant.
Results
Our study included 76 patients admitted to the PICU and NICU at the CHRISTUS Children’s Hospital of San Antonio between 2014 and 2023. In total, 32/76 (42.1%) patients experienced a hemorrhagic event. Among those who experienced a hemorrhagic event, 15 patients (46.9%) survived. A larger proportion of survivors, 30/44 (68.2%), was observed among those who did not experience a hemorrhagic event (Figure 1). Flowchart diagram of study selection.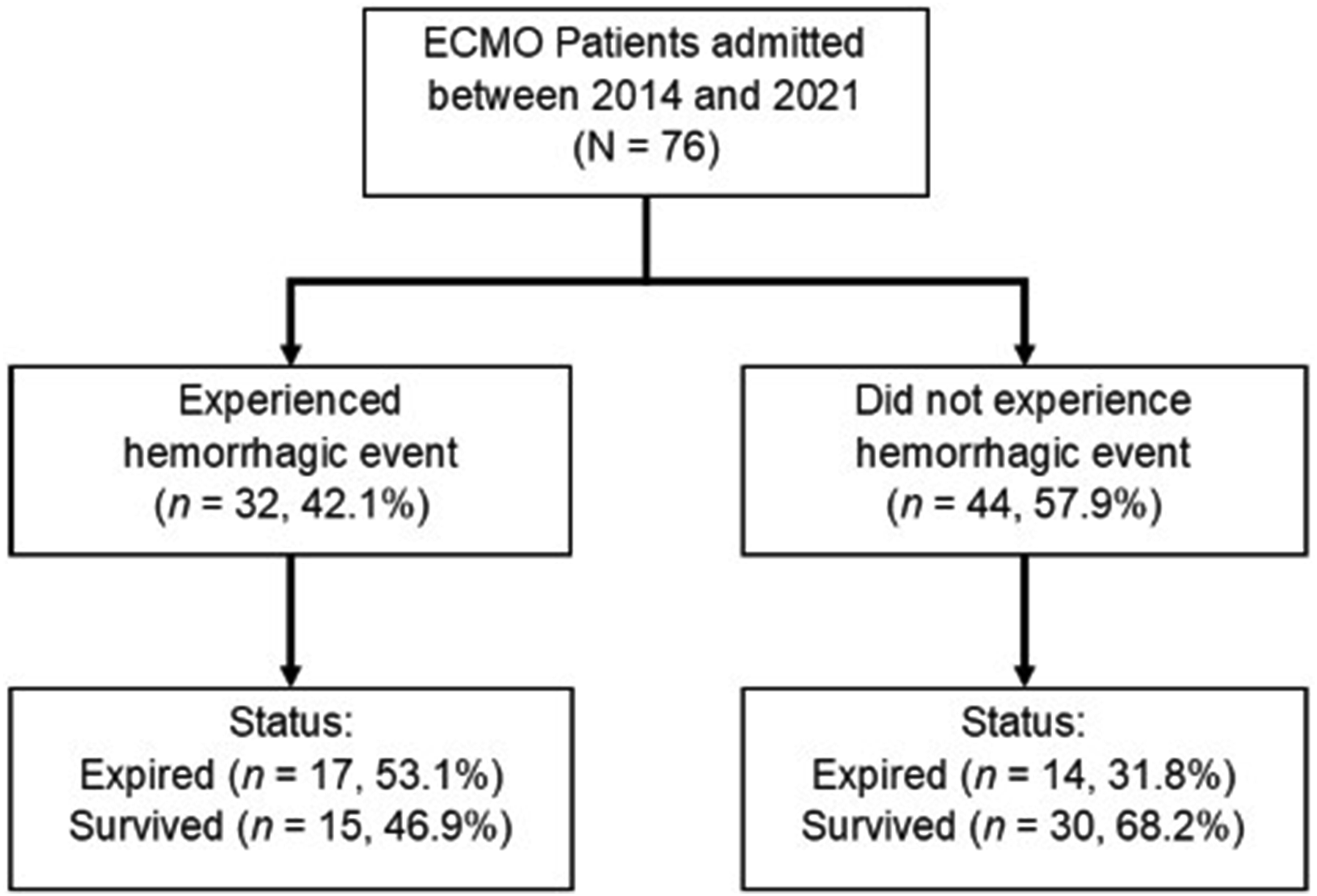
Patient characteristics, by bleeding event.
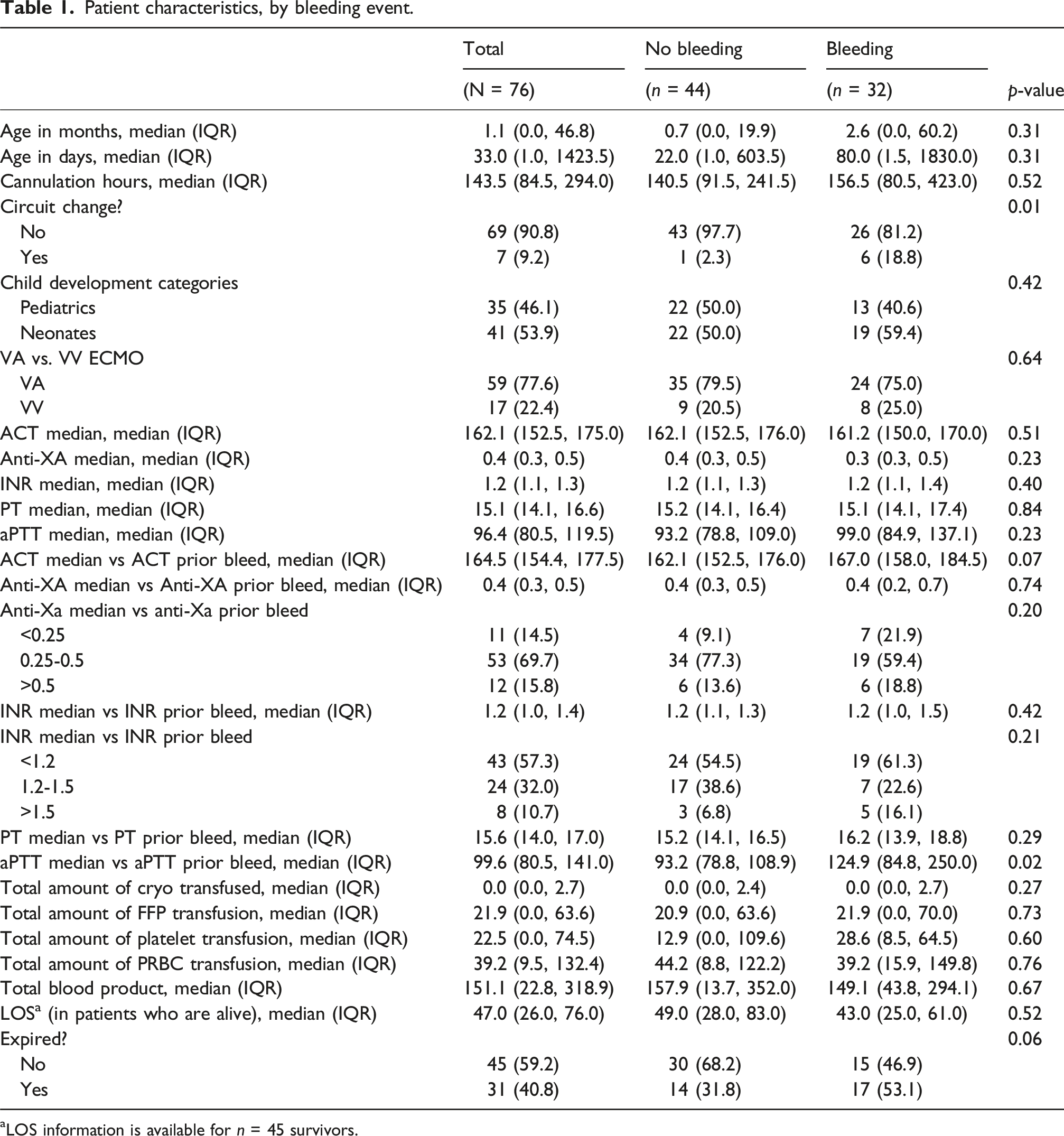
aLOS information is available for n = 45 survivors.
Figure 2 presents the time to events estimated by the Kaplan-Meier statistic. The estimated cumulative incidence of the hemorrhagic event at 10-, 20-, and 30-days post-cannulation was 36.9% (26.2%-50.3%), 58.8% (41.9%-76.5%), and 72.5% (47.2%-92.7%), respectively (Figure 2, Table S1). Sub-analysis indicated that patients with circuit change had 85.7% bleeding compared with 37.7% in those who did not have circuit change (Table S2). No difference in the bleeding proportion was found between VV versus VA ECMO groups (Table S3) nor between neonates versus pediatrics (Table S4). Kaplan-Meir plot of hemorrhagic events.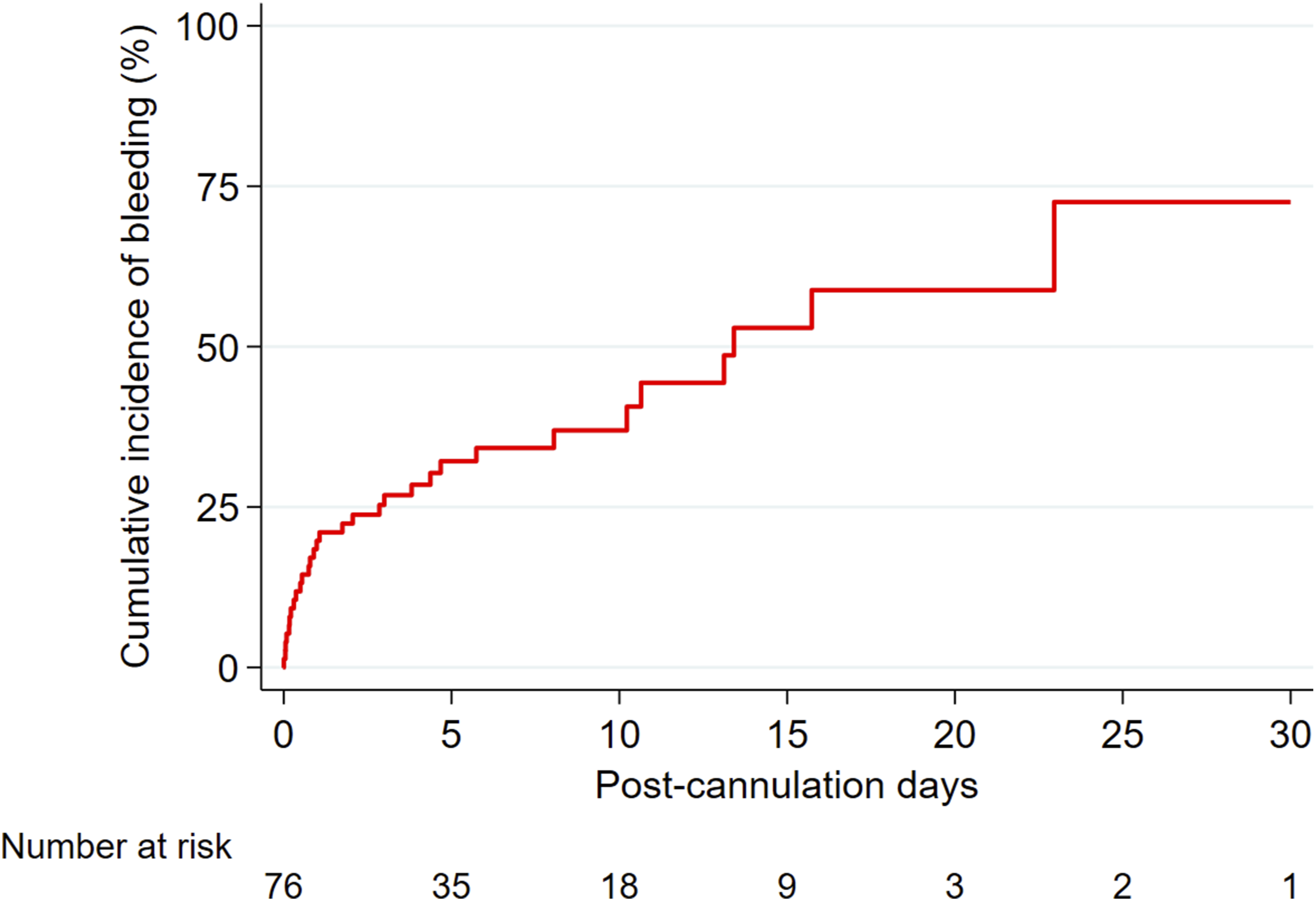
We compared the median values of anticoagulant parameters, starting 24 h after ECMO cannulation, between patients who experienced a hemorrhagic event and those who did not. We found no significant differences between the two groups (Table 1). The medians excluded the values collected in the first 24 h after cannulation because a heparin bolus is given right before cannulation; thus, anticoagulation parameters are expected to be elevated during the first 24 h after cannulation.
We subsequently compared the median anticoagulant parameters measured from patients who did not experience a hemorrhagic event to the median anticoagulant parameters measured prior to the hemorrhagic event (within 6 h of the event) for those who experienced a bleeding event. Our results indicated a difference in median aPTT and median aPTT prior to the bleeding event (p = 0.02) (Figure 3). Patients having hemorrhagic events appeared to have a higher median ACT level compared with the median ACT prior to the bleeding event in the group without hemorrhagic events. However, the difference did not reach statistical significance (p = 0.07) (Figure S1). The remaining comparisons showed no significant differences (Table 1). Boxplot of aPTT by bleeding event; (A) median aPTT from non-bleed patients versus median aPTT for bleed patients; (B) median aPTT from non-bleed patients versus aPTT-prior-bleeding-event from bleed patients.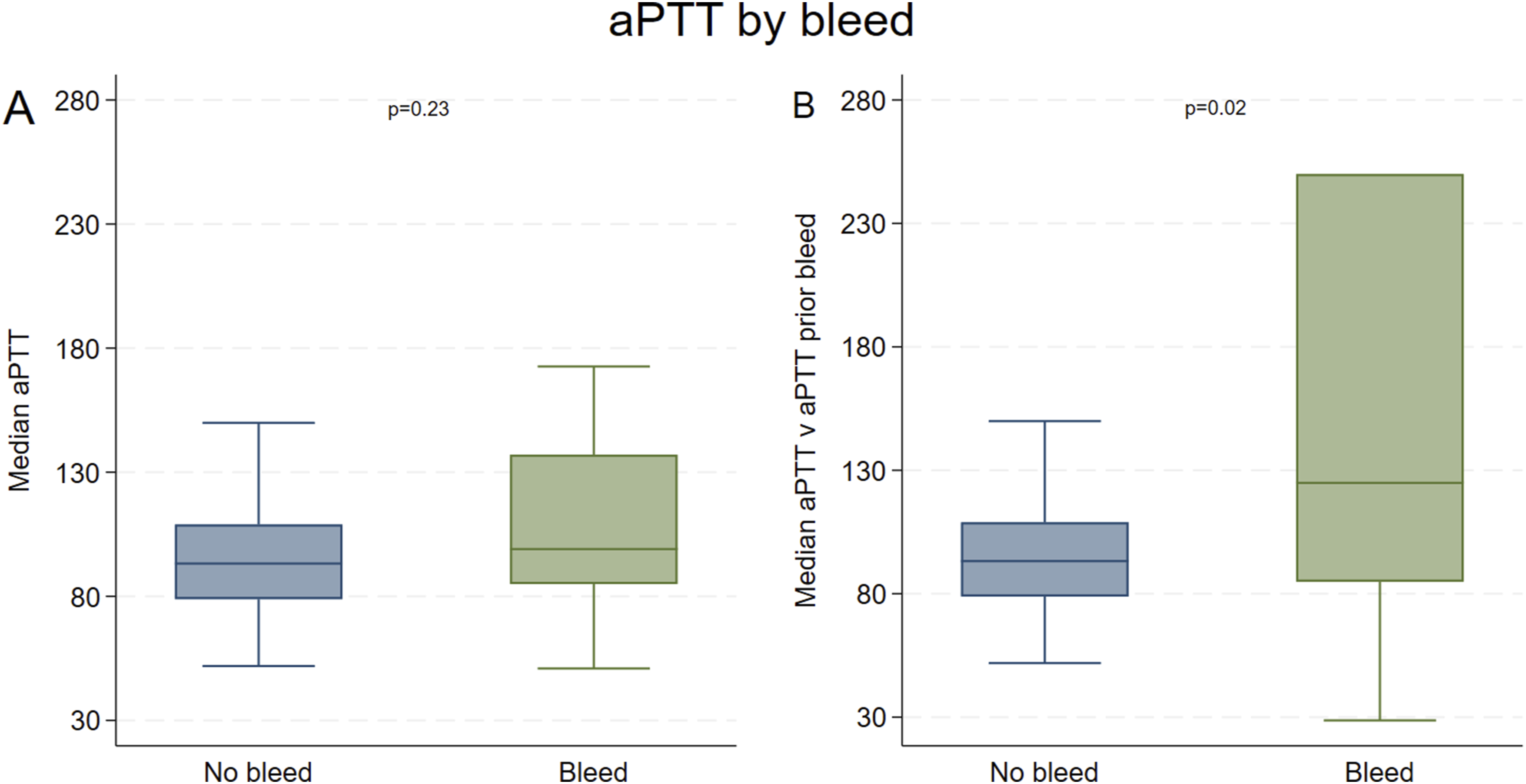
Logistic regression analysis of hemorrhagic event
Multivariable logistic regression for bleeding event.
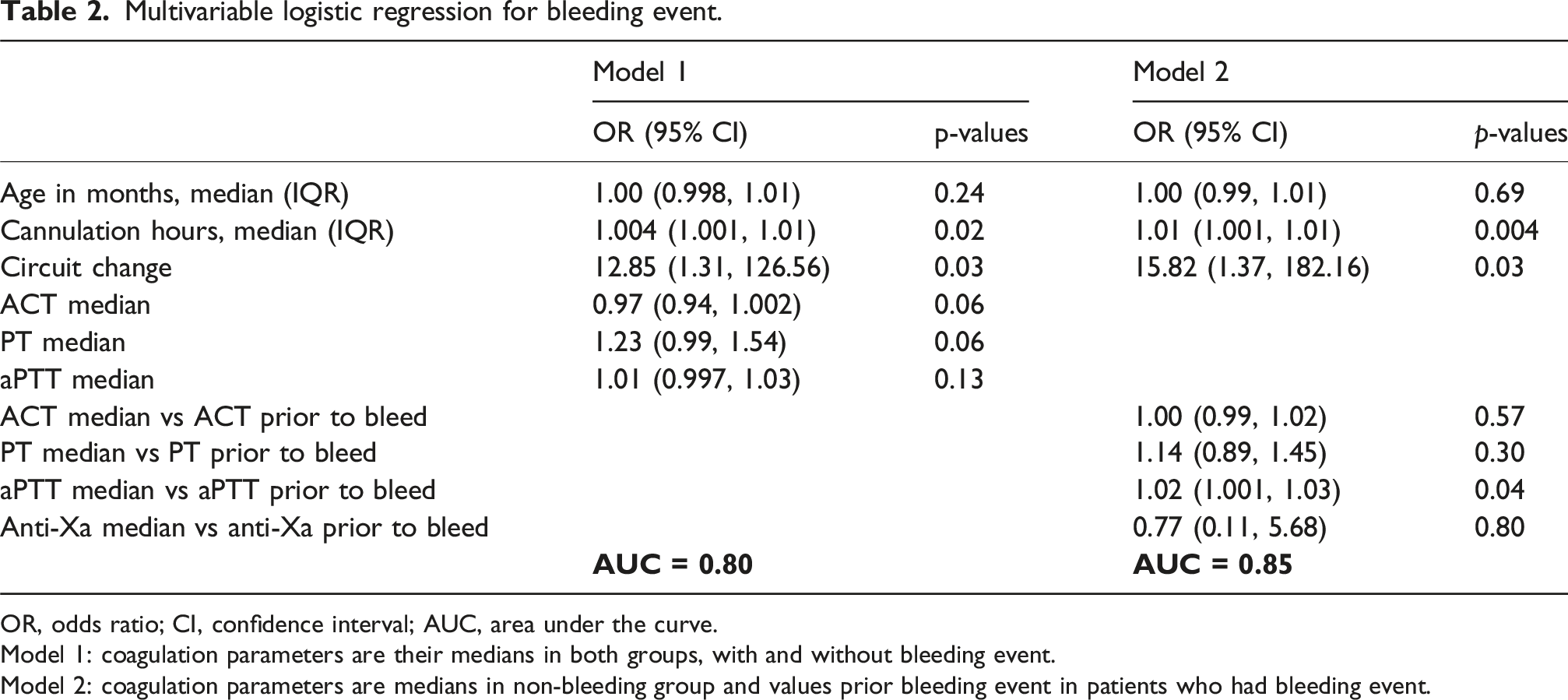
OR, odds ratio; CI, confidence interval; AUC, area under the curve.
Model 1: coagulation parameters are their medians in both groups, with and without bleeding event.
Model 2: coagulation parameters are medians in non-bleeding group and values prior bleeding event in patients who had bleeding event.
Logistic regression analysis for mortality
Multivariable logistic regression for mortality.
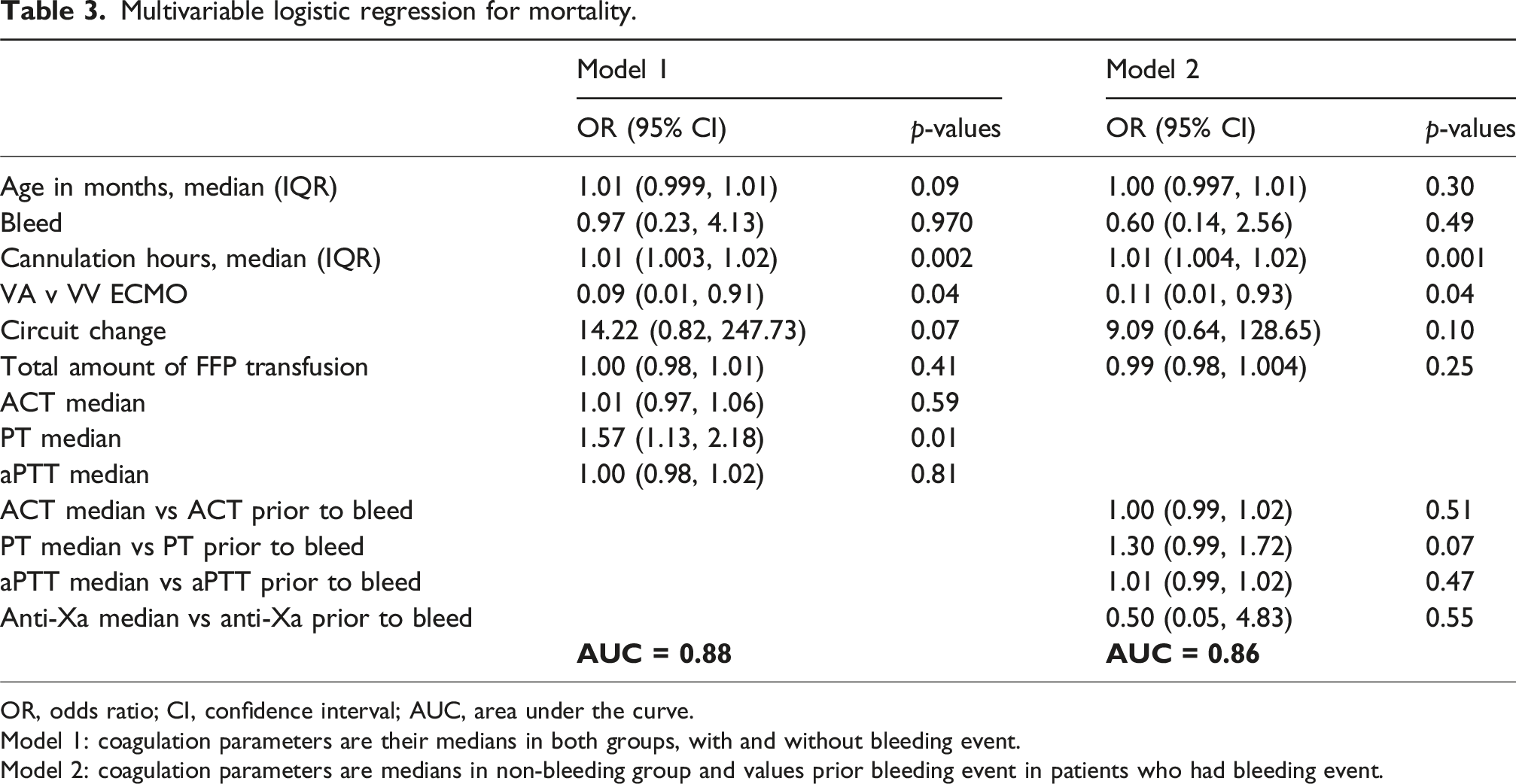
OR, odds ratio; CI, confidence interval; AUC, area under the curve.
Model 1: coagulation parameters are their medians in both groups, with and without bleeding event.
Model 2: coagulation parameters are medians in non-bleeding group and values prior bleeding event in patients who had bleeding event.
Linear regression analysis for LOS
Multivariable linear regression for length of stay (includes only those that did not expire, n = 45).
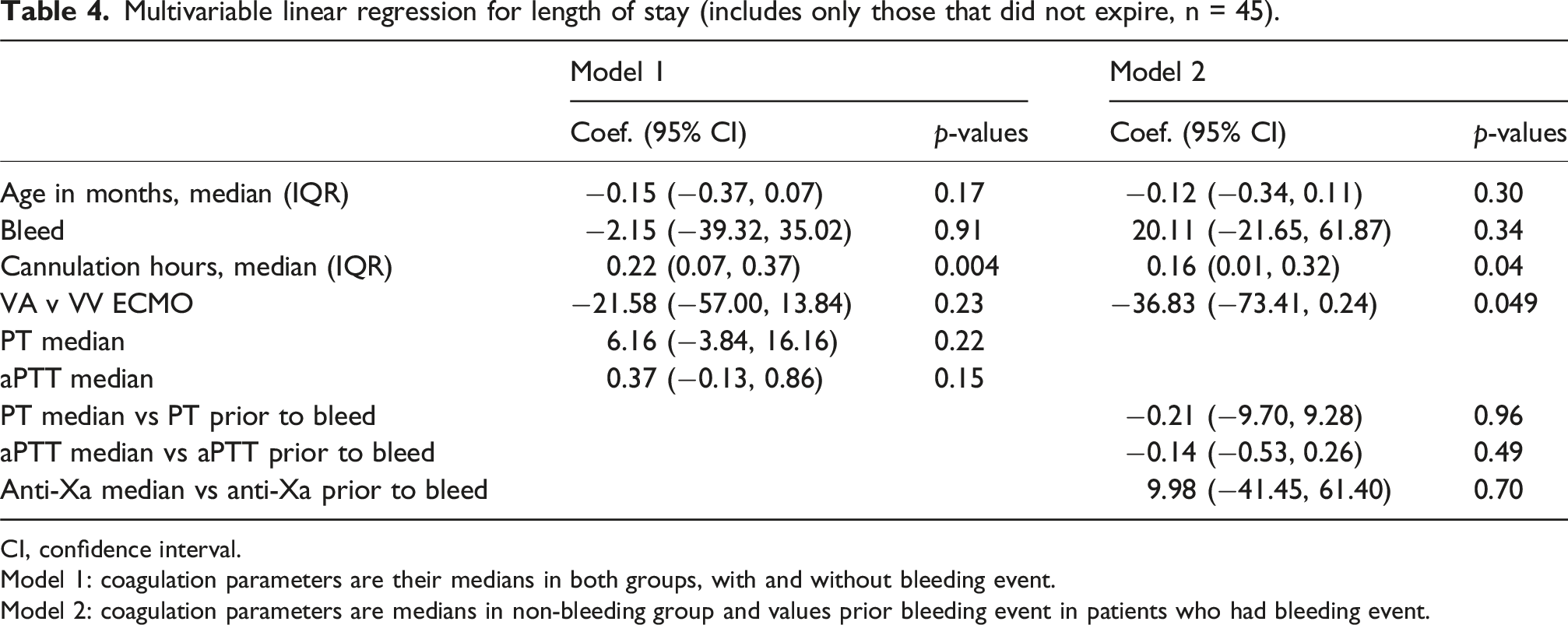
CI, confidence interval.
Model 1: coagulation parameters are their medians in both groups, with and without bleeding event.
Model 2: coagulation parameters are medians in non-bleeding group and values prior bleeding event in patients who had bleeding event.
Linear regression analysis for the length of ECMO
Univariable and multivariable linear regression for cannulation hours (i.e., ECMO run hours).
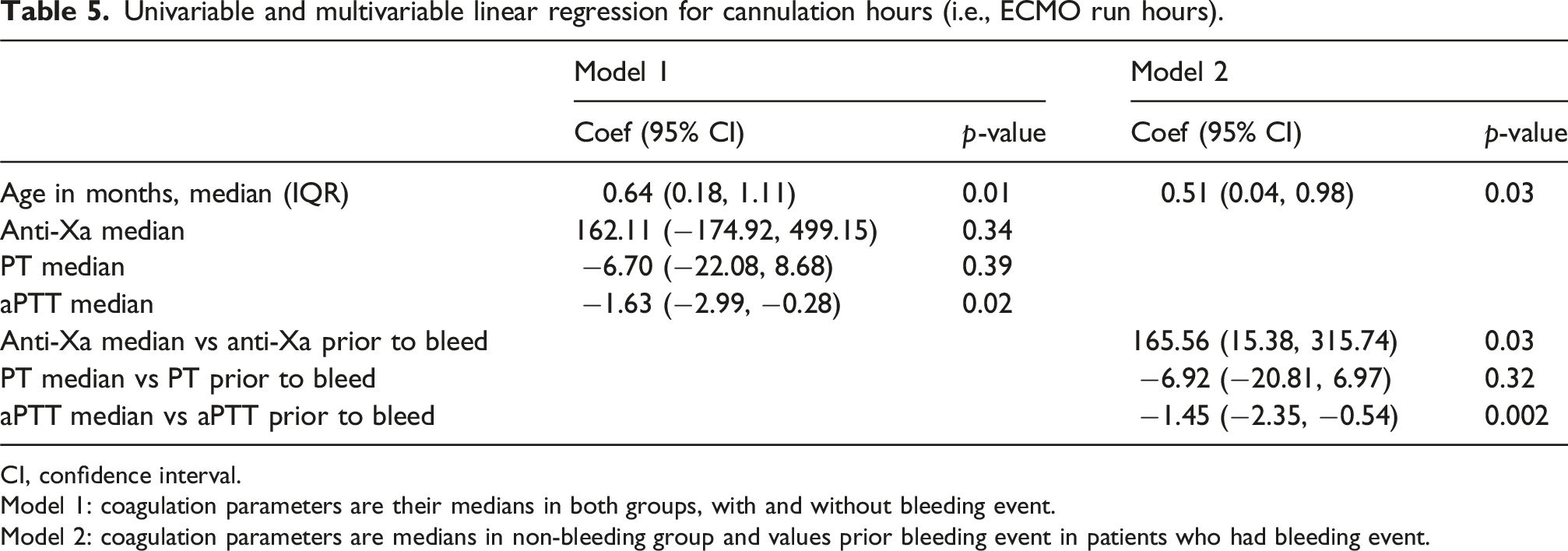
CI, confidence interval.
Model 1: coagulation parameters are their medians in both groups, with and without bleeding event.
Model 2: coagulation parameters are medians in non-bleeding group and values prior bleeding event in patients who had bleeding event.
Among the anticoagulant parameters, both models suggested a significant negative association with aPTT (−1.63 [−2.99, −0.28], p = 0.02; −1.45 [−2.35 to −0.54], p = 0.002). In addition, model 2 indicated a significant effect from median anti-Xa and median anti-Xa prior to bleeding (165.56 [15.38-315.74], p = 0.03) (Table 5).
Discussion
Our study demonstrated that, for patients who were placed on VV or VA-ECMO, a higher aPTT was associated with a significantly higher risk of bleeding. A higher ACT and PT were also associated with a higher risk of bleeding, but the difference was not statistically significant. More importantly, the study shows that the longer the patient was on ECMO, the higher the probability of having a hemorrhagic event. Mortality was higher in the bleeding group, but no statistical significance was reached. Interestingly, circuit change was associated with bleeding events. This could be due to our practice of giving a heparin bolus after a circuit change. This suggests that hemorrhagic events are multifactorial, and future studies should consider other variables.
Since the inception of extracorporeal life support, anticoagulation has been the cornerstone of managing the inevitable thrombotic risk as part of using this modality of treatment. After successfully performing cardiopulmonary bypass (CPB) using a membrane oxygenator, extracorporeal support was maintained for 4 days in dogs in the 1960s.12–14 As in CPB, heparin was used to prevent clotting in the pump and circuit. The bleeding was significant, and the heparin dose was titrated in stages while monitoring using only ACT. 15 The first successful clinical cases of ECMO were reported in the early 1970s.4,16
The pathophysiology of clotting during ECMO is considerably complex. Despite significant advances, our current understanding of the biological mechanisms underlying clotting remains limited. This gap in knowledge likely contributes to the ongoing challenges in developing effective anticoagulation strategies. Once the blood contacts the plastic tubing of the ECMO circuit, a cascade of systemic and cellular events will trigger an inflammatory response. These include several humoral and cellular systems, including contact, intrinsic, and extrinsic coagulation, complement systems, endothelial cells, leukocytes, platelets, and cytokines. 2 In the absence of anticoagulation, this will inevitably lead to thrombin deposition and the formation of clots. Clotting is not confined to the circuit; it can also occur in the patient’s blood vessels and cause significant morbidity and possibly mortality.
ELSO acknowledges that a balance between anticoagulation and pro-coagulant function is necessary to avoid issues with inappropriate hemorrhage and thrombosis. 7 In a multicenter study in 8 children’s hospitals, the authors reported bleeding events in 70.2% of children receiving ECMO, including intracranial hemorrhage in 16.0%. The bleeding events were independently associated with a higher daily risk of mortality. 17 A systematic review reported a third of the patients had a bleeding event of any severity. 18 Sy et al. reported the incidence of major bleeding to range between 13% and 50% in a meta-analysis performed on observational studies that included 1496 patients supported with VA-ECMO, and it was correlated to the strategy used to monitor heparin. 19 Bleeding complications were reported at 27.6% in an observational study of the ELSO registry. 20 Two-thirds of those who experienced bleeding did not survive to hospital discharge, compared to less than half of those who did not experience bleeding. 20 We report 42.1% hemorrhagic events associated with ECMO in our cohort. Less than half of those patients survived to hospital discharge. Other studies have reported that bleeding has been associated with worse outcomes.17,20,21
ELSO Guidelines were published in 2022 to assist physicians in managing anticoagulation on ECMO. 7 Ideally, monitoring would be conducted in vivo, which captures a comprehensive instantaneous view of a patient’s coagulation. However, out of necessity, the assays commonly employed are in vitro and typically assess only part of the coagulation cascade. This, in turn, leads to an imperfect assessment of the patient’s true coagulation potential. Additionally, the recommendation from ELSO for optimal anticoagulation states that there is no universally accepted best practice, and each facility must find what serves its patients best. 7 This is supported by several other studies that cite the inter-facility differences in anticoagulation goals, titration, and dosing of anticoagulants.22–28
The challenge is higher when caring for neonates undergoing ECMO. There is a poor correlation among laboratory assays used in anticoagulation monitoring. This has led to center-specific algorithms for coagulation monitoring labs and the frequency of obtaining them. To help guide decision-making, it is important to evaluate the influential factors that will affect coagulation.24,29–31
There are two ways in which anticoagulation can be assessed. The first is measuring the time it takes the blood to form a clot, i.e., clotting time. This is based on either standard laboratory testing: aPTT or the whole blood point-of-care ACT. The second measures heparin concentration by chromogenic approach, based on anti-Xa activity. 32 Many of the standard tests used to assess anticoagulation are site-dependent. This could be attributed to the variability among labs when performing those tests. For example, anti-Xa assay results vary depending on whether the performing lab adds antithrombin to the sample. 33 This will lead to site-dependent variability and reproducibility. Thus, the standard of care anti-Xa levels at one site cannot be reliably translated as a gold standard to other sites. 33
Similarly, aPTT is another commonly used test to assess anticoagulation, and based on what reagents are used in the performing laboratory, the reported time can be affected. 32 This makes inter-facility comparisons challenging.23,26 Similar to our results, Sklar et al. suggested that a lower aPTT may aid in reducing the risk of life-threatening hemorrhagic events on VV-ECMO. This systemic analysis found that, while there was an increased risk of developing thrombotic events, this risk was mainly related to the circuit. They concluded that an aPTT under 60 s should be targeted in this population of ECMO patients. 23
Regarding ACT, the activator used will affect its reproducibility and clot detection.34,35 Also, variability was observed within the same facility, as the technique is dependent on the operator. 35 Finally, variables such as temperature, hemodilution, hematocrit, fibrinogen level, and platelet count and function will affect the results as the assay is performed on whole blood.34,35
Vescoelastography (TEG® and ROTEM®) has been used to assess ECMO anticoagulation in recent decades. Those assays provide a detailed analysis of the blood’s ability to form a clot. They also measure the time to clotting initiation and clot strength. Their use is limited by the time and cost consumed to perform the assay. Not enough studies correlate those assays with outcomes or the risk of bleeding on ECMO. Those assays can be beneficial in detecting underlying coagulopathy after adding a heparinase.36–40 This subset of patients could have other abnormal parameters, such as higher INR and a prolonged aPTT. Because these parameters could be altered due to various reasons, clinical management decisions based on TEG results were primarily applied to patients with suspected or confirmed underlying bleeding disorders at our institution, who represented a small subset of the overall cohort.
Specific goals of anticoagulation will vary depending on the perceived risk of bleeding. This leads to complexity in comparing one patient to another in terms of what anticoagulation goals should be accepted.7,24,25 In a recent study, COVID-19 patients receiving ECMO were found to have a higher frequency of coagulation disorders. 21
Moreover, children and neonates are at higher risk of bleeding due to the different concentrations of coagulation factors and their response to heparin.24,29–31 As such, no one test can reliably coordinate optimal anticoagulation while on ECMO.7,24,26,27
Conclusion
Our study identified several factors associated with hemorrhagic complications, mortality, and ECMO duration. Elevated aPTT and prolonged ECMO duration were independently associated with an increased risk of bleeding events. Cannulation time and circuit change were also found to increase bleeding risk, while PT showed a trend toward significance. For mortality, prolonged cannulation duration was associated with higher odds of death, whereas VV-ECMO configuration was linked to improved survival. Although coagulation parameters had limited predictive value for mortality, PT demonstrated a modest association. Additionally, older age and higher anti-Xa levels correlated with longer ECMO duration.
These findings highlight the multifactorial nature of coagulation and bleeding risks in pediatric ECMO. Tailoring anticoagulation strategies to patient-specific and pathology-specific factors remains essential. Future research should focus on differentiating ECMO phenotypes and addressing thromboinflammatory pathways rather than relying solely on traditional coagulation assays.
Study limitations
This is a retrospective study conducted by a single institution on the available database. It should be interpreted with some limitations in mind. Observational studies play a significant role in health research, particularly when evidence from randomized controlled experiments is not available or feasible. However, retrospective analyses are prone to recall, or misclassification bias and chronological relationships are often difficult to assess. Moreover, some risk factors may be present but cannot be measured, predisposing such studies to misinterpretation. A limitation of this study is the complexity of the pathophysiology of clotting during ECMO, which is not fully understood. The underlying pathologies were not consistently documented during the data collection and were thus not included. Variability in underlying diagnoses could influence coagulopathy and bleeding risk. Future investigation in prospective studies with more granular diagnostic data collection. Despite advances in technology and clinical practice, the limited knowledge of the biological mechanisms of clotting remains a challenge, complicating the development of effective anticoagulation strategies.
Supplemental material
Supplemental material - Correlation of anticoagulation assays with bleeding during extracorporeal membrane oxygenation at a tertiary pediatric hospital
Supplemental Material for Correlation of anticoagulation assays with bleeding during extracorporeal membrane oxygenation at a tertiary pediatric hospital by Mohammed Salameh, Jazmin Delgado, Ryan Killebrew, Cody Henderson, Elumalai Appachi in Perfusion
Footnotes
Acknowledgments
The authors acknowledge the essential contributions of the Biostatistics team at Baylor College of Medicine, namely Duc T Nguyen, MD, PhD, Kenneth Jhune Nobleza, and Ankona Banerjee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
