Abstract
Extracorporeal Membrane Oxygenation (ECMO) is commonly associated with a high blood transfusion requirement. Jehovah’s Witness patients present a particular challenge. The impossibility of transfusing blood cells and starting anticoagulation treatment are common contraindications for this supportive measure. Here we report the case of a Jehovah’s Witness patient with refractory hypoxemia due to influenza A H1N1 pneumonia who required venovenous ECMO for 11 days. We describe the use of a bloodless approach to reduce the waste of blood, avoiding anticoagulation, and improving red blood cell production. We then summarize the current literature on the use of ECMO in Jehovah’s Witness patients and, finally, we propose some recommendations for their management.
Keywords
Introduction
Extracorporeal Membrane Oxygenation (ECMO) is a complex support measure that is commonly associated with high blood transfusion requirements due to bleeding, hemolysis, and the need to maintain adequate oxygen delivery in the hypoxic patient. 1 The use of anticoagulant therapy is the gold standard during ECMO therapy, but this increases the risk of bleeding. The impossibility of anticoagulation is still a contraindication for extracorporeal life support at many ECMO centers. As Jehovah’s Witness patients (JW) reject blood transfusions, many centers consider these patients to be absolutely contraindicated for ECMO. However, experienced ECMO centers have reported good results after extracorporeal life support in JWs while respecting their desires and accepting the risks. Here we present a case report of a JW patient with refractory hypoxemia due to influenza A H1N1 pneumonia, who was successfully supported with ECMO without transfusions and without anticoagulant therapy for 11 days. We then present the results of a literature review (from 2002 to 2021) of JWs supported with either venovenous (VV) or venoarterial (VA) ECMO. Finally, we discuss the key points in the management of these patients.
Case report
A 47 year-old female Jehovah’s Witness with obesity Class II (168 cm and 110 kg) was admitted to the Emergency Department at a secondary level hospital for flu symptoms and shortness of breath. Bilateral lung infiltrates were observed on the chest X-ray. High flow nasal cannula and empiric antibiotic treatment with ceftriaxone, azithromycin, and oseltamivir were started. Influenza A H1N1 was identified by polymerase chain reaction (PCR) in nasopharyngeal smear. The patient progressed to respiratory failure requiring mechanical ventilation (MV) and was admitted to the Intensive Care Unit (ICU). Sedation, positive end expiratory pressure (PEEP) adjustment, neuromuscular blockade, and prone positioning were established but no improvement in gas exchange was observed; in view in the progressive worsening of oxygenation with a PaO2/FiO2 ratio of 48 mmHg, our ECMO team was activated. After discussion with the referral center and her relatives about a potential contraindication due to her rejection of blood transfusions, we finally considered the case to be suitable for extracorporeal support. Signed informed consent was obtained from the family and all the associated risks were explained. VV ECMO support was started at the referral center (CardioHelp®, Getinge AG, Sweden) by our team with a femoro-jugular configuration (25 F, 55 cm right femoral vein drainage and 19 F, 15 cm right jugular vein return). The procedure was performed percutaneously under ultrasound guidance. The left femoral vein was assessed but was considered to be too small, and so the right jugular vein was chosen. Pre-ECMO hemoglobin was 7.7 mmol/L. The system was primed with saline (total priming volume of 600 mL). The patient was transferred on ECMO to our hospital by ground ambulance without complications (110 km). Unfractionated heparin bolus (50 UI/kg) was administered during cannulation, but heparin infusion was not started.
During the ICU stay, MV was adjusted measuring transpulmonary pressure using an esophageal balloon during the first days, adjusting PEEP between 12 and 16 cmH2O with inspiratory pressure of 10 cmH2O in the Pressure Control Ventilation mode, and achieving tidal volumes below 1 mL/kg of Ideal Body Weight (60.3 kg). Percutaneous tracheostomy was performed on day 4 to allow awake ECMO. No life-threatening bleeding was observed. Neuromuscular blockers were withdrawn and sedatives were switched to remifentanil and dexmedetomidine, reaching a Richmond Agitation-Sedative Scale level of −1. Intensive rehabilitation therapy was started. Broncho-alveolar lavage confirmed the presence of Influenzas A H1N1 infection, with negative bacterial cultures. Antibiotic treatment consisted of 5 days of oseltamivir and azithromycin and 10 days of cefotaxime. Iron deficiency was found in lab tests on the second day of ECMO support. Blood samples were minimized to just one per day. Pediatric sample tubes and a closed in-line blood sample system were used. No anticoagulation was used at any time during the ECMO support. Blood flow was maintained between 4 and 4.5 L/minute, with drainage pressure kept at maximum of −70 cmH2O. The heater-cooler was set to maintain a blood temperature of 37°C. Intravenous treatments with erythropoietin (EPO) 150 UI/kg three times per week, iron, vitamin B12, and folic acid were started. Hemoglobin results are shown in Figure 1. Lactate levels remained stable during the support and were considered a good surrogate of adequate oxygen delivery. Table 1 shows the average values of PaO2, SpO2, serum lactate, and urine output during ECMO support.
Evolution of variables related to oxygen delivery and renal perfusion throughout the ECMO run.
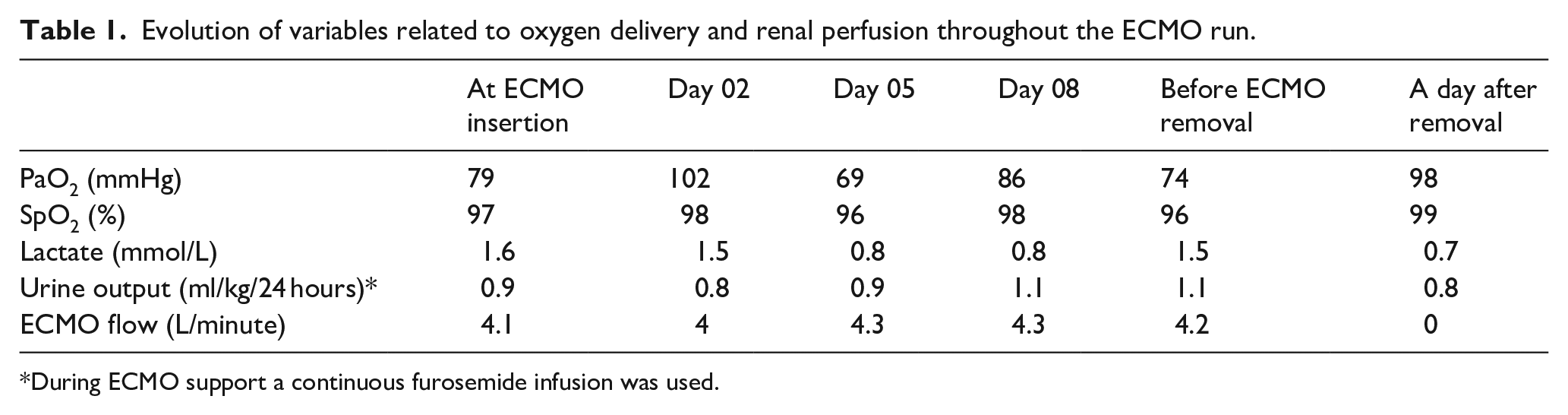
During ECMO support a continuous furosemide infusion was used.
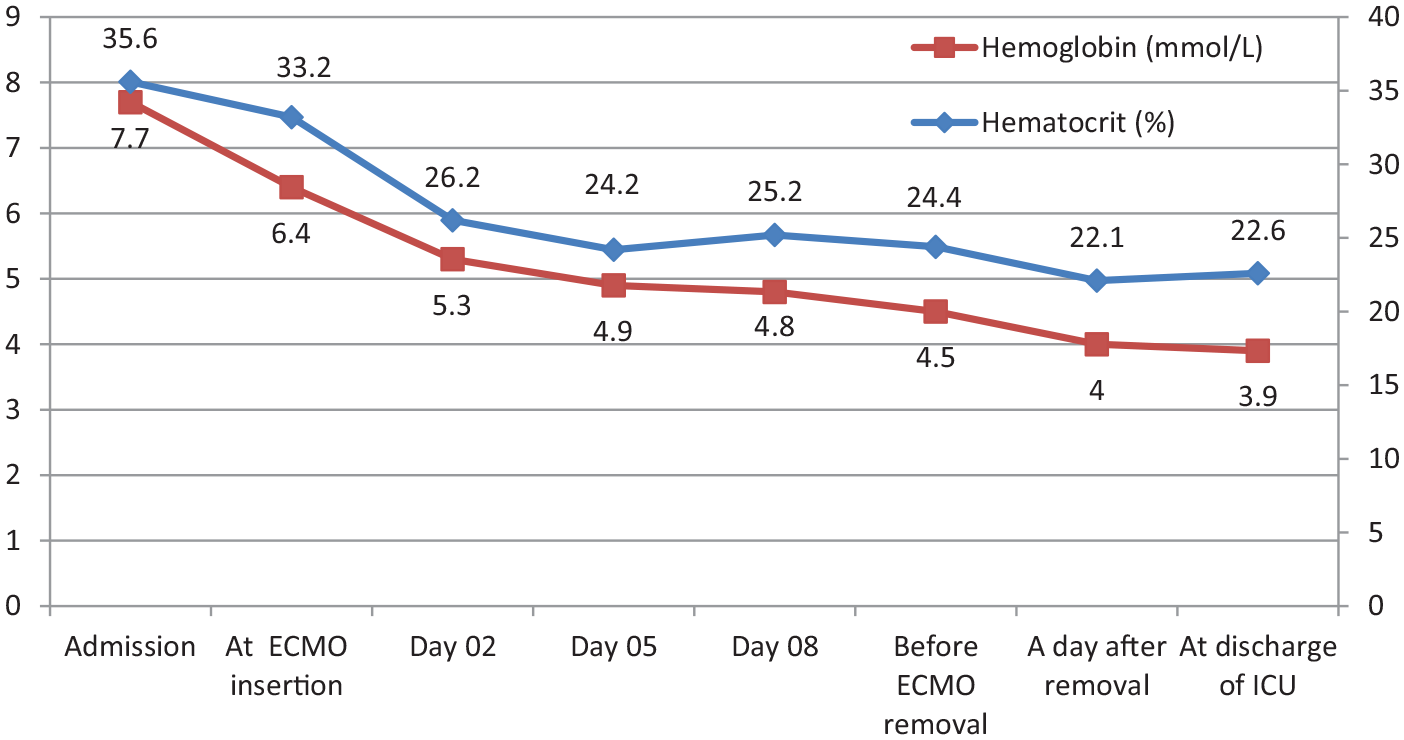
Serial changes in hemoglobin and hematocrit levels.
After 10 days of a heparin-free ECMO run, she developed hypofibrinogenemia, with normal platelets value and other coagulation tests, and non-life-threatening bleeding from the tracheostomy. Fibrinogen replacement therapy was performed with fibrinogen concentrate. The circuit was checked each 12 hours in order to identify clots in the system. Some insignificant clots were identified in the oxygenator, but no dysfunction was evidenced (post-oxygenator PaO2 above 400 mmHg and normal levels of post-pre differential pressure). The patient showed progressive respiratory improvement with a significant increase in respiratory compliance and a reduction in radiological infiltrates. She was successfully weaned off ECMO at day 11. To avoid wasting the circuit blood, a surgical cell-saver was used. After clamping and cutting the circuit, a luer 3/8 connector was added. Residual blood in the circuit was flushed to the cell-saver blood reservoir so as to prevent blood loss. A unit of self-blood was transfused after obtaining family consent (Figure 2). Hemoglobin concentration after ECMO decannulation was 4 mmol/L. After decannulation, the circuit was checked, and we identified the presence of clots only in the stagnant areas of the oxygenator. The patient was transferred to the referral hospital. Before her transfer, a venous ultrasound of the sites of insertion of cannula was performed and venous thrombosis was ruled out. Evolution was satisfactory. The patient was discharged after 26 days of hospitalization with a hemoglobin value of 5.8 mmol/L.
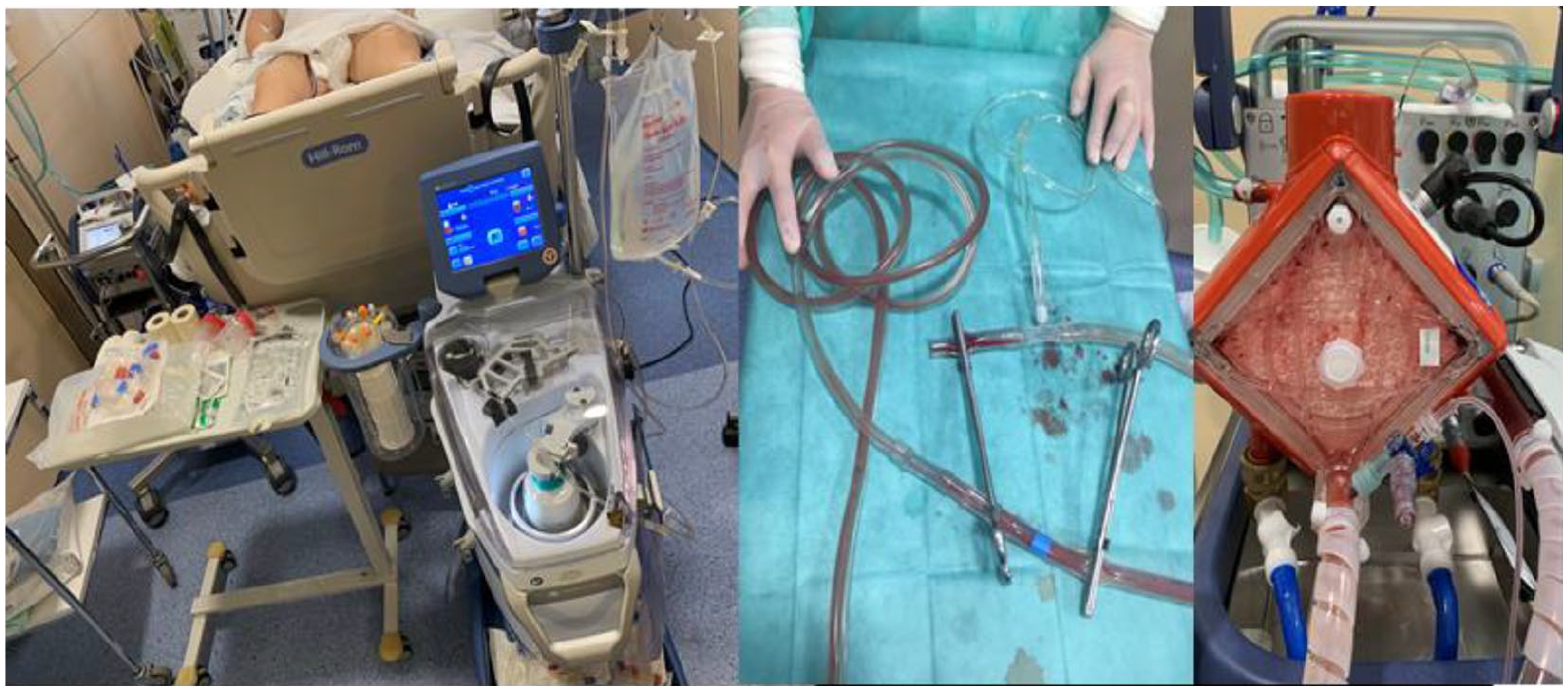
Cell-saver and circuit preparation for self-transfusion.
Discussion
Our literature search identified six articles, all of them case reports. Two articles were ruled out due to ethical concerns regarding the administration of ECMO to children of JW parents. The other four case reports2–5 are summarized in Table 2. Three cases were respiratory ECMO patients with VV ECMO, one of whom required conversion to VA due to right ventricular failure. The fourth case was a circulatory support with VA ECMO in a patient with cardiogenic shock after heart transplant. In all cases, femoral veins were used as the site for drainage cannula. The administration of erythropoietin and the return of the blood from the circuit after ECMO decannulation were common to all cases. A surgical cell-saver was used in three cases to auto-transfuse blood from the circuit. Park et al. 5 described the return of blood by introducing saline into the circuit after clamping the drainage cannula with a connector and using the drive of the centrifugal ECMO pump, without complications. Despite all these and other measures, in all cases a constant fall in hemoglobin levels was found. The threshold for ECMO initiation in JW patients may differ from that in the rest of the population. Achieve the correct time to start ECMO support in JW patients is a challenge. Conservative management in these patients can be deleterious. Prolonging the indication for ECMO therapy in these patients—for fear of associated complications—can be catastrophic. A late indication has been shown to be associated with worse outcomes6,7 and this situation might be considered.
Review of cases of ECMO use in Jehovah’s Witnesses.
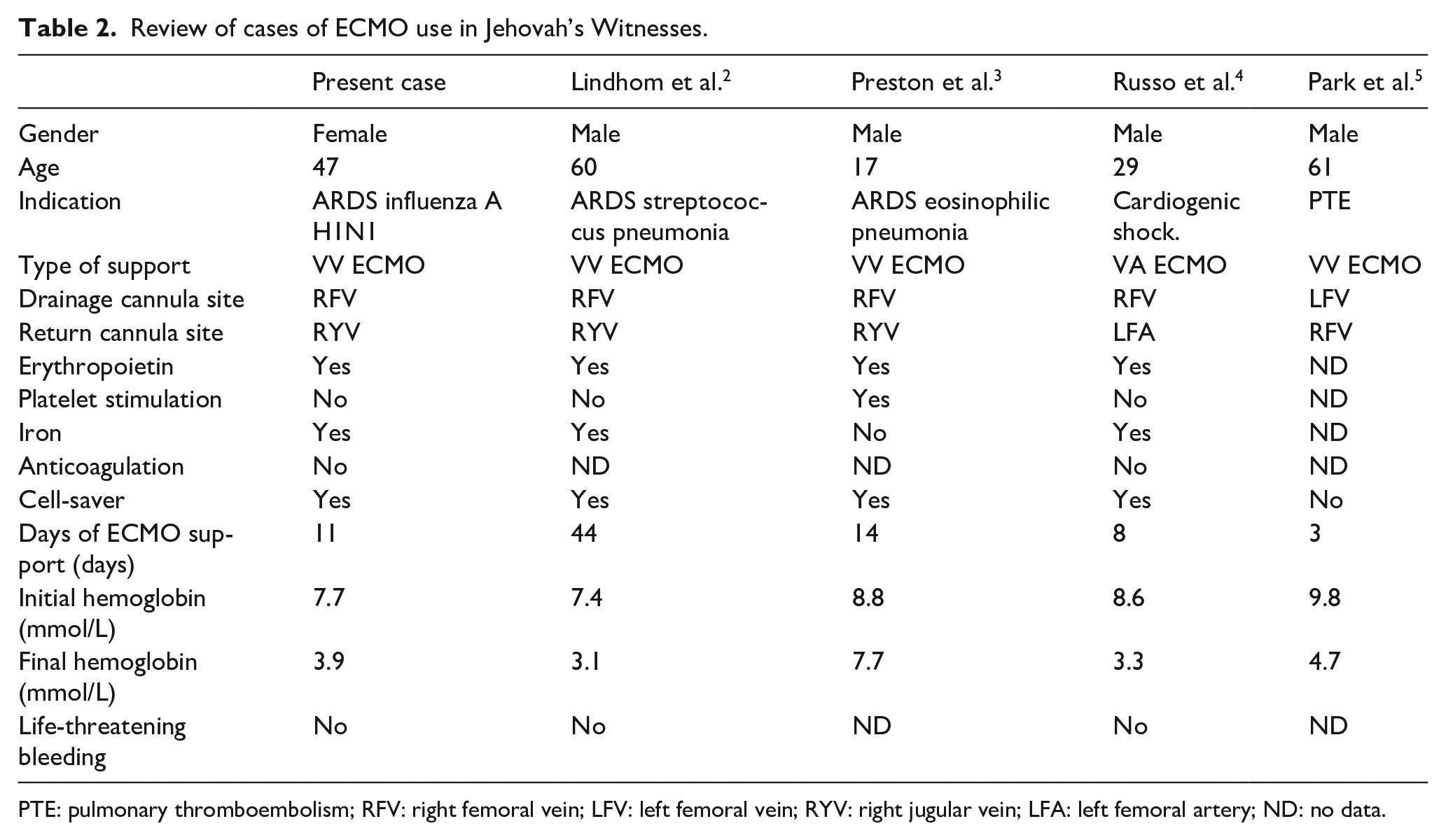
PTE: pulmonary thromboembolism; RFV: right femoral vein; LFV: left femoral vein; RYV: right jugular vein; LFA: left femoral artery; ND: no data.
The treatment of Jehovah’s Witnesses undergoing extracorporeal therapy is a challenge. The evidence for pharmacological therapies is poor, 8 and only a few cases have been reported. However, it is important to consider certain features in the management of these patients that can increase the chances for a successful ECMO therapy. Based on our experience, the literature, and the rationale of ECMO physiology, we present some recommendations in Table 3:
Optimize red blood cell production: Erythro-poiesis stimulants (intravenous recombinant human erythropoietin) and intravenous iron supplements have been used in almost all the cases reported and seem to be safe and useful in JW with acute anemia. 9 In case of deficiency, folate and vitamin B12 supplements can be administered.
Optimize oxygen delivery and consumption: High ECMO blood flows should be maintained to provide higher oxygen content. This is also useful in heparin-free ECMO runs in order to avoid clotting. Low oxygen saturations should be avoided. Patients under ECMO support commonly tolerate lower saturation values than other standard ICU patients. In JWs one should look very carefully for tissue hypoxia due to anemia if arterial saturations drop below 90%. Oxygen consumption should be reduced with sedation or neuromuscular blockage if an adequate ratio cannot be maintained.
Minimize blood loss: The frequency of blood samples extractions should be minimized in order to avoid iatrogenic anemia. The use of pediatric sample tubes and installed closed in-line blood sample system may be useful in this regard.9,10 If anticoagulation monitoring is needed, activated clotting time (ACT) monitoring requires less blood in each sample. When a blood sample is taken for analysis, coagulation factors including fibrinogen, and the level of platelets should be monitored; especially in the case of heparin-free long ECMO runs. Blood gas monitoring and pre-oxygenator circuit calibrations should be reduced. Post-oxygenator samples should be used only if oxygenator dysfunction is suspected. A system with the least priming volume should be used in order to minimize the initial dilution. A surgical cell-saver should be used to retrieve the blood from the circuit and auto-transfuse the patient after ECMO weaning. The use of a cell-saver is feasible and safe; in comparison with other blood-sparing techniques, it reduces the risk of air embolism, and clot mobilization. 5
Minimize bleeding risk: Anticoagulation therapy should be avoided at all times during the ECMO run. Although anticoagulation is routinely used in ECMO,11,12 in many instances ECMO has been safely run without heparin and does not cause major thrombotic complications, 13 especially if high blood flows can be achieved. Nevertheless, cannula related venous thrombosis is common if heparin is not used 14 and routine venous ultrasound is recommended after decannulation. All other coagulation factors (like calcium or temperature) should be controlled and replaced in case of coagulopathy. A safe conservative cannulation approach should be used and less aggressive sites and thin and short cannulas are preferable in order to reduce bleeding risk. However, an excessively short diameter may increase the risk of thrombosis and compromise the possibility of achieving high flow. The use of bi-lumen cannulas is controversial. Using a single puncture site may be an option; however, the wider caliber and the greater technical difficulty of canulation technique may be a problem in case of bleeding. A safe approach might be femoro-femoral cannulation because of the easier bleeding control at the time of cannulation. Ultrasound-guided cannulation plays a vital role in these patients and keeping invasive procedures to the minimum is mandatory. In this regard, ultrasound of the insertion sites after decannulation is also advisable. There is a high risk of venous thrombosis, 15 especially in patients with heparin-free runs. It is reasonable to put off tracheostomy if a short ECMO run is expected, due to the higher risk of bleeding and transfusion requirements in tracheostomized patients on ECMO. 16 If possible, VV ECMO over VA due to the lower risk of bleeding at the cannulation site.
Talk with relatives: Patient’s wishes should be respected but their relatives must be fully informed of the risks of each intervention and the lower chance of survival without the standard treatment, especially if a bleeding complication occurs. Our case was presented to the hospital ethics committee, which decide that in case of life-threatening bleeding, the autonomy of the patient should be respected.
Summary of recommendations for supporting Jehovah’s Witnesses with ECMO.

Conclusions
Jehovah’s Witnesses patients present a challenge and an ethical concern in ICUs when they require highly invasive therapies such as ECMO. By using a bloodless approach and following certain recommendations, it is feasible to achieve good outcomes with ECMO without blood transfusions in selected JW patients.
Footnotes
Acknowledgements
We thank all the members of the intensive care unit Vall d’Hebron Hospital who assisted in the care of this patient.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ note
This case report was presented to the Clinical Research Ethics Committee of the Vall d’Hebron Hospital. The registration number was 491 (June 11, 2021). Authors have obtained patient’s approval to publish this case report.
