Abstract
Background
Mediastinal masses in children secondary to malignancy can cause significant airway and great vessel compression, leading to respiratory and cardiovascular compromise. Extracorporeal membrane oxygenation (ECMO) has been described as a bridge to diagnosis and treatment for masses causing cardiopulmonary instability.
Objectives
To evaluate outcomes of pediatric oncologic patients requiring ECMO for mediastinal masses.
Methods
We conducted a sub-analysis of a previously published multicenter cohort study examining ECMO outcomes in pediatric hematologic and oncologic patients from 2009 to 2021. Patients less than 19 whose disease presented as a mediastinal mass were included. Presenting features, ECMO characteristics, and outcomes were analyzed.
Results
Eleven patients with mediastinal masses were identified, 7 with solid tumors and 4 with hematologic malignancies. The indications for ECMO were combined cardiac and respiratory failure in 64%, respiratory failure in 27%, and cardiac failure in 9%. ECMO survival was 72% (8/11), and survival to hospital discharge was 45% (5/11). Upon separation into solid tumor and hematologic malignancy groups, the ECMO survival was 71% (5/7) and 75% (3/4), and survival to hospital discharge was 29% (2/7) and 75% (3/4), respectively. Patients with solid tumors had longer intensive care unit (ICU) lengths of stay (LOS) and hospital LOS.
Conclusions
Our study supports ECMO cannulation for patients with mediastinal masses secondary to hematologic malignancies that require advanced cardiopulmonary support. However, due to poorer outcomes in solid tumors, ECMO candidacy should be carefully considered in this population.
Keywords
Introduction
Mediastinal masses, both anterior and posterior, in the pediatric population may be secondary to malignancy, most commonly T cell lymphoblastic leukemia and lymphoma, Hodgkin lymphoma, non-Hodgkin lymphoma, neuroblastoma, thymoma, or germ cell tumors.1–3 These masses can cause significant compressive effects on adjacent structures, including the airway and great vessels, often leading to severe respiratory distress and cardiovascular compromise.4–6 If the point of airway obstruction is distal enough, it may not be possible to rescue the patient with intubation or tracheostomy.7–11 Orthopnea and stridor on presentation are associated with increased risk of airway compromise.1,12 Compression of the great vessels, such as the superior vena cava (SVC), which can be worse when lying flat, may lead to impairment of cardiac filling and cardiac output.13–15 Patients who exhibit features such as orthopnea, respiratory distress, tracheal compression, or great vessel compression may be considered high risk for anesthetic administration.2,6,12 In some cases, the distress is exacerbated during procedures such as biopsy or resection of the mass, particularly under general anesthesia, which directly leads to physiologic changes such as loss of muscle tone in the neck and airway.2,6,12 Those with high-risk features may progress quickly to respiratory arrest, cardiovascular collapse, or combined cardiorespiratory failure.2,6
Extracorporeal membrane oxygenation (ECMO) may be considered when the risk of the above outcomes is high. If the patient’s anatomy permits [5], ECMO can serve as a bridge to diagnosis and treatment in this patient population, after which the mass would presumably shrink and compressive effects decrease. The current literature includes case reports describing both venovenous (VV) and venoarterial (VA) ECMO rescue for mediastinal masses with cardiopulmonary instability.4,5,7,8,10,11,13–26 However, no large studies focusing on use of ECMO in mediastinal masses have yet been published.
Our goal in this study was to examine the outcomes of pediatric oncologic patients with mediastinal masses that required ECMO support.
Methods
The “Pediatric Hematology and Oncology Patients on Extracorporeal Membrane Oxygenation: Outcomes in a Multicenter, Retrospective Cohort, 2009–2021” study was a multicenter, retrospective cohort study that evaluated the mortality outcomes for children with oncologic diseases that required ECMO support. 27 The study was reviewed and approved by the institutional review board at the University of Texas Southwestern (UTSW). All contributing sites had a data use agreement with UTSW. Informed consent was waived. Patients less than 19 years of age at the time of receiving ECMO and had an oncologic diagnosis or hematopoietic stem cell transplant at 16 pediatric intensive care units (PICUs) in the United States and Israel between 2009 and 2021 were included in the study. The data was collected as previously published.
For this sub-analysis, patients whose oncologic disease presented as a mediastinal mass were included. Baseline clinical data collected included demographics, oncologic diagnoses, mass related complications, and comorbidities. ECMO-specific data collected included ECMO indication, ECMO configuration, time between intubation and ECMO cannulation, cardiopulmonary resuscitation (CPR) prior to ECMO initiation, and occurrence of extracorporeal cardiopulmonary resuscitation (ECPR). ECMO complications were also collected, including bleeding, clotting, and mechanical difficulties. Laboratory data was collected for the time of ECMO initiation and for the first 7 days of the ECMO course. Outcomes collected included ECMO survival, hospital survival, duration of ECMO course, ICU length of stay (LOS), hospital LOS, and indication for ECMO decannulation.
Descriptive statistics were utilized for this analysis to describe demographic and clinical characteristics and outcomes. Due to non-parametric data, medians and interquartile ranges (IQR) were used to analyze the data. Categorial variables were compared using Fisher’s exact test and continuous variables were compared using unpaired t-tests.
Results
Patient demographics and presenting features
Patient demographics and presenting features.
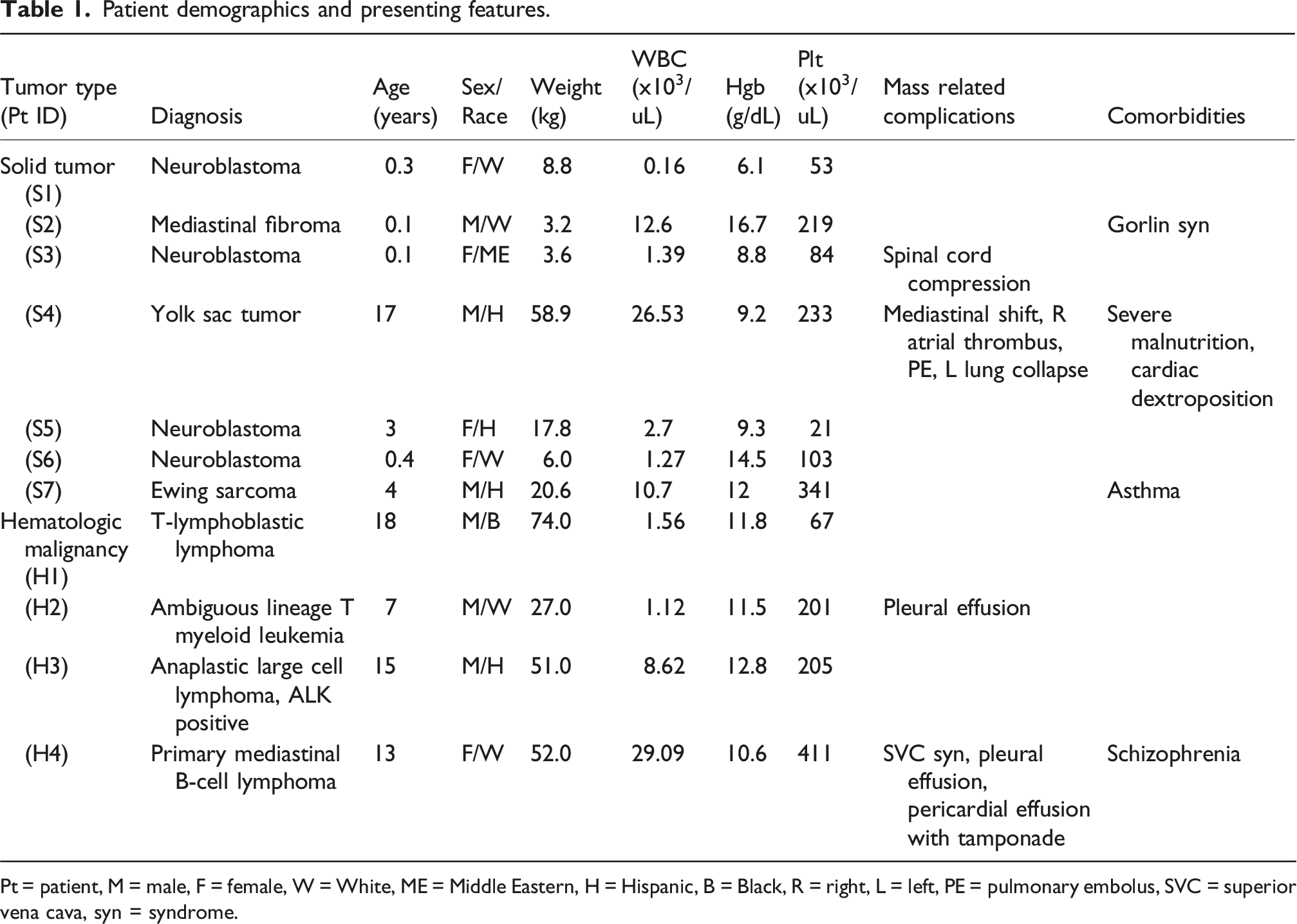
Pt = patient, M = male, F = female, W = White, ME = Middle Eastern, H = Hispanic, B = Black, R = right, L = left, PE = pulmonary embolus, SVC = superior vena cava, syn = syndrome.
ECMO characteristics
ECMO characteristics.
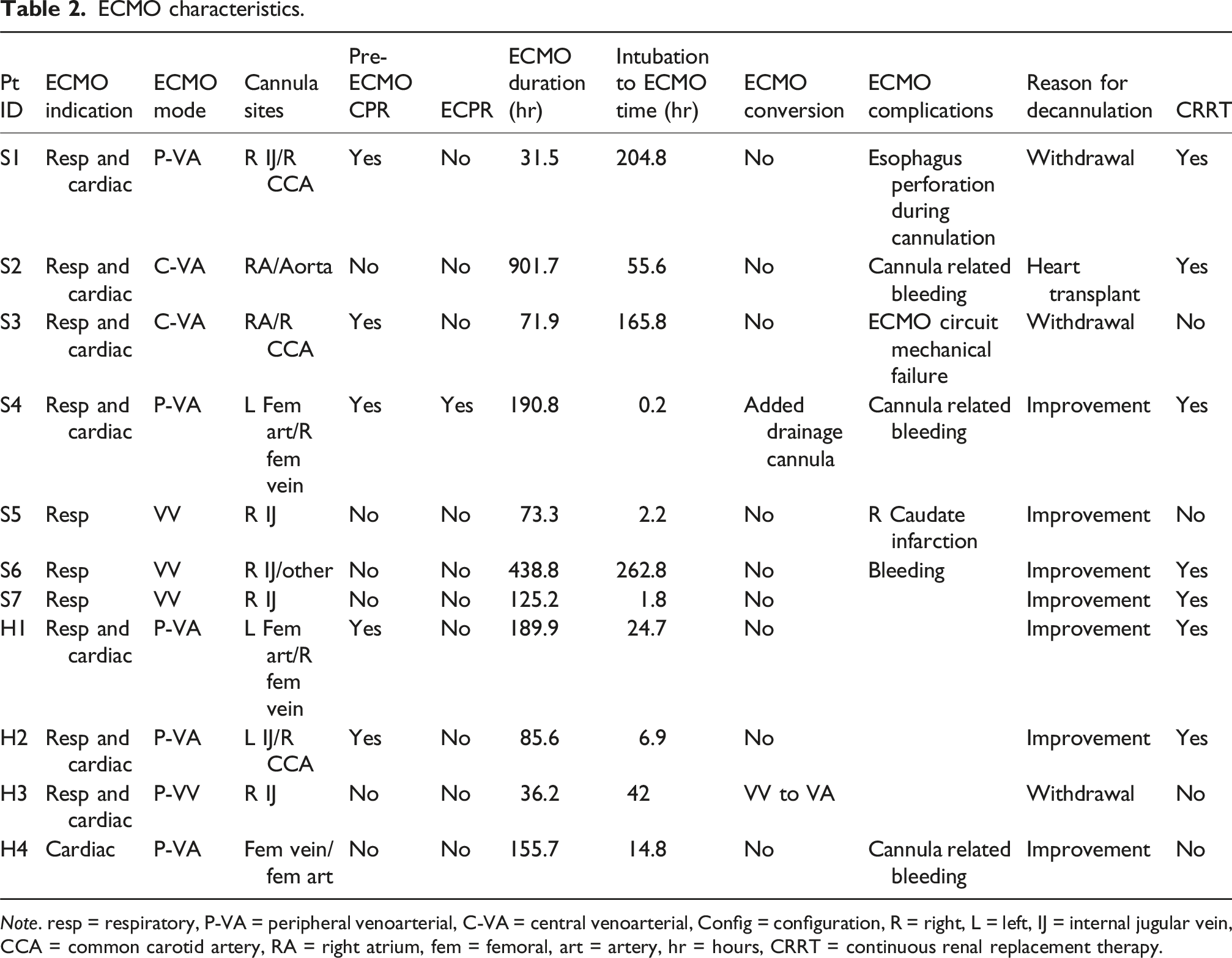
Note. resp = respiratory, P-VA = peripheral venoarterial, C-VA = central venoarterial, Config = configuration, R = right, L = left, IJ = internal jugular vein, CCA = common carotid artery, RA = right atrium, fem = femoral, art = artery, hr = hours, CRRT = continuous renal replacement therapy.
Outcome data
Patient outcomes.
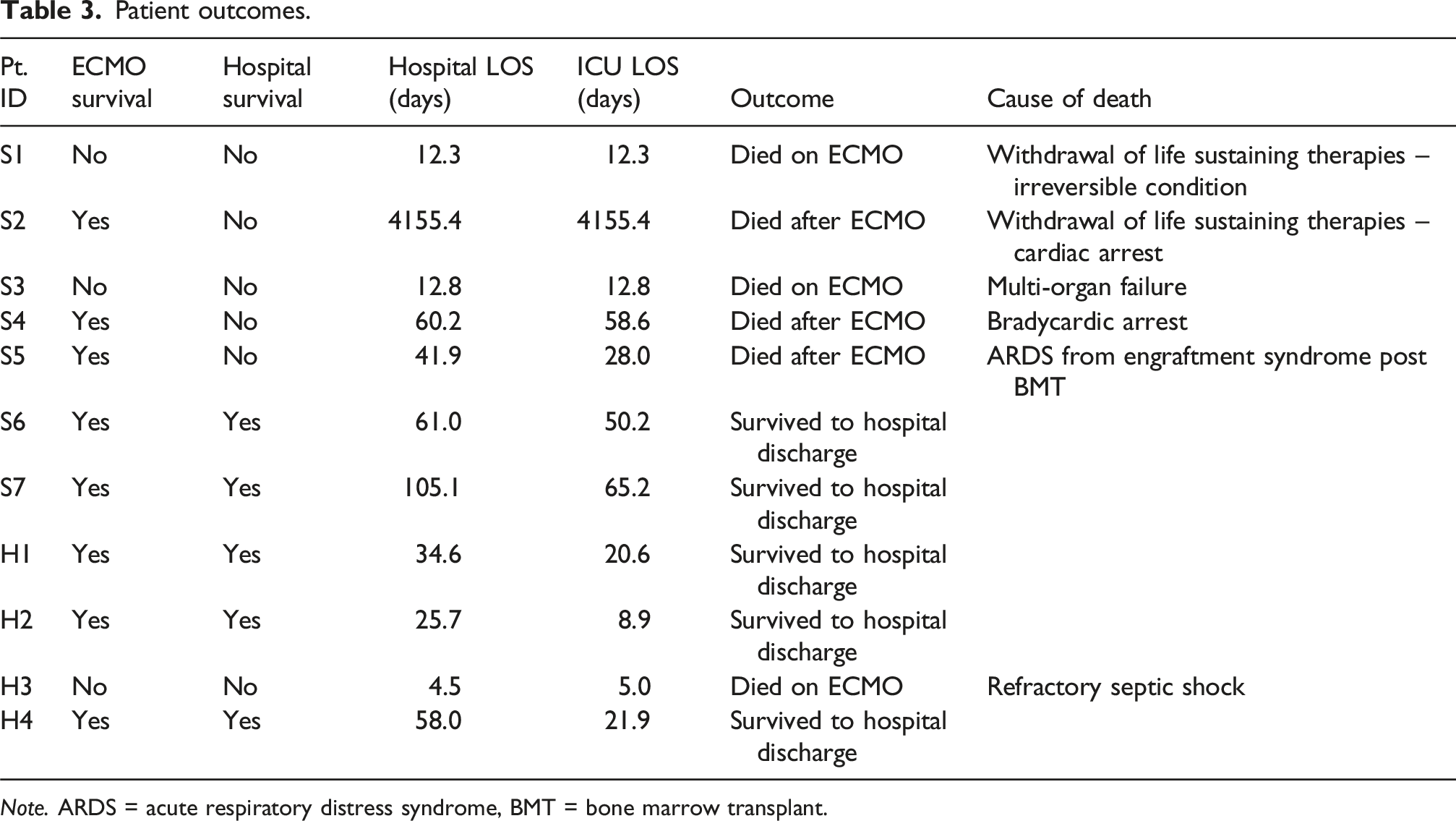
Note. ARDS = acute respiratory distress syndrome, BMT = bone marrow transplant.
Outcomes, overall and by tumor type.
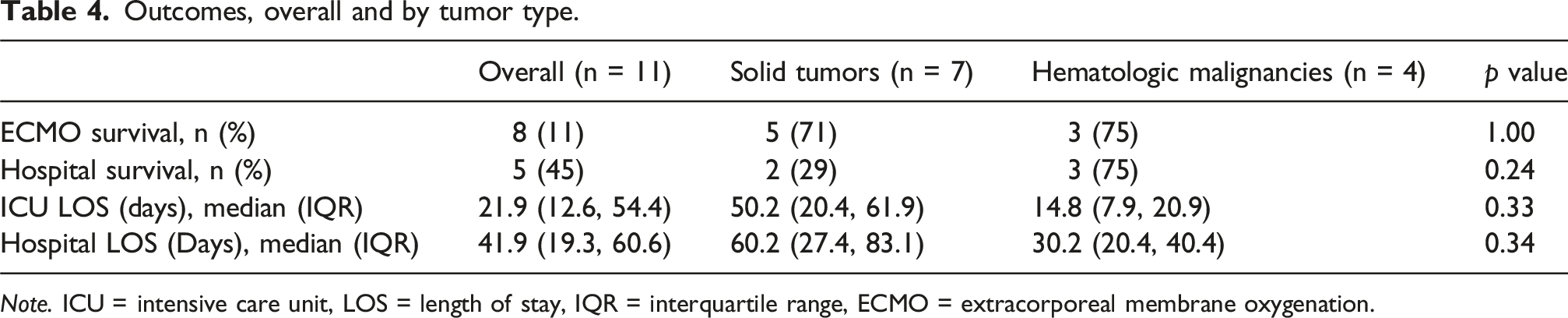
Note. ICU = intensive care unit, LOS = length of stay, IQR = interquartile range, ECMO = extracorporeal membrane oxygenation.
Discussion
In this investigation of mediastinal masses within a larger subset of hematology and oncology patients who require ECMO, we were able to identify 11 patients for further analysis. Although the number of patients included was small, it is still the largest study comparing pediatric mediastinal masses requiring ECMO to date. Our ECMO survival outcomes between both hematologic malignancies and solid tumor malignancies was 75%. However, survival to hospital discharge remained low at 45% overall. The discrepancy between ECMO survival and survival to hospital discharge is similar to the larger pediatric hematology and oncology study from which this study is derived, which reported a 55% ECMO survival and a 37% survival to hospital discharge. 27 An ELSO registry study reported a similar survival to hospital discharge in pediatric patients with neoplasm as approximately 40% across 2 decades. 28 In comparison, overall pediatric ECMO survival is 55-62% depending on ECMO indication. 29 Our study supports the current literature in demonstrating that post ECMO survival to hospital discharge in oncologic patients is lower than that of the overall pediatric population.
We found that mortality in patients who required CPR prior to ECMO cannulation was 60%. In our study, all of the patients supported by ECMO who died after CPR had underlying solid tumor malignancies. The larger overall study by Mowrer et al. demonstrated an association between CPR and mortality both on and after ECMO of 75% in pediatric patients with hematologic and oncologic disease. 27 However, due to small sample size, it is difficult to correlate the mortality in our sub-analysis with CPR given the high mortality in the solid tumor group overall.
The malignancies that most frequently cause mediastinal masses including acute lymphoblastic leukemia (ALL), Hodgkin lymphoma, neuroblastoma, thymoma, and germ cell tumors.1–3 Childhood and adolescent cancer 5-years survival in hematologic tumors varies from 90% in ALL to 95% in Hodgkin lymphoma.30,31 Solid tumor survival is generally lower reaching around 80%, with a 5-years survival rate of 50% in those with high-risk neuroblastoma. 32 Hematologic cancers generally respond robustly to chemotherapy. 33 Solid tumors may require prolonged courses of chemotherapy or debulking to achieve tumor shrinkage. 34
Several case reports have been published regarding the utilization of ECMO in both adult and pediatric patients with mediastinal masses.4,5,7,8,10,11,13–26 The majority of case reports included hematologic malignancies. While nearly all patients survived to hospital discharge, we recognize the potential for inherent bias in submitting cases with favorable outcomes for publication. We postulate that the high ECMO survival rate reflected in the literature as well as our study in hematologic malignancies is due to the rapid reversibility of the compressive effects of mediastinal masses with chemotherapy. ECMO cannulation can be safely considered in patients with hematologic malignancies as a rescue therapy until they respond to treatment. As solid tumors may take several weeks or months to respond to chemotherapy and may ultimately require surgical resection, ECMO may be considered as a bridge to treatment in this population with careful patient selection on a case-by-case basis due to high mortality following ECMO.
The limitations of this study include its retrospective nature and small sample size, which is reflected in the lack of statistical significance of our findings. In addition, there were missing variables in the data set, including CT imaging and echocardiogram findings demonstrating radiographic high risk features (such as tracheal or great vessel compression) for mediastinal masses.
In this sub-analysis of a multicenter study of pediatric oncologic patients with mediastinal masses supported on ECMO in 16 centers over a 12-year period, we found that overall patient survival was 45%. Upon further separation into subgroups based on oncologic disease, hematologic malignancy patients demonstrated favorable outcomes, and our study therefore supports ECMO candidacy as a rescue for these patients. However, patients with solid tumors had worse outcomes in comparison to hematologic malignancies in terms of mortality, hospital LOS, and ICU LOS, and clinicians should carefully consider ECMO candidacy in this population given the high post-ECMO mortality rate. Future studies should focus on the post-ECMO period to better support these patients. A prospective data registry would be useful in identifying risk factors that may contribute to the discrepancy between ECMO survival and overall mortality. Further management strategies and therapies could then be targeted to improve outcomes.
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of the investigators involved in the original multicenter cohort study (Mowrer et al., Pediatr Crit Care Med. 2024; PMID: 39028213), whose efforts in data collection and study design made this sub-analysis possible. We specifically recognize the following collaborators for their role in the original paper: Lisa Lima, MD; Xilong Li, MD; Hitesh Sandhu, MD; Brian Bridges, MD; Ryan Barbaro, MD; Raymond Nkwantabisa, MD; Taylor Olson, MD; Matthew Malone, MD; Neel Shah, MD; Matt Zinter, MD; Jon Gehlbach, MD; Laura Hollinger, MD; Briana Scott, MD; Reut Kassif Lerner, MD; Thomas V Brogan, MD; Renee M Potera, MD.
Ethical considerations
This study is a sub-analysis of a previously conducted study approved by the Institutional Review Board of University of Texas Southwestern. No additional ethical approval was required for this sub-analysis. All original data collection complied with ethical standards.
Consent to participate
Informed consent was waived.
Author contributions
All ten authors have participated sufficiently and intellectually and fully contributed to the work to take public responsibility for the content of this article, including the conception, design, and conduction of the study. Substantial contributions to the conception of design, acquisition of data, or analysis and interpretation of the data were made by N.C., S.N., A.O., M.C.M., S.G., R.N., A.R., L.R., and S.B. Drafting the article and revising it critically for important intellectual content was by N.C., S.N., A.O., J.T., M.C.M., S.G., A.R., L.R., and S.B. Final approval of the version to be published was provided by N.C., S.N., A.O., J.T., M.C.M., S.G., R.N., A.R., L.R., and S.B.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
