Abstract
Negative pressure ventilation (NPV) has been used to assist patients with respiratory failure. Venovenous extracorporeal membrane oxygenation (V-V ECMO) supports patients with acute lung injury who do not respond to conventional therapy. Some patients remain on prolonged ECMO support without access to lung transplantation. We report a 40-year-old woman cannulated for V-V ECMO due to refractory hypoxemia (PaO2/FiO2 of 58) on hospital day 20 for ARDS from bacteremia and native mitral valve endocarditis. After 108 days, she continued to require ventilator and ECMO support. Applying a negative-pressure Cuirass device in combination with positive pressure ventilation (PPV) improved her carbon dioxide clearance, enhanced overall ventilation/perfusion matching, and facilitated decannulation. Using NPV to assist in V-V ECMO weaning is a novel application of a legacy technology. This case shows NPV as an effective adjunct to PPV for patients on prolonged ECMO support with no other options.
Keywords
Introduction
Venovenous extracorporeal membrane oxygenation (V-V ECMO) is used to support patients failing conventional therapy in acute respiratory distress syndrome (ARDS). Approximately 30% of cases are reported as requiring prolonged ECMO support, with approximately 50% of those patients never improving to the point of decannulation. 1 In the face of quiescent native lung recovery, absent access to lung transplantation, we are left with the difficult decision regarding continuing ECMO support indefinitely or pursuing end-of-life withdrawal. 2 When faced with these difficult decisions, it is our duty to use all available resources to enhance native lung mechanics.
Negative-pressure breathing is physiological. Dr Philip Drinker is known as the “father of mechanical ventilation” for inventing the iron lung in 1928. His negative pressure ventilator (NPV) relied on an external device to apply negative pressure around the thorax, reducing pleural pressure, increasing transpulmonary pressure, and aiding air entry and gas exchange. 3 Since the birth of the modern intensive care units in the 1960s, there has been a universal shift to the use of positive pressure ventilation (PPV). 4 PPV can improve safety and support by offering enhanced control over respiratory variables to achieve a desired minute ventilation and mean airway pressure. Recently, there has been renewed interest in NPV, with devices like the Cuirass BCV (Hayek Medical, USA). This device is used predominantly in the treatment of patients with neuromuscular weakness. 5 The combination of PPV with NPV has been shown to enhance global pulmonary mechanics. 6 Combined NPV and PPV has been applied in the treatment of ARDS. 7 To our knowledge, there have been no cases of NPV with PPV in treating patients on ECMO. The case described here provides an example where the combination of NPV to PPV allowed for the successful decannulation of V-V ECMO in a patient with ARDS. Written consent from the patient’s health care proxy was obtained before publication.
Case history
A 40-year-old female with systemic lupus erythematosus, common variable agammaglobulinemia, and severe gastroparesis requiring total parenteral nutrition (TPN) presented with Staphylococcus Aureus bacteremia and native mitral valve endocarditis.
Her course was complicated by hypoxemia needing intubation on hospital day-5, progressing to ARDS. Despite the implementation of ARDS best practices, she required V-V ECMO cannulation on day-20 due to refractory hypoxia and hypercarbia. A chest x-ray at the time of cannulation showed bilateral infiltrates (sFigure 1A). Her ECMO course included bilateral pneumothoraces (sFigure 1B) and multi-drug resistant infections. She underwent tracheostomy on ECMO day-30. She maintained a two-site cannulation strategy (internal jugular-femoral), working daily with physical therapy under our institutional ECMO mobility protocol.
On hospital day-128 (ECMO day-108), she was on pressure control (21 cm H2O over positive end expiratory pressure of 5 cm H2O, resulting in a tidal volume of 170 mL) with ECMO support (Flow 3.2 lpm, Sweep Gas Flow (SGF) 3.0 lpm, FdO2 of 0.5). Her arterial blood gas showed pH 7.31, PaCO2 70 mmHg, PaO2 88 mmHg. Due to TPN dependence and chronic fraility, she was not a candidate for lung transplantation.
The NPV device was then applied (sFigure 2) with a continuous negative pressure of −20 cmH20f while maintaining pressure control ventilation. Her tidal volume increased from 170 mL to 250 mL, reaching 350 mL in 24 h. This allowed for weaning of the SGF while maintaining PaCO2 and arterial pH, prompting decannulation from ECMO on day-114 (Figure 1(a)). Figure 1(b) and (c) also illustrates the improvement in chest x-ray aeration, along with the trends in tidal volume and SGF (Figure 1(d)), following the initiation of Cuirass NPV with PPV. (a) Plot of ECMO sweep gas flow and tidal volume illustrating the increase in native lung tidal volume and subsequent decrease in ECMO sweep gas flow following the initiation of Cuirass NPV + PPV (b) Chest-Xray prior to the initiation of Cuirass NPV + PPV (c) Chest – Xray following the initiation of Cuirass NPV + PPV (d) Table illustrating the SGF requirements against arterial PaCO2 and pH pre and post Cuirass NPV + PPV initiation.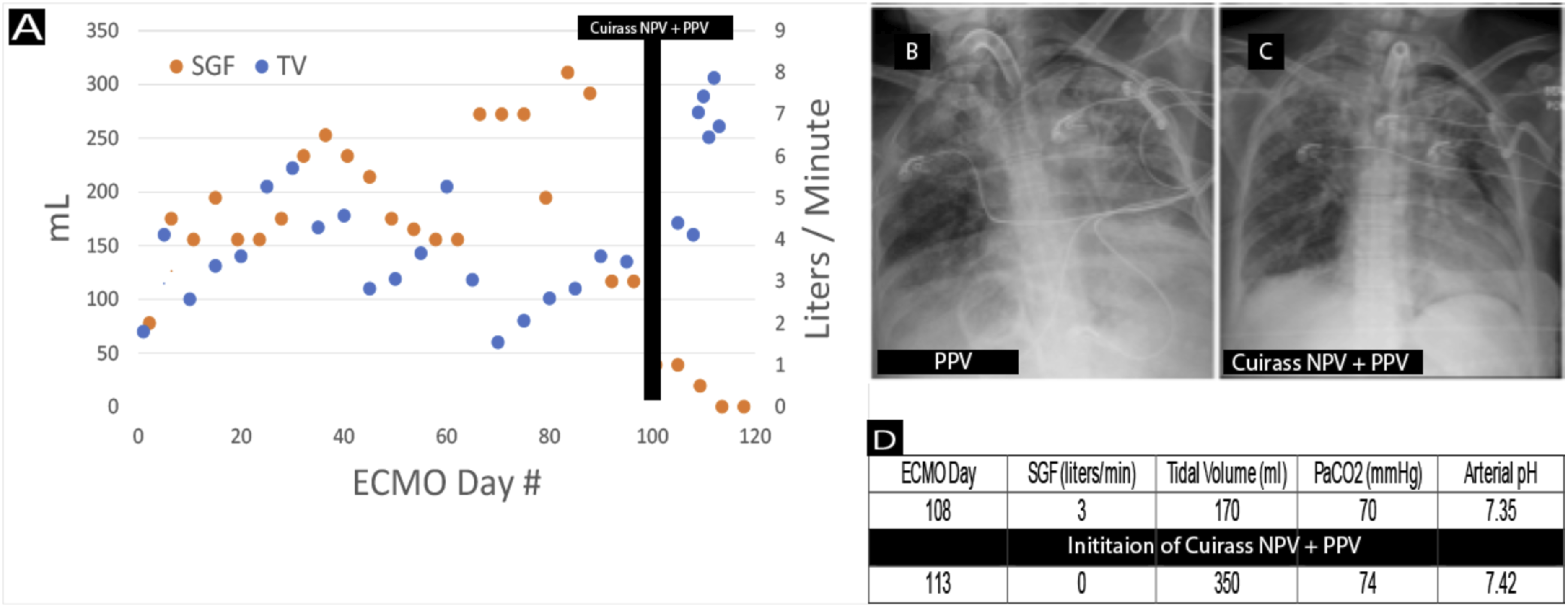
The patient’s lung mechanics continued to show evidence of improvement. However, 1 month after ECMO decannulation, she developed septic shock from pneumonia progressing to multi-organ failure. Her family opted for comfort measures, and she died after a 7-months hospital stay.
Discussion
In ARDS, heterogeneous injury contributes to ventilation–perfusion mismatch, persistent dead-space fraction, and limits effective gas exchange. 8 The conventional treatment with PPV is pressure-limited, preferentially delivering tidal volume to areas of high compliance, often leading to alveolar over-distention, while areas of low compliance (atelectatic regions) remain under-ventilated. 9 This impaired gas exchange can be a rate-limiting step in the effective weaning of ECMO. NPV applies a uniform negative pressure across the chest, effectively pulling air into lung regions previously quiescent to gas exchange, allowing a more even ventilatory distribution and improving lung compliance by recruiting regions of alveolar collapse.3,10
The use of combined PPV and NPV promotes more homogeneous lung inflation, improved ventilatory distribution, reduces alveolar over-distention, and assists diaphragmatic movement, offloading the muscular work of breathing. 11
In our patient, ARDS produced heterogeneous lung injury, dependent atelectasis, and critical illness-associated weakness. We suspect the primary benefit of NPV with PPV in this case was the redistribution of ventilation and recruitment of atelectatic lung regions, thereby improving gas exchange, reducing dead space fraction, all while lowering the patient’s overall work of breathing.
The overall goal of mechanical support with V-V ECMO is to extend treatment time to allow native lung recovery and to take back control over the work of breathing. In patients not eligible for lung transplantation, prolonged ECMO support remains a center-dependent strategy, and some patients may never be liberated successfully with standard mechanical ventilation. As international ECMO use expands, clinicians are increasingly encountering patients who remain ECMO-dependent for extended periods, underscoring the need for novel adjunctive strategies to support ECMO weaning. 12 This case describes the successful decannulation of V-V ECMO in a patient with severe ARDS and prolonged ECMO use, achieved through a combination of positive and negative pressure ventilation. Further studies are needed to define optimal timing, patient selection, and standardized protocols for its use.
Supplemental Material
Supplemental Material - Novel use of negative pressure ventilation for weaning of venovenous extracorporeal membrane oxygenation
Supplemental Material for Novel use of negative pressure ventilation for weaning of venovenous extracorporeal membrane oxygenation by Adam Green, Sandhya Ashokkumar, Nafisa Wadud, Ben Feng, Nitin Puri, and Jason Bartock in Perfusion
Footnotes
Consent for publication
Written informed consent was obtained from the patient’s guardian for the publication of any potentially identifiable images or data included in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
