Abstract
Introduction
Optimal sedation strategies for patients with respiratory failure requiring venovenous extracorporeal membrane oxygenation (VV ECMO) are not well defined.
Methods
Physicians caring for patients on ECMO in an intensive care unit were invited to complete an international survey about sedation and analgesia practices for adult patients undergoing VV ECMO for severe Acute Respiratory Distress Syndrome (ARDS).
Results
The survey was completed by 234 physicians, most commonly identifying as critical care (29.9%) and practicing in North America (38.9%). The Richmond Agitation Sedation Scale (88.5%) and Clinical Pain Observation Tool (40.2%) were commonly used for monitoring. Agents of choice were propofol (73.9% prior to ECMO and 56% within 48 h after initiation) and fentanyl (57.3% and 40.2%), with increasing preference for dexmedetomidine after ECMO initiation (14.5% prior to and 25.6% after initiation). Routine neuromuscular blockade use beyond 48 h after cannulation was rare (1.3%). Common strategies to reduce sedatives included sweep gas titration (74.4%) and minimizing nocturnal interventions (63.7%). Most respondents believe antipsychotics also work to this effect (57.3%).
Conclusions
Sedation strategies for adult ARDS patients receiving VV ECMO seem to be influenced by time from cannulation and patient factors. Future research should focus on identifying optimal sedation strategies and developing a consensus for best practice.
Keywords
Introduction
Sedation practices in adult patients with respiratory failure requiring invasive mechanical ventilation have changed significantly over the past 20 years. Current guidelines prioritize analgesia and recommend targeting symptom-guided sedation. 1 Despite these guidelines, current clinical practices for patients with respiratory failure requiring venovenous extracorporeal membrane oxygenation (VV ECMO) are not well defined. Variability in practice stems from the competing concerns of oversedation and the consequences of paralysis leading to ICU acquired weakness (ICUAW) and diaphragm atrophy on the one hand, and patient safety with ECMO cannulas, patient self-induced lung injury (P-SILI), and challenges achieving lung protective ventilation on the other. Concerns about ECMO circuit induced changes in volume of distribution, dilution, and sequestration of commonly used medications likely also influences practice. 2
Prior studies have explored defining current clinical practices through provider surveys.3–5 The most recent survey, by Dzierba et al., found that validated sedation and pain scales were commonly used and that sedative preferences varied based on the targeted level of sedation.
While these studies provided valuable information at the time of their publication, the impact of the subsequent coronavirus pandemic and resulting rapid increase in ECMO centers worldwide on practice patterns remains unknown.
In this study, we aimed to characterize current analgesia and sedation practices in adult patients with respiratory failure secondary to severe Acute Respiratory Distress Syndrome (ARDS) requiring VV ECMO support in an intensive care unit (ICU) setting, with the goal of understanding current practices and identifying areas for future research.
Other ECMO modes such as venoarterial ECMO (VA ECMO) and non-ARDS etiologies for respiratory failure (e.g. chronic lung disease with ECMO as bridge to lung transplant) were excluded in order minimize confounding from practice variability related to ECMO modality and patient pathology.
Methods
Survey creation
Data were collected through an online survey developed by the authors using REDCap electronic data capture tools, distributed from Cooper University Healthcare.6,7
A writing group was established that included authors from multiple disciplines and included international representation and Extracorporeal Life Support Organization (ELSO) involvement. Study questions were created through consensus among the authors and feedback received after pilot testing from physicians at the authors’ centers who were not involved in the study.
Questions targeted physician practices and perceptions regarding analgesia and sedation for patients requiring VV ECMO for severe ARDS. We explicitly excluded cases of venoarterial, venopulmonary, extracorporeal CO2 removal and VV ECMO for non-ARDS causes of respiratory failure. Questions pertaining to the percentage of patients achieving a specified highest level of mobility were optional. All other questions were required. Questionnaires were considered complete if all required questions were answered. The survey was determined to be exempt from the Cooper University Hospital Institutional Review Board approval as the board determined that it did not qualify as human subjects research.
Survey administration
Physicians who serve as the providers for adult patients receiving VV ECMO for respiratory support in an ICU were invited to complete the survey. Survey respondents were identified using a survey that was distributed through multiple venues, including the ELSO electronic newsletter (>20,000 users & center members), 8 ELSO Adult Respiratory WhatsApp (Meta, Menlo Park, CA) group chat (>1000 members), International Society for Heart and Lung Transplantation (ISHLT) mechanical support discussion board (>3800 members in >45 countries), 9 Europe (EURO) ELSO newsletter, EURO ELSO app, and word of mouth by the authors. The survey was also directly emailed to all ELSO member center program coordinators and medical directors whose contact information was publicly available on the ELSO website (693 centers, including non-adult centers, at the time of survey distribution) 10 with a request to invite physicians in their respective ECMO programs to complete it. Respondents were offered a $10 Amazon Gift Card (Amazon, Seattle, Washington) as a stipend for their time.
By distributing the survey through newsletters and other online communication platforms, it was not possible to track the response rate of eligible physicians.
Statistical analysis
Qualitative variables are reported as numbers and percentages, while quantitative variables are reported as median and interquartile range unless otherwise specified. To assess general sedation practices, a descriptive analysis was performed to determine the frequency and percentage of all responses. Practice patterns were then further compared by center case volume, center location, and physician specialty. All statistical analyses were performed using R version 4.1.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 376 survey responses were collected between September 2024 and January 2025, of which 136 were excluded due to being incomplete. Six were excluded because they were provided by non-physician providers, leaving 234 complete physician responses for analysis. Incomplete survey responses (i.e., not electronically submitted) were excluded because they represented abandoned surveys. The overall response rate could not be determined due to the dissemination process.
Respondent characteristics
The majority of surveys, 85.5% (n = 200), were completed by physicians practicing at ELSO member centers. A total of 143 ELSO centers participated, with a wide range of annual case volumes. There was also international representation with a broad distribution.
Characteristics of survey respondents.

Sedation and analgesia monitoring
Sedation and analgesia monitoring.
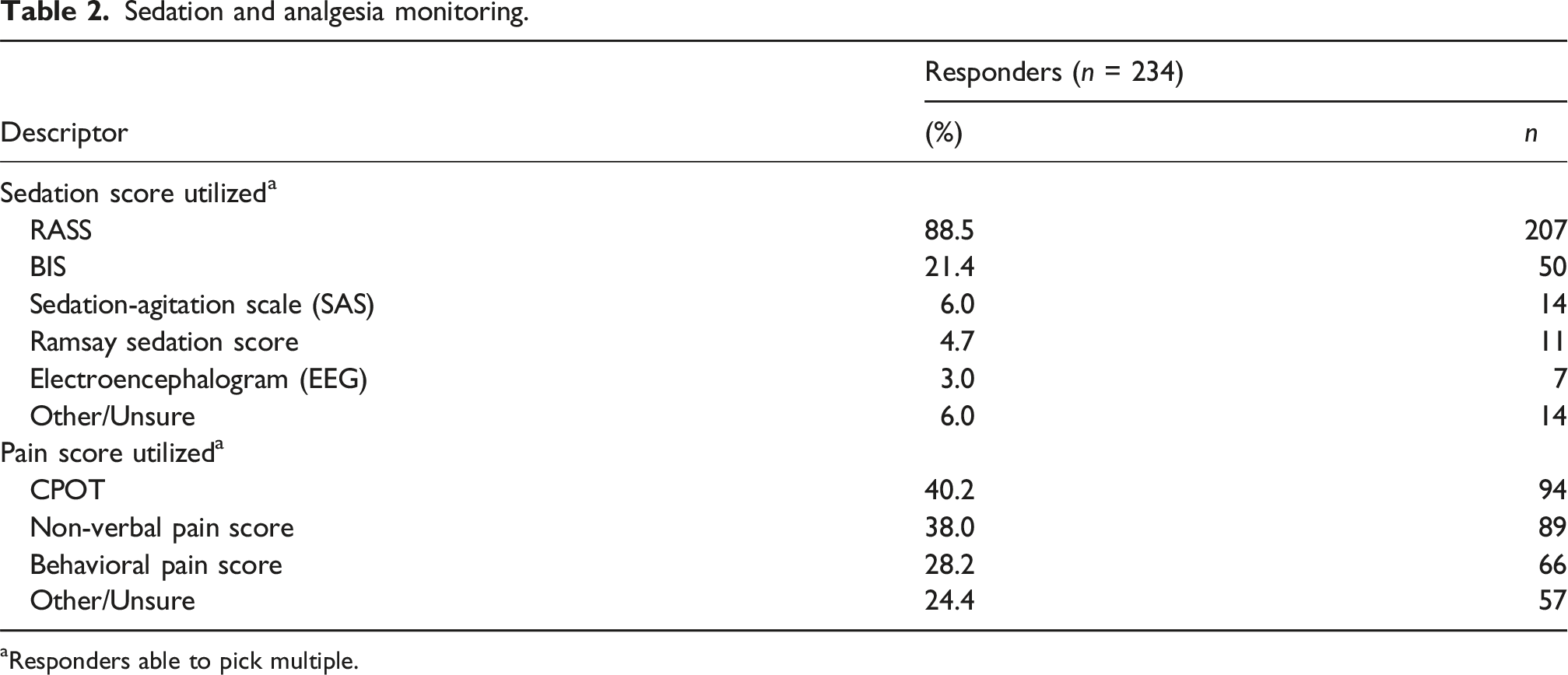
aResponders able to pick multiple.
Neuromuscular blockade use
In non-ECMO severe ARDS patients, 47.0% (n = 110) of respondents stated neuromuscular blockade was used most (38.5%, n = 90) or all (8.5%, n = 20) of the time. However, in the patients with severe ARDS at the time of cannulation that number increased to 71.4% (n = 167) with a similar number responding to most of the time (42.3%, n = 99) but a significantly higher number stating all (29.1%, n = 68) of the time.
Despite a high percentage of centers using neuromuscular blockade, there was evidence that these centers attempted to discontinue paralysis after cannulation. Very few stated neuromuscular blockade was used routinely for >48 h after cannulation (1.3%, n = 3). Most centers appear to discontinue within 48 h of cannulation (<8 h: 23.9%, n = 56, 8–48 h: 23.5%, n = 55) (Supplemental Material Table 1).
Sedation goals
Sedation goals & sedation weaning.
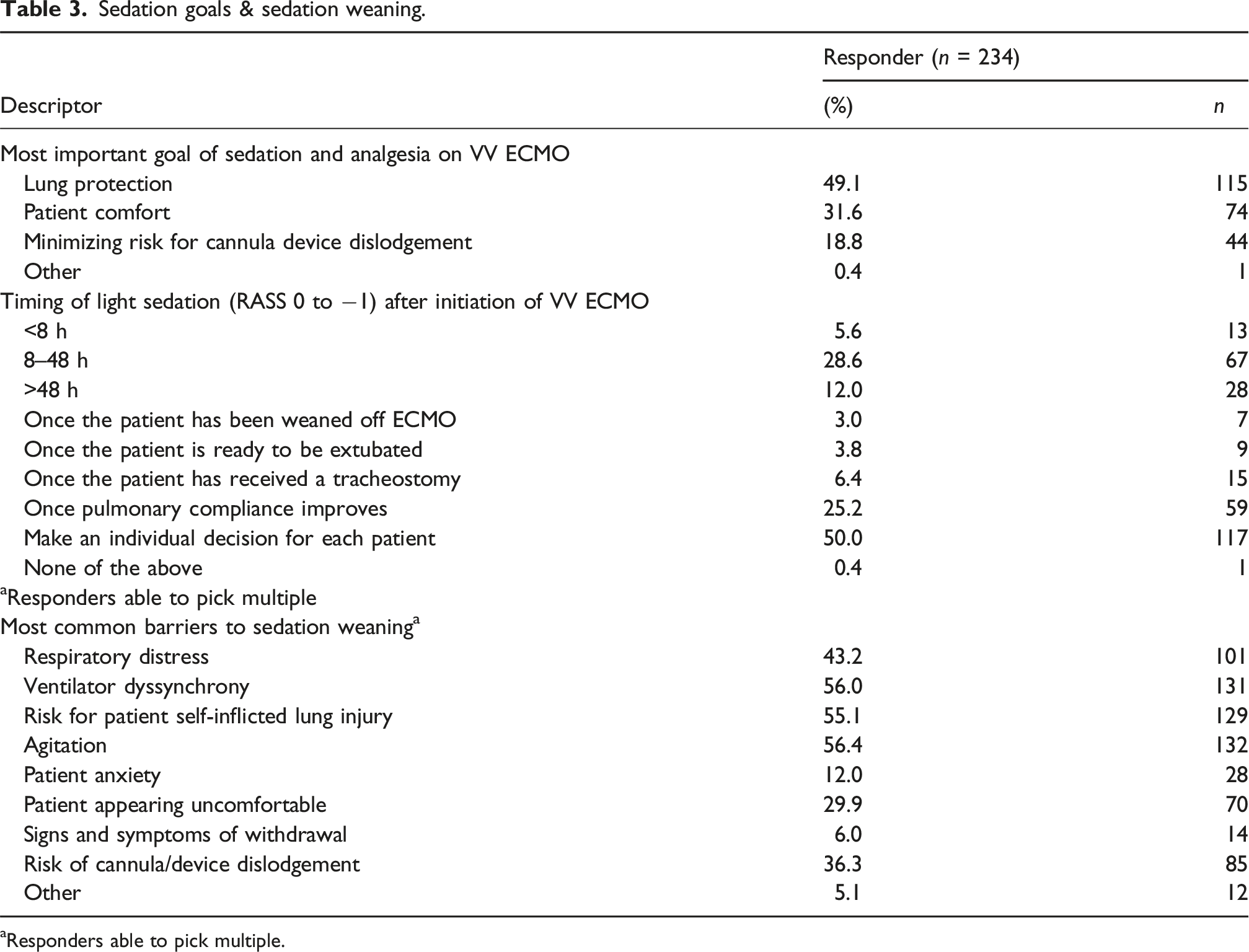
aResponders able to pick multiple.
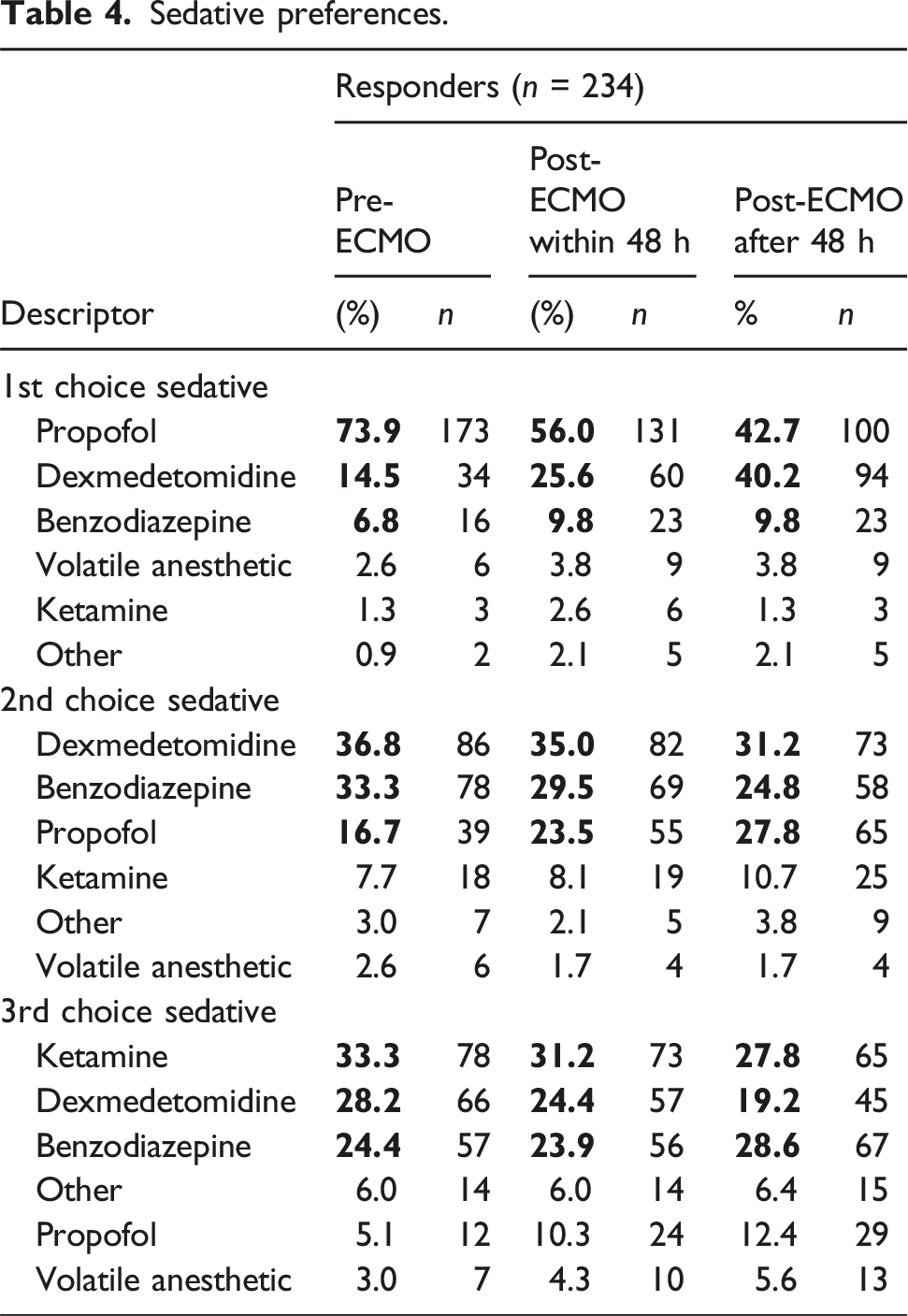
Preferences in sedatives
Sedative preferences.
Preferences in analgesics
Analgesic preferences.
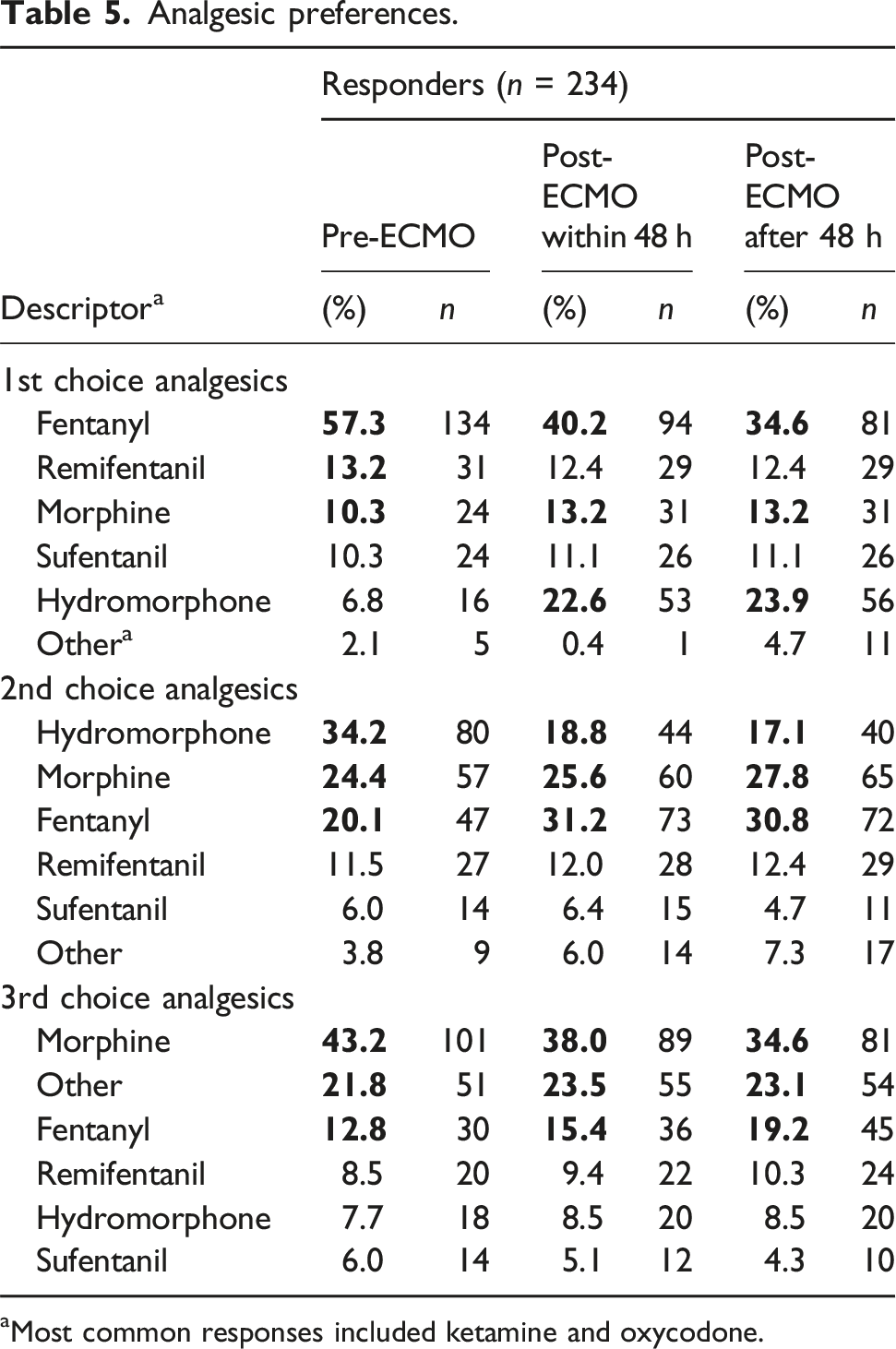
aMost common responses included ketamine and oxycodone.
Analgesic administration
Analgesia appears to be given largely as a continuous infusion during the period surrounding cannulation: 85% (n = 199) pre-ECMO and 88.5% (n = 207) within 48 h of cannulation. However, similar to neuromuscular blockade, there was a decrease in reported use of continuous opioids more than 48 h after cannulation (68.4%, n = 160) (Supplemental Material Table 2).
Adjunct medications and strategies
There was an inconsistent response regarding the proportion of patients who received scheduled antipsychotics. Interestingly, over half of the participants (57.3%, n = 134) either agree or strongly agree that antipsychotics are effective.
The most common interventions to reduce sedation included increasing sweep gas to decrease respiratory drive (74.4%, n = 174), minimizing nocturnal interventions (63.7%, n = 149), and implementing bundled care (51.3%, n = 120) (Supplemental Material Table 3).
Liberation from sedation and mechanical ventilation
Barriers to sedation weaning included respiratory distress (43.2%, n = 101), ventilator dyssynchrony (56%, n = 131), risk of patient self-inflicted lung injury (55.1%, n = 129), patient agitation (56.4%, n = 132) and cannula dislodgement (36.3%, n = 85) (Table 4).
Most patients remain on mechanical ventilation while being supported with VV ECMO, with two-thirds of survey responses stating either none (13.7%, n = 32) or few (37.2%, n = 87) are liberated (Supplemental Material Table 4).
Mobility
Most respondents aim to mobilize patients beyond sitting over the edge of the bed (87.2%, n = 204). However, it is clear that many patients do not achieve significant mobilization, with 10.9% (n = 22 out of 201 respondents) reporting that none of their patients are mobilized (Figure 1). Highest Level of Mobility Achieved on VV ECMO*. Reported median percentage of patients achieving each category as their highest level of mobility on VV ECMO. Patients were only counted in the category representing their highest level of mobility. Bars represent the Q1–Q3 interquartile range. *Responses optional to this survey question.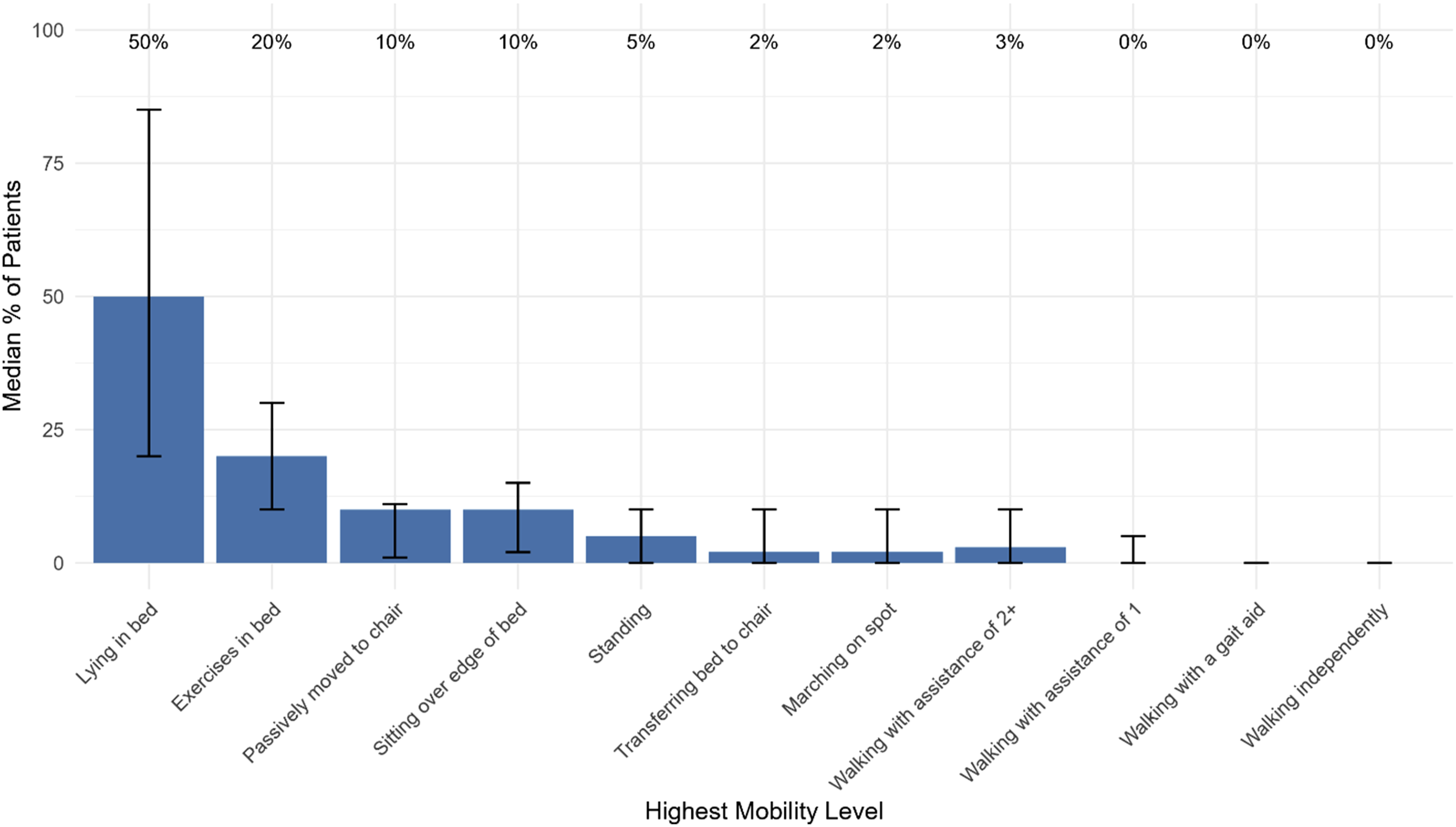
Responses stratified by annual case volume, physician specialty, and location
No significant trends were observed when responses were stratified by estimated annual VV ECMO case volume, physician specialty, or center location. The number of responses in each category was limited to any statistical comparison. These results are included in the Supplement Material.
Discussion
While critical care guidelines continue to offer a general practice framework, 1 the best strategies for patients needing VV ECMO for respiratory support are not clearly defined. Providers must weigh often conflicting goals to find the most suitable approach.
In this international survey of physicians caring for adult ICU patients requiring VV ECMO for ARDS, we aimed to understand their practice patterns regarding sedation and analgesia.
Two main themes were identified: first, although individual physicians may have preferred practice patterns, decisions must be made at the patient level; second, there is a general trend toward decreasing sedation as the ECMO course progresses. However, we also observed significant variation in goals and practices for analgesia and sedation that differed among providers and at different points in the ECMO timeline.
In general, propofol for sedation and fentanyl for analgesia were the preferred agents throughout the ECMO course. This finding aligns with prior research on ECMO patients and suggests that pharmacodynamic concerns about circuit sequestration are less significant considerations for agent selection.4,11
Previous surveys have characterized analgesia and sedation practices in ECMO, including patients requiring VV ECMO for ARDS.3–5 The most recent, published in 2019 by Dzierba, found that the sedation goal determined the choice of agent: propofol for deeper sedation, dexmedetomidine for lighter sedation. Our study, however, offers much more insight into how physicians approach these practices. As patients progress through their ECMO course, physicians tend to minimize sedation. This is reflected in stopping neuromuscular blockade and switching from propofol to dexmedetomidine. Additionally, adjuncts—such as antipsychotics and various non-pharmacological strategies—are used with differing levels of supporting evidence to reduce sedation needs.12,13
The most common intervention used, titration of the sweep gas to reduce respiratory drive, is important to understand. Achieving a PaCO2 ≤ 40 mm Hg has been associated with a greater chance of reaching a sedation goal in patients on VV ECMO for ARDS. 14 Therefore, this common clinical practice may help to minimize unnecessary use of sedatives.
A more alert ECMO patient can breathe independently, mobilize, and - in some cases-be liberated from mechanical ventilation. Excellent outcomes have been reported by some centers that prioritize liberation from mechanical ventilation while on VV ECMO. 15 While this approach is not common in prior work or in our study, 16 allowing patients to be awake and safely participate in their care is a reasonable goal. Understanding these barriers to this can help improve future care strategies. Our survey indicated that perceived barriers to weaning sedation mainly focused on pulmonary condition and mechanics, including respiratory distress, ventilator dyssynchrony, risk of self-inflicted lung injury, and patient anxiety.
The variation in reported preferences and practices among respondents is notable but similar to prior work, despite our respondents being limited to physicians.3–5 This may be partly due to the characteristics of this patient cohort. Severe ARDS is a heterogeneous clinical syndrome with many causes. 17 Maintaining sedation and analgesia in these patients can be challenging, as evidenced by the number of agents respondents reported using.
The common use of antipsychotics was surprising, with 41% of respondents indicating that they are administered to all or most patients to reduce other sedatives. The use of antipsychotics in ECMO patients has been previously described. 4 However, this was for delirium prevention and treatment, not to meet a sedation goal, as specified in our study. While general critical care guidelines recommend against routine antipsychotic use for delirium prevention and treatment, they do not address their use for sedation. 1
This study has several strengths. First, it has strong representation from international centers—61.1% of respondents were from non-North American centers—enhancing the validity of the findings as a reflection of current clinical practice. Second, it provides new information on the use of antipsychotics, sweep gas titration, and adjunct therapies that have not been previously examined. Third, it gathers a relatively high number of physician responses. Finally, understanding how practice patterns evolve during the ECMO course offers valuable insights. Prior work that gathered information on analgesic and sedative preferences by light or deep level of sedation – with propofol most commonly preferred for targeting deep sedation and dexmedetomidine preferred for light sedation. 4 Our study uniquely gathered information on agent preferences prior to, within 48 h, and greater than 48 h from ECMO cannulation. Propofol was preferred over dexmedetomidine at all time points, but the difference was smallest 48 h after cannulation (43% choosing propofol and 40% of choosing dexmedetomidine) by which time 47% respondents indicated they target light sedation.
This study also has several limitations. First, survey responses were voluntary and could be influenced by reporting bias and the provided survey completion award of $10 gift cards. Second, by distributing the survey through newsletters and online communication platforms, it was not possible to track the response rate of all eligible physicians. This limitation has been cited in prior work on this subject. 4 Our survey did include responses from physicians at 143 different ELSO centers out of over 750 centers worldwide. 18 Third, our study considered each medication and therapy separately, without examining combinations or bundled strategies. Fourth, we did not collect data on the causes of ARDS, and physicians’ approaches might be more consistent in a more narrowly defined patient group. Fifth, we limited the survey to physician responses and relied on their recollections. While this allowed our study to capture the preferences of this specific provider group, patient management decisions are also informed and made by other members of the care team. Finally, our study did not investigate the management of delirium, and practices may differ when delirium is present.
Conclusions
Overall, this study enhances our understanding of current clinical practice. There is ongoing variation among physicians in how they manage analgesia and sedation for adult ARDS patients on VV ECMO. However, common themes include the use of propofol and fentanyl-based regimens, implementation of validated sedation and pain scoring, and a trend toward decreasing neuromuscular blocking agents and lighter sedation targets over time. The choice of agents, use of paralytics, and sedation goals seem to be influenced by timing of cannulation, respiratory physiology, and patient agitation.
Variation can be expected in a growing field with evolving technology and applications. However, following specified analgesia and sedation strategies can greatly influence patient outcomes. 19 Therefore, future research should focus on identifying the best sedation strategies and reaching a consensus on best practices.
Supplemental material
Supplemental Material - Sedation practices in adult patients with severe ARDS on extracorporeal respiratory support using VV ECMO – An international survey
Supplemental Material for Sedation practices in adult patients with severe ARDS on extracorporeal respiratory support using VV ECMO – An international survey by Christopher Noel, Adam Green, Abhimanyu Chandel, Joseph Delio, Nitin Puri, Chase Donaldson, Craig Rackley, Steven . Conrad, Peter Rycus, Joseph E. Tonna, Akram Zaaqoq, Justyna Swol and Christopher King in Perfusion
Supplemental material
Supplemental Material - Sedation practices in adult patients with severe ARDS on extracorporeal respiratory support using VV ECMO – An international survey
Supplemental Material for Sedation practices in adult patients with severe ARDS on extracorporeal respiratory support using VV ECMO – An international survey by Christopher Noel, Adam Green, Abhimanyu Chandel, Joseph Delio, Nitin Puri, Chase Donaldson, Craig Rackley, Steven A. Conrad, Peter Rycus, Joseph E. Tonna, Akram Zaaqoq, Justyna Swol and Christopher King in Perfusion
Supplemental material
Supplemental Material - Sedation practices in adult patients with severe ARDS on extracorporeal respiratory support using VV ECMO – An international survey
Supplemental Material for Sedation practices in adult patients with severe ARDS on extracorporeal respiratory support using VV ECMO – An international survey by Christopher Noel, Adam Green, Abhimanyu Chandel, Joseph Delio, Nitin Puri, Chase Donaldson, Craig Rackley, Steven A. Conrad, Peter Rycus, Joseph E. Tonna, Akram Zaaqoq, Justyna Swol and Christopher King in Perfusion
Footnotes
Ethical considerations
The survey was determined to be exempt from Institutional Review Board approval as the board determined that it did not qualify as human subjects research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Justyna Swol, MD: Dr. Swol received honorariums from Getinge and Abiomed, is a member of ECPR Advisory Board of Getinge, and is an Associate Editor of Perfusion. Joseph E. Tonna, MD, MS: Dr Tonna is the Chair of the Registry Committee of The Extracorporeal Life Support Organization (ELSO) and receives funding from the NIH/NHLBI on: R01HL168510.
Data Availability Statement
Data can be provided upon written request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
