Abstract
Objective
We sought to compare early postoperative outcomes between mitral valve repair (MVr) and replacement (MVR) in patients with mitral valve endocarditis.
Background
The optimal surgical approach for mitral valve endocarditis remains controversial, with some studies suggesting that mitral valve repair may be associated with better outcomes than mitral valve replacement and others suggesting no significant differences. This study compares the early postoperative outcomes between repair and replacement in this cohort using a large national database.
Methods
A retrospective, propensity-score matched analysis was conducted using the national data from the United Kingdom, comparing 1381 patients who underwent repair and 3276 patients who underwent replacement between 2000 and 2019. The primary outcome was in-hospital mortality, and secondary outcomes included prolonged admission (>10 days), re-exploration for bleeding, postoperative stroke, and postoperative dialysis. Binary logistic regression models were conducted in the matched group to further examine the relationship between procedure and outcomes.
Results
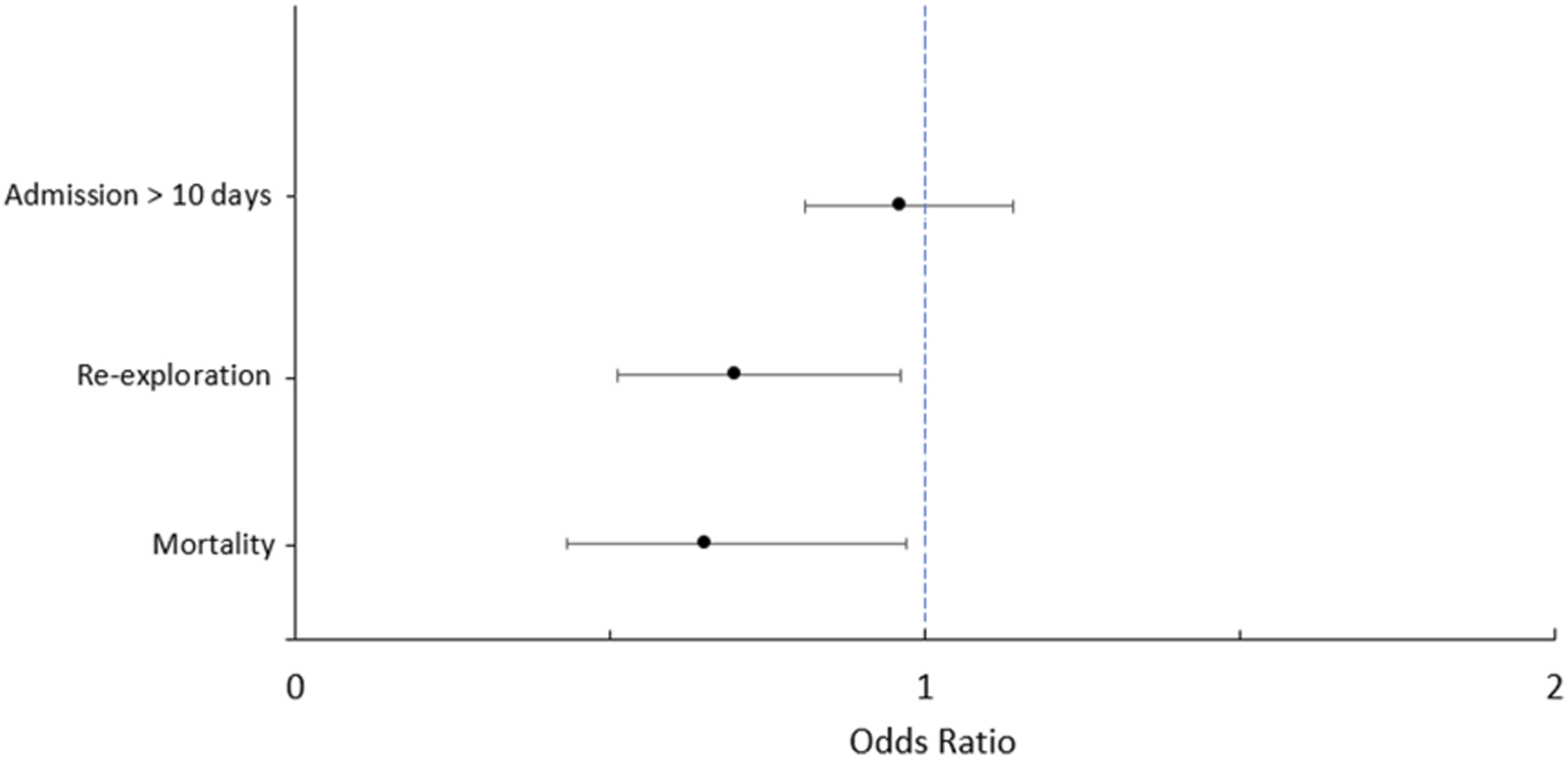
After propensity-score matching, 1249 pairs were identified. In-hospital mortality was significantly lower in the repair group (4% vs 7%, p < .001). Rates of re-exploration for bleeding (6% vs 9%, p = .019), postoperative stroke (1% vs 3%, p = .030), and postoperative dialysis (5% vs 7%, p = .016) were also significantly lower in the repair group. Binary logistic regression analyses demonstrated repair to be independently associated with lower risk of both mortality (OR:0.65, 95% CI:0.43-0.97, p = .034) and re-exploration for bleeding (OR:0.70, 95% CI:0.51-0.96, p < .028).
Conclusions
This study suggests that patients receiving repair for mitral valve endocarditis have significantly lower mortality and better early postoperative outcomes than those receiving replacement.
Introduction
Rates of infective endocarditis in the general population are rising, having increased from 26.6 cases per million in 2009-2010 to 50 cases per million in 2018-191. Infective endocarditis affecting the mitral valve (MV) is a serious condition that can result in significant morbidity and mortality if not promptly and effectively treated. The optimal surgical treatment for patients with this condition remains controversial, with mitral valve repair (MVr) and replacement (MVR) representing the two surgical options. Repair rates for MV endocarditis remain low. A large study in the United States demonstrated a repair rate of 20%. 2 There are, however, significant variations in practice, with one European centre quoting repair rates of 80%. 3
Given the relatively low repair rates and therefore, lack of consensus on whether to repair or replace in the context of MV endocarditis, there is a need for further research to better understand the optimal surgical treatment for patients with MV endocarditis. The aim of this study is therefore to compare early postoperative outcomes between MVr and MVR in a large cohort of patients using data from the National Adult Cardiac Surgery Audit (NACSA) database.
Methods
Data sources
Our analyses were conducted on the National Adult Cardiac Surgery Audit (NACSA) data, maintained by the National Institute for Cardiovascular Outcomes Research (NICOR). This national database contains clinical information on demographics, pre- and post-operative clinical information including mortality, for all major adult cardiac surgery procedures performed in the United Kingdom. Maintenance and validation are regularly undertaken by the use of reproducible cleaning and maintenance algorithms, with return to individual centres for local validation. 4 We obtained approval to conduct this study from NICOR and from our institution, the University Hospitals Bristol and Weston National Health Service Foundation Trust Clinical Audit Team, to carry out this research without requiring patient informed consent. The data was received with all identifiable patient information removed; the requirement for patient informed consent was waived and the dataset was adapted for use with statistical analysis software.
Missingness
The nature of missingness in the dataset was investigated using Little’s test, which returned a significant result (p < .001), indicating that data were not completely missing at random. In the following variables, we determined that missingness was likely to be related to the true value (percentage missingness stated in brackets): coronary disease (29%), preoperative AKI (28%), concomitant tricuspid surgery (65%), concomitant coronary artery bypass graft (32%), concomitant ablation (26%), in-hospital mortality (2%), postoperative stroke (12%), postoperative dialysis (11%), re-exploration for bleeding (8%). We therefore imputed missing values with ‘0’ in these variables. In the remaining variables, no attempts were made to replace missing values.
Ethics statement
The register-based cohort study is part of a research approved by the Health Research Authority (HRA) and Health and Care Research Wales and a need for patients’ consent was waived, as all patients in the database were anonymised (HCRW) (IRAS ID: 257758,23.7.2019).
Study population
In order to conduct this retrospective study, we obtained data on 4657 patients undergoing mitral valve surgery (MVS) for endocarditis between 2000 and 2019. We included concomitant coronary artery bypass graft (CABG) surgery, concomitant tricuspid valve (TV) surgery and concomitant ablation for atrial fibrillation (AF). We excluded concomitant aortic valve (AV) surgery, as well as patients with missing data for aetiology of MV disease or procedure date.
Outcomes
The primary outcome was in-hospital mortality. Secondary outcomes included re-exploration for bleeding, prolonged hospital stay (defined as greater than 10 days), post-operative stroke and post-operative dialysis.
Statistical analysis
Unmatched cases: univariable comparisons of baseline characteristics.
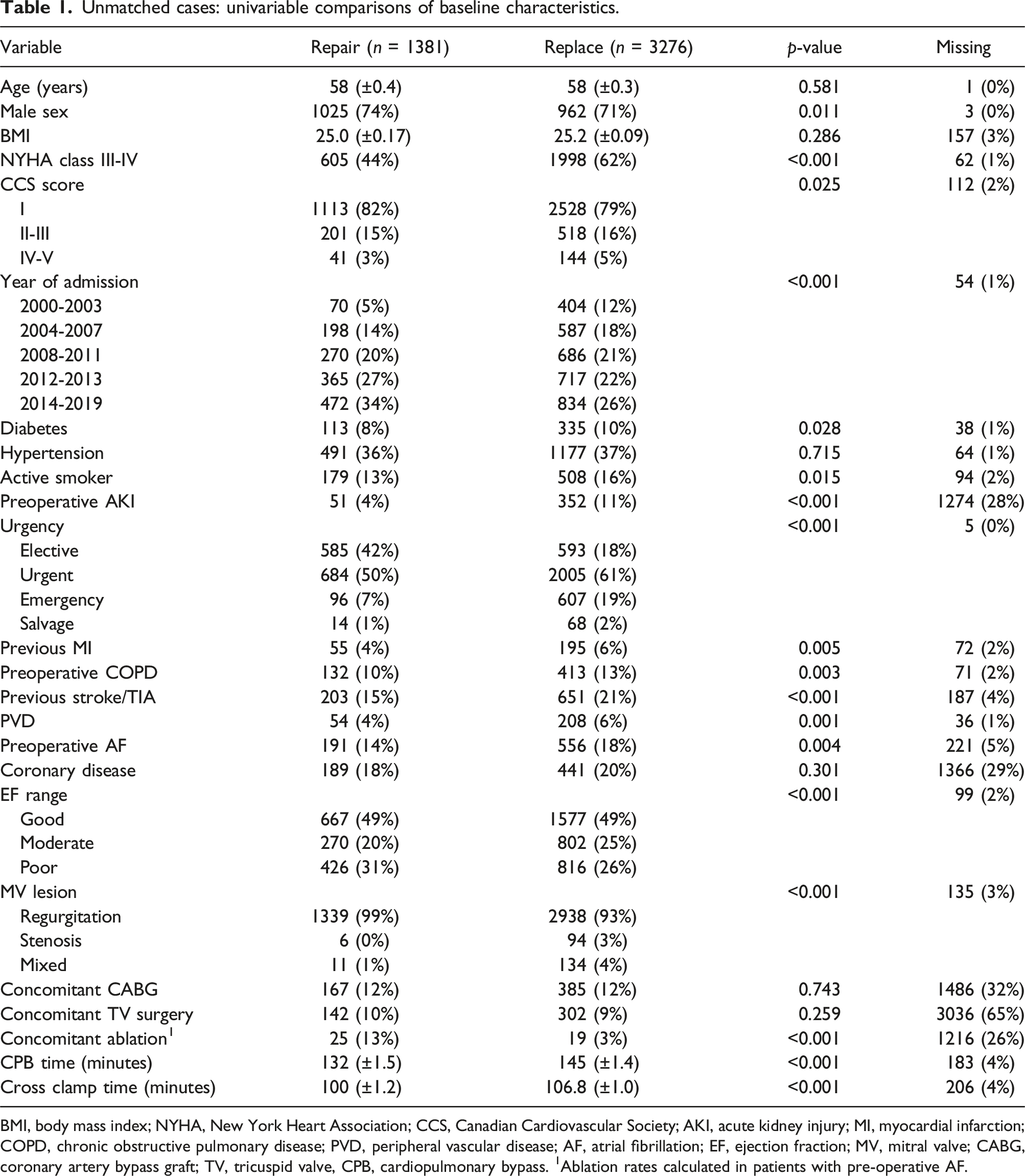
BMI, body mass index; NYHA, New York Heart Association; CCS, Canadian Cardiovascular Society; AKI, acute kidney injury; MI, myocardial infarction; COPD, chronic obstructive pulmonary disease; PVD, peripheral vascular disease; AF, atrial fibrillation; EF, ejection fraction; MV, mitral valve; CABG, coronary artery bypass graft; TV, tricuspid valve, CPB, cardiopulmonary bypass. 1Ablation rates calculated in patients with pre-operative AF.
Matched cases: Univariable comparisons of baseline and intraoperative characteristics.
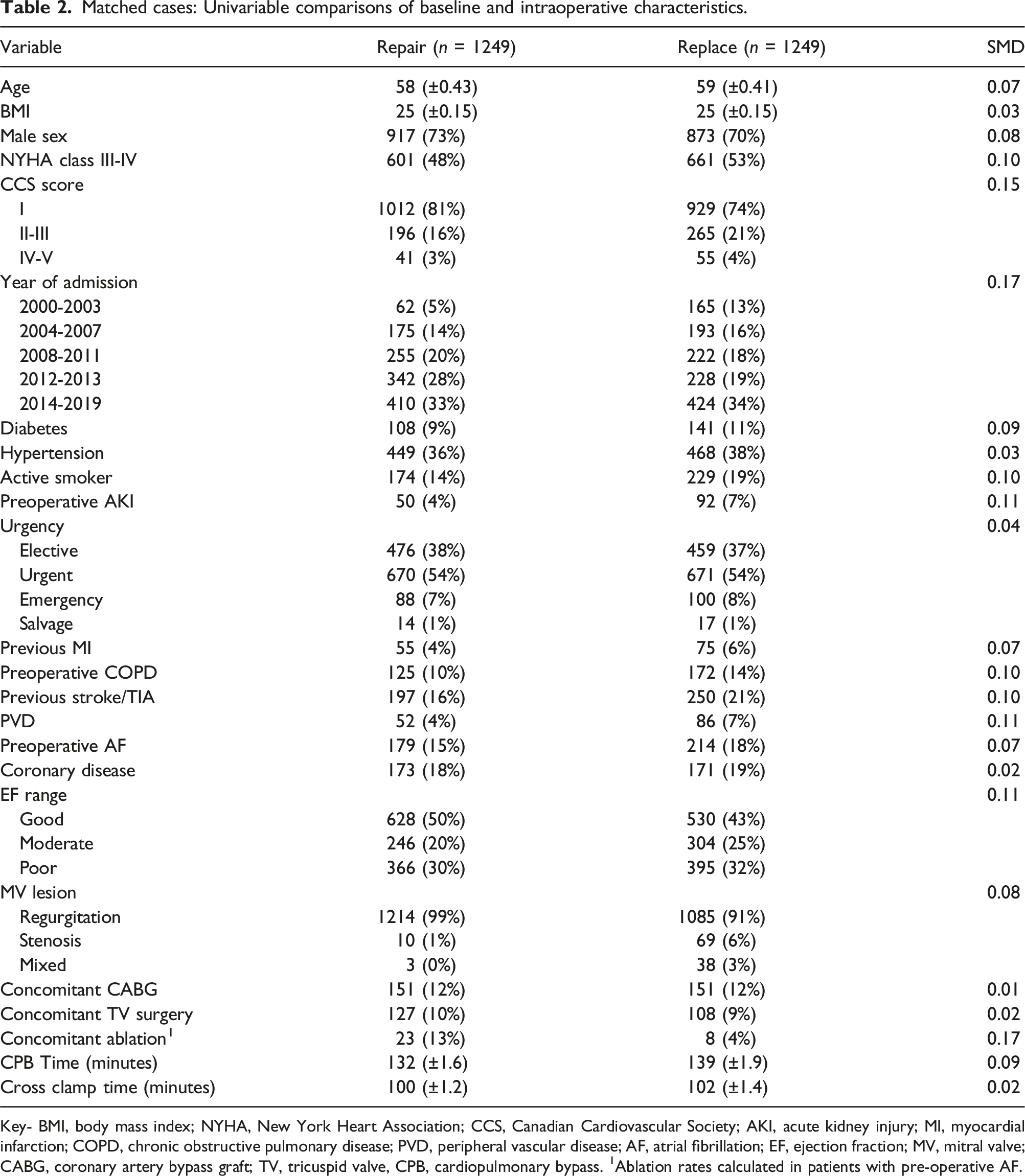
Key- BMI, body mass index; NYHA, New York Heart Association; CCS, Canadian Cardiovascular Society; AKI, acute kidney injury; MI, myocardial infarction; COPD, chronic obstructive pulmonary disease; PVD, peripheral vascular disease; AF, atrial fibrillation; EF, ejection fraction; MV, mitral valve; CABG, coronary artery bypass graft; TV, tricuspid valve, CPB, cardiopulmonary bypass. 1Ablation rates calculated in patients with pre-operative AF.
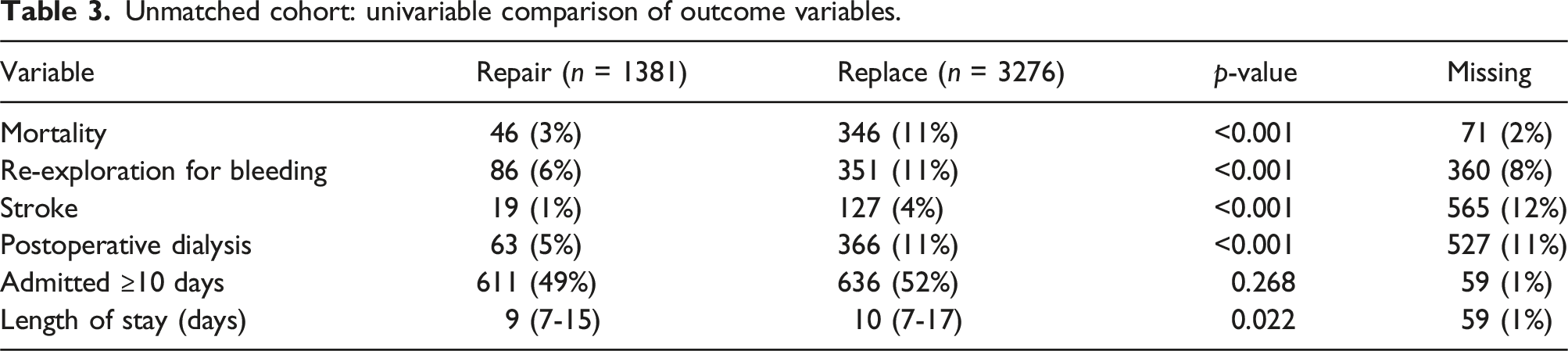
Unmatched cohort: univariable comparison of outcome variables.
Univariable outcomes in matched cohort.
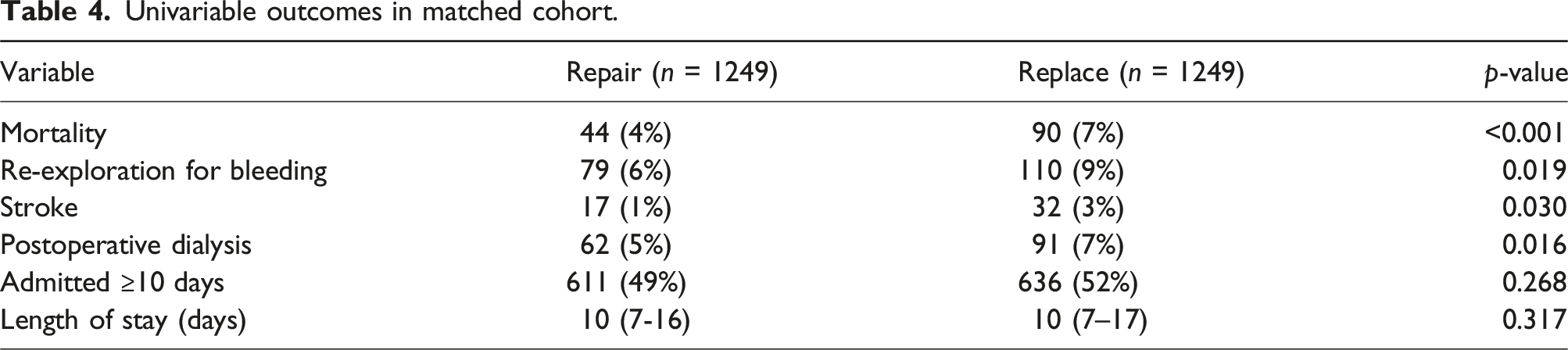
To minimize selection bias, a propensity-score matching analysis was planned. Propensity-score matching was performed using the ‘MatchIt’ package in R Core Team (2021). R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Version 4.0.5. All descriptive and regression analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, NY, USA).
Propensity scores were calculated using a logistic regression model that included the following covariates: sex, New York Heart Association (NYHA) class, Canadian Cardiovascular Society (CCS) score, diabetes, hypertension, smoking status, preoperative acute kidney injury (AKI), urgency, previous myocardial infarction (MI), previous chronic obstructive pulmonary disease (COPD), previous stroke or transient ischaemic attack (TIA), peripheral vascular disease (PVD), preoperative AF, coronary artery disease, ejection fraction (EF) range, MV lesion, body mass index (BMI) and age. Patients in the MVr group were matched to patients in the MVR group using a nearest-neighbour matching algorithm in a 1:1 ratio, based on the logit of the propensity score using calipers of width equal to 0.2 of the standard deviation of the logit of the propensity score.
Multivariable outcomes in matched cohort.
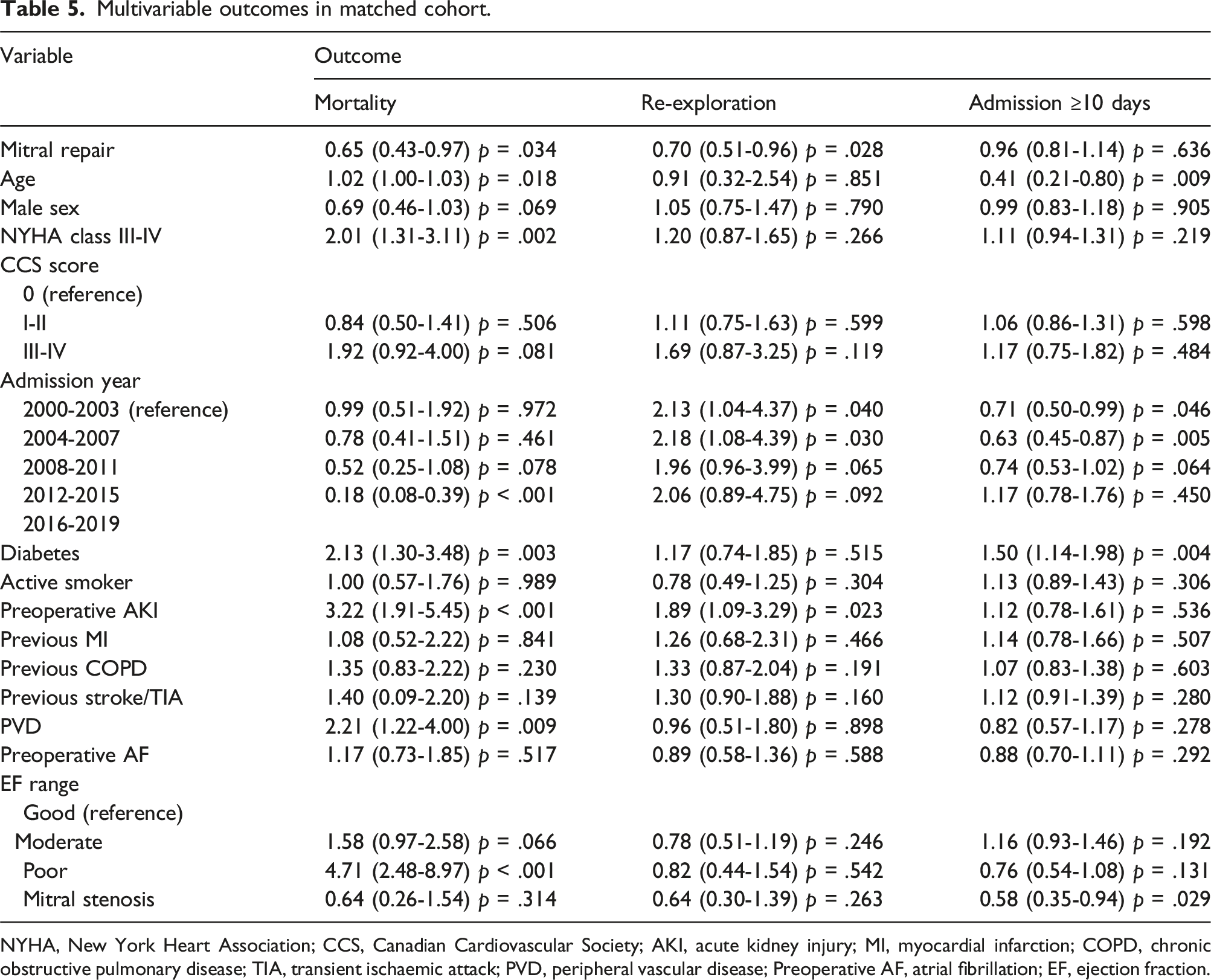
NYHA, New York Heart Association; CCS, Canadian Cardiovascular Society; AKI, acute kidney injury; MI, myocardial infarction; COPD, chronic obstructive pulmonary disease; TIA, transient ischaemic attack; PVD, peripheral vascular disease; Preoperative AF, atrial fibrillation; EF, ejection fraction.
Multivariable associations with mortality in MVr and MVR (matched cohorts).
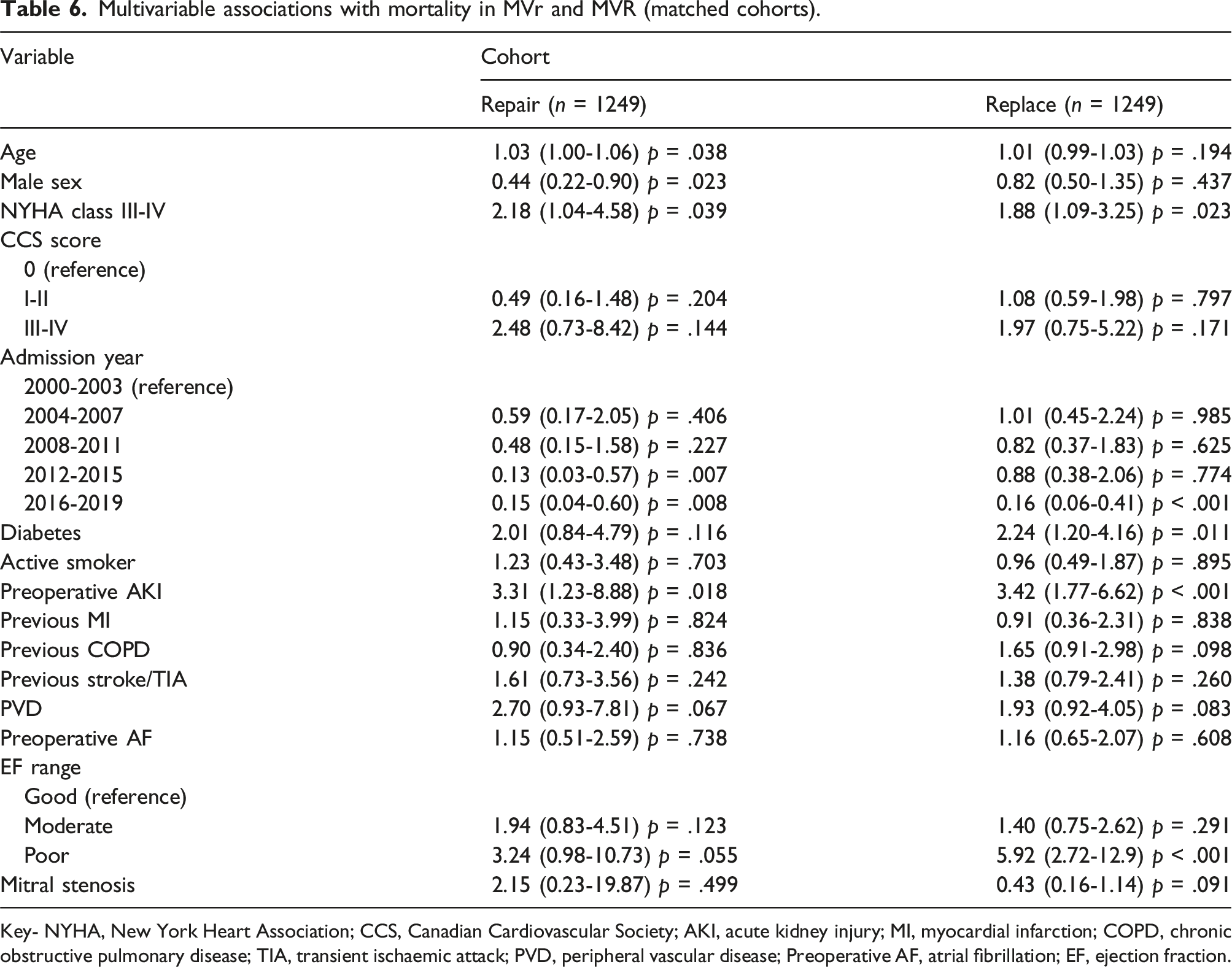
Key- NYHA, New York Heart Association; CCS, Canadian Cardiovascular Society; AKI, acute kidney injury; MI, myocardial infarction; COPD, chronic obstructive pulmonary disease; TIA, transient ischaemic attack; PVD, peripheral vascular disease; Preoperative AF, atrial fibrillation; EF, ejection fraction.
Results
Unmatched cohort
In terms of baseline characteristics and intraoperative variables, we observed significant differences between the unmatched MVr (n = 1381) and MVR (n = 3276) groups. Patients undergoing MVr were more likely to be male (74% vs 71%, p = .011). They were less likely to present with NYHA class III-IV symptoms (44% vs 62%, p < .001) and had lower Canadian Cardiovascular Society (CCS) scores (p = .025). MVr patients also had lower rates of preoperative AKI (4% vs 11%, p < .001), previous stroke/TIA (15% vs 21%, p < .001) and preoperative AF (14% vs 18%, p = .004). They were more likely to undergo concomitant AF ablation (13% vs 3%, p < .001), had shorter cardiopulmonary bypass (CPB) times (132 minutes vs 145 minutes, p < .001) and shorter aortic cross-clamp (AoX) times (100 minutes vs 107 minutes, p < .001) (see Table 1).
There were statistically significant differences in early post-operative outcomes in the unmatched cohorts. Patients undergoing MVr had lower rates of mortality (3% vs 11%, p < .001), re-exploration for bleeding (6% vs 11%, p < .001), stroke (1% vs 4%, p < .001) and post-operative dialysis (5% vs 11%, p < .001). Patients undergoing MVr also had shorter overall hospital stays (9 days vs 10 days, p < .001), however there were no significant differences in rates of patients staying in hospital beyond 10 days (49% vs 52%, p = .022).
Matched cohort
Propensity-score matching yielded 1249 pairs of matched MVr and MVR cases (see Table 2). There were no significant differences between the two groups for most variables, as standardized mean differences (SMD) were less than 0.10 for all but five variables (see Table 2). The variables that differed between the two groups despite matching were CCS scores (SMD = 0.15), year of admission (SMD = 0.17), preoperative AKI (SMD = 0.11), PVD (SMD = 0.11) and EF range (SMD = 0.11).
After propensity-score matching, patients undergoing MVr had significantly lower rates of mortality (4% vs 7%, p < .001), re-exploration for bleeding (6% vs 9%, p = .019), stroke (1% vs 3%, p = .030), and postoperative dialysis (5% vs 7%, p = .016) (see Table 4). There were no significant differences in the proportion of patients admitted for greater than 10 or the length of stay between the two groups.
Multivariable regression analysis
Binary logistic regression in the matched cohort revealed MVr to be independently associated with a lower risk of mortality (OR:0.65, 95%CI:0.43-0.97, p = .034) or re-exploration for bleeding (OR:0.70, 95%CI:0.51-0.96, p = .028). Poor EF had the strongest independent association with mortality (OR:4.71, 95%CI:2.48-8.97, p < .001). Other independent predictors of mortality included preoperative AKI (OR:3.22, 95%CI:1.91-5.45, p < .001), NYHA class III-IV (OR:2.01, 95%CI:1.31-3.11, p = .002), diabetes (OR:2.13, 95%CI:1.30-3.48, p = .003), PVD (OR: 2.21, 95%CI:1.22-4.00, p = .009) and advancing age (OR:1.02, 95%CI:1.00-1.03, p = .018) (see Table 5). In terms of re-exploration for bleeding, preoperative AKI was an independent predictor (OR:1.89, 95%CI:1.09-3.29, p = .023). Diabetes was the only independent predictor for hospital stay greater than 10 days (OR:1.50, 95%CI:1.14-1.98, p = .004). Advancing age (OR:0.41, 95%CI:0.21-0.80, p = .009) and mitral stenosis (OR:0.58, 95%CI:0.35-0.94, p = .029) were found to be protective factors.
Separate modelling demonstrated common and different predictors of early mortality in MVr and MVR. In patients undergoing repair, advancing age (OR:1.03, 95%CI:1.00-1.06, p = .038), NYHA class III-IV (OR:2.18, 95%CI:1.04-4.58, p = .039) and preoperative AKI (OR:3.31, 95%CI:1.23-8.88, p = .018) were independent predictors for mortality, whilst male sex was a protective factor (OR:0.44, 95%CI:0.22-0.90, p = .023). In patients undergoing replacement, NYHA class III-IV (OR:1.88, 95%CI:1.09-3.25, p = .023), diabetes (OR:2.24, 95%CI:1.20-4.16, p = .011), preoperative AKI (OR:3.42, 95%CI:1.77-6.62, p < .001) and poor LVEF (OR:5.92, 95%CI:2.71-12.9, p < .001) were independent predictors of mortality. There was no relationship between sex and mortality in MVR.
Discussion
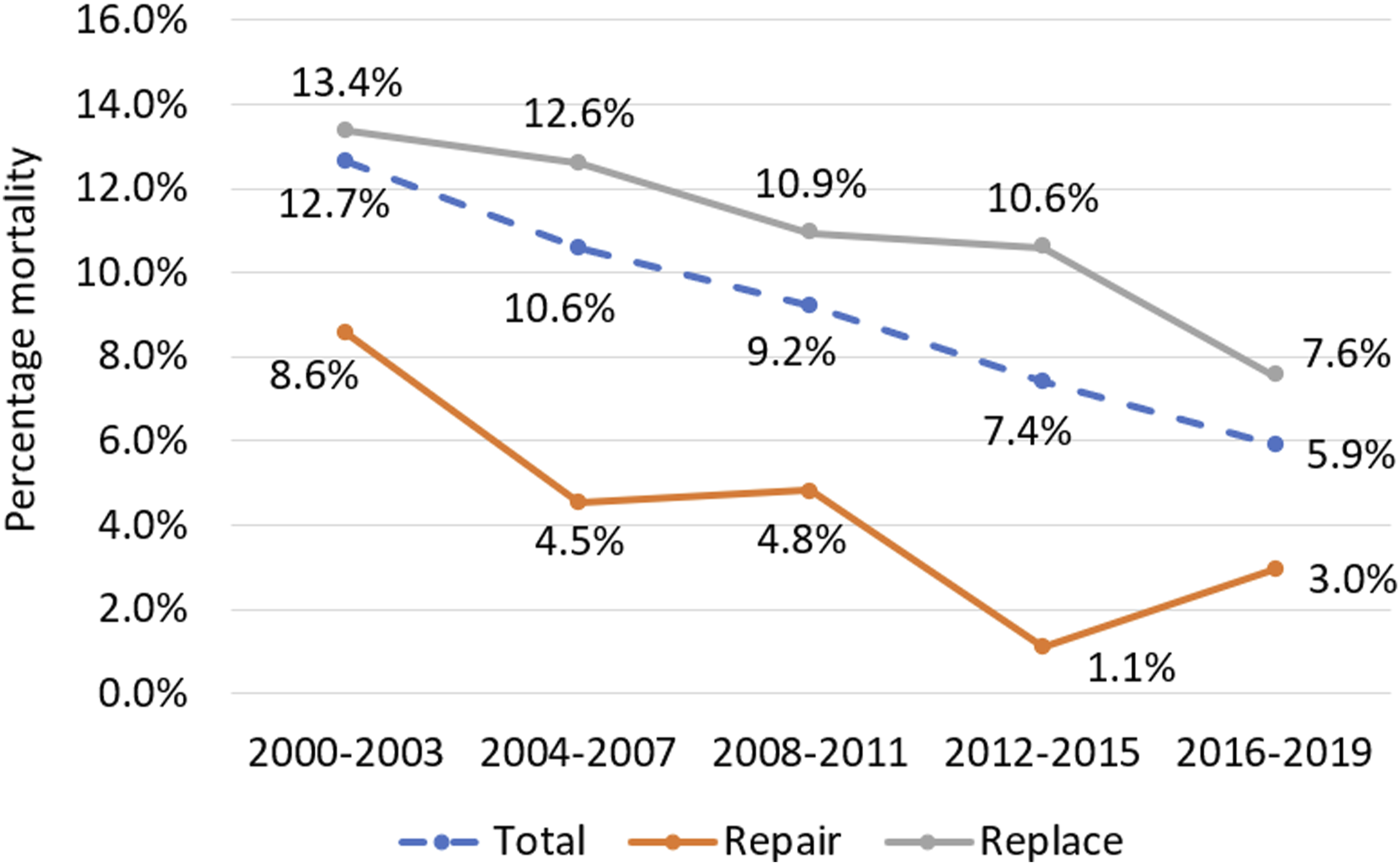
There are significant differences between patients with MV endocarditis receiving MVr and MVR. Our analysis reveals patients receiving MVr to be significantly less comorbid than those undergoing MVR. They had shorter CPB and AoX times, shorter hospital stays and superior postoperative outcomes. After propensity-score matching, these significant outcome differences persist, with MVr resulting in reduced mortality, re-exploration for bleeding, stroke and postoperative dialysis compared to MVR. Regression analysis in the matched cohort was designed to minimise the impact of confounding variables. Despite this, MVr emerged as independently associated with a lower risk of both mortality and re-exploration for bleeding. Whilst this is encouraging data supporting the repair of mitral valve endocarditis where possible, this may also relate to unmeasured risk factors associated with patients receiving a replacement. Trends in MV surgery for endocarditis shifted significantly in the UK between 2000 and 2019. Repair rates have risen consistently from 15% in 2000-2003 to 36% in 2016-2019 (see Figure 1). Furthermore, early postoperative mortality rates have dropped significantly, from 8.6% to 3.0% post-repair and 13.4% to 7.6% post-replacement (see Figures 2 and 3). Trends in repair rates for mitral valve endocarditis in overall cohort. Trends in mortality by procedure: total, mitral repair and mitral replacement. Forest plot demonstrating the independent relationship between mitral repair and outcomes in matched population. Odds ratios ±95% confidence intervals.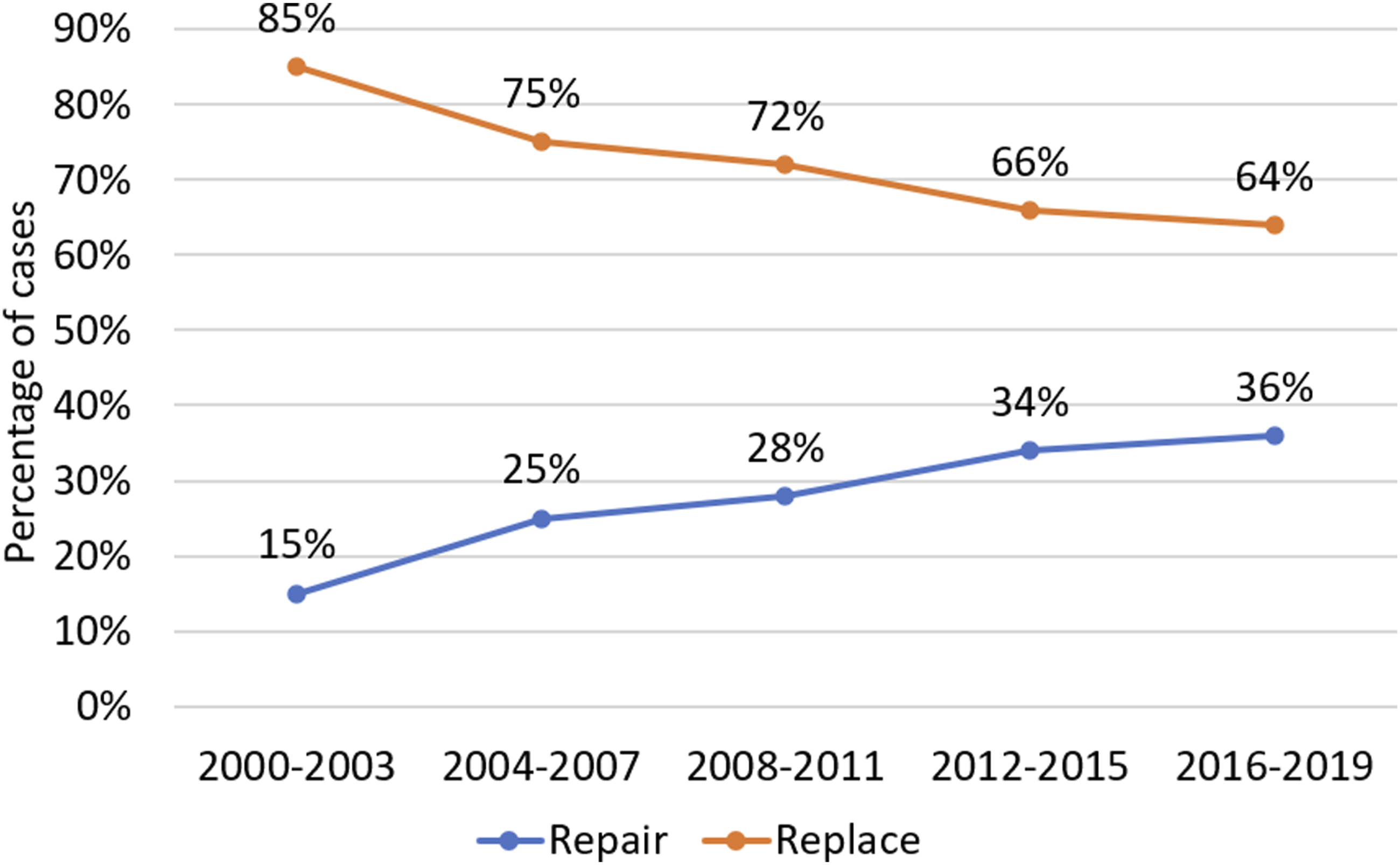
Our findings show that UK practice is steadily shifting and embracing evidence in the literature which we have corroborated with our results, demonstrating the superiority of MVr over MVR where appropriate. In terms of early outcomes, our results support a repair-orientated approach for MV endocarditis. This confirms the findings of a range of smaller studies which yielded similar results,3,5–8 A pertinent follow-on question is, in which patients should we seek to perform repair? Clearly, the extent of infection is one of the most important considerations. We were unable to elucidate further on this given the nature of national registry data, however Omoto et al suggested based on findings in their paper that the extent of the infected lesion is the key variable when deciding whether to repair or replace. 8 Liang et al demonstrated the importance of early intervention, showing that early surgery was associated with better short and long-term postoperative outcomes. 9 In a large systematic review and meta-analysis, Narayanan et al. looked at over 21 studies and also concluded that early intervention is particularly crucial in reducing postoperative mortality in patients with endocarditis. 10 Furthermore, El Khoury’s group have repeatedly demonstrated that it is possible to achieve repair rates of 80% for endocarditis when an aggressive repair-orientated, early intervention approach is taken.3,11–13 Both patch and non-patch repair techniques were demonstrated to result in safe, durable results compared to MVR.
Interestingly, when comparing our separate multivariable mortality models for mortality in the MVr and MVR cohorts (see Table 6), age emerged as an independent predictor of mortality in patients undergoing MVr but not in those undergoing MVR. The odds ratio suggests a 3% increased mortality risk associated with each additional year in age, which translates to a 30% increased risk of mortality per decade. This is a clinically significant difference and suggests that we should take a cautious approach to repairing MV in the context of elderly patients with endocarditis. This may be caused by poorer native tissue quality being less amenable to support a repair following the damage caused by endocarditis. Other important predictors of poor outcomes include NYHA class III-IV, preoperative AKI, preoperative diabetes and poor LVEF. Patients fulfilling these criteria should be optimized as much as possible prior to intervention. Recent surgery (2016-2019) emerged as an independent protective factor against mortality, suggesting improving surgical practices per se have reduced mortality rates over time. This is encouraging and, in line with univariable trends, confirms that the UK is heading in the right direction in terms of reducing mortality following surgery for MV endocarditis.
The uniquely large UK sample size based on nationally collected data provides strength to our study. Multivariable analysis allowed us to account for the impact of covariates on observed outcomes. It is important to acknowledge the limitations of our study design, which must be taken into account when drawing conclusions from our findings. Whilst the NICOR database undergoes regular maintenance and validation, given its size we cannot rule out the possibility of some data being recorded incorrectly. There are also areas in our analysis where a lack of granularity in the data limited the conclusions we were able to make; data on the different types of repairs performed, as well as information on why surgeons chose not to repair in MVR cases, would have allowed us to comment further on the reasons behind the observed outcome differences. Crucially, lack of long-term follow up data limited out study to looking at early post-operative outcomes. Given that we made no attempt to systematically replace all missing data, our multivariable analyses were conducted as complete-case analyses. The size of the dataset and the relatively low levels of missingness in the data limit the impact of these factors. Whilst recognising these limitations, our study confirms that there is indeed a relationship between MV procedure and post-operative outcomes for patients with MV endocarditis.
Conclusions
(1)- Early postoperative outcomes are superior post MVr for endocarditis. (2)- Age is a poor prognostic factor for repairing MV endocarditis. (3)- Overall mitral repair rate for endocarditis in the UK has increased over time. (4)- Early surgery and repair-orientated strategy combined yield high repair rates and good postoperative outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Access to The National Adult Cardiac Surgery Audit (NACSA) data was obtained from NICOR through funding from the Bristol Heart Institute department of cardiothoracic surgery. This study was also funded by a grant contribution from the charity Heart Valve Voice.
Central message
Mitral valve repair is a viable option in endocarditis with superior early outcomes, however repair rates remain low. Repair should be prioritized when technically possible in experienced centres.
Perspective statement
The superiority of mitral valve repair over replacement has been established in patients with degenerative disease; repair rates have thus risen significantly in that cohort. In terms of endocarditis we are yet to reach a consensus; rates of repair remain low. Our results demonstrate mitral valve repair to be a safe approach, with superior early outcomes in patients with endocarditis.
