Abstract
Introduction
This study aimed to characterize the severity of bleeding and its association with short-term neurologic outcomes in pediatric ECMO.
Methods
Multicenter retrospective cohort study of pediatric ECMO patients at 10 centers utilizing the Pediatric ECMO Outcomes Registry (PEDECOR) database from December 2013-February 2019. Subjects excluded were post-cardiac surgery patients and those with neonatal pathologies. A novel ECMO bleeding scale was utilized to categorize daily bleeding events. Poor short-term neurologic outcome was defined as an unfavorable Pediatric Cerebral Performance Category (PCPC) or Pediatric Overall Performance Category (POPC) (score of >3) at hospital discharge.
Results
This study included 283 pediatric ECMO patients with a median (interquartile range [IQR]) age of 1.3 years [0.1, 9.0], ECMO duration of 5 days [3.0, 9.5], and 44.1% mortality. Unfavorable PCPC and POPC were observed in 48.4% and 51.3% of patients at discharge, respectively. Multivariable logistic regression analysis included patient’s age, cannulation type, duration of ECMO, need for cardiopulmonary resuscitation, acute kidney injury, new infection, and vasoactive-inotropic score. As the severity of bleeding increased, there was a corresponding increase in the likelihood of poor neurologic recovery, shown by increasing odds of unfavorable neurologic outcome (PCPC), with an adjusted odds ratio (aOR) of 0.77 (confidence interval [CI] 0.36–1.62), 1.87 (0.54–6.45), 2.97 (1.32–6.69), and 5.56 (0.59–52.25) for increasing bleeding severity (grade 1 to 4 events, respectively). Similarly, unfavorable POPC aOR (CI) was 1.02 (0.48–2.17), 2.05 (0.63–6.70), 5.29 (2.12–13.23), and 5.11 (0.66–39.64) for bleeding grade 1 to 4 events.
Conclusion
Short-term neurologic outcomes in pediatric ECMO are proportional to the severity of bleeding events. Strategies to mitigate bleeding events could improve neurologic recovery in pediatric ECMO.
Take-Home Message
Increasing severity of bleeding events is associated with adverse short-term neurologic outcome at discharge in children requiring ECMO.
Introduction
Although pediatric extracorporeal membrane oxygenation (ECMO) use is increasing, neurologic outcomes remain suboptimal, with 10%–50% of children who have undergone ECMO support experiencing some degree of neurologic disability.1–6 Neurologic complications during ECMO, such as seizures, hypoxemic ischemic encephalopathy, ischemic stroke, and intracranial hemorrhages are associated with poor neurologic outcomes.1,2,7–10 Potential risk factors for neurologic complications during ECMO include metabolic acidosis, inotrope or vasopressor requirement, utilization of a ventricular assist device (VAD) prior to ECMO initiation, ECMO cannulation during cardiopulmonary resuscitation (ECPR), a rapid decrease in PaCO2, and a rapid increase in blood pressure.1,2,8,11
Bleeding is also a common complication in pediatric patients supported on ECMO, affecting 38%–70% of patients.4–7 Although severe bleeding events have been associated with adverse neurologic outcomes and increased mortality, it’s unclear whether mild-to-moderate bleeding events are also associated with poor neurologic outcomes.4,5,8 Despite being two of the most common complications encountered in pediatric patients supported on ECMO, the relationship between the severity of bleeding events and neurologic outcomes is not well-defined.
Estimating the severity of bleeding events reported during the ECMO course and their association with neurologic outcomes has been challenging due to variability in defining these events. Studies have used various definitions of clinical bleeding events on ECMO, including bleeding at the cannula or surgical site, receiving a blood transfusion, or experiencing intracranial hemorrhage.12,13 It is essential to have a standard, universally accepted definition of bleeding events to compare the severity of bleeding, the associated clinical outcomes, and the effectiveness of interventions to avoid bleeding complications in pediatric ECMO patients. A new bleeding scale specifically designed for pediatric ECMO patients was utilized in this retrospective study conducted across 10 centers to evaluate daily bleeding severity in pediatric ECMO patients. Our study aimed to evaluate the association between the severity of bleeding complications and short-term neurologic outcomes in these patients.
Materials and methods
Cohort
In this multicenter retrospective cohort, patients less than 19 years of age supported on ECMO in 10 pediatric intensive care units from December 2013 to February 2019 were included. Patients with cardiac surgery during their admission and neonatal pathologies (persistent pulmonary hypertension of the neonate, congenital diaphragmatic hernia, and meconium aspiration syndrome) were excluded to minimize heterogeneity of the cohort, and the possibility of post-surgical bleeding and exposure to cardiopulmonary bypass as confounders. Subjects were also excluded if they never had a bleeding scale recorded in the database. Each clinical site has either IRB approval to participate in the Pediatric ECMO Outcomes Registry Database (PEDECOR) or a waiver for non-human subject research.
Data collection
Data were extracted from the PEDECOR Database.14–16 PEDECOR is a web-based data platform housed at Baylor College of Medicine (BCM), Houston, Texas. Data are stored in a secure, HIPAA-compliant database maintained by BCM (IRB number H-37253). Detailed pre-cannulation and hospital course variables are collected, in addition to daily laboratory and blood product transfusion data.
Texas children’s hospital (TCH) extracorporeal membrane oxygenation (ECMO) bleeding scale.

ACT: activated clotting time; RBC: red blood cells; IV: intravenous; CNS: central nervous system.
Daily TCH ECMO Bleeding Scale values were assigned retrospectively by research coordinators, ECMO specialists, or ECMO physicians and recorded in PEDECOR. Short-term neurologic outcome was evaluated using Pediatric Cerebral Performance Category (PCPC) and Pediatric Overall Performance Category (POPC), which were routinely collected in the PEDECOR database.21,22 We defined unfavorable PCPC and POPC as a score greater than three at hospital discharge based on prior literature.21–24 For patients whose baseline PCPC and POPC scores were four or greater, a favorable outcome was defined as having no change from baseline scores. Mortality was defined as in-hospital mortality.
Statistical analysis
Demographic and clinical characteristics were summarized by median with interquartile range (IQR) or frequency with percentage. Receiver Operating Characteristic (ROC) curves were created for the maximum, mean, median, and mode of the TCH ECMO Bleeding Scale values during the ECMO run for the cohort to determine threshold values using sensitivity, specificity, and area under the curve (AUC) to discriminate adverse clinical outcomes. Based on the ROC analysis, we further stratified the cohort into five groups by maximum bleeding severity grade on the TCH ECMO Bleeding Scale and compared these groups using analysis of variance (ANOVA), Kruskal-Wallis’s test, Fisher’s exact test, or Chi-square test.
Both univariable and multivariable logistic regression analyses were performed to assess the unadjusted and adjusted odds for unfavorable PCPC and unfavorable POPC at hospital discharge adjusting for maximum severity of bleeding events and other risk factors while additionally accounting for correlation within study sites using generalized estimating equations. A 0.3 mg/dL increase in creatinine was used in the analyses in alignment with Kidney Disease Improving Global Outcomes (KDIGO) Stage 1 Acute Kidney Injury (AKI) criteria. 25 Multivariable regression selected risk factors with a p-value <.05 and less than 15% missing data on univariable regression, in addition to severity of bleeding, age, new infection, and duration of ECMO. Similar logistic regression analyses were performed to assess the unadjusted and adjusted odds for in-hospital mortality. All analyses were performed using Stata v 15.1 (StataCorp, College Station, TX, USA).
Results
Cohort’s clinical characteristics and subgroup comparison by maximum bleeding severity.
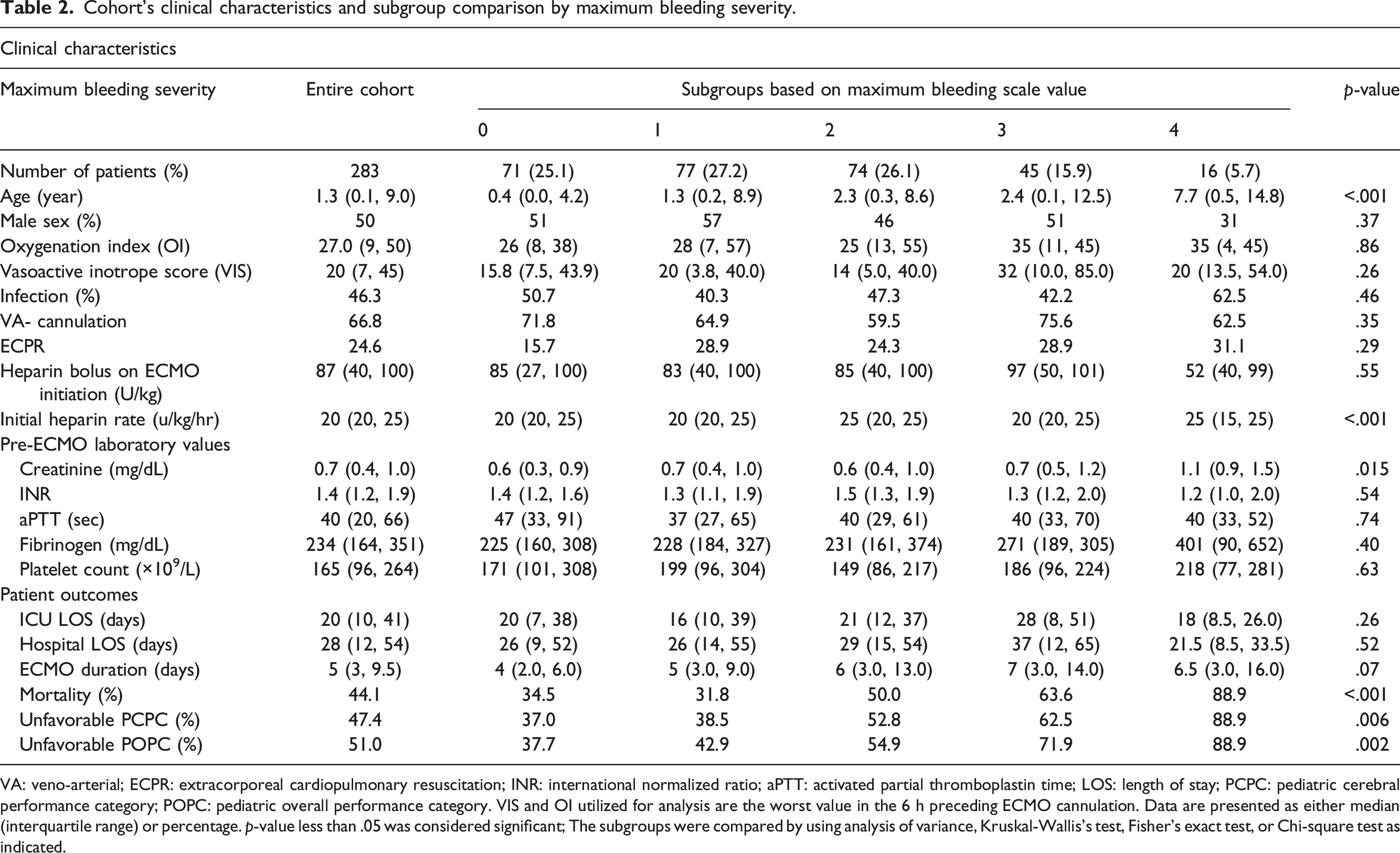
VA: veno-arterial; ECPR: extracorporeal cardiopulmonary resuscitation; INR: international normalized ratio; aPTT: activated partial thromboplastin time; LOS: length of stay; PCPC: pediatric cerebral performance category; POPC: pediatric overall performance category. VIS and OI utilized for analysis are the worst value in the 6 h preceding ECMO cannulation. Data are presented as either median (interquartile range) or percentage. p-value less than .05 was considered significant; The subgroups were compared by using analysis of variance, Kruskal-Wallis’s test, Fisher’s exact test, or Chi-square test as indicated.
Seventy-five percent of patients experienced at least one bleeding event during their ECMO course with the following distribution of maximum bleeding severity: 27.2% mild (grade 1), 26.1% moderate (grade 2), 15.9% severe (grade 3), and 5.7% catastrophic (grade 4) (Supplemental Figure 1). The daily proportion of bleeding events based on severity remained similar over the first 28 ECMO days, with the incidence of severe and catastrophic bleeding events ranging from 0 to 9% during the first 28 days of ECMO (Supplemental Figure 2). Most severe or catastrophic bleeding events occurred early in the ECMO course, with 50% of severe or catastrophic events occurring by ECMO day 2 and 75% by day 6. Of all severe and catastrophic bleeding events, nearly half (42.6%) of the events were preceded by either mild (16.4%) or moderate (26.2%) clinical bleeding events the day prior (Figure 1). Of all severe and catastrophic bleeding events, nearly half (42.6%) of the events were preceded by either mild (16.4%) or moderate (26.2%) clinical bleeding events 24 h prior to the severe or catastrophic bleeding event.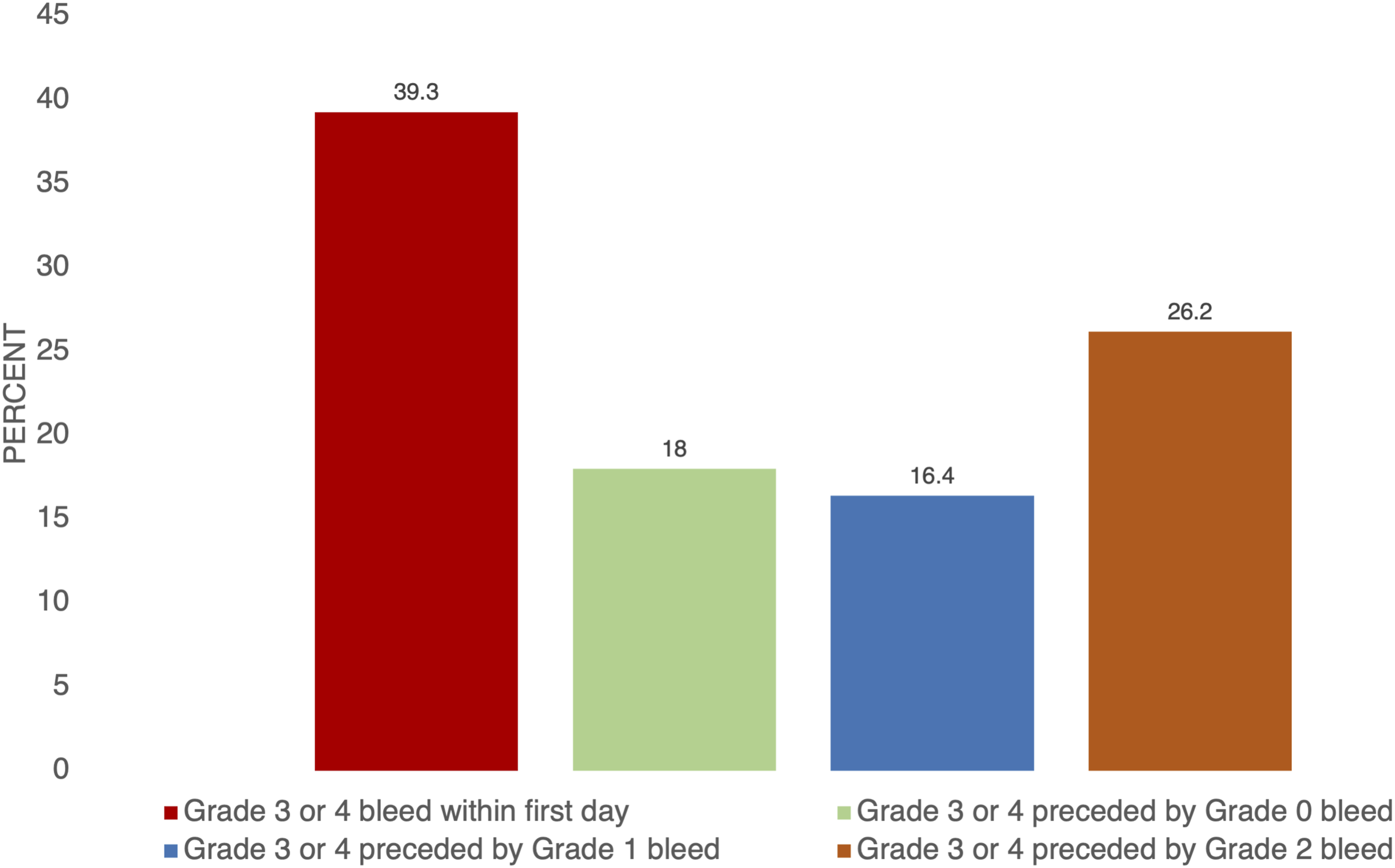
ROC were obtained to determine the threshold TCH ECMO Bleeding Scale value to best discriminate patients with adverse outcomes. We used the cohort’s maximum, mean, median, and mode bleeding scale values for the ROC analysis. Of all values, the maximum TCH ECMO Bleeding Scale value during the ECMO run was found to best discriminate patients with adverse outcomes (Supplemental Tables 2–4). Moderate (grade 2) maximum bleed severity on the TCH ECMO Bleeding Scale had an area under the curve (AUC) of 0.63 (95% confidence interval (CI) 0.56–0.70, 55% sensitivity, and 66% specificity) for unfavorable PCPC, AUC of 0.65 (95% CI 0.57–0.72, 56% sensitivity, and 68% specificity) for unfavorable POPC, and AUC of 0.61 (95% CI 0.55–0.68, 60% sensitivity, and 63% specificity) for in-hospital mortality (Supplemental Figure 3). The proportion of patients with unfavorable PCPC, unfavorable POPC, and in-hospital mortality increased with maximum bleeding severity during the ECMO course (Figure 2). The proportion of patients with unfavorable PCPC, unfavorable POPC, and in-hospital mortality increased with maximum bleeding severity during the ECMO course.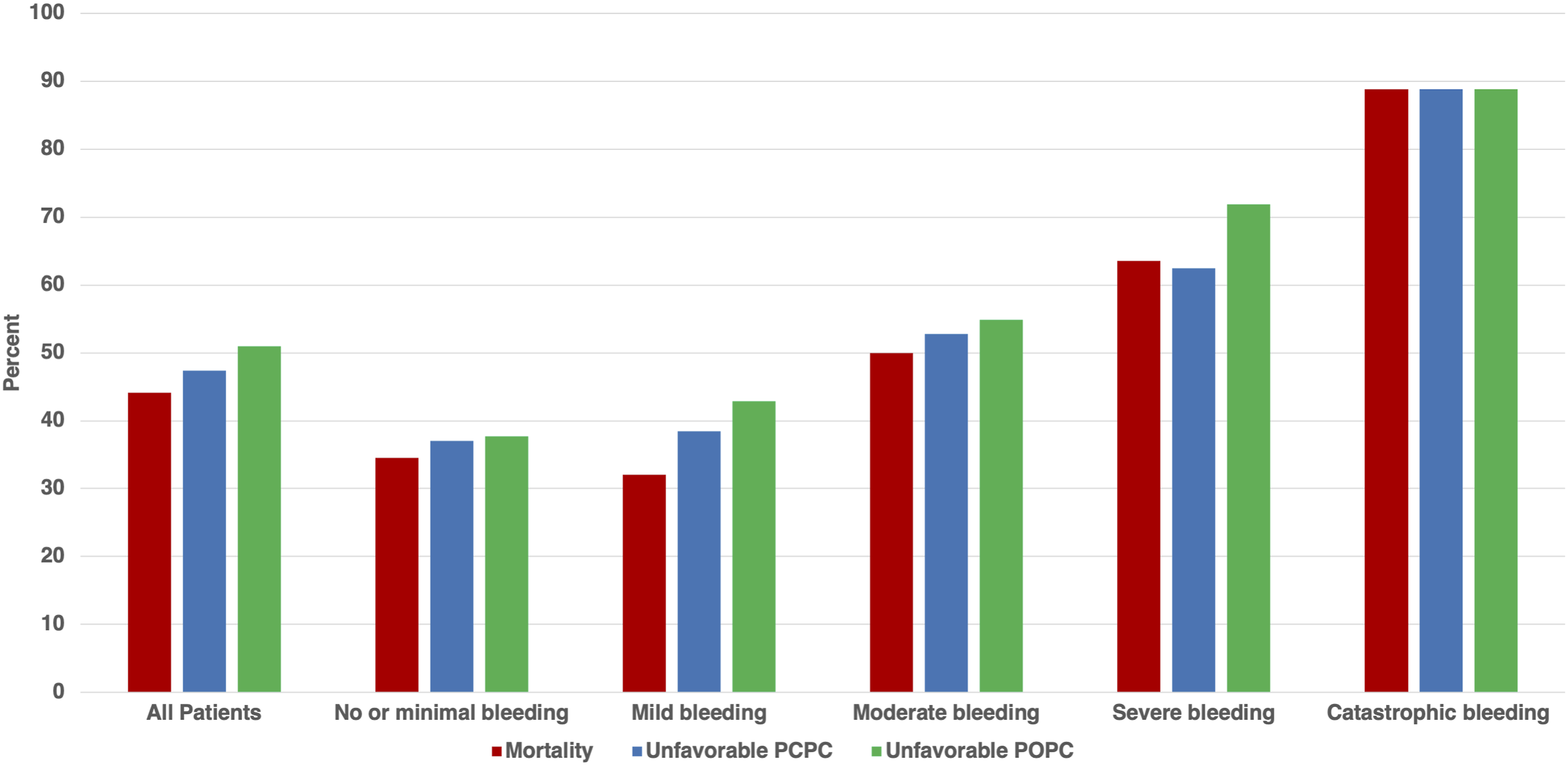
The univariable analyses for individual variables are available in Supplemental Tables 5–7. On multivariable logistic regression, the odds of unfavorable PCPC increased with bleeding severity (adjusted odds ratio (aOR) 0.77, 95% CI 0.36–1.62 for mild; aOR 1.87, 95% CI 0.54–6.45 for moderate; aOR 2.97, 95% CI 1.32–6.69 for severe; and aOR 5.56, CI 0.59–52.25 for catastrophic bleeding) and unfavorable POPC (aOR 1.02, 95% CI 0.48–2.17 for mild; aOR 2.05, 95% CI 0.63–6.70 for moderate; aOR 5.29, 95% CI 2.12–13.23 for severe; and aOR 5.11, 95% CI 0.66–39.64 for catastrophic bleeding) (Figure 3). Increasing severity of bleeding was also associated with mortality (aOR 0.43, 95% CI 0.26–0.71 for mild; aOR 1.47, 95% CI 0.56–3.84 for moderate; aOR 1.98, 95% CI 1.15–3.40 for severe; and aOR 5.64, 95% CI 1.72–18.56 for catastrophic bleeding severity). Any cardiopulmonary resuscitation (CPR) (out-of-hospital CPR, in-hospital CPR, or E-CPR) was also associated with unfavorable PCPC (aOR 2.30, 95% CI 1.07–4.94) and mortality (aOR 2.49, 95% CI 1.20–5.17) (Figure 3). The multivariable logistic regression analyses for in-hospital mortality, unfavorable PCPC, and unfavorable POPC are available in Supplemental Tables 8–10. In multivariable logistic regression, severity of bleeding events categorized by the TCH ECMO Bleeding Scale increased with the likelihood of in-hospital mortality and unfavorable PCPC and POPC at discharge in children requiring ECMO. PCPC: pediatric cerebral performance category; POPC: pediatric overall performance category; Yr: years of age; CPR: cardiopulmonary resuscitation; VIS: vasoactive inotrope score; OI: oxygenation index.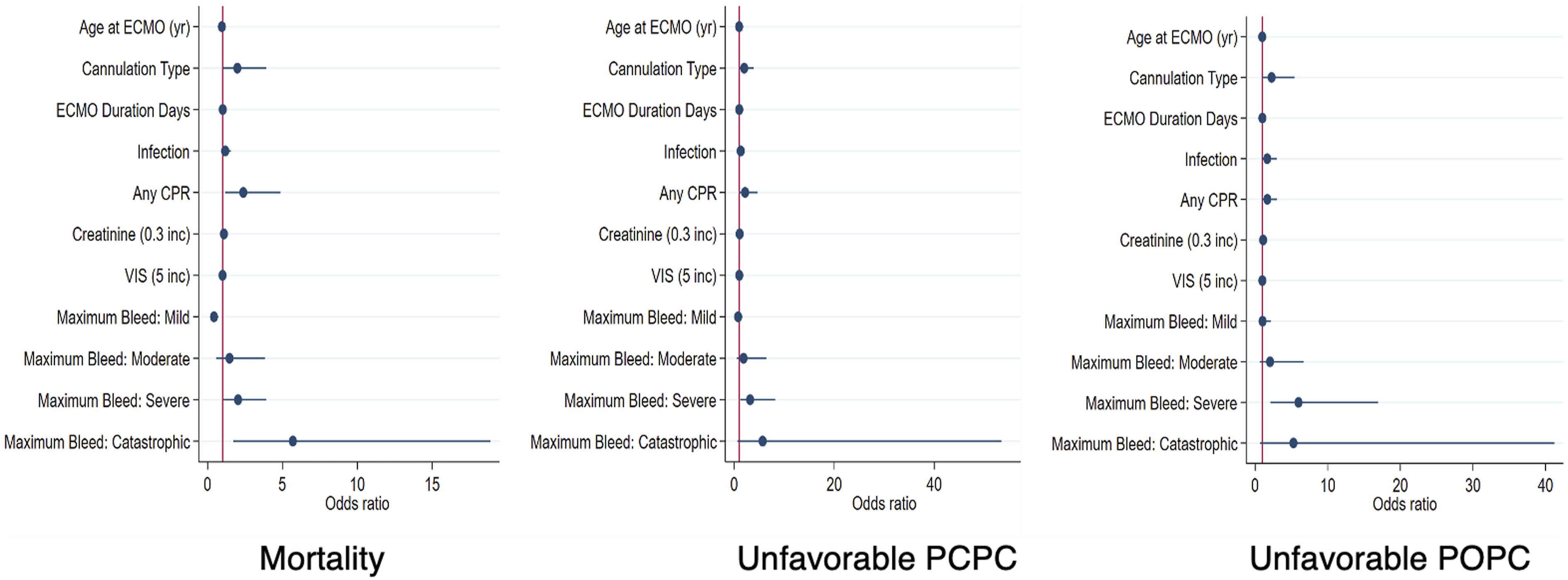
Discussion
Severe bleeding events have been associated with mortality and adverse neurologic outcomes, but clinical outcomes stratified by daily bleeding severity on ECMO have not been previously reported.4,17,18 In this study, we show that the maximum severity of bleeding during the ECMO course was the most discriminatory for clinical outcomes when compared to the mean, median, and mode values of the TCH Bleeding Scale. We also described the pattern of daily distribution of the severity of bleeding events. Notably, the daily distribution of bleeding events based on severity remained similar over the first 28 ECMO days, with severe and catastrophic bleeding events ranging from 0 to 9% during the first 28 days of ECMO. Most severe or catastrophic bleeding events occurred early, with 50% of events occurring within 48 h after ECMO initiation. Additionally, nearly half (42.6%) of severe and catastrophic bleeding events were preceded by either mild (16.4%) or moderate (26.2%) clinical bleeding events. Lastly, we show that short-term neurologic outcomes and in-hospital mortality in pediatric patients supported on ECMO are proportional to the severity of bleeding events, utilizing the TCH ECMO Bleeding Scale.
There is insufficient data available on the severity of bleeding and its impact on clinical outcomes in critically ill children. In a study of critically ill children conducted at a single center, Sequeira et al. found that bleeding severity was independently associated with an increased risk of mortality (aOR for each bleeding category was 2.4, 95% CI 1.5–3.7), fewer days spent free of pediatric ICU (PICU), and fewer days free of ventilator support. 26 In their study cohort, only one patient required ECMO support and experienced no bleeding complications. Dalton and colleagues reported that bleeding events in pediatric ECMO patients were associated with an increased risk of mortality (Hazard ratio of 1.75, 95% CI 1.2–2.55). 27 However, there was no bleeding severity stratification and the need for red blood cell transfusion, regardless of clinical bleeding, was considered a bleeding complication. In their study, laboratory blood sampling contributed to significant blood loss, and at least one transfusion was given solely due to blood sampling in 42.2% of their subjects.
In our study, 47.4% of patients had unfavorable PCPC, and 51% had unfavorable POPC at discharge, consistent with the previously reported cumulative outcome of 48% (321/643) in a systematic review. 5 The proportion of patients with unfavorable neurologic outcomes was lower in those with less severe bleeding events (37% in patients with minimal bleeding and 38% with mild bleeding) compared to those with severe (66%) and catastrophic (88%) events. On multivariable logistic regression, any bleeding event was associated with unfavorable short-term neurological outcomes with a proportional increase in aOR from minimal to catastrophic bleeding (Figure 3). This finding is significant because although mild and moderate bleeding events had lower odds of poor neurologic outcomes, about 40% of severe and catastrophic bleeding were preceded by these milder events. Possible mechanisms behind the association between increased bleeding severity and poor neurologic outcome include hemodynamic instability and decreased hemoglobin levels, leading to decreased oxygen delivery. 28 Additionally, patients experiencing cannula site bleeding, intracranial hemorrhage, or pulmonary hemorrhage may require deeper levels of sedation for a longer duration, potentially necessitating higher doses of sedatives and neuromuscular blockade to minimize patient movement. 29 Lastly, exposure to red blood cell and other hemostatic transfusions may have indirect detrimental effects. 30 In future studies, it is important to assess these factors to understand the link between bleeding severity and neurocognitive outcomes.
Likely due to the lack of standardized bleeding assessment tools, there is much variability in reporting of bleeding events in pediatric ECMO patients.31–33 A recent systematic review found that only two pediatric studies reported bleeding assessment tools for critically ill neonates and children.31,34,35 They found no report of using bleeding assessment tools in pediatric ECMO patients. An ideal bleeding assessment scale for pediatric ECMO patients should be able to grade severity of bleeding, guide therapeutic interventions, assess response to these therapeutic interventions, and allow for comparison of bleeding events across institutions. The most challenging aspect of developing such bleeding assessment tool is associating the severity of bleeding with measurable clinical consequence. Recently, a multicenter- and multispecialty- expert panel created the Bleeding Assessment Scale in critically Ill Children (BASIC) using the Delphi consensus process. 36 They define bleeding as minimal, moderate, and severe, using organ dysfunction, proportional changes in vital signs, anemia, and quantifiable bleeding. This group has also recognized the challenge in defining the clinical relevance of bleeding as it can vary for each population subset. It is yet to be determined whether the inclusion of the Pediatric Logistic Organ Dysfunction Score-2 (PELODS-2) in the BASIC criteria will hinder bedside application in ECMO patients.
The TCH ECMO Bleeding Scale was developed specifically for patients requiring ECMO, incorporating common therapeutic interventions to mitigate the risk of bleeding, and designed to be easily performed at the bedside. Our multicenter retrospective cohort study demonstrated that a standardized clinical bleeding scale could feasibly be used to quantify the severity of bleeding events. Often patients have multiple bleeding events of varying severity during their ECMO course. It is unclear whether frequency, duration, or severity of bleeding events is most correlated with patient outcomes. Our study found that the maximum assigned bleeding scale value (with cutoff of grade 2) on the TCH ECMO Bleeding Scale showed moderate sensitivity and specificity and was superior in discriminating for mortality and adverse neurologic outcome compared to median, mode, and mean of the bleeding scale values during the ECMO course (Supplemental Figure 3). The moderate sensitivity and specificity of maximum bleeding severity for mortality and adverse neurologic outcome can be explained by acknowledging that there are many other clinical factors that contribute to morbidity and mortality on ECMO beyond bleeding complications. Most severe and catastrophic bleeding events occurred early in the ECMO course, but the daily proportion of severe and catastrophic bleeding events remained relatively constant (0%–9%) throughout the duration of ECMO (Supplemental Figure 2). These findings were consistent with the previously reported rate of bleeding events on ECMO.27,37 In this study, because the bleeding severity was stratified daily, we could further discern the timing of severe or catastrophic bleeding in relation to mild or moderate bleeding events. We found that nearly half (42.6%) of severe and catastrophic events were preceded by either mild (16.4%) or moderate (26.2%) clinical bleeding events (Figure 1). Although neither mild nor moderate bleeding events were independently associated with mortality or poor neurologic outcome, recognizing these events and preventing progression of bleeding severity could improve patient outcomes. Studies are needed to elucidate risk factors specifically associated with mild to moderate bleeding events, and whether early therapeutic interventions to control these bleeding events can prevent severe and catastrophic bleeding events.
Limitations
This study has several limitations. As with any retrospective design, this study is limited to associations rather than causation. The multivariable models adjusted for some confounding factors, but have excluded many factors, including red blood cell and hemostatic transfusions, coagulation abnormalities, ECMO flow rate, hemolysis, and circuit components. We also excluded patients after reparative cardiac surgery and neonatal pathologies to minimize the heterogeneity of the cohort. Post-surgical bleeding and exposure to cardiopulmonary bypass can influence the association between bleeding severity and clinical outcomes. Another major limitation of this study is that the TCH ECMO Bleeding Scale has not been validated for inter-user reliability and was recorded retrospectively. Therefore, variability in assessments and documentation at different centers could lead to variation in data collection. Additionally, missing data in this retrospective cohort could contribute to selection bias as about 10% of patients in the cohort had missing bleeding scale values for three or more ECMO days. Lastly, while PCPC and POPC have been utilized to describe neurofunctional disability in ECMO patients, these scores can only be utilized to describe short-term neurologic prognosis as they are assessed at discharge and cannot account for more subtle developmental deficits that may manifest later.38,39 A more comprehensive neurologic outcome assessment tool is needed in future studies.
Despite the limitations mentioned above, this study has shown that daily standardized grading of bleeding events is feasible, and there is an incremental association between the severity of bleeding and short-term neurologic outcomes.
Conclusions
Neurologic outcomes in pediatric ECMO patients are proportional to the severity of bleeding events graded by the novel TCH ECMO Bleeding Scale. Strategies to mitigate bleeding events could improve neurologic recovery in these patients. Furthermore, a standardized grading of bleeding events on ECMO is feasible to improve consistency and accuracy in reporting bleeding events on pediatric ECMO.
Supplemental Material
Supplemental Material - Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale
Supplemental Material for Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale by Katherine F Doane, Danielle Guffey, Laura L Loftis, Trung C Nguyen, Matthew A Musick, Amanda Ruth, Ryan D Coleman, Jun Teruya, Christine Allen, Melania M Bembea, Brian Boville, Jamie Furlong-Dillard, Santosh Kaipa, Mara Leimanis, Matthew P Malone, Lindsey K Rasmussen, Ahmed Said, Marie Steiner, Deanna T Tzanetos, Heather Viamonte, Linda Wallenkamp and Arun Saini in Perfusion
Supplemental Material
Supplemental Material - Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale
Supplemental Material for Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale by Katherine F Doane, Danielle Guffey, Laura L Loftis, Trung C Nguyen, Matthew A Musick, Amanda Ruth, Ryan D Coleman, Jun Teruya, Christine Allen, Melania M Bembea, Brian Boville, Jamie Furlong-Dillard, Santosh Kaipa, Mara Leimanis, Matthew P Malone, Lindsey K Rasmussen, Ahmed Said, Marie Steiner, Deanna T Tzanetos, Heather Viamonte, Linda Wallenkamp and Arun Saini in Perfusion
Supplemental Material
Supplemental Material - Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale
Supplemental Material for Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale by Katherine F Doane, Danielle Guffey, Laura L Loftis, Trung C Nguyen, Matthew A Musick, Amanda Ruth, Ryan D Coleman, Jun Teruya, Christine Allen, Melania M Bembea, Brian Boville, Jamie Furlong-Dillard, Santosh Kaipa, Mara Leimanis, Matthew P Malone, Lindsey K Rasmussen, Ahmed Said, Marie Steiner, Deanna T Tzanetos, Heather Viamonte, Linda Wallenkamp and Arun Saini in Perfusion
Supplemental Material
Supplemental Material - Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale
Supplemental Material for Short-term neurologic outcomes in pediatric extracorporeal membrane oxygenation are proportional to bleeding severity graded by a novel bleeding scale by Katherine F Doane, Danielle Guffey, Laura L Loftis, Trung C Nguyen, Matthew A Musick, Amanda Ruth, Ryan D Coleman, Jun Teruya, Christine Allen, Melania M Bembea, Brian Boville, Jamie Furlong-Dillard, Santosh Kaipa, Mara Leimanis, Matthew P Malone, Lindsey K Rasmussen, Ahmed Said, Marie Steiner, Deanna T Tzanetos, Heather Viamonte, Linda Wallenkamp and Arun Saini in Perfusion
Footnotes
Acknowledgements
The authors thank the contributing sites and the Research Informatics Core in the Research Resources Office (RRO) of the Department of Pediatrics at Baylor College of Medicine for the design, development, and maintenance of the Pediatric ECMO Outcomes Registry (PEDECOR).
Author contributions
LL, TN, JT, and MM developed the TCH ECMO Bleeding Scale. KD, LL, AS, TN, RC, AR, and MM contributed to conceptual design of the study. DG was responsible for statistical analysis. Writing by KD and AS with editing by all authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jun Teruya: Evaheart member of DSMB and STAGO honoraria for presentation: Ahmed Said: supported by the Children’s Discovery Institute Faculty Development Award at Washington University in St. Louis: Marie Steiner: Teaching contract (managing coagulation) with Medtronic and study advisory board for Octapharma (AT in Adult CPB heparin resistance). All other authors report no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Code availability
Available on request.
Data availability statement
Available on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
