Abstract
Introduction
For Jehovah’s Witness (JW) patients requiring cardiac surgery, various strategies such as preoperative use of erythropoietin stimulating agents (ESAs), intravenous iron (IVI), and non-pharmacologic interventions have emerged to prevent complications from blood loss given transfusion is not acceptable in this population.
Methods
Retrospective case-control of cardiac surgeries performed by the same surgeon between 1/1/2011 and 8/30/2021. JW patients were matched to non-JW who received blood products and non-JW who did not receive blood products on a 1:2:2 basis. Patients were matched on procedure, age, gender, and Society of Thoracic Surgeons morbidity score. Eligible patients were aged
Results
A total of 27 JW, 52 non-JW transfused, and 53 non-JW not transfused patients were included in the analysis. JW patients had significantly higher mean hematocrits at every time point when compared to non-JW transfused patients and at all time points except clinic and the last recorded operating room value when compared to non-JW not transfused patients. No significant differences in thrombotic rates were found between groups, however there was a numerically higher incidence in the JW population (JW: 7.4%; non-JW transfused: 0%; non-JW not transfused: 1.9%; p = .106).
Conclusion
A blood conservation protocol in a JW population was associated with higher perioperative hematocrit values when compared to matched controls. Further prospective study is warranted before applying similar protocols to other populations given the possibility for an increased rate of venous thromboembolism.
Keywords
Introduction
Cardiac surgery is a leading cause of blood transfusion in the United States. Blood transfusion is required in as many as 90% of cardiac procedures, and the risk of transfusion increases as the complexity and duration of the procedure increases. 1 Transfusion of blood products has been associated with short- and long-term morbidity, and blood transfusion is independently associated with mortality rate of 2.3-5.6 deaths per million components transfused. 2 Patients of the Jehovah’s Witness (JW) faith decline transfusion of red cells on religious grounds, even if transfusion would be lifesaving. Because of product refusal, these patients present a major challenge for management when undergoing cardiovascular procedures in which significant blood loss is anticipated. For JW patients requiring cardiac surgery, various protocols have emerged to bolster pre-operative hematocrit levels to prevent complications associated with anemia during and after the procedure, to control bleeding and prevent blood loss during the procedure, and to promote recovery following the procedure.3–15
Strategies to bolster pre-operative hemoglobin (Hgb) include pre-surgical treatment with intravenous iron (IVI) and erythropoiesis stimulating agents (ESAs). Both of these agents are associated with increasing red blood cells through unique mechanisms without requiring exposure to blood products, which is deemed an acceptable strategy for patients of the JW faith. ESAs promote erythropoiesis by stimulating progenitor cells in the bone marrow to mature and release into the blood stream as reticulocytes. Supplementation of iron ensures that there are sufficient iron stores for incorporation into hemoglobin of the released reticulocytes.16,17 Preoperative optimization of hematocrit with these agents mitigates the risk of a lower nadir hematocrit (Hct) intra-operatively, which is associated with need for blood transfusion and therefore associated complications. 1 ESAs have been associated with a risk of hypertension and increased thrombosis in chronic kidney disease populations, although this risk is increased with longer drug exposure and when targeting higher hemoglobin thresholds (particularly when targeting a Hgb higher than 11 g/dL over a period of months to years). 16 Recent meta-analyses have demonstrated that the use of ESAs and iron supplementation perioperatively in non-cardiac and cardiac populations did not result in significantly increased rates of venous thromboembolism, however, many of the trials included in these analyses were small and did not report data on thromboembolism rates.18,19 Intraoperative measures to mitigate blood loss include the use of blood conservation devices (e.g. CellSaver) and some centers also utilize pharmacologic adjuncts such as tranexamic acid. Post-operative measures to prevent blood loss include minimization of blood draws and minimizing volume when phlebotomy is necessary. These aforementioned protocols have been successful at various JW surgery centers and is well described in the present literature, although no trial has evaluated JW outcomes when compared to non-JW patients who receive blood products and non-JW patients who do not receive blood products, in which the present case control study aims to evaluate.3–15
Materials and methods
This investigation was a retrospective, case-matched study comparing JW patients, non-JW patients who received blood products, and non-JW patients who did not receive blood products for cardiac surgeries matched in a 1:2:2 ratio based on surgery type, age, gender, and Society of Thoracic Surgeons (STS) morbidity and mortality score prior to the procedure between the dates of 1/1/2011 and 8/30/2021. After institutional review board review and with waiver of informed consent, eligible patients were retrospectively identified through a combination of chart-review and data extraction from the University of Kentucky (UK) Albert B. Chandler hospital’s STS database (IRB Number/Approval Date: 71257, 8/22/21). Eligible patients were defined as: adults aged
All JW patients were enrolled in a protocol to conserve blood volume perioperatively, which included strategies such as pre-operative use of ESAs, IVI, utilization of intraoperative blood salvage, and post-operative minimization of blood draws/blood volume. The blood conservation protocol delineated patients to receive ESAs if preoperative hemoglobin values were less than 14 g/dL (approximate Hct 42%) and patients were also supplemented with IVI if determined to be appropriate after assessment of iron studies at the discretion of the cardiac surgeon and consulted nephrologist. While not all JW patients qualified to receive ESAs and IVI, every JW patient received non-pharmacologic measures to limit iatrogenic blood loss.
All JW patients, at initial encounter, had a hematocrit performed. This was used to calculate their first “on pump” hematocrit using a “perfusion calculator,” which takes into account age, sex, height, weight, and prime volume of the cardiopulmonary bypass circuit (CPB). The hemodilution volume is initially 1,800 mL (prime volume). Priming volume for the CPB circuit is 800 mL, with the initial cardioplegia volume being 1000 mL, the standard solution for all patients was Wechsler-Peyton cardioplegia solution. All patients had the same intraoperative protocol and procedures were performed by the same cardiac surgeon. Inflow temperature during the bypass run was at moderate hypothermia (26°C), procedures were performed using a single aortic cross-clamp, hemo-concentration was initiated if reservoir volume was above 2 L. An initial 1 L dose of cardioplegia was administered (up to 1.2 L depending on patient weight) and subsequent doses of 300 mL delivered every 20 min throughout the cross-clamp time. Systemic rewarming was initiated when approximately 20 min of cross-clamp time remained. Prior to cross clamp removal, 300 mL of warm cardioplegia solution was administered. Once off pump, if any volume remained in the reservoir, it was hemoconcentrated and delivered back to the patient.
Outcomes were collected by retrospective chart-review. Primary outcomes included assessment of efficacy: change in perioperative hematocrit values at various time points; as well as safety: incidence of thrombosis events including venous thromboembolism (VTE) and arterial thromboembolism (ATE) up to 30 days post-operatively. Other outcomes collected included total utilization of blood products in each group, hospital/intensive care unit length of stay, duration of mechanical ventilation, renal outcomes, sternal wound infections, bleeding requiring re-operation, valve dysfunction requiring re-operation, and in-hospital/30-day mortality.
Statistical analysis
The twenty-seven JW patients were matched to fifty-two non-JW patients who were transfused and fifty-three patients not transfused who underwent cardiac surgeries by the same surgeon on the basis of procedure, gender, STS Morbidity score, and age. Binary variables are reported as counts and percent of groups; normally distributed continuous variables as group means and standard deviations, and non-normally distributed continuous variables as medians and ranges. Risk factors and outcomes were compared across the three matched groups using chi-square, Kruskal-Wallis, or analysis of variance (ANOVA) as appropriate. Hematocrit levels were compared between groups over several time points within the perioperative period using a linear mixed model to control for intra-patient covariance over time. SPSS (Statistical Package for the Social Sciences) version 28 (International Business Machines Corporation, Armonk, NY) was used for all calculations. Values were considered statistically significant if they fell below 0.05.
Results
Patient and operative characteristics.
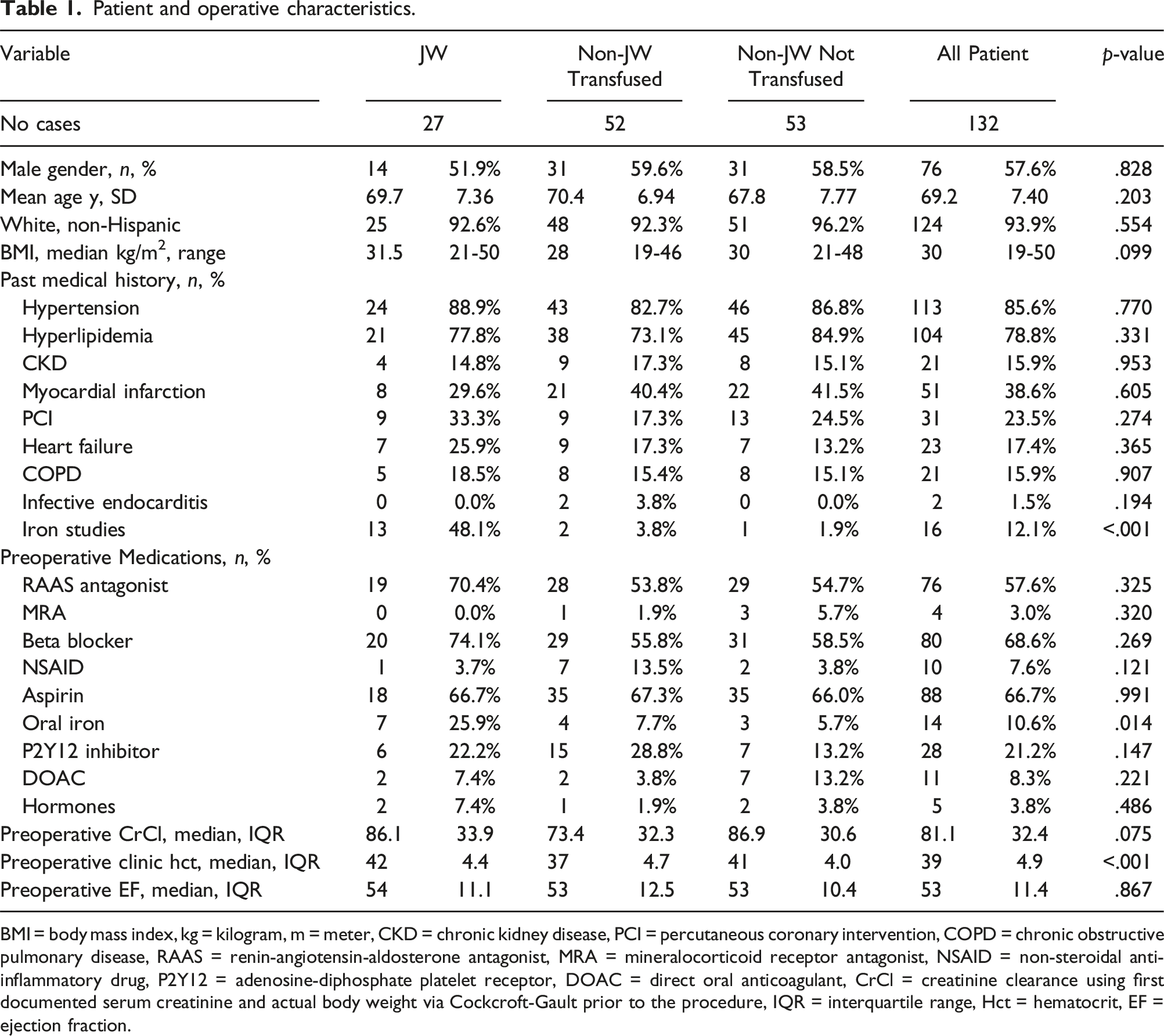
BMI = body mass index, kg = kilogram, m = meter, CKD = chronic kidney disease, PCI = percutaneous coronary intervention, COPD = chronic obstructive pulmonary disease, RAAS = renin-angiotensin-aldosterone antagonist, MRA = mineralocorticoid receptor antagonist, NSAID = non-steroidal anti-inflammatory drug, P2Y12 = adenosine-diphosphate platelet receptor, DOAC = direct oral anticoagulant, CrCl = creatinine clearance using first documented serum creatinine and actual body weight via Cockcroft-Gault prior to the procedure, IQR = interquartile range, Hct = hematocrit, EF = ejection fraction.
Operative characteristics.
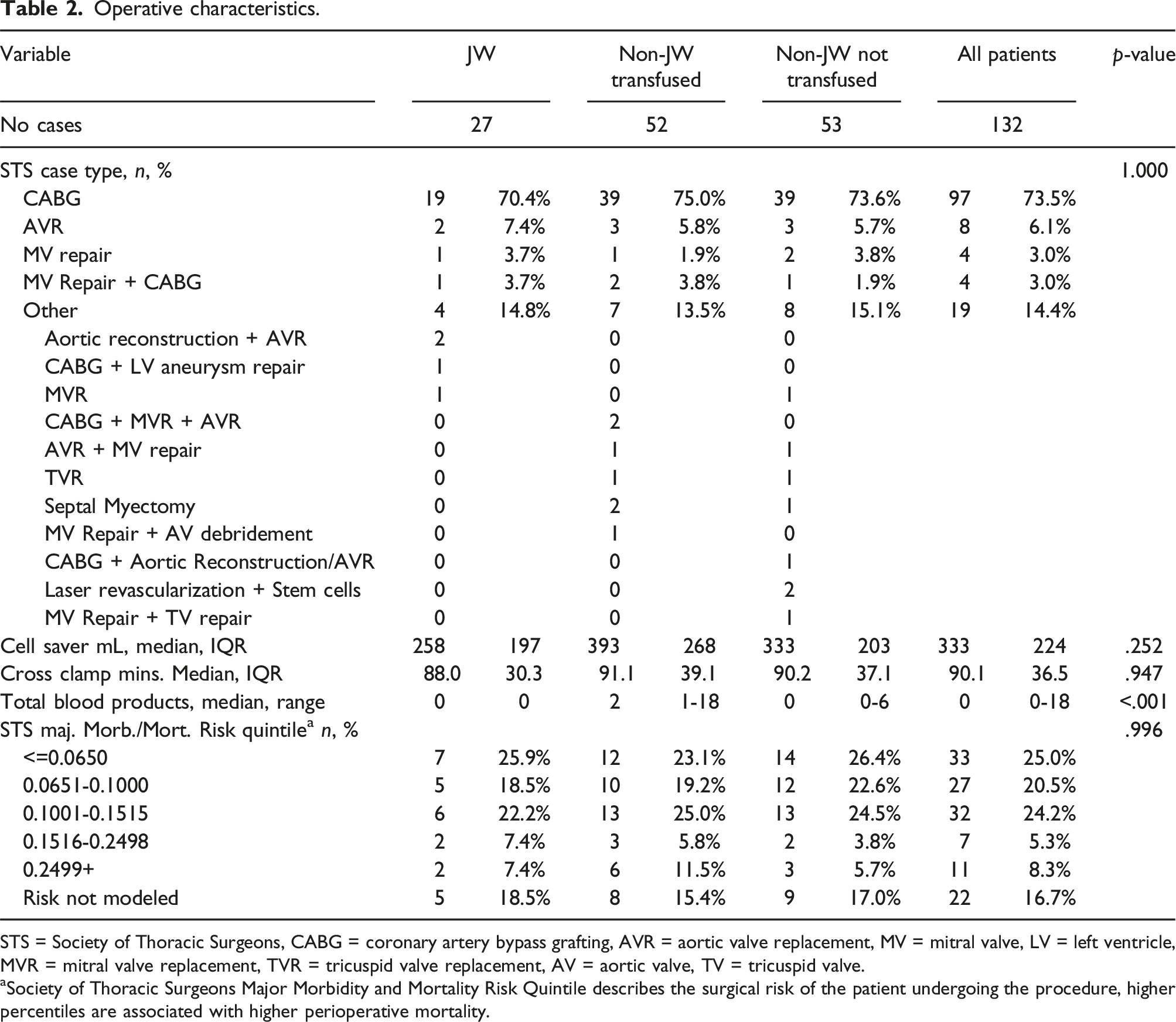
STS = Society of Thoracic Surgeons, CABG = coronary artery bypass grafting, AVR = aortic valve replacement, MV = mitral valve, LV = left ventricle, MVR = mitral valve replacement, TVR = tricuspid valve replacement, AV = aortic valve, TV = tricuspid valve.
aSociety of Thoracic Surgeons Major Morbidity and Mortality Risk Quintile describes the surgical risk of the patient undergoing the procedure, higher percentiles are associated with higher perioperative mortality.
For the primary efficacy outcome, mean perioperative hematocrit values were numerically higher in the JW group than at any other time point when compared to non-JW transfused and non-JW not transfused patients. When statistics were applied through the linear mixed regression model that accounted for patient variance over time, JW patients had significantly higher perioperative hematocrit values at every time point except clinic and last OR JW not transfused patients (Figure 1). Jehovah’s Witnesses had significantly higher mean hematocrit at each time point except for clinic and last OR not-transfused patients. Based on estimates from a linear mixed regression model accounting for intra-patient covariance over time. There were significant differences at every time point between groups except for the Clinic and Last OR values for the JW and Non-JW Not Transfused patients. OR = Operating Room, JW = Jehovah’s Witness.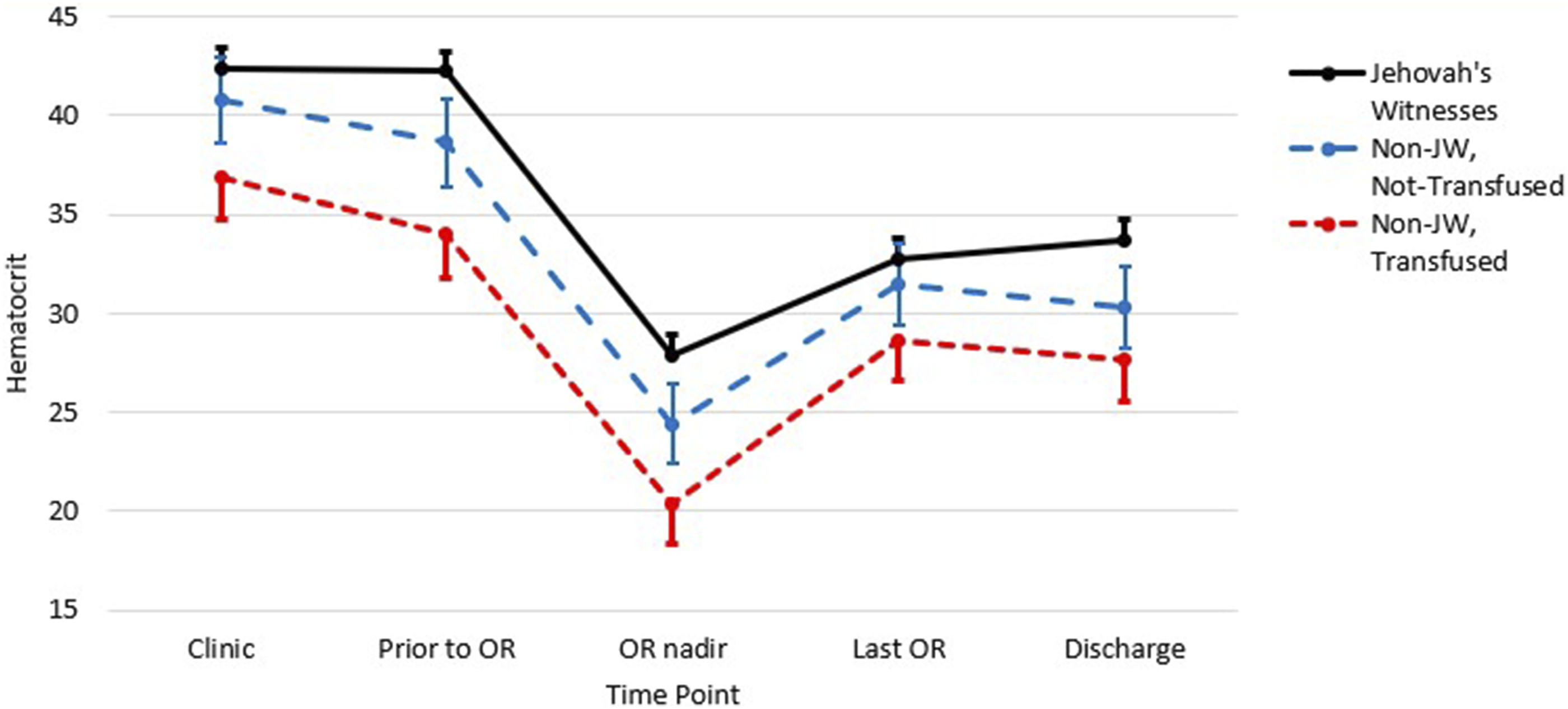
For the primary safety outcome, there were no significant differences between groups in regards to ATE (JW: 3.7%; non-JW transfused: 5.8%; non-JW not transfused: 3.8%; p = .872) or VTE (JW: 7.4%; non-JW transfused: 0%; non-JW not transfused: 1.9%; p = .106). There were, however, numerically higher events of VTE in the JW group. This number was higher in the subgroup of patients who received ESAs/IVI (ESA/IVI VTE: 2/19 [10.5%], no ESA/IVI VTE: 0/8 [0%]). There was no difference in inpatient mortality between groups, however there was a significant difference in the percentage of patients alive at 30 days in favor of patients who did not receive blood transfusions (JW: 96.3%, non-JW transfused: 86.5%, non-JW not transfused: 100%, p = .029).
Outcomes.
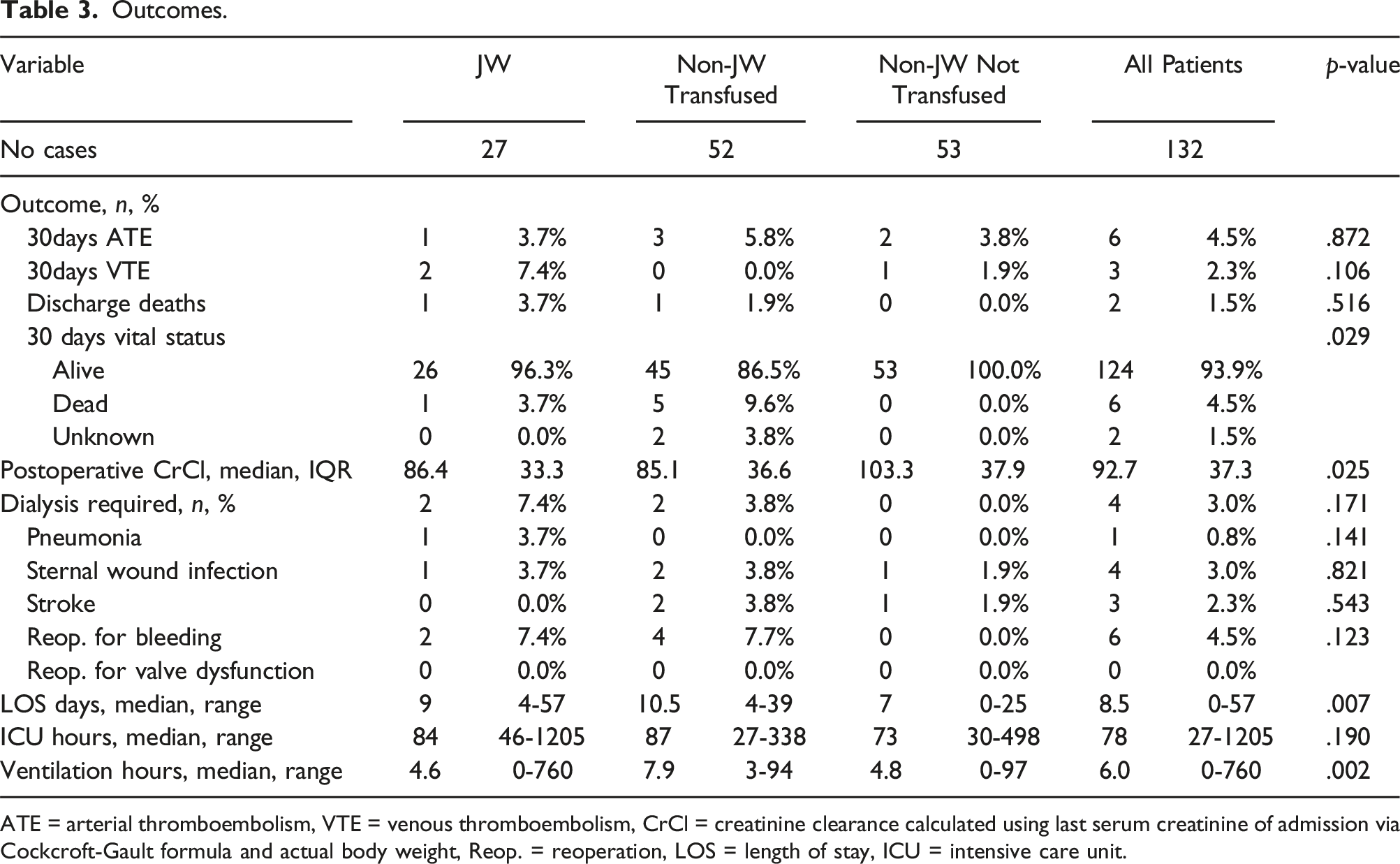
ATE = arterial thromboembolism, VTE = venous thromboembolism, CrCl = creatinine clearance calculated using last serum creatinine of admission via Cockcroft-Gault formula and actual body weight, Reop. = reoperation, LOS = length of stay, ICU = intensive care unit.
Discussion
In this study, we observed that JW patients who decline blood products and participated in a blood conservation protocol had improvements in perioperative hematocrit values when compared to non-JW patients who received blood products and non-JW patients that did not receive blood products. Our study was unique in that no previous study had evaluated clinical outcomes when comparing control patients who received blood and those who did not, and provides further data into the efficacy of this intervention relative to patients who did not require blood transfusion who were not the of the Jehovah’s Witness faith. Hematocrit was consistently higher at most study time points when values were compared with the linear mixed model, and were also numerically higher at each time point studied. This finding is consistent with previous literature describing success when implementing a combined pharmacologic and non-pharmacologic blood conservation protocol at other centers.3–15
The present study also provides insight into the safety of a blood conservation protocol when compared to each population. A non-significant increase in the rate of venous thromboembolism was observed, which has also been reported by other centers. Duce et al. performed a matched control study that compared JW patients who received ESA to non-JW patients who did not receive blood. In that study there was a non-significant but numerical increase in the cumulative mortality or thrombosis (JW, ESA Group 5.66% v. non-JW, non-ESA 0.94%, p = .119). 3 Our study had a similar rate of thrombosis for patients in the JW population compared to controls up to 30 days post discharge (JW: 7.4%; non-JW transfused: 0%; non-JW not transfused: 1.9%; p = .106). Because this study was not powered to detect a difference in the rate of thrombosis, this non-significant finding cannot rule out a higher incidence of thrombosis for patients treated with ESA/IVI. Of the venous thrombotic events observed, there was one pulmonary embolism and one deep venous thrombosis in the JW population and one deep venous thrombosis in the non-JW non-transfused group. Of the arterial thrombotic events observed, there was one patient in the JW group who developed lower extremity arterial thrombi while on extracorporeal support, one transfused patient who developed a LV thrombus, two transfused patients who developed acute ischemic strokes, and two non-transfused patients who developed acute ischemic strokes. Despite this finding of possible increased VTE, it was reassuring that there were positive significant findings regarding vital status at 30 days, hospital length of stay, and total hours ventilated for the JW population when compared to patients who received blood products. Additionally, the utility of ESA/IVI therapy may reduce the periprocedural risk for receipt of hemostatic agents such as recombinant factor VIIa and four-factor prothrombin complex concentrate (4-F PCC), which have been associated with thrombosis in non-cardiac surgery populations given their mechanism of action.20-21 Several retrospective trials and meta-analyses have demonstrated increased thrombosis with recombinant factor VIIa in the setting of cardiac surgery, although these trials are difficult to extrapolate risk given a wide variance in dosing strategies (ranges with estimates of up to 20% of patients experiencing thrombotic events).22–24 Data regarding thrombosis risk with 4-F PCC is lacking in the cardiac surgery population, however several small retrospective studies and one prospective study have suggested the risk of thrombosis is similar when used as first line therapy or when compared to FFP.25–28 One retrospective study has described a center’s experience with utilization of 4-F PCC in JW patients undergoing cardiac surgery, with a trend towards higher rates of thrombosis in patients who received 4-F PCC compared to those who did not (4-F PCC group: 10.5% v. no 4-F PCC group: 3.0%, p = .24). 29 The Average Wholesale Price of these agents are also significant when compared to ESA/IVI therapy (4-F PCC, 2500 units [25 units/kg for a 100 kg patient with no repeat dosing] $8,525; recombinant factor VIIa 5000 mcg [50 mcg/kg for a 100 kg patient with no repeat dosing] $14,900; ESA 10,000 units [100 units/kg for a 100 kg patient] $320.70/dose three times weekly until appropriate Hct achieved; IVI 200 mg $88.30/dose for five doses).16,17,20,21 Additionally, receipt of clotting factor concentrates is not acceptable for every person of the JW faith, and is often a personal choice whether or not to receive these products. Products that are considered unacceptable include whole blood, packed red blood cells, platelets, and fresh frozen plasma. Products that are a personal choice include immune globulins, clotting factor concentrates, and albumin. Products that are generally deemed acceptable include recombinant agents, erythropoietin, vasopressin, and antifibrinolytics. 30
This study has several strengths, with the first being this is the only study to our knowledge to compare JW patients to matched controls who received blood and who did not receive blood. The primary outcomes related to efficacy and safety offer insight into the utility of such a protocol for patients who cannot receive blood products (e.g. JW patients and transplant patients in which avoidance of allosensitization is desired) and generates research questions for the utility of such a protocol in patients who can receive blood when there is a desire to limit transfusions due to possible adverse effects. Patients were also well matched for surgical risk, as demonstrated by the use of the STS morbidity and mortality index as well as the procedure they underwent, which is reassuring when examining efficacy and safety amongst the groups studied.
There are also limitations to our study, with the first being that this was a retrospective chart review and causation cannot be extrapolated from the results. The second being that there were patients in which the STS risk score was not available to be modeled, however there were similar rates of non-modeled patients in each group. There were also several cases that qualified as “Other” when examining the procedure type, however there were similar amounts of “Other” cases in each study group. Additionally, not every patient in our study received pharmacologic modification through the use of ESAs and IVI, which were prescribed at discretion of the treating physician based on their baseline hemoglobin. However, this does provide real world data for patients who may not be eligible for these interventions when their baseline Hct is sufficient without pharmacologic intervention. Finally, the sample size of our study is relatively small, however this is consistent with the size of other published studies on perioperative JW blood conservation protocols.
Conclusions
Use of a blood conservation protocol in a Jehovah’s Witness population was associated with higher perioperative hematocrit values compared to matched controls as well as a higher 30-day survival and lower hospital length of stay and hours requiring mechanical ventilation when compared to patients who required blood products. The utility of blood conservation protocols must be weighed with the potential for increased thromboembolic risk from erythropoietin stimulating agents. Prospective, larger trials are needed before recommending application of similar protocols to populations in which receipt of blood products is considered warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Deidentified data may be available upon reasonable request to the primary investigator.
