Abstract
Introduction
Left ventricular pseudoaneurysm is a rare but serious clinicopathologic entity.
Materials and results
This article describes a case report of 51-year-old man who experienced recurrence of chest pain and dyspnea 4 months later after anterior ST elevation myocardial infarction of first diagonal branch. Anterior basal left ventricular pseudoaneurysm was diagnosed and successful surgical treatment was performed. One year after operation, patient has no cardiovascular events and remains in NYHA class II.
Conclusion
Cardiac magnetic resonance should be performed, if there is a suspicion of left ventricular pseudoaneurysm from transthoracic echocardiography. Surgery is the treatment of choice in case of left ventricular pseudoaneuryms because untreated lesions carry a significantly high risk of rupture.
Keywords
Introduction
Left ventricular pseudoaneurysm (LVP) is a serious clinicopathologic entity.1–3 More than a half of all LVPs are due to myocardial infarction (MI), but LVP could also occur after cardiac surgery (mostly mitral valve), endovascular interventions, trauma and infections.1,2,4,5 This disease carries a significant risk of rupture therefore, an early diagnosis and treatment are crucial. 2 We present a successful case of anterior LVP surgical treatment in a 51-year-old man 4 months after anterior ST elevation MI in a single vessel disease. Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Case presentation
A 51-year-old man was treated after ST elevation anterior MI. Invasive coronary angiography showed coronary artery (CA) stenoses: S1 – 20%, S6 – 20%, S7 – 20%, S9 – 100%. Angioplasty and stenting of the first diagonal vessel were performed. Four days later the course was complicated by stent thrombosis. Repeated first diagonal vessel angioplasty and stenting were performed. Several transthoracic echocardiography (TTE) examinations were performed and no mechanical complications of MI were observed. Left ventricular ejection fraction (LV EF) was 40% because of akinesis in the base of anterior wall, hypokinesis in mid and apical segments of anterior and lateral walls. Mild pericardial effusion was detected, but it was not hemodynamically significant.
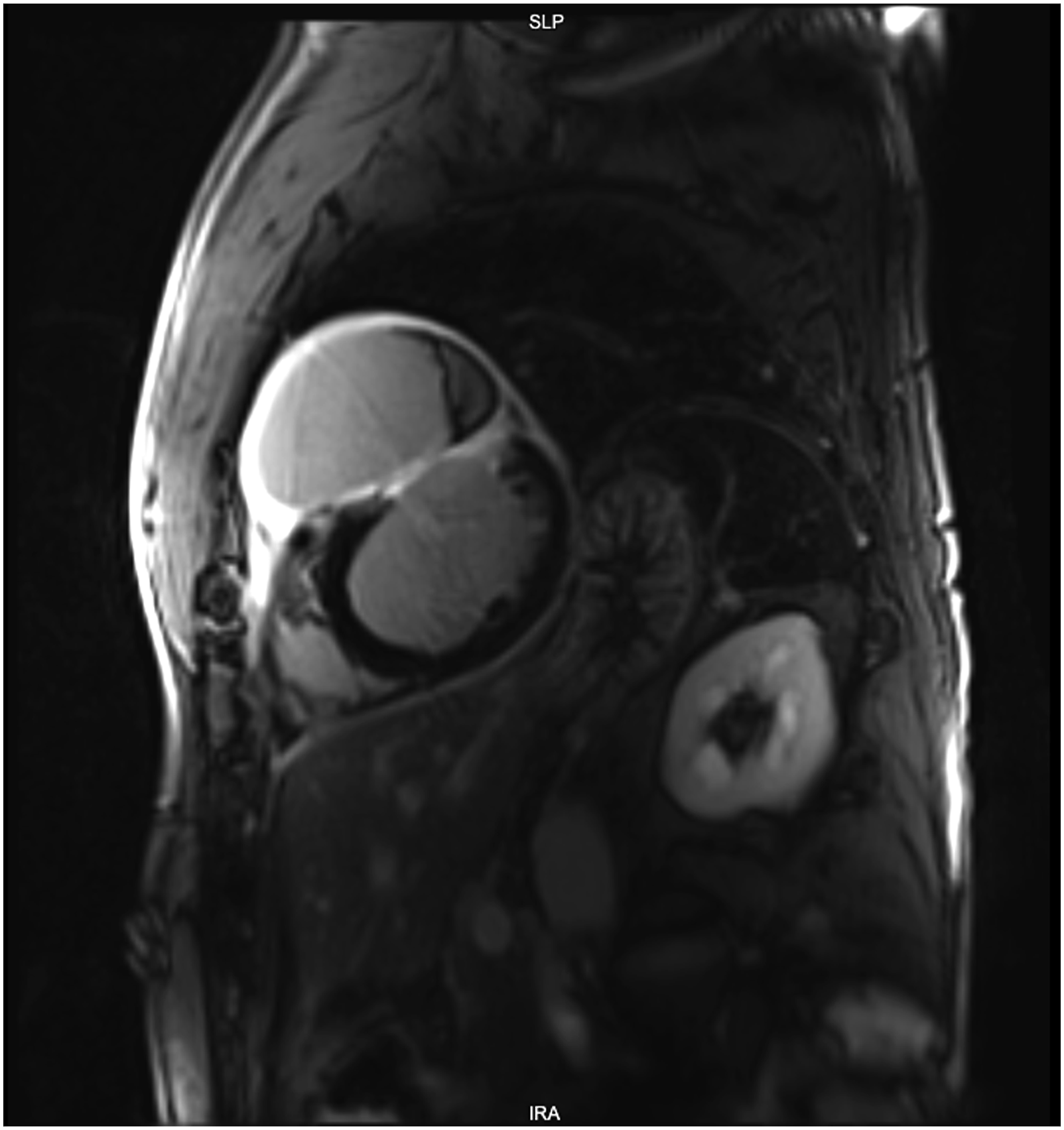
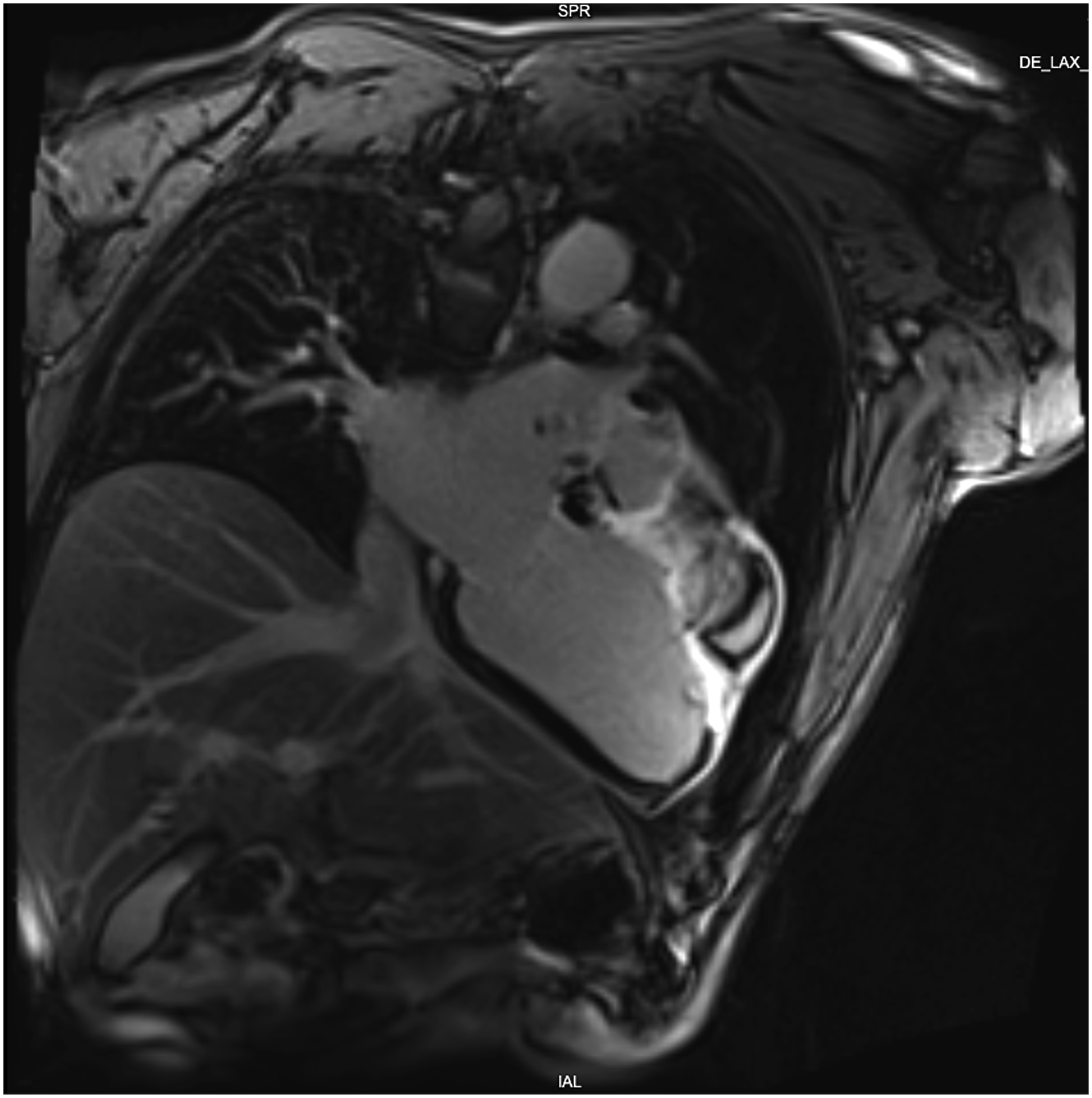
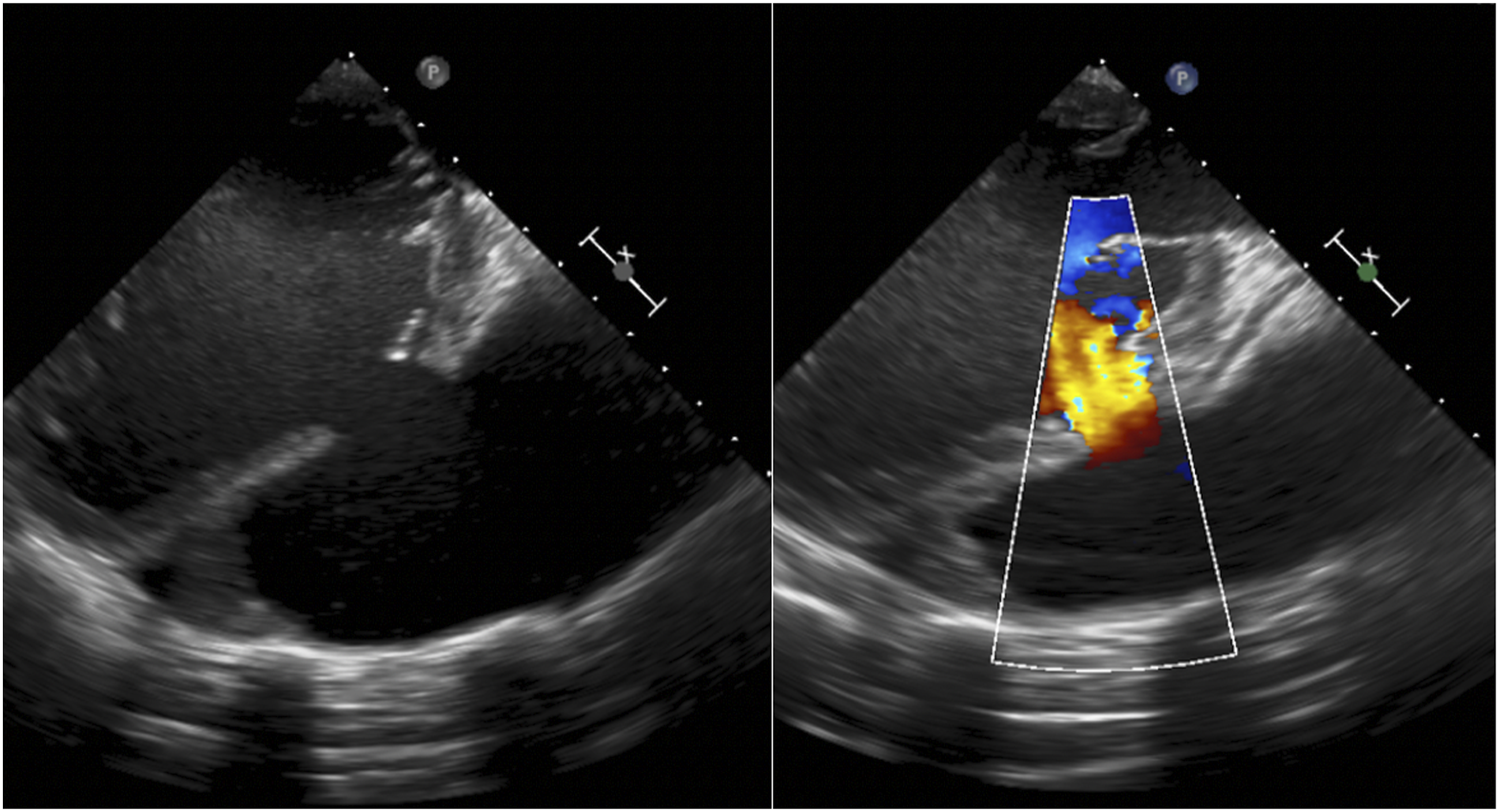
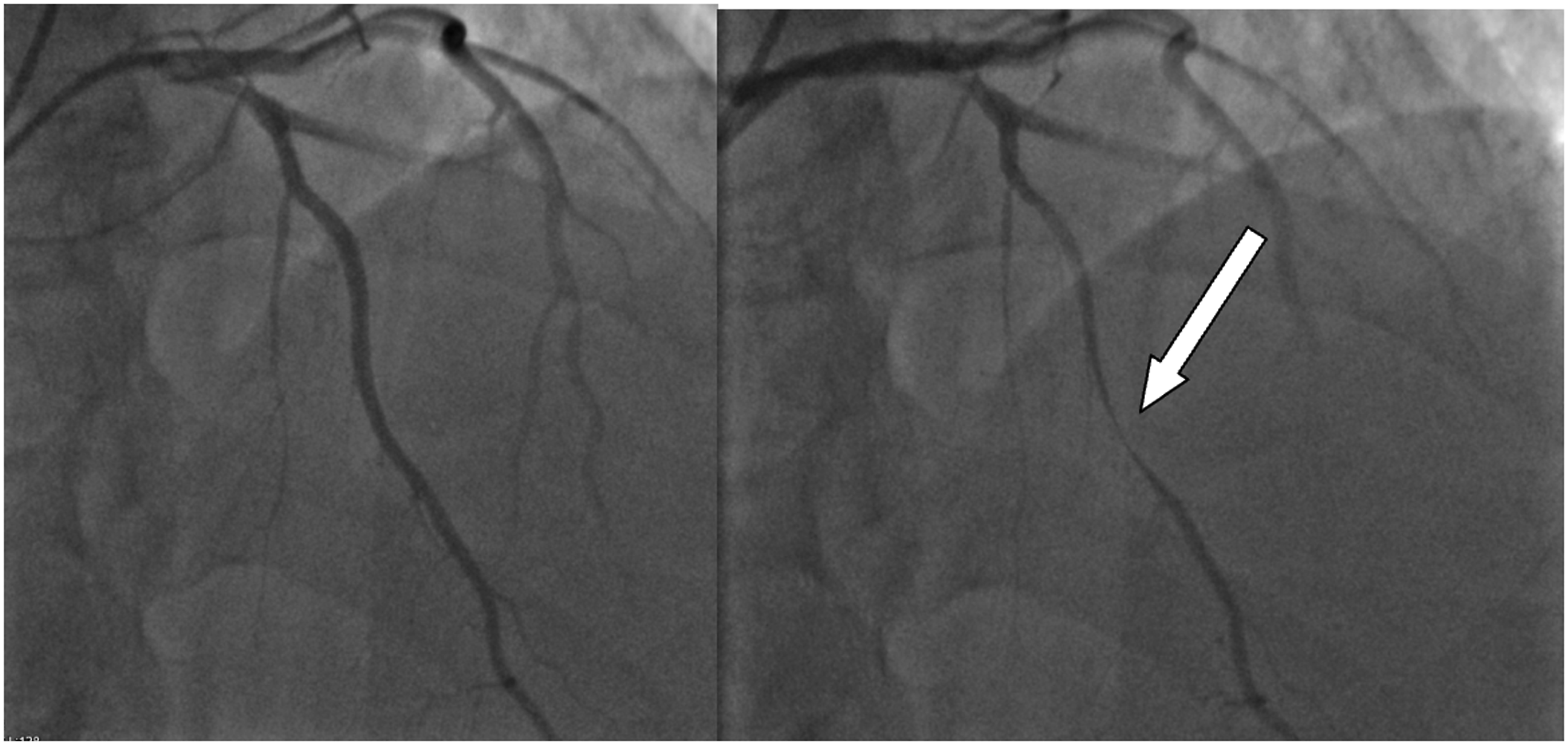
Four months later, after recurrence of chest pain and heart failure (HF) symptoms (NYHA III), he was urgently hospitalized. Chest X-ray showed the displacement of the left ventricle (LV), the left contour was unevenly convex (Figure 1). According to TTE data, LVP in the anterior LV wall was suspected. Cardiac magnetic resonance imaging (MRI) (Figures 2 and 3) showed LVP of ∼7.0 × 5.2 × 11.4 cm size, ∼250 mL volume, in the basal-middle part of the LV anterior wall. LV was dilated with reduced LV systolic function (LV EF 30%). Transesophageal echocardiography (TEE) also confirmed anterior LVP (Figure 4). Coronary angiography was repeated. No CA stenosis were detected, but at mid of left anterior descending (LAD) (S7 segment) was with signs of bridge, pressing of LAD by LVP (Figure 5). Patient data were discussed by the Heart team and surgical treatment was planned. Chest X-ray: displacement of the left ventricle. Left ventricular pseudoaneurysm in cardiac MRI. Left ventricular pseudoaneurysm in cardiac MRI. Left ventricular pseudoaneurysm in transesophageal echocardiography (TEE). Coronary angiography: signs of bridge at mid of left anterior descending artery.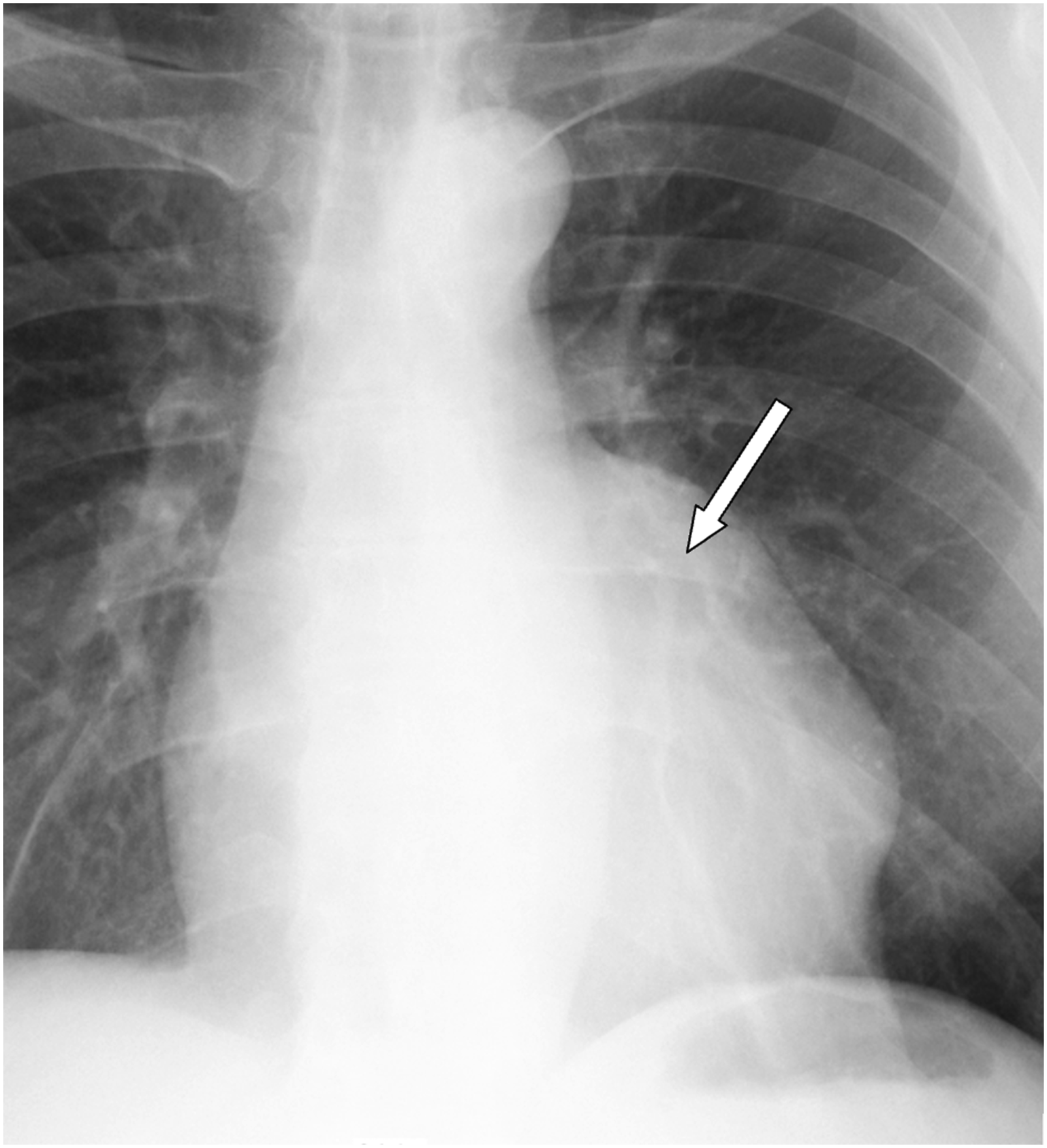
Complete dissection of the heart was performed after cross-clamping the aorta, in an effort to avoid systemic embolization. The dissection was initially limited to the anterior surface of the heart. The heart was then gently mobilized from pericardium and loose adhesions were taken down. The tissues of the LVP sac were resected. The ventricular defect was closed with a patch of Gore-Tex 4.0 × 1.5 cm to avoid potential distortion of the heart structures or excessive traction on the edges of the defect. A second layer over the patch was closed using a patch of autologous pericardium for reinforcing the reconstructed ventricular wall and for hemostasis (Figure 6). The ventricular defect closed with a patch. 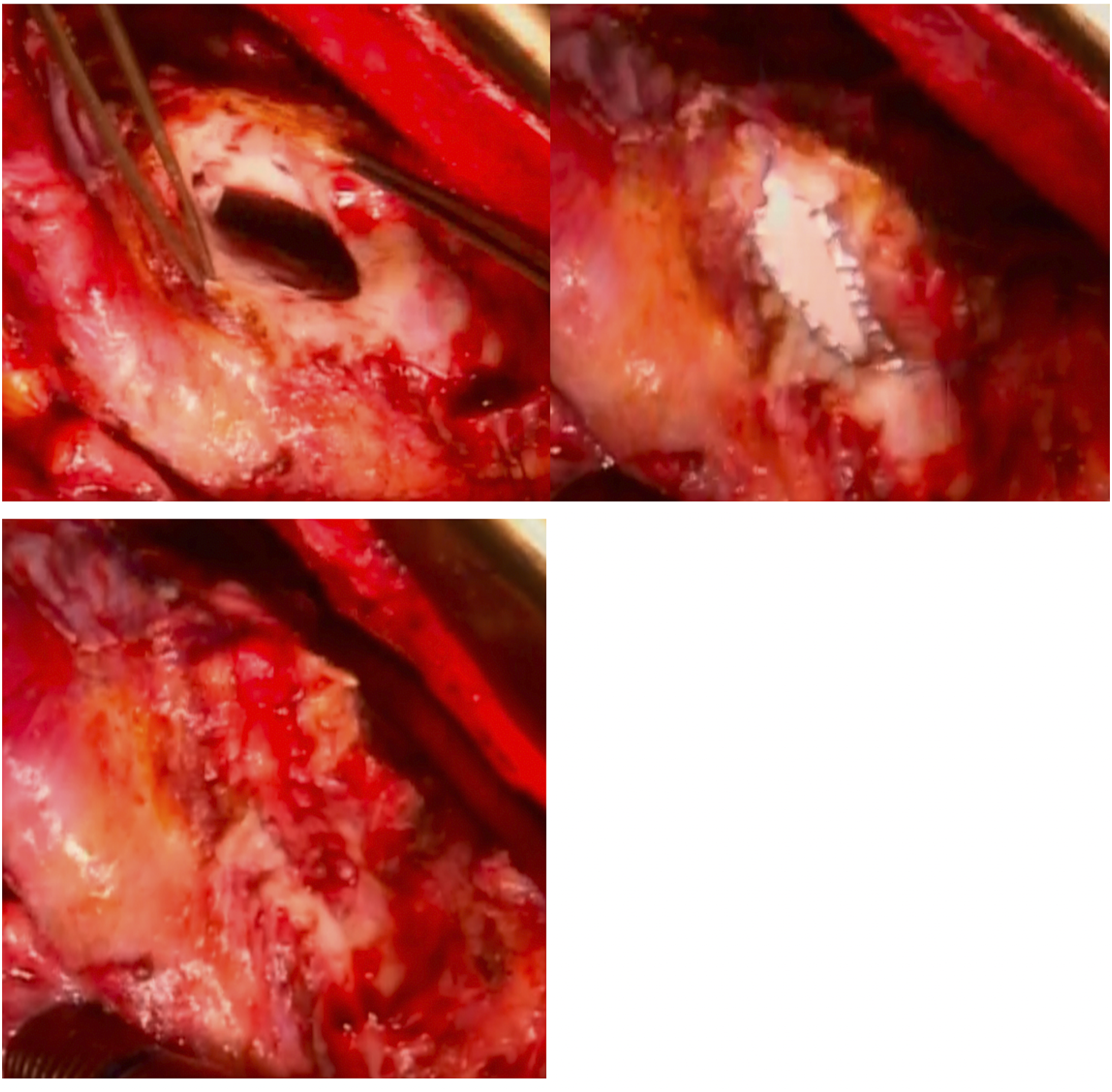
The patient’s postoperative course was smooth and after 12 days patient was referred for rehabilitation. One year after operation he remains in good clinical condition (NYHA class II) without cardiovascular events. TTE and cardiac MRI (Figure 7) were repeated 1 year after surgery. Both exams revealed that LV is not dilated and systolic LV function improved (LV EF 42%). Cardiac MRI one year after the surgery.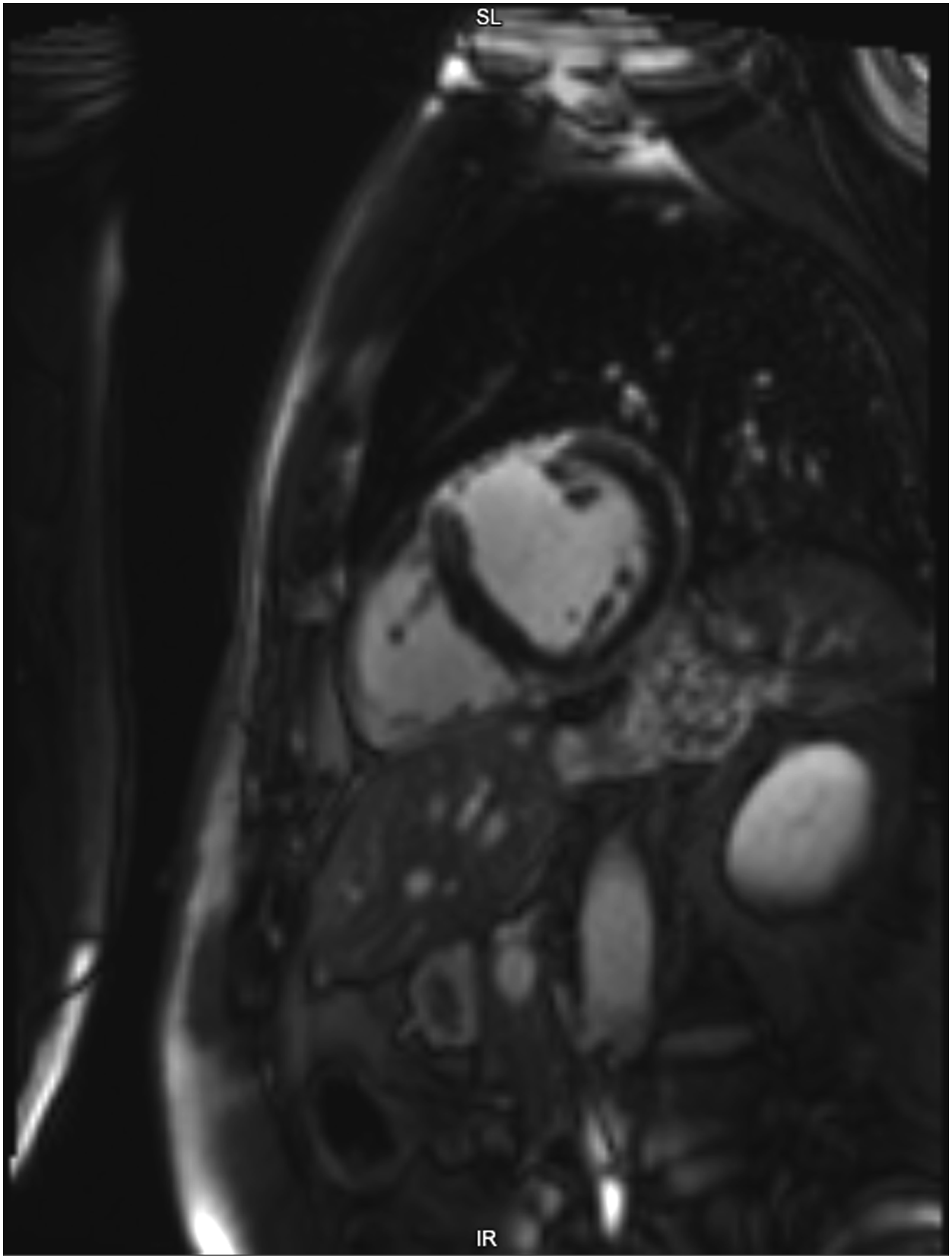
Discussion
The incidence of LVPs after MI is reported to be around 0.2–0.5%. 6 LVP form when cardiac rupture is contained by adherent pericardium or scar tissue, that is why LVP contains no endocardium or myocardium. 1 Wall stress and loss of myocardial integrity (because of MI) are the likely determinants of cardiac rupture which usually results in cardiac tamponade and death. 1 The most common site for the development of LVP is inferior or posterolateral wall. 6 The example presented in this case report is interesting because it was very large anterior LVP, which was diagnosed 4 months after anterior ST elevation MI (as usual LVP forms 2 months after MI) in a single vessel disease. The clinical presentation of LVPs are nonspecific, including HF, chest pain, dyspnea or arrhythmia. It also must be added that a thin or disrupted myocardium moves dyskinetically and can contribute to dangerous ventricular arrhythmia, on the other hand stasis of blood flow inside the LVP cavity can form thrombus and create embolic events. 7 In a systematic literature review, 10% (n = 290) cases were asymptomatic 1 while in another case series 48% (n = 52) cases were asymptomatic. 6 In our case, patient presented with non-specific symptoms like recurrent chest pain and progressive HF 4 months after acute anterior MI.
LVP is a rare complication of MI in which the role of imaging is paramount.1,8,9 In our case LVP was suspected according TTE and confirmed by MRI and TEE data. Although distinguishing true aneurysms from LVP might be difficult, it is essential, since LVPs have much greater risk of rupture.10,11 Historically angiography was considered to be the first-choice test, however non-invasive imaging methods such as TTE, MRI and TEE are preferred now. 10 TEE is useful for assessing LVPs location, size, the presence of thrombus, involvement of mitral apparatus. 7 MRI allows to evaluate structures and tissues, especially pericardium, thrombi and distinguish LVP from true aneurysm.12,13
LVP requires immediate intervention with either surgery or minimally invasive percutaneous transcatheter closure. 8 The surgery itself has high mortality ranging from 20 to 36%, but some of the recent studies have shown improvement in mortality to 10%.13,14 The risk of LVP rupture is estimated to be between 30 and 45%.15,16 The risk of rupture is greater when LVP is large (>3 cm), acute (<3 months after MI) or located in the anterior or lateral wall. 17 However, cases, where surgery cannot be considered because of high risk, and medical management is preferred, mortality is even high and it’s close to around 48%. 18 In last two decade, enhanced transcatheter procedural approaches and advanced device technology has made the percutaneous closure of LVP an increasingly practical option, especially in a patient with an unfavorable predicted surgical outcome.19–21 However, post-MI LVPs benefit the most from the surgical closure.22,23 Because of large anterior LVP patch of autologous pericardium was chosen in our case. To improve curative effect, coronary artery bypass graft could be included in postinfarction LVP treatment 23 but was not needed in our case.
Conclusions
Most LVPs patients report symptoms of HF or chest pain. Cardiac MRI should be performed, if there is a suspicion of LVP from TTE. Surgery is the treatment of choice in case of LVP because untreated lesions carry a significantly high risk of rupture.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
