Abstract
Despite increasing early rehabilitation and mobilisation (ERM) in paediatric intensive care, current evidence for ERM of neonatal and paediatric patients receiving extracorporeal membrane oxygenation (ECMO) is limited. The proposed benefits of ERM in neonatal and paediatric ECMO patients are multifaceted, including reduced immobility related issues and maintenance of functional ability. However, ECMO presents additional safety and logistical challenges, and currently there are no published neonatal or paediatric guidelines. A consensus document was developed to provide guidance for ERM with neonatal and paediatric ECMO patients. The document was developed by specialist physiotherapists from nine ECMO centres within the UK and Ireland, together with the UK Paediatric Critical Care Society ECMO group and members of the multidisciplinary team. The document covers key considerations and practicalities for completing ERM in this population including, acuity level measurement, activity level guidance, safety and risk assessment, and goal setting. Risk assessment and safety checklist bedside tools are also included and designed to be adapted as required to meet specific unit policies and protocols.
Introduction
Extracorporeal membrane oxygenation (ECMO) supports the gas exchange and/or cardiac function of critically ill children. Twelve centres provide ECMO for children within the United Kingdom (UK) and Ireland. The number of centres commissioned to provide ECMO has increased over recent years, in part due to NHS England’s review of cardiac services, and the drive for specialist care closer to patient’s homes. Approximately 270 paediatric patients receive ECMO in the UK and Ireland each year. Children can be supported on ECMO for several weeks and even months in some situations. Historically patients remained ventilated, sedated, and immobile during this time. However, there is increasing evidence of the harmful effects of prolonged bed rest in critically ill children. 1
Early mobilisation is recommended practice within paediatric intensive care units (PICUs), to prevent morbidity and optimise function. 1 Early mobilisation programmes have been shown to reduce time from admission to mobilisation and improve activity levels in the PICU population.2–4 Recent studies have demonstrated the safety of early mobilisation in PICU.5,6 LaRosa et al. 5 studied 4658 mobility events across 82 PICUs in the United States and concluded that medical equipment dislodgement was rare (0.3%). Authors recommend early mobilisation should be considered within 24 hours of admission. 1 Within paediatric intensive care literature early mobilisation is described as part of a rehabilitation bundle, including delirium prevention, family engagement, nutrition, and sleep hygiene.1,2
Rehabilitation strategies are also well established in adult intensive care units and early mobilisation is increasingly common in adult patients supported on ECMO. Early mobilisation in ventilated adults has been shown to reduce the incidence of intensive care unit (ICU) acquired weakness, improve functional capacity, and increase ventilator free days.7,8 Eden et al. 9 published a protocol which provides guidance for rehabilitation of adults receiving veno-venous (VV) ECMO. Several retrospective studies have reported that it is feasible and safe to deliver early rehabilitation in adult patients receiving veno-arterial (VA) and VV ECMO.10–13 No adverse events were reported, and interventions included standing and ambulation. Early mobilisation, started within the first week of ECMO cannulation, has also been associated with shorter duration of ECMO support and ICU length of stay. 14 An exploratory analysis, including 107 adults, reported that physiotherapy whilst on ECMO was associated with reduced ICU mortality when controlled for baseline and severity-of-illness characteristics. 15
Whilst the strategies and benefits described in the adult ECMO population may be extrapolated to older teenagers, neonatal and paediatric ECMO provides additional safety and logistical challenges. At present there are no published guidelines to support rehabilitation of neonatal and paediatric ECMO patients. The proposed benefits of early mobilisation of neonatal and paediatric ECMO patients include reduced immobility related issues, maintenance of functional ability, avoidance of delirium, and improved psychological well-being. Current evidence is limited to single-centre case reports. Authors have described active rehabilitation of paediatric patients supported on VV ECMO as safe, but requiring a coordinated, multidisciplinary approach (age range 8–16 years).16–19 Rehabilitation activities included in-bed cycling, sitting on the edge of the bed, standing and walking. The concept of ‘awake’ ECMO has been described in three single-centre studies, although minimal information was provided regarding the rehabilitation conducted.20–22 In these studies, extubation was reported as safe during VV and VA ECMO in a variety of neonatal and paediatric populations. Extubation also resulted in reduced sedation requirements and increased opportunity for early mobilisation.
The terminology used within the literature is inconsistent. Early mobilisation has been defined as
Purpose
This document was developed to provide guidance for ERM of neonatal and paediatric ECMO patients. Specialist physiotherapists from all 12 UK and Ireland ECMO centres were invited to collaborate. Nine centres were actively involved in establishing this consensus agreement. The document was developed in conjunction with the UK Paediatric Critical Care Society ECMO group and other members of the multidisciplinary team (MDT). It is based on extensive clinician experience and current evidence, and includes key considerations, activity guidance, and risk assessment. The tools included are designed to be adapted by individual centres to meet specific policies/protocols.
Multidisciplinary team
Early rehabilitation and mobilisation of patients on ECMO requires an experienced MDT.
24
The ERM core team will differ between centres, but will most commonly include: • ECMO co-ordinator • ECMO specialist nurse/perfusionist • Senior Nurse • Intensive care/ECMO Consultant • Physiotherapist
Consulting the ECMO surgeon is recommended for any activities of level 4 and above (refer to Activity Level Tool, Table 3). The provision of ERM input varies between hospitals, however allied and other health professionals are vital and can include many of the following: • Occupational Therapist • Speech and language Therapist • Play Therapist • Psychologist • Dietitian • Teacher
Equipment
Desirable equipment for neonatal and paediatric ECMO ERM.

Completing early rehabilitation and mobilisation
For ERM to be successfully carried out with neonatal and paediatric ECMO patients, a robust and structured process needs to be in place prior to any activity. In line with current literature for ERM on PICU, assessment of appropriateness for ERM should occur within 24 hours of ECMO cannulation.
1
An overview of the process is displayed in Figure 1. Neonatal and paediatric ECMO ERM flowsheet.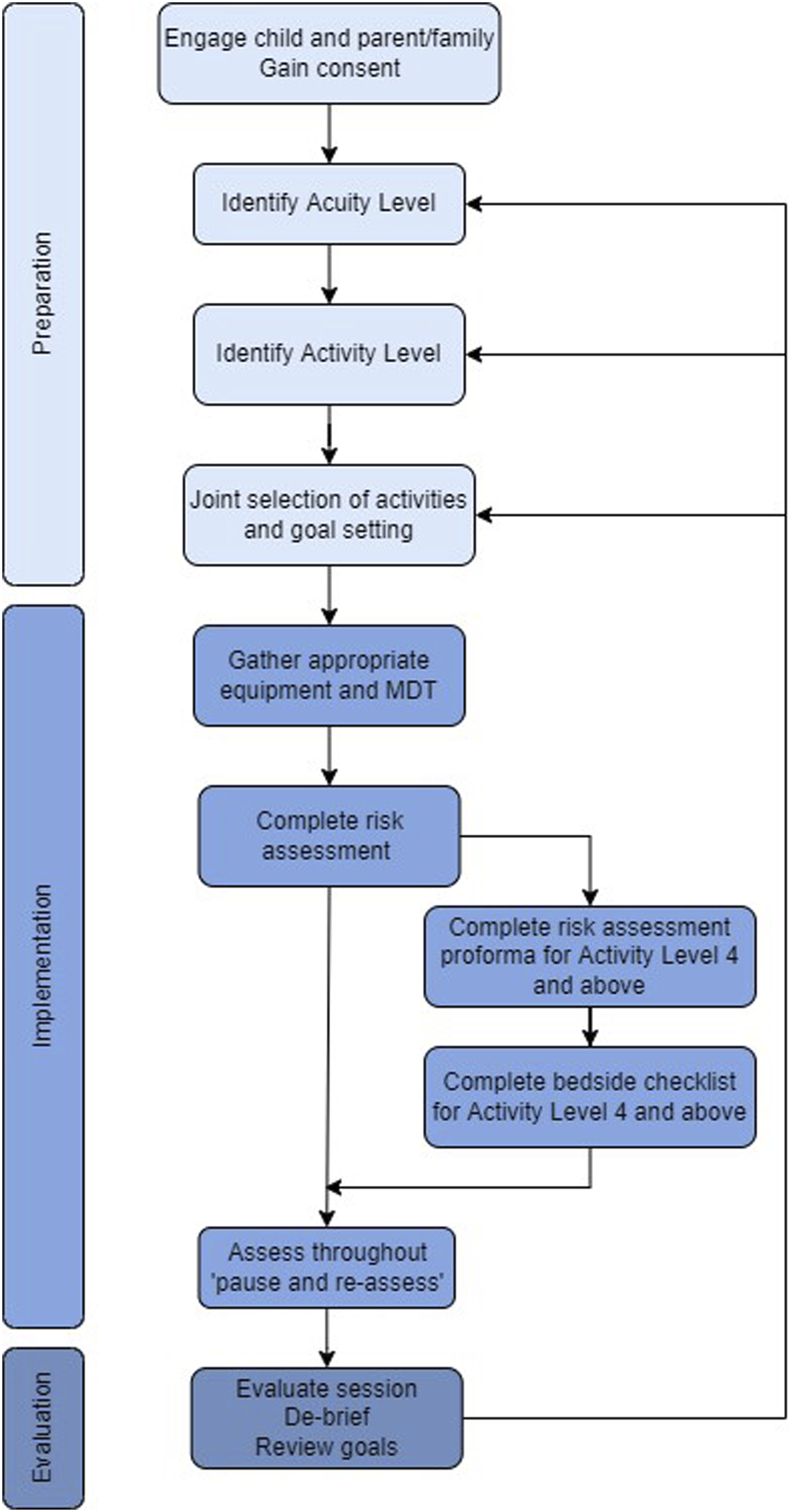
Acuity levels and clinical status
The decision to initiate ERM is dependent on the clinical status and acuity of the patient. This is defined as the amount of support the patient is requiring to maintain stability in key systemic areas and the stability of the ECMO provision itself. It is also important to consider patient age, baseline mobility level, cognitive/developmental status, admitting diagnosis, and anticipated length of ECMO run/PICU stay, prior to commencing ERM.
Neonatal and paediatric ECMO ERM Acuity Level Tool.
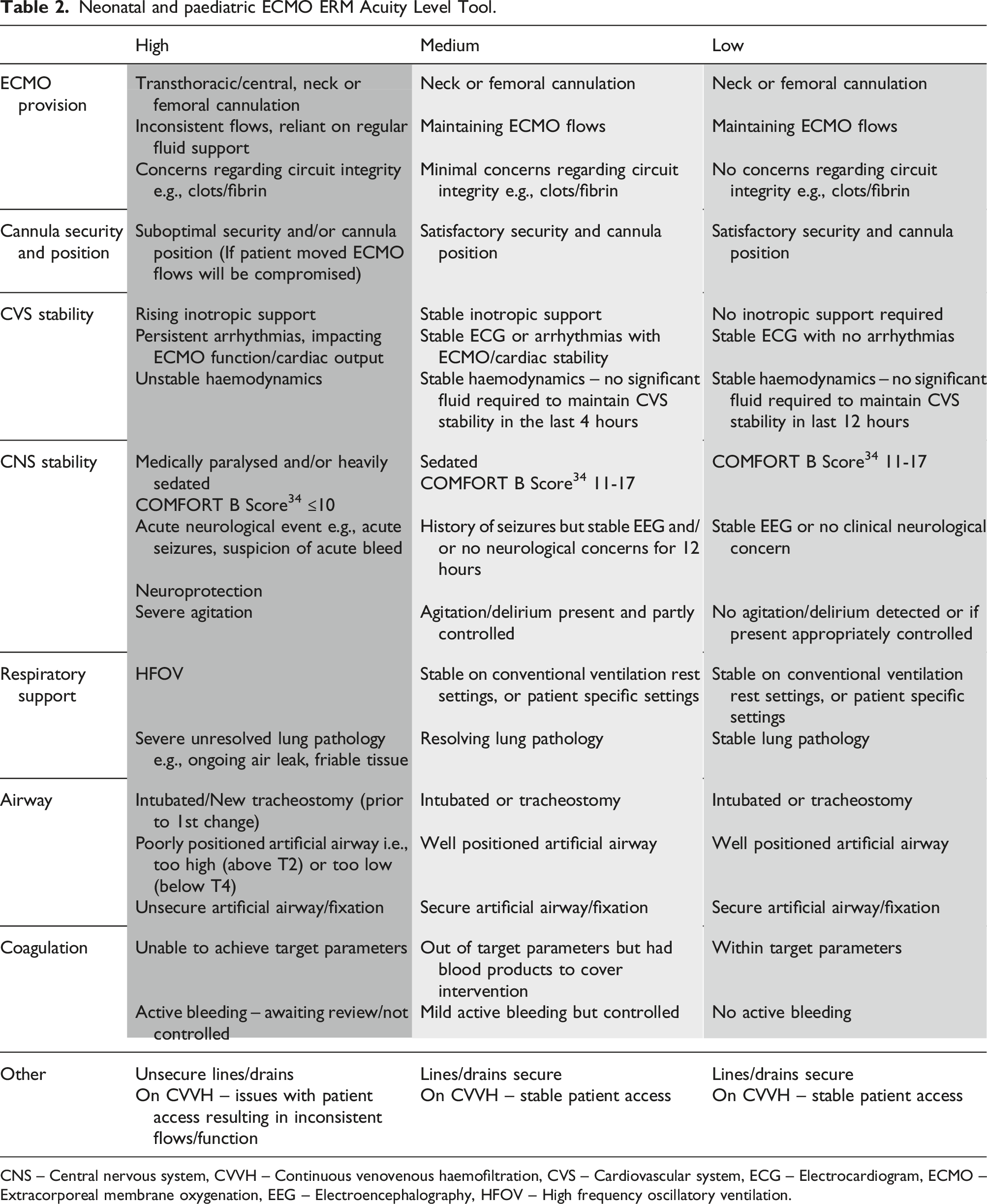
CNS – Central nervous system, CVVH – Continuous venovenous haemofiltration, CVS – Cardiovascular system, ECG – Electrocardiogram, ECMO – Extracorporeal membrane oxygenation, EEG – Electroencephalography, HFOV – High frequency oscillatory ventilation.
Clinical acuity categorisation should be a joint decision involving appropriate members of the ERM core team. It is advised that the determined acuity level is clearly documented together with the ERM intervention in the patients’ clinical notes. It can also be documented on the Checklist for ECMO ERM (Supplementary file I). Clinical acuity level should be re-evaluated prior to each ERM session.
Selecting early rehabilitation and mobilisation activities
Neonatal and paediatric ECMO ERM Activity Level Tool.
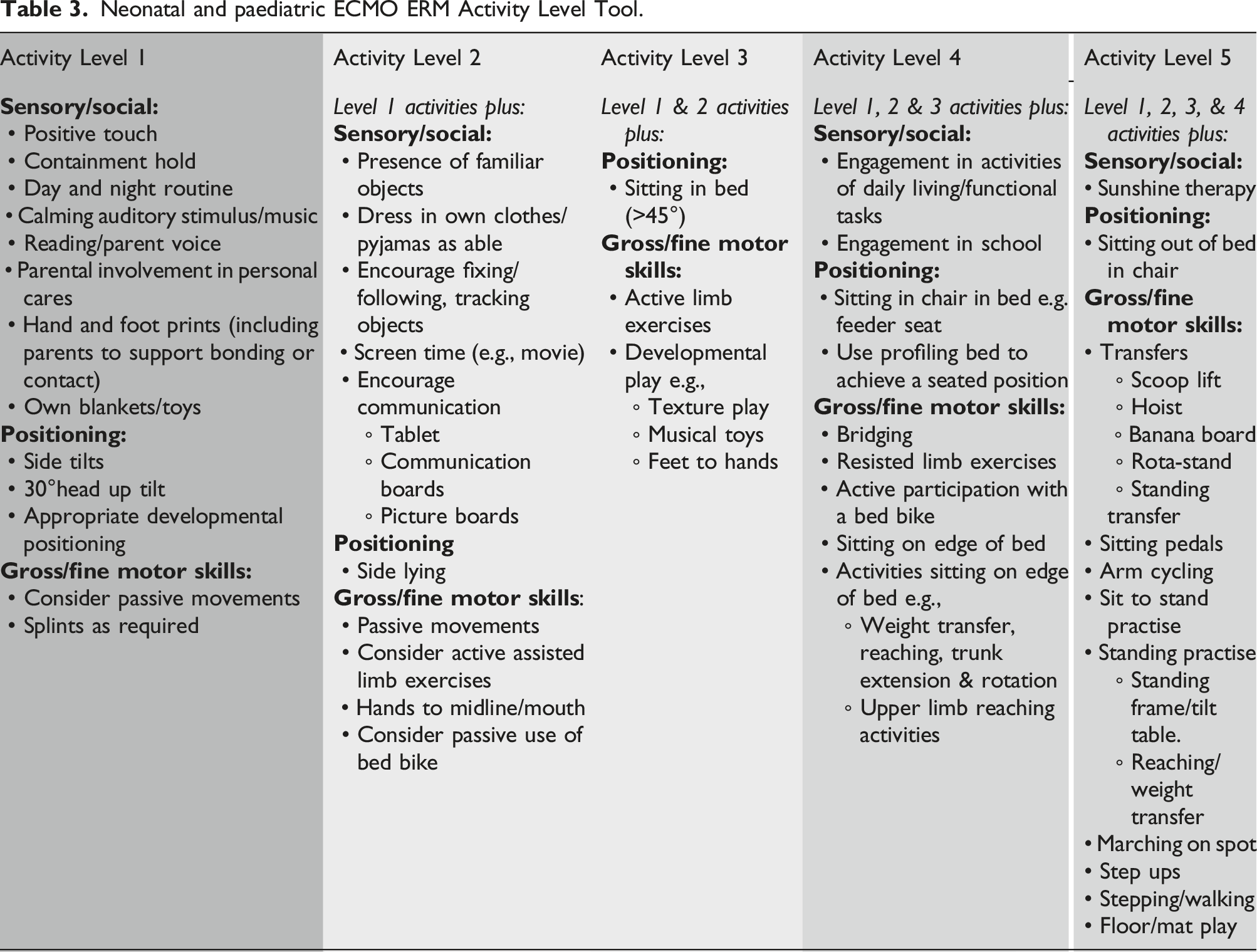
The Activity Level Tool comprises of a selection of sensory, social, positioning, and gross/fine motor activities, although it is not designed to be an exhaustive list. All members of the MDT can be involved in activity selection, and parental/family and patient engagement should be encouraged. Completing a maximum of three activities of mixed genre, spaced out, within a 24-hour period is recommended. For example, a patient in acuity level 3, sits up in bed and participates in a school lesson in the morning, completes a sit on the edge of the bed with physiotherapists in the afternoon and watches a movie with parents in the evening.
The activity levels are suggested as a guide, and it is recognised there may be exceptions. In such circumstances an individual patient assessment and a collaborative MDT approach should be used to select appropriate activities whilst ensuring safety. Prone positioning has not been included in this document as its purpose in neonatal and paediatric ECMO is to influence respiratory function. It would not be completed primarily for developmental or rehabilitation purposes.
Preparation for early rehabilitation and mobilisation
In addition to determining patient acuity and activity levels, it is recommended that a comprehensive, patient specific, risk assessment is completed prior to undertaking ERM. A Risk Assessment Proforma has been created for ERM of activity level 4 and above (Supplementary file II). It is designed to be completed with the ECMO MDT, as a one-off exercise prior to commencement of a new ERM activity. Once completed it does not need to be repeated for the same ERM activity. Completion of the risk assessment allows consideration and mitigation of the following risks/hazards: • ECMO cannula movement, advancement and/or rotation • Patient airway mismanagement • Risk of bleeding/increased bleeding • Patient agitation/hypertension • Respiratory compromise due to cardiovascular exertion • Dislodgement/mismanagement of additional hardware e.g., lines or chest drains • Inadequate personnel number or experience
To provide further opportunity for safety consideration a Checklist for ECMO ERM has also been developed for activity level 4 and above (Supplementary file I). This should be used at the bedside, immediately prior to each ERM session. These documents have been designed to be adapted by individual centres to suit specific hospital policies/practices.
Safety and re-assessment
Continuous assessment should occur during ERM to ensure safety is maintained and both the ECMO circuit and the patient are tolerating the activity. The same considerations outlined in the Checklist for ECMO ERM (Supplementary file I) need to be reassessed throughout the activity. These include security of the ECMO cannula, ETT and other attachments, baseline ECMO and physiological patient parameters, and sedation/pain levels. Concerns during ERM sessions should be raised using the ‘pause and reassess’ strategy used in PICU ERM. 1 Whereby the ERM activity is either continued after a period of assessment and stabilisation or stopped if the concern persists. The situation should then be re-evaluated in 24 hours, or when deemed appropriate. A post-ERM review is advised and included within the Checklist for ECMO ERM; to reassess safety considerations, de-brief and provide recommendations for future ERM sessions.
Staff must adhere to individual hospital moving and handling protocols. Team members completing ERM should be familiar with the emergency event ECMO protocols and adhere to individual hospital policies/protocols.
Goal setting and progression
SMART (Specific, Measurable, Achievable, Realistic, Timed/Timely) goals should be used when ERM is commenced. These can be linked to the International Classification of Functioning (ICF), Disability and Health - Children and Youth version activity/participation domain. 25 Joint goal setting involving the parent/family, and child when possible, will enable therapy to be targeted to the individual. 26 It is recommended that goal setting and progress should be reviewed weekly, or more frequently if indicated by changes in clinical status. 9 The use of Goal Attainment Scaling (GAS) can assist with measuring progress against individual set goals. 27
Using the Activity Level Tool (Table 3) progression of ERM activity can be achieved by increasing the number or intensity of the activities, or by progressing to the next activity level within the same acuity level. This can be done within the same session or considered in planning of future sessions. If progression to the next activity level falls outside the current assessed acuity level, then this should only be attempted once the patient is assessed and found to be within the corresponding acuity level. For example, a patient in the high acuity level cannot progress to activity level 2 until assessed to be within the medium acuity level. However, a patient in the medium acuity level, can progress from activity level 2 to level 3.
Outcome measures
To aid evaluation and progression of ERM the use of outcome measures is encouraged. However, there are limited outcome measures available for use in the PICU setting and none have been identified specifically for neonatal or paediatric ECMO patients. Available options are:
Additional neurodevelopmental outcome measures can be used to assess long term progress and for continuation of monitoring following PICU discharge (e.g., Bayley Scales of Infant and Toddler Development, Hammersmith Infant Neurological Examination).
Additional considerations
• Sedation and delirium management – The use of a standardised sedation scoring system is recommended for neonatal and paediatric ECMO patients.
31
This should be reviewed frequently by the MDT and sedation minimised where possible.
32
Delirium screening should also be considered, this can be completed using the Cornell Assessment of Pediatric Delirium (CAPD).
33
• Dietitian input – It is essential to ensure the patient is receiving adequate nutrition to facilitate recovery and meet the additional energy requirements for ERM. High level ERM may not be appropriate for patients that have a poor nutritional status, despite being stable in other elements of their medical care. Dieticians can also advise on feed timings which will help facilitate ERM. • Timing of ERM – This should be considered to avoid fatigue or over stimulation, especially if several activities are planned for the same day. Completion of ERM in relation to planned procedures or ECMO weaning should be avoided. • Parental/family involvement – This should be encouraged during the planning, completion, and evaluation of ERM activity. • Holistic approach – A holistic and patient centred approach should be adopted throughout ERM. • Additional services involvement (e.g., psychological support, chaplaincy) should be considered throughout ERM planning and evaluation. • Patients suffering acute neurological events – Consideration of additional tone/spasticity management may be required.
Supplemental Material
Supplemental Material - Early rehabilitation and mobilisation of neonatal and paediatric extracorporeal membrane oxygenation patients: A UK and Ireland consensus document
Supplemental Material for Early rehabilitation and mobilisation of neonatal and paediatric extracorporeal membrane oxygenation patients: A UK and Ireland consensus document by Emma Shkurka, Hannah Child, Jennie Balls, Ellie Melkhuln, Lee Carter, Vanessa Compton, Ross Marscheider, Rachel McConnell, Samantha Meenaghan, Natalie Milburn, Gillian Nugent, Gemmasi Peabody and Laura Rimmer in Perfusion
Footnotes
Acknowledgements
We would like to thank the UK Paediatric Critical Care Society ECMO group for their support and advice during this project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
