Abstract
Fine particulate matter <2.5 μm in diameter (PM2.5) has been validated to associate with cardiovascular diseases (CVD) incidence and mortality. So far, no study has quantitatively evaluated the relationship between the atmospheric PM2.5 exposure and ischemic heart disease (IHD). We conducted a meta-analysis to illustrate the relationship between PM2.5 and IHD. Published articles were systematically searched (until June 2022) from PubMed, EMBASE, Cochrane Library. A random-effect model was performed to summarize the total relative risks (RRs) and 95% confidence intervals (CIs). Meta-analysis was performed using Stata 12.0 software. A total of 28 studies among 23 cohorts (23.38 million individuals and 256256 IHD cases) were included. With PM2.5 increasing 10 μg/m3, the total RRs of IHD incidence and mortality were 1.07 (95% CI: 0.99–1.17), 1.21 (95% CI: 1.15–1.28), respectively. In sub-analyses, our study revealed that the combined RRs of exposure to PM2.5 on IHD mortality in Asian and European population [1.11 (95% CI: 0.93–1.33); 1.06 (95% CI: 1.02–1.11)] were much lower compared with American and Canadian people [1.27 (95% CI: 1.17–1.37); 1.30 (95% CI: 1.24–1.35)]. Furthermore, study duration, size and some adjustments were related with the total RR. Our findings indicated that exposure of an increase in the concentration of atmospheric PM2.5 may increase the risk of IHD incidence and mortality. Further evidence is needed to confirmed the association.
Introduction
Due to the continuous development of social economy, the lifestyle has been greatly changed, and the number of cardiovascular diseases and deaths has increases by year. As one of the important diseases of cardiovascular diseases, ischemic heart disease (IHD) represents a large burden on individuals and health care resources worldwide. 1 Globally, in 2019, IHD was the world’s leading cause of morbidity and mortality, accounting for 16% (8.8 million) of the global disease deaths. 2 The high cost of disease prevention presents a growing challenge for health care systems, and cardiovascular diseases contribute substantially to these costs. In America, the estimated direct and indirect cost of heart disease from 2014 to 2015 was $218.7 billion. 3
In large prospective trials, several risk factors have been empirically linked to an increased risk of IHD, include physical inactivity, tobacco use, diet, “bad fats” in the blood, hypertension, diabetes mellitus and being overweight.4–6 There is clear evidence for the contribution of fine particulate (PM2.5) to cardiovascular outcomes, including IHD. In 2017, IHD attributable to PM2.5 resulted in 977140 deaths and 21.93 million disability-adjusted life years (DALYs) globally. 7 Exposure to PM2.5 may plausibly develop IHD through multiple pathways, such as through aggravating myocardial ischemia/reperfusion (I/R) injury, 8 triggering inflammatory responses, 9 and upregulating the endothelin system. 10
The role of PM2.5 in the development of cardiovascular diseases has been investigated for many years. A growing body of epidemiological literature shows that PM2.5 is a well-defined risk factor for IHD, earlier studies of long-term PM2.5 exposure focused more the United States and Europe,11–13 whereas in the past decade more studies have been published in Asia and Oceania.14,15 While there was an evidence-based meta-analyses also revealed that long-term exposure of PM2.5 may be related to a higher risk of cardiovascular disease event. 16 However, the results were still limited and unclear. Therefore, there is an urgent need to summarize and compare the IHD mortality in relation to atmospheric PM2.5 exposure. To systematically explore the association between PM2.5 and IHD, we carried out a meta-analysis to draw a more convincing conclusion with respect to PM2.5 and IHD.
Methods
Search strategy
The present analysis was performed according to the Preferred Reporting Items for Meta-analyses (PRISMA) guideline. We searched the eligible publications in the PubMed, EMBASE, and Cochrane Library up to June 2022. The following keywords were used: “ultrafine fibers” or “airborne particulate matter” or “ambient particulate matter” or “ultrafine particulate matter” or “ultrafine particles” combined with “cohort” and “ischemic heart disease”. Two investigators searched the studies independently. We obtained data by screening titles and abstracts, and examined full text. Reference lists were searched for additional relevant publications. The identified articles should be published in English. According to the flow diagram of literature selection is shown in Figure 1. Flow diagram of literature selection. IHD, ischemic heart disease; RR, relative risk.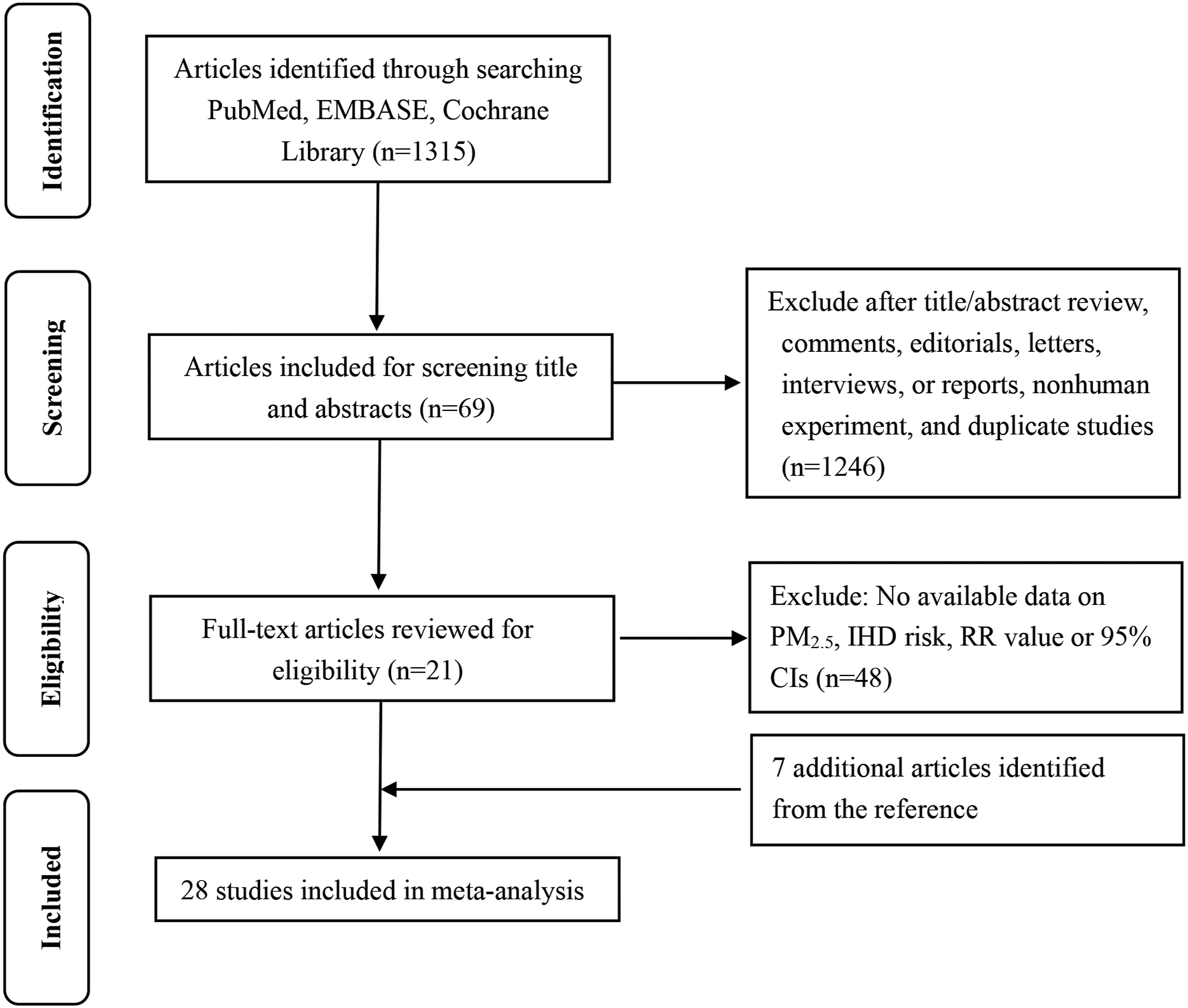
Selection criteria
Studies were eligible if they satisfied the following conditions: (1) cohort studies; (2) exposed from PM2.5; (3) outcome was IHD incidence or mortality; (4) had available data of relative risk (RR) or odds ratio (OR) or hazard ratio (HR) with corresponding 95% CI. Articles were excluded if they satisfied these criteria: (1) nonhuman experiment; (2) reviews, comments, editorials, letters, interviews, or reports; (3) duplicate studies; (4) incomplete data.
Data extraction
Study basic information and characteristic data included the author, year, design, location, sex, duration, study size, number of cases, source of outcome data, outcome event, relative risk (RR) or hazard ratio (HR) or odds ratio (OR) with 95% confidence intervals (95%CI), and adjusted variables. Two investigators extracted data independently.
Literature quality assessment
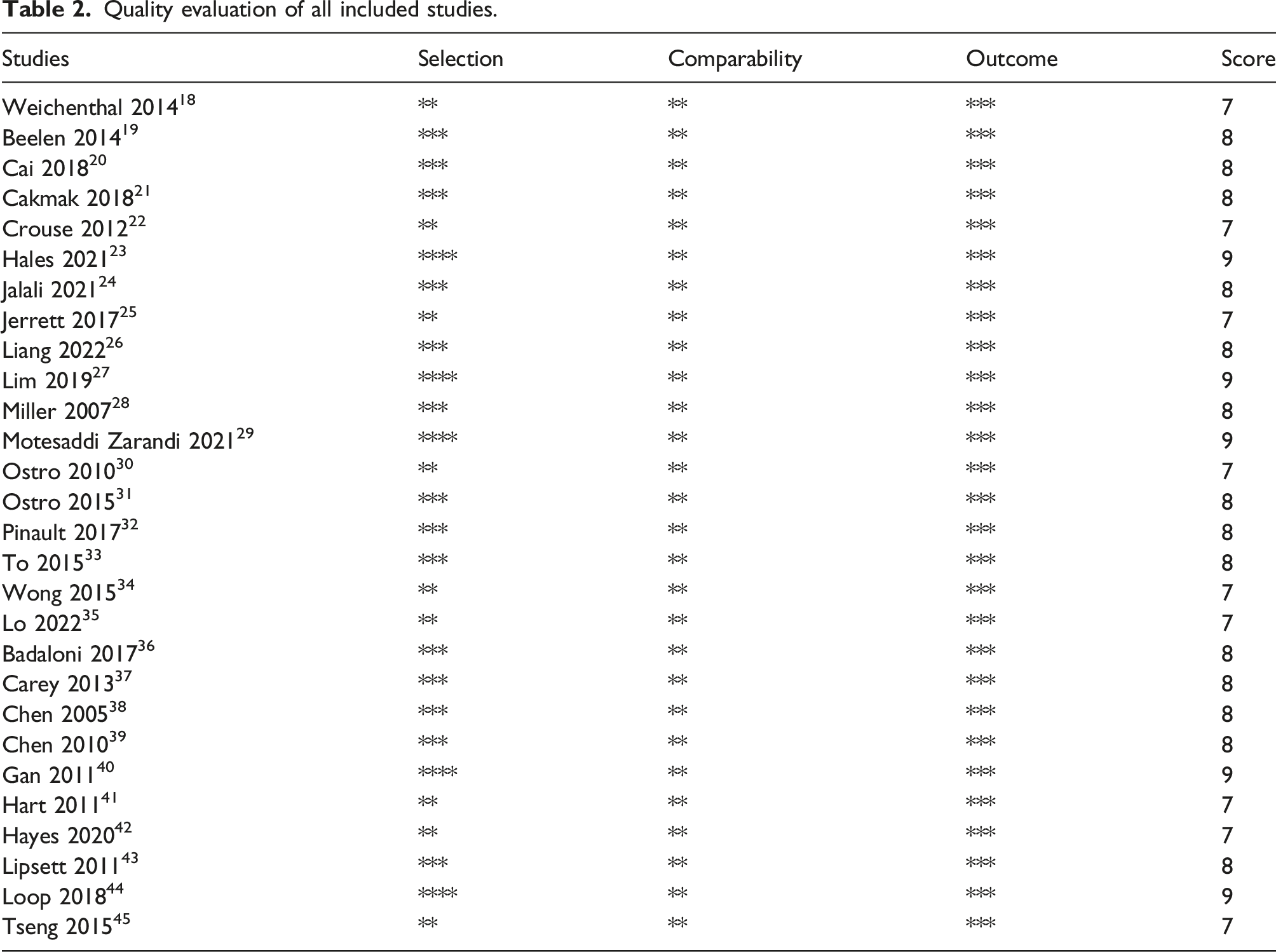
We estimated the quality of studies by the Newcastle–Ottawa Quality Assessment Scale with nine questions. 17 One score represented a satisfactory answer, which a maximum point was 9. Only those studies for which the majority of the questions were deemed satisfactory (ie, which a score of 6 or higher) were defined as high methodological quality.
Statistical analysis
All studies which examined the relationship between the exposure to PM2.5 and IHD with RR or HR or OR and 95% CI were included. All study effect estimates were converted to present a change of 10 μg/m3. Heterogeneity of effect size across the studies was assessed by the Cochran’s Q test and I2 statistics. Generally, I2 < 30% was defined as no or acceptable heterogeneity, 30–75% as mild heterogeneity, and over 75% as notable heterogeneity. The random-effects model was used only when there existed significant heterogeneity; otherwise, the fixed-effects model was applied for further analysis. Sensitivity analysis was used to determine the stability and reliability of the results by sequentially deleting one study. Subgroup analyses and meta regression analyses were conducted to investigate the heterogeneity sources. The funnel plots, Egger’s linear regression and Begg’s rank correlation test were carried out to assess the publication bias, p < .05 was considered a potential small study bias. All statistical analyses were conducted using STATA statistical package version 12.0.
Results
Studies characteristic for mata-analysis
Characteristics of studies examining the association between long-term PM2.5 exposure and ischemic heart disease events.
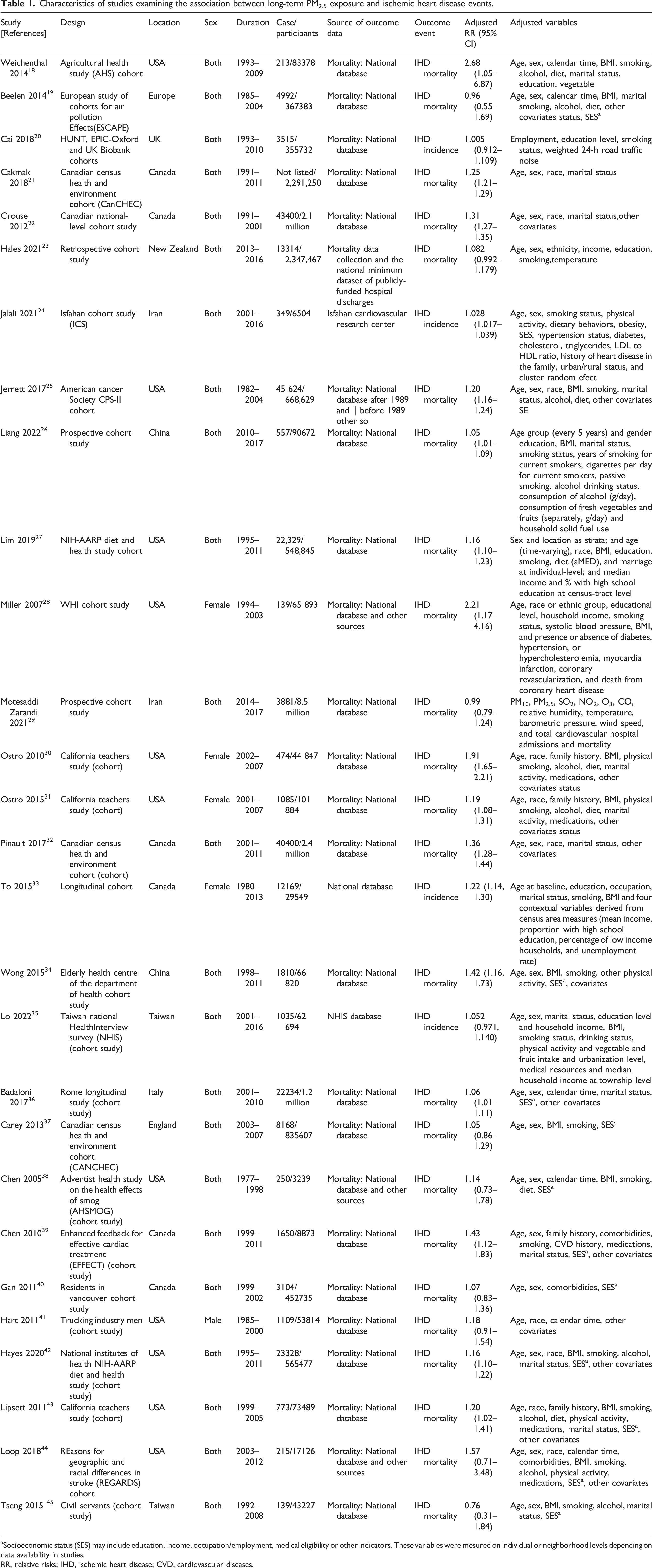
aSocioeconomic status (SES) may include education, income, occupation/employment, medical eligibility or other indicators. These variables were mesured on individual or neighborhood levels depending on data availability in studies.
RR, relative risks; IHD, ischemic heart disease; CVD, cardiovascular diseases.
Quality evaluation of all included studies.
PM2.5 and IHD risk
We identified 28 studies of PM2.5 exposure and IHD risk with study populations restricted to patients without a previous IHD. As shown in Figure 2, the multivariable-adjusted RRs from 28 articles were extracted. A random-effect model was used to calculate the combined RR due to the high heterogeneity (I2 = 94.8%). The total RR was determined to 1.18 (95% CI: 1.12–1.25) per 10 μg/m3 increase in PM2.5 by combining the IHD mortality and incidence. The RR of the IHD mortality in the random effects meta-analysis was 1.21 (95% CI: 1.15–1.128) and the RR of an incident IHD was 1.07 (95% CI: 0.99–1.17). The total RRs of IHD incidence and mortality. IHD, ischemic heart disease; RR, relative risk.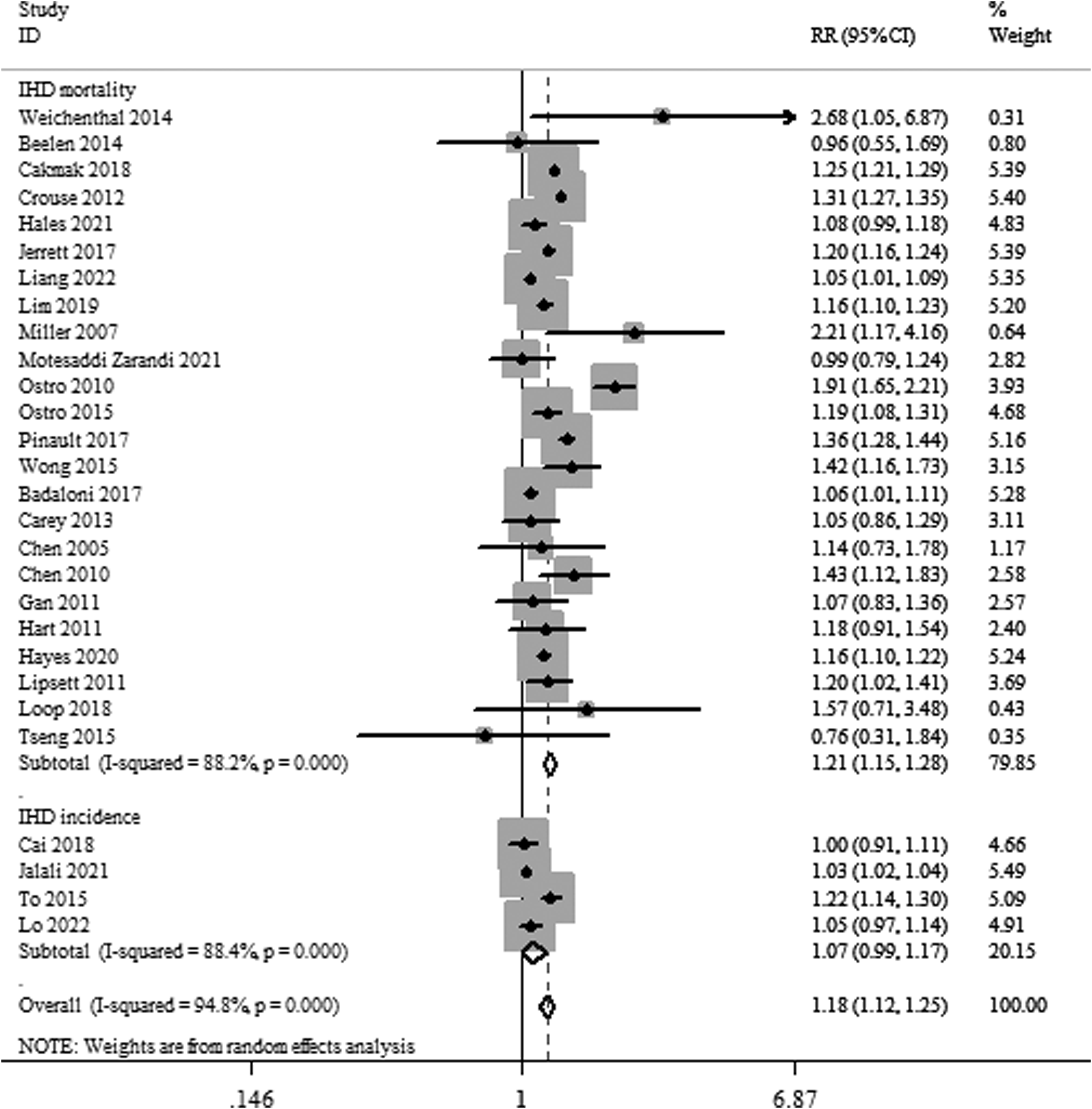
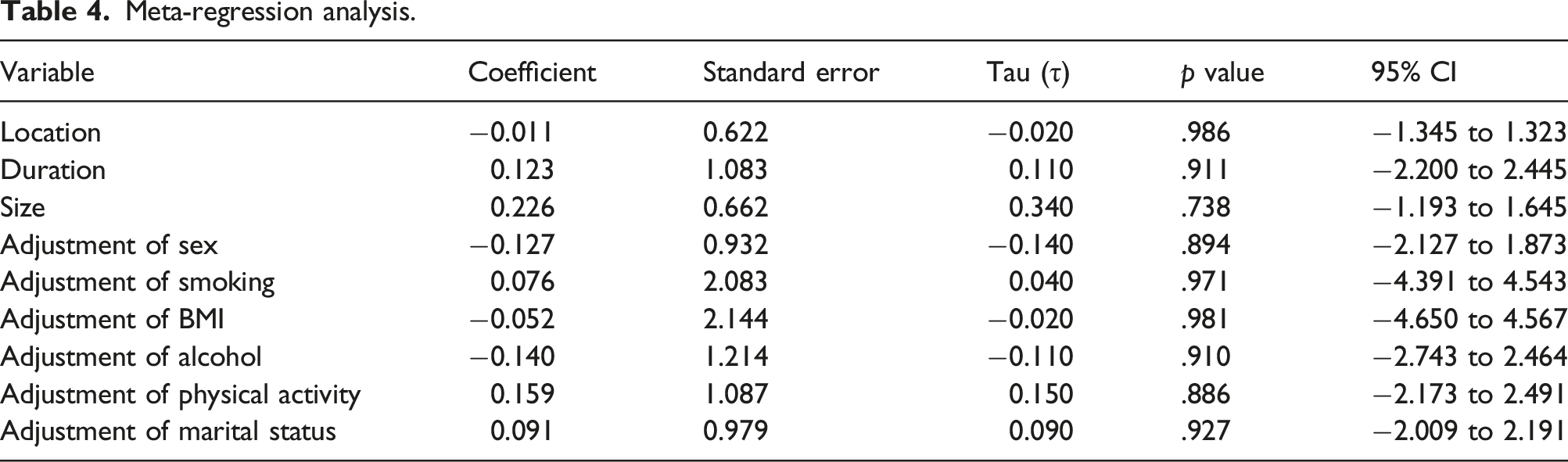
Subgroup and meta-regression analysis
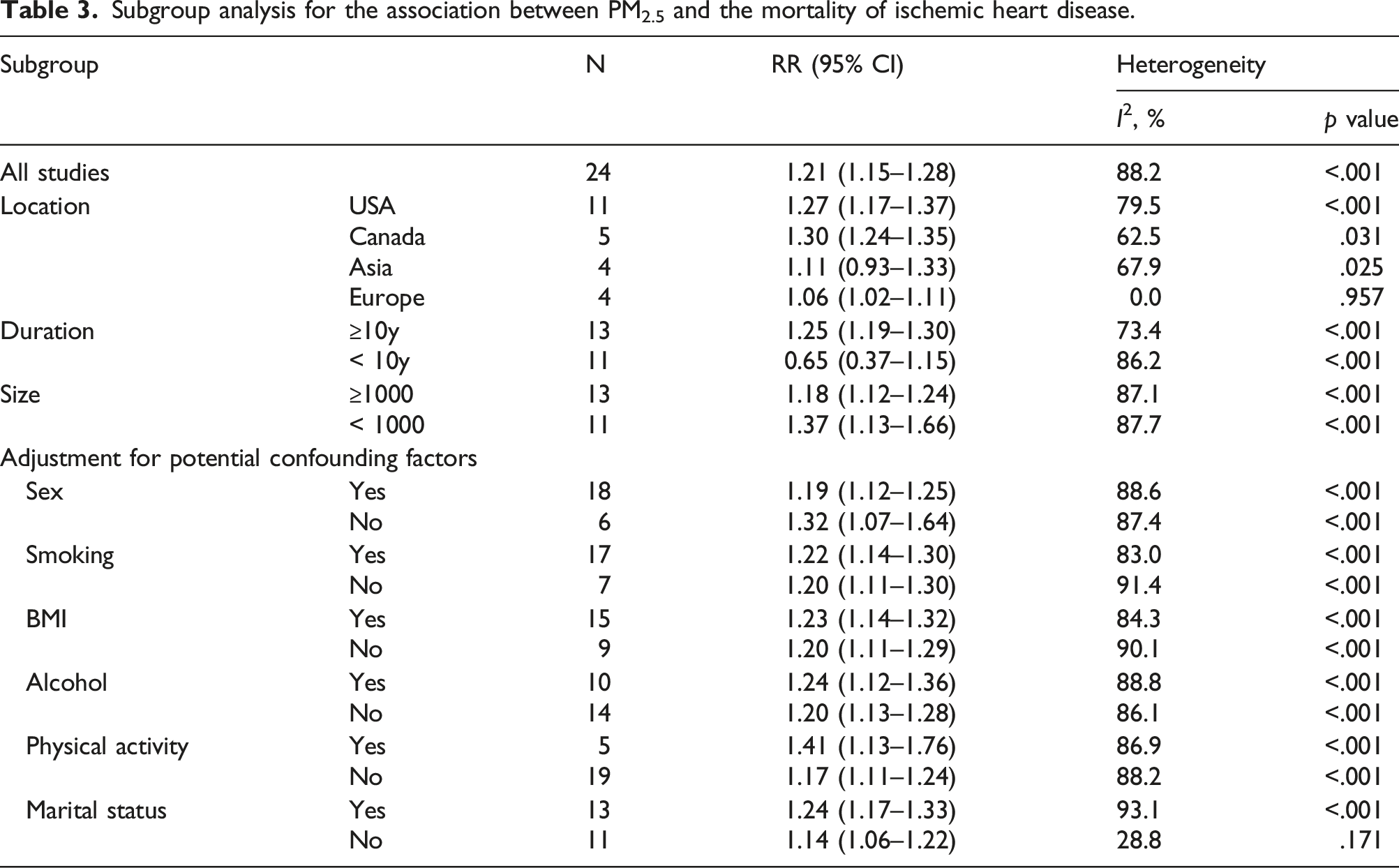
Subgroup analysis for the association between PM2.5 and the mortality of ischemic heart disease.
Meta-regression analysis.
Publication bias and sensitivity analysis
There was no indication of publication bias in the reporting of results on PM2.5 and risk of skin cancer, from either visualization of Begg’s funnel plot (p = .395) or the Egger’s test (p = .395). In sensitivity analysis, each study was sequentially deleted at a time, and the remaining data were re-calculated, which estimated showed that the result was statistically robust.
Discussion
In our research, we reviewed 28 published articles among 23 cohorts regarding the effect of exposure to PM2.5 on IHD incidence and mortality, covering 256256 IHD cases. Our findings indicated that IHD incidence and IHD mortality had obvious increases in risk. In our meta-analysis, the results showed that a 10 μg/m3 increase in PM2.5 exposure was related to a 21% increased risk of IHD mortality and 7% increased risk of IHD incidence. Our results are consistent with data from other meta-analyses. 16 Additionally, we found that the combined RRs of exposure to PM2.5 on IHD mortality in Asian and European population [1.11 (95% CI: 0.93–1.33); 1.06 (95% CI: 1.02–1.11)] were much lower compared with American and Canadian people [1.27 (95% CI: 1.17–1.37); 1.30 (95% CI: 1.24–1.35)]. Furthermore, study duration, size and some adjustments were related with the total RR.
Atmospheric particulate matter (PM) is segmented into four categories based on aerodynamic diameter: total suspended particulate (TSP ≤ 100 μm), particulate matter (≤ 10 μm), fine particulate matter (≤ 2.5 μm) and ultrafine particles (≤ 0.1 μm). Due to the small particle size, light quality and a relatively large specific surface area, PM2.5 are considered as ambient air quality management worldwide based on some known cardiorespiratory health effects. 45 Several potential mechanisms might exit to account for the health effect. First, PM2.5 enters the respiratory system causing systemic inflammation, oxidative stress and reactive oxygen species. A vivo model experiment revealed that PM2.5 exacerbated inflammation, vascular remodeling and right ventricular hypertrophy. 46 PM2.5 exposure induced systemic inflammation, cerebrovascular oxidative injury, and intracranial atherosclerosis in rat models. 47 In numerous vitro studies have showed that exposure to PM2.5 induces apoptosis and oxidative stress in human cells.48,49 Second, PM2.5 has the potential to reduce cell viability, induce oxidative DNA damage and induce global DNA methylation. 50 Extractable organic matters from PM2.5 significantly enhanced 8-OHdG and γH2AX signals and caused DNA damage.51,52 Recent studies have demonstrated that PM2.5 causes cardiovascular pathological damage and functional changes, involving DNA methylation, non-coding RNA, histone modification and chromosome remodeling.53–55 Next, PM2.5 may affect the coagulation system, change autonomic nerve function, injure the vascular endothelium and affect vasomotor function. A recent study has demonstrated that PM2.5 exposure induced more serious inflammation and oxidative stress in the circulation system and promoted a hypercoagulable state by JNK/P53 pathway. 56 PM2.5 enters the human body through various pathways to affect central nervous system (CNS) diseases. PM2.5 activates the microglia in the central nervous system, which can lead to inflammatory and neurological damage. 57 Moreover, exposure to ambient levels of PM2.5 were related to chronic reductions in brachial endothelial function. In a mouse model, exposure to low concentration of PM2.5 altered vasomotor tone, induced vascular inflammation and potentiated atherosclerosis. 58 PM2.5 causes other reaction through entering into the circulatory system via the digestive tract. A potential mechanism was found that PM2.5 was associated with increase serum levels of hormones, PM2.5 exposure may affect the HPA axis through the gastrointestinal tract microbiota pathway (GBA mechanism). 59 Additionally, clinical studies have shown that exposure to PM2.5 can increase insulin resistance and blood pressure. Diabetes and hypertension resulted in higher magnitude associations between PM2.5 and CVD mortality.60,61
Some previous studies have synthesized the overall evidence of IHD and PM2.5 in the past decade. The data from the Global Burden of Disease Study (GBD) in 2017 had revealed that IHD attributable to PM2.5 resulted in big burdens, especially in Asia, Oceania and sub-Saharan Africa. 7 While there have been two meta-analyses of long-term PM2.5 on the risks of IHD mortality. Results from the first meta-analysis of PM2.5 and incident AMI and IHD mortality, which showed a 23% increased risk of IHD mortality per 10 μg/m3 increase in long-term average PM2.5 exposure. 16 Another meta-analysis found that acute effect of PM2.5 on IHD mortality was less than the chronic effect, and the combined effect values were much lower in Asian population. 62 We observed an obvious difference in the risk of IHD mortality in Asian and European population (11%, 6% increased risk, respectively) compared with American and Canadian people (27%, 30% increased risk, respectively). Furthermore, study duration, size and some adjustments were related with the total RR. There are some reasonable explanations. Different regions have different demographics, and the compositions of atmospheric particulate matter are diverse from each other. In addition, the concentration of particulate matter in the air in Asian countries is general higher than in Europe and the USA, so the expose-reaction relationship curve tends to be flat at high concentration. In sensitivity analysis, we found the combined RRs was robust. Considering that the positive results are often easier to publish than negative results, therefore, there is still a certain publication bias in analyzing the effect of PM2.5 on IHD mortality. In addition, there was a high heterogeneity in the literature included in this study, which may be caused by different assessment methods of exposure to PM2.5, population susceptibility and adjustment methods of confounding factors in different studies.
In our meta-analysis, there is several shortcomings. Primarily, other air pollutants such as sulfur dioxide (SO2), nitrogen dioxide (NO2), carbon monoxide (CO) and ozone (O3) may also contribute to IHD death.63,64 However, only the effect estimates of the single pollutant model were included, and the potential joint effect and collinearity between multiple pollutants were not considered. Second, high heterogeneity exists in the including literatures, which may be caused by the different exposure levels and different measurement methods, and the differences in susceptibility of populations in different regions. Moreover, we identified characteristics of 28 studies among 23 cohorts. Results from different study designs should be expected to differ systematically, resulting in increased heterogeneity. Therefore, we excluded the randomized controlled trials (RCTs). In addition, studies on PM2.5 contribution to IHD deaths are still insufficient, thus further evidence will be obtained from well-designed and large-scale prospective studies.
Conclusions
In summary, the current systematic review and meta-analysis demonstrated that exposure of PM2.5 may increase the risk of IHD incidence and mortality. Given to the limited number of included studies and the potential biases in the present study, the result is still uncertain. Hence, high-quality prospective cohort studies and double-blinded randomized-controlled trials were supposed to investigate the influence of PM2.5 on IHD. In addition, it’s necessary that more accurate evaluation of PM2.5 exposure was moved forward in time.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
