Abstract
Introduction
Although venovenous extracorporeal membrane oxygenation (VV ECMO) is a reasonable salvage treatment for acute respiratory distress syndrome (ARDS), it requires sedating the patient. Sevoflurane and propofol have pulmonary protective and immunomodulatory properties. This study aimed to compare the effectiveness of sevoflurane and propofol on rats with induced ARDS undergoing VV ECMO.
Methods
Fifteen sprague-dawley (SD) rats were randomly divided into three groups: Con group, sevoflurane (Sevo) group and propofol (Pro) group. Arterial blood gas tests were performed at time pointsT0 (baseline), T1 (the time to ARDS), and T2 (weaning from ECMO). Oxygenation index (PaO2/FiO2) was calculated, and lung edema assessed by determining the lung wet:dry ratio. The protein concentration in bronchial alveolar lavage fluid (BALF) was determined by using bicinchoninic acid assay. Haematoxylin and eosin staining was used to evaluate the lung pathological scores in each group. IL-1β and TNF-α were also measured in the BALF, serum and lung.
Results
Oxygenation index showed improvement in the Sevo group versus Pro group. The wet:dry ratio was reduced in the Sevo group compared with propofol-treated rats. Lung pathological scores were substantially lower in the Sevo group versus the Pro group. Protein concentrations in the BALF and levels of IL-1β and TNF-α in the Sevo group were substantially lower versus Pro group.
Conclusion
This study demonstrates that compared with propofol, sevoflurane was more efficacious in improving oxygenation and decreasing inflammatory response in rat models with ARDS subject to VV ECMO treatment.
Keywords
Introduction
Acute respiratory distress syndrome (ARDS), a common condition encountered in the intensive care unit, is characterised by an acute inflammatory response in the pulmonary parenchyma that results in alveolar edema and reduced lung compliance. 1 Venovenous extracorporeal membrane oxygenation (VV ECMO) is a salvage therapy used in patients with ARDS for lung protective mechanical ventilation and to provide adequate time for treating the cause of ARDS, facilitating lung healing. 2 According to the Extracorporeal Life Support Organization (ELSO), the patient should be thoroughly sedated with light anaesthesia during the first 12–24 h of cannulation and management. 3 Respiratory paralysis is seldom required and is only induced during venous cannula placement to avoid spontaneous breathing. 4 Post induction of respiratory paralysis, however, the sedation and analgesia requirements of patients with ARDS receiving VV ECMO often increase simultaneously.3, 4
Sevoflurane and propofol are commonly used general anaesthetics in clinical practice. Propofol is widely used for sedation in mechanically ventilated adult patients. 5 The use of inhaled anaesthetics such as sevoflurane has also, in recent times, extended from the operating room to long-term sedation of critically ill patients. 6 Furthermore, increasing evidence has shown that sevoflurane and propofol have anti-inflammatory effects.7-10 ARDS refers to an acute inflammatory response associated with diffuse lung infiltrates resulting in a systemic inflammatory response syndrome (SIRS). 11 Apart from lung disease, VV ECMO is also initiated in instances of rapid and complex inflammatory response, similar to that seen in SIRS. A variety of coagulative and inflammatory reactions cascade when blood first enters the extracorporeal circuit. 12 However, the effect of these anaesthetic agents on the inflammatory response in patients with ARDS during VV ECMO has not yet been studied.
Considering the pharmacokinetic alterations caused by the VV ECMO circuit, 13 investigating the effects of sevoflurane and propofol on inflammatory responses in patients with ARDS subject to VV ECMO are warranted. This study aimed to evaluate the effect of propofol and sevoflurane on the inflammatory response during VV ECMO. The findings of this pre-clinical study provide a valuable theoretical basis for clinical application.
Methods
The protocols were reviewed and approved by the institutional animal experimental ethics committee of Lanzhou University Second Hospital (Lanzhou, Gansu, China). The investigation was in accordance with the recommendations of the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Institutes of Health publication No. 85–23, revised 1996).
Animals
Fifteen Sprague-Dawley (SD) rats (male, aged 10–12 weeks, 300–400 g body weight) were used in this study. All animals were housed in a temperature- and humidity-controlled room on a 12-h light/dark cycle with free access to food and water.
Experiment protocols
Animals were randomly assigned to the different experimental groups: Con group (n = 5), Sevoflurane (Sevo) group (n = 5), and Propofol (Pro) group (n = 5). Rats were first anaesthetised with 1% pentobarbital sodium 50 mg/kg intraperitoneally, followed by fixation on the operating table in the supine position. Endotracheal intubation with a 14G needle was performed under direct vision, and animals were connected to a rodent ventilator (Shanghai Alcott Biotech Co. Ltd, China). The ventilator parameters were set as follows: tidal volume, 6 mL/kg; respiratory rate, 70-75 breaths/min; positive end-expiratory pressure, two cmH2O; and inhalation:exhalation ratio, 1:2. Inhalation oxygen concentration was maintained at 40%. Body temperature of the rats was maintained at 36 ± 0.5°C by using a heating lamp and was monitored by rectal thermometry. The right femoral artery and vein were exposed and free under local anaesthesia with 1% lidocaine. A 24 G intravenous indwelling needle was punctured into the femoral artery and a monitor was connected for continuous haemodynamic monitoring.
Although the ARDS rat model of VV ECMO has been well explained in our previous studies,
14
a brief description of the methodology is described here: Post the above procedure, ARDS was induced by intravenous injection of 100 mg/kg oleic acid (OA) (99%, Sigma-Aldrich, USA). One hour after the injection when arterial blood gas indicated partial pressure oxygen by fractional inspired oxygen ratio (PaO2/FiO2) < 300, VV ECMO was established at a flow rate of 80–90 mL/kg/minutes. Inspired oxygen concentration was maintained at >90% After initiating VV ECMO, Sevo group rats were made to inhale 1.5% sevoflurane (Maruishi Pharmaceutical Co. Ltd, Japan), whereas Pro group rats were subject to intravenous infusion with 1% propofol 10 mg/kg/h (Corden Pharma S.P.A., USA). Other respiratory parameters remained unchanged. The flow diagram of the experiment is summarized in Figure 1. Flow diagram showing the experimental protocol. Created with BioRender (https://biorender.com/).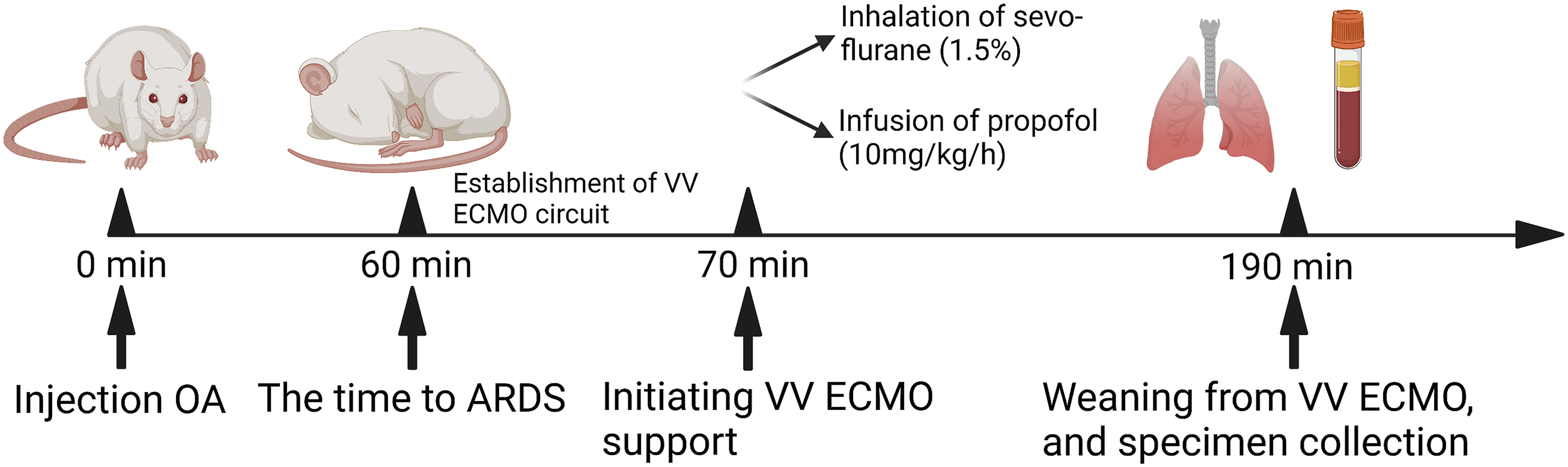
Arterial blood gas analysis and collection of bronchoalveolar lavage fluid
Blood was collected from the femoral artery and arterial bloody gas analysis was performed at the following three time points: T0 (baseline), T1 (the time to ARDS initiation), and T2 (weaning from ECMO). At the end of the experiment, the left hilum was clipped and extraction of bronchoalveolar lavage fluid (BALF) was performed with 2.0 mL pre-cooled normal saline (0.9%). The lavage was repeated thrice and was considered successful if the recovery rate was above 80%. The total protein content in the BALF was measured using bicinchoninic acid assay (BCA) Protein Quantitation Kit (Solarbio, China) according to the manufacturer’s protocol.
Lung wet:dry ratio
Rats were sacrificed when the experimental period was complete. The left lung was isolated and weighed immediately (wet weight) after the lung surface liquid was absorbed using an absorbent paper. Dry weight was measured by placing lungs in an oven at 60°C for 72 h until a constant weight was observed. The lung wet:dry ratio was then calculated.
Haematoxylin-eosin staining
The middle lobe of the left lung was fixed with 4% paraformaldehyde. After fixation and dehydration, the specimens were embedded in paraffin and cut into 5 μm-thick sections. Sections were stained with haematoxylin and eosin in accordance with routine procedures of haematoxylin-eosin (H&E) staining, followed by observation of the histopathological changes in the lung specimen sections under a light microscope (Olympus, Japan). The extent of lung tissue damage was observed and scored in the sections using the double-blind method. Scores were determined based on the following four parameters: congestion/edema in the alveolar cavity; lung tissue haemorrhage and thickening; inflammatory cell infiltration in the alveolar cavity and blood vessel cavity; and alveolar wall congestion, edema, thickening, and hyaline membrane form. 14 The scores of these four aspects were added to form a visual field lung injury score: 0 points for no pathological changes, one point for mild pathological changes, two for moderate changes, three for severe changes, and four for very severe changes. The 200 alveoli of lung tissue samples were examined at 400X magnification to determine the lung injury score.
Immunohistochemistry
Sections were dehydrated in ovens at 60°C for 60 min. After dewaxing and rehydrating, antigen retrieval was initiated with 10 mM citrate buffer (ZSGB-BIO, China) for 5 min. Sections were incubated with the rabbit anti-rat Myeloperoxidase (1:2000 dilution, Abcam, USA) primary antibody overnight at 4°C. On the next day, the sections were incubated with the biotinylated sheep anti-rabbit IgG secondary antibody at room temperature for 20 min, followed by using horseradish peroxidase (ZSGB-BIO, China). Sections were sealed with neutral resin and analysed under a light microscope (Olympus, Japan). Myeloperoxidase-positive nuclei were counted in three randomly selected areas per section from each individual rat, and the percentage of Myeloperoxidase-positive nuclei was calculated for each area.
Enzyme-linked immunosorbent assay
BALF and serum and lung specimens were used to evaluate the immunomodulatory function of the sedatives. Cervical venous blood was collected and centrifuged at 3500 rpm/min for 15 min at 4°C. The supernatant was then collected and stored at −80°C until use. Lung tissues were homogenised and the supernatant was collected for analysis by enzyme-linked immunosorbent assay (ELISA). The levels of TNF-α and IL-1β in BALF, serum and lung were measured using the ELISA kit (Shanghai Enzyme-linked Biotechnology Co. Ltd, China) according to the manufacturer’s instructions.
Statistical analysis
All statistical analyses were performed using SPSS statistical software 22.0 (SPSS Inc, Chicago, IL, USA) and graphs were presented using GraphPad Prism 7.0 (GraphPad, USA). Measurement data were expressed as mean ± SD. The normality of distribution was studied using the Shapiro-Wilk test. Data from perioperative physiological parameters were analysed using repeated measurements of analysis of variance (ANOVA). Data between the two groups were analysed using the unpaired Student’s t-test for normally distributed data or the non-parametric Mann-Whitney U-test for non-normally distributed data. For comparisons across the three groups, ANOVA was used followed by Tukey’s multiple comparisons test. p-values of <0.05 (2-tailed) were considered statistically significant.
Results
Physiologic parameters
Physiological parameters.
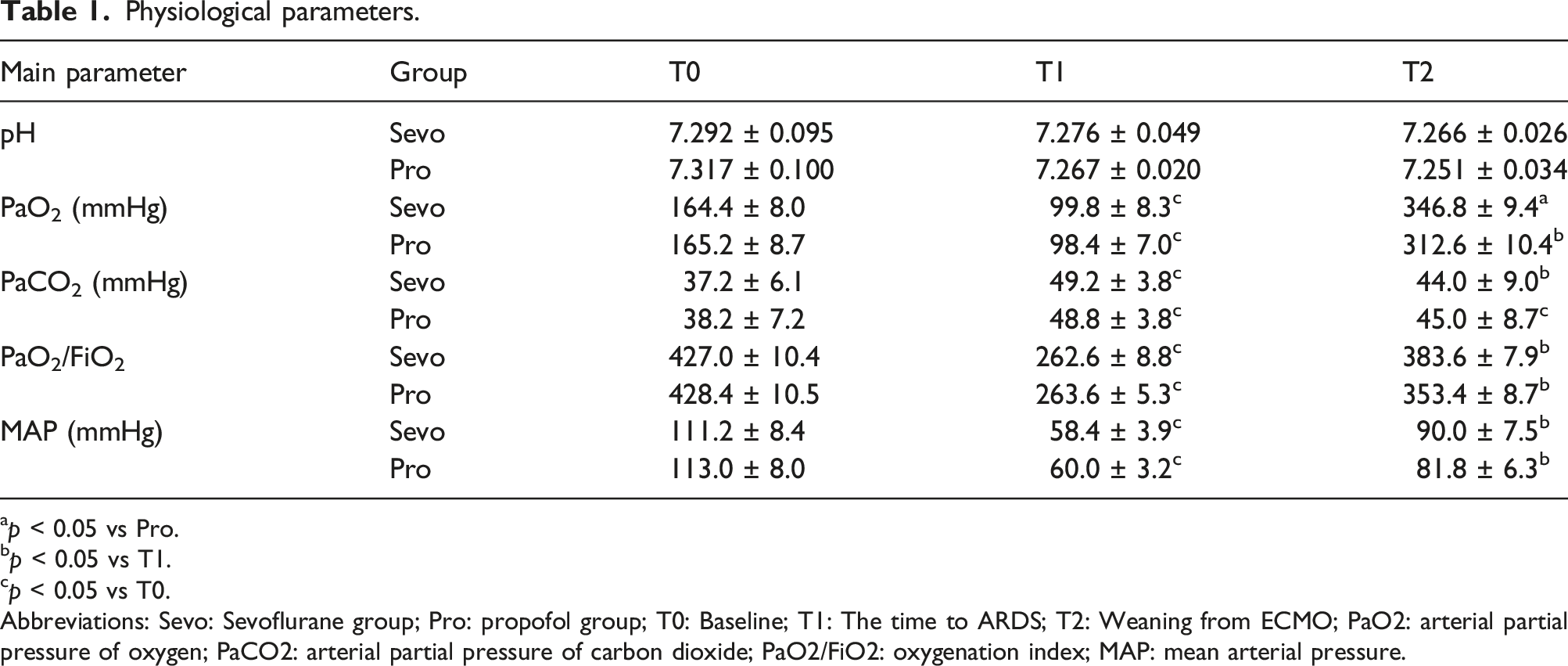
ap < 0.05 vs Pro.
bp < 0.05 vs T1.
cp < 0.05 vs T0.
Abbreviations: Sevo: Sevoflurane group; Pro: propofol group; T0: Baseline; T1: The time to ARDS; T2: Weaning from ECMO; PaO2: arterial partial pressure of oxygen; PaCO2: arterial partial pressure of carbon dioxide; PaO2/FiO2: oxygenation index; MAP: mean arterial pressure.
Histopathology
Hematoxylin-eosin staining revealed the extent of pathological changes in the lung tissues of the two groups. The pathological changes observed in the lung tissue sections included mild interstitial hyperplasia, moderate vascular congestion, and moderate inflammatory cell infiltration in the Pro group (Figure 2(a)). The extent of these pathological changes was reduced in the sections receiving sevoflurane treatment (Figure 2(b)). Quantification indicated that sevoflurane was more effective than propofol in reducing the lung injury score (Figure 2(c)). Pathological evaluation of lung tissues. Representative images of lung sections stained with haematoxylin-eosin (H&E); the alveolar exudation was marked by a red star and blood cell was marked by a black arrow. Scale bars: 200X, 50 μm; 400X, 20 μm (a, b). The lung injury score of each group was graded from 0 (no injury) to 4 (maximum injury) (c).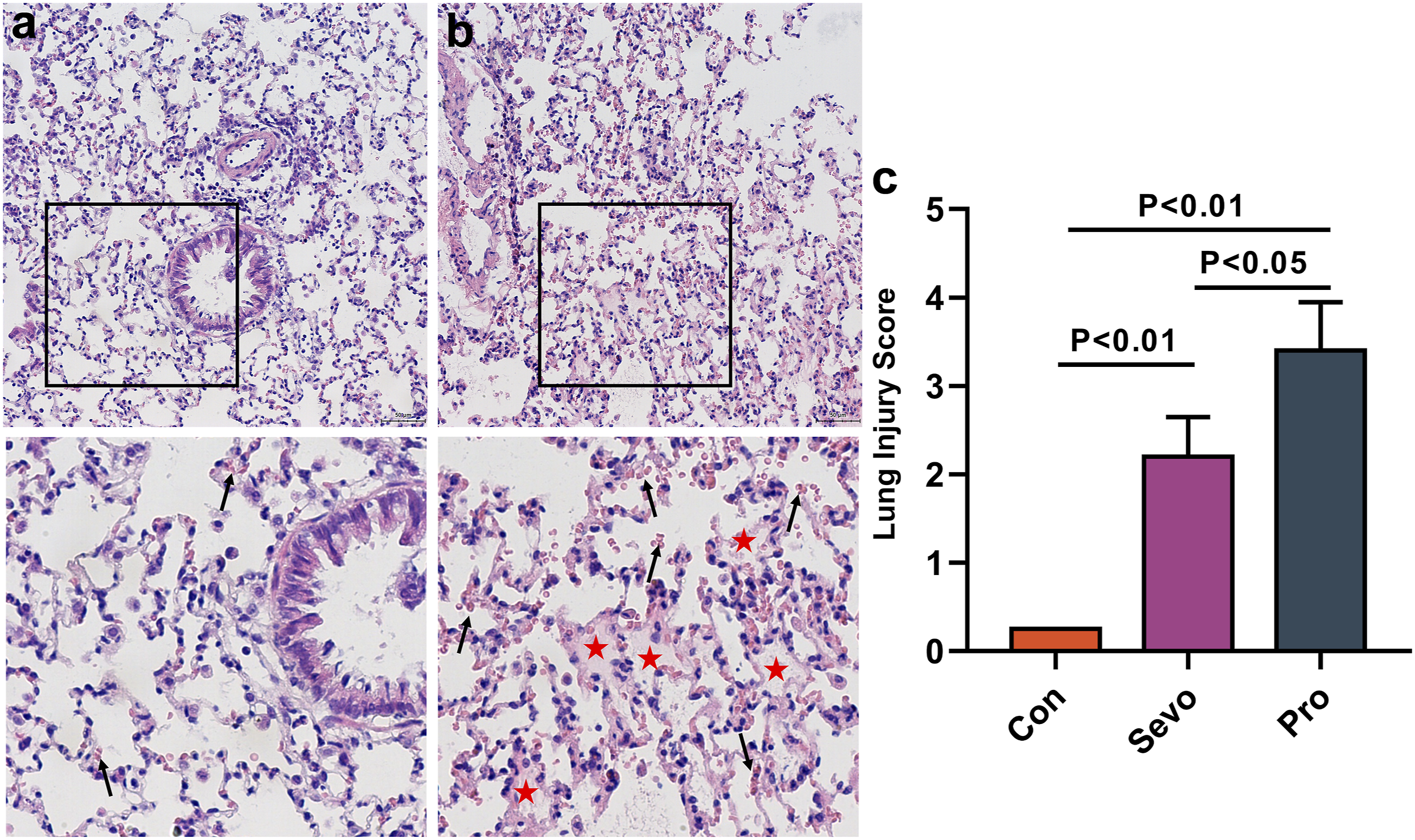
Pulmonary edema and bronchoalveolar lavage fluid
The wet:dry ratio of the Pro group was significantly increased compared with the Sevo group (Figure 3(a)). Considering the protein content in BALF (Figure 3(b)), the Sevo group showed significant reduction in pulmonary capillary alveolar leakage compared to the Pro group. The water content in lung tissue was determined using the wet:dry ratio (a). The alveolar capillary barrier was determined by measuring the total protein content in BALF (b).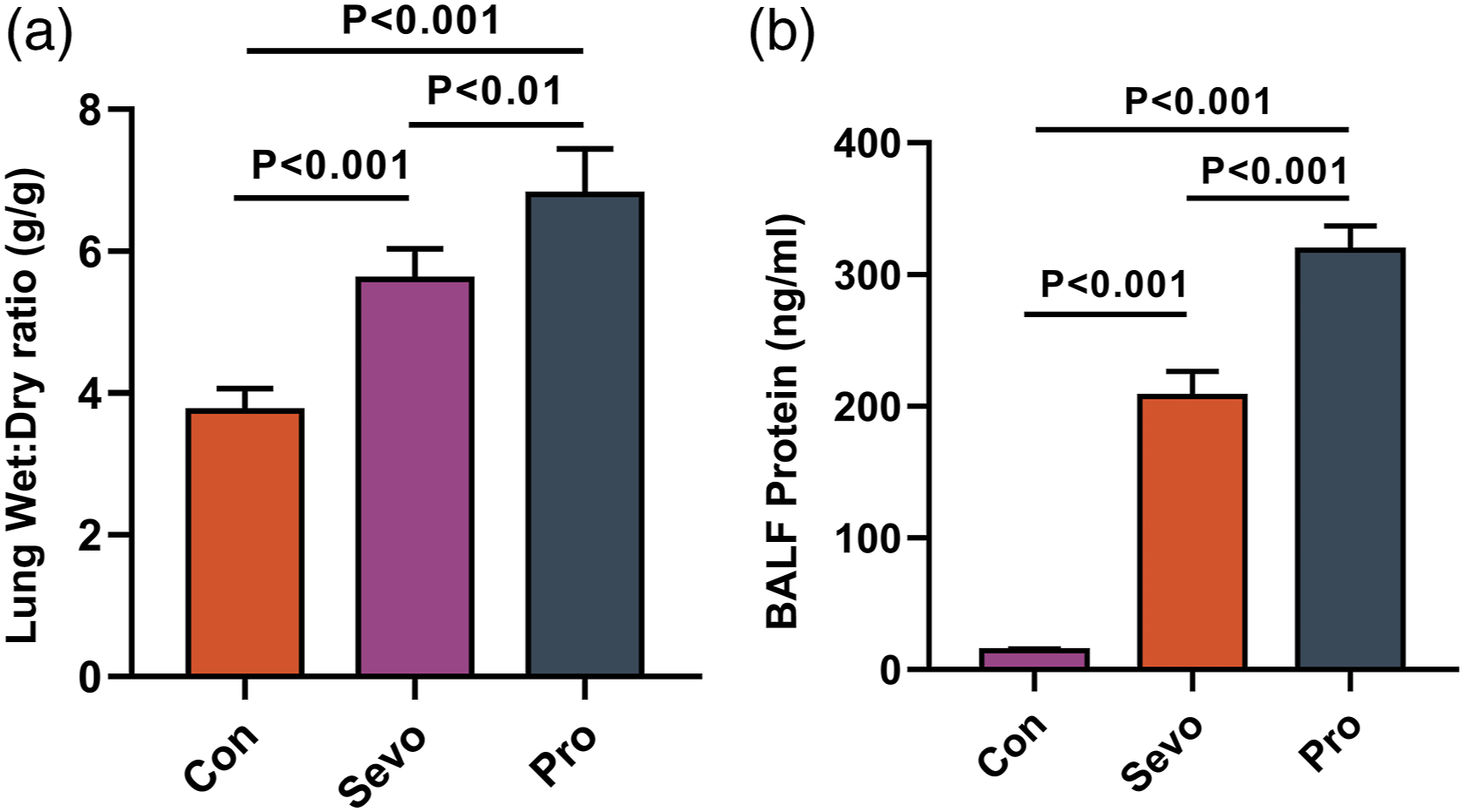
Neutrophil infiltration and inflammatory factor
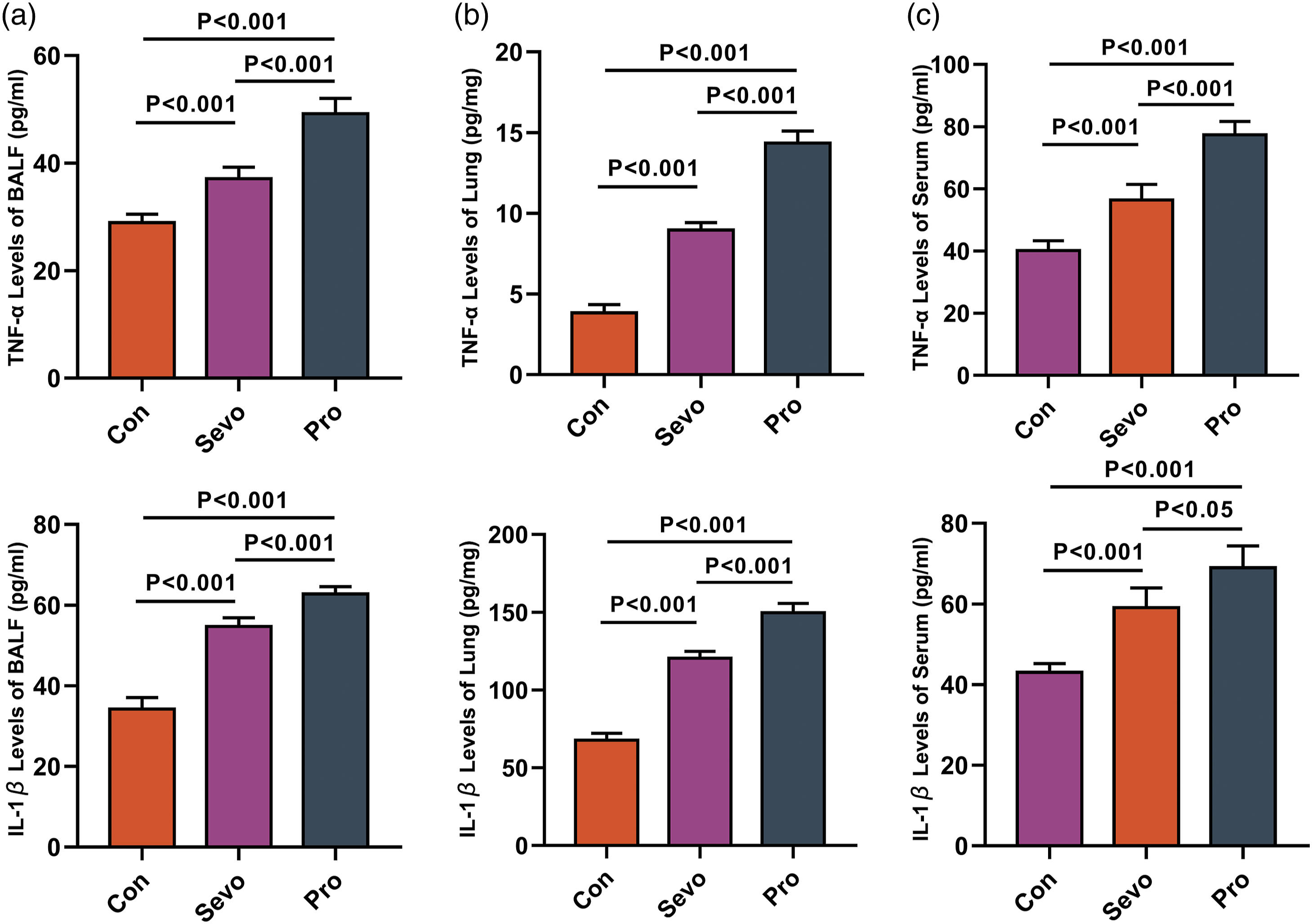
Myeloperoxidase staining in lung tissues of the Sevo and Pro groups are shown in Figures 4(a) and (b). Neutrophil numbers were significantly lower in the Sevo group compared to the Pro group (Figure 4(c)). The BALF (Figure 5(a)), lung (Figure 5(b)) and serum (Figure 5(c)) levels of TNF-α and IL-1β in the Pro group were higher than in the Sevo group. Myeloperoxidase staining was used to detect neutrophil infiltration in lung tissue. Immunohistochemistry staining was used to detect Myeloperoxidase, a marker of neutrophil in lung tissue. Scale bars: 200X, 50 μm; 400X, 20 μm (a, b). Three fields were randomly selected to quantitatively analyse the activity of Myeloperoxidase (c). BALF, lung, and serum levels of TNF-α and IL-1β in each group of rats. The levels of IL-1β and TNF-α were decreased in BALF, lung and serum of Sevo group rats compared to the Pro group rats.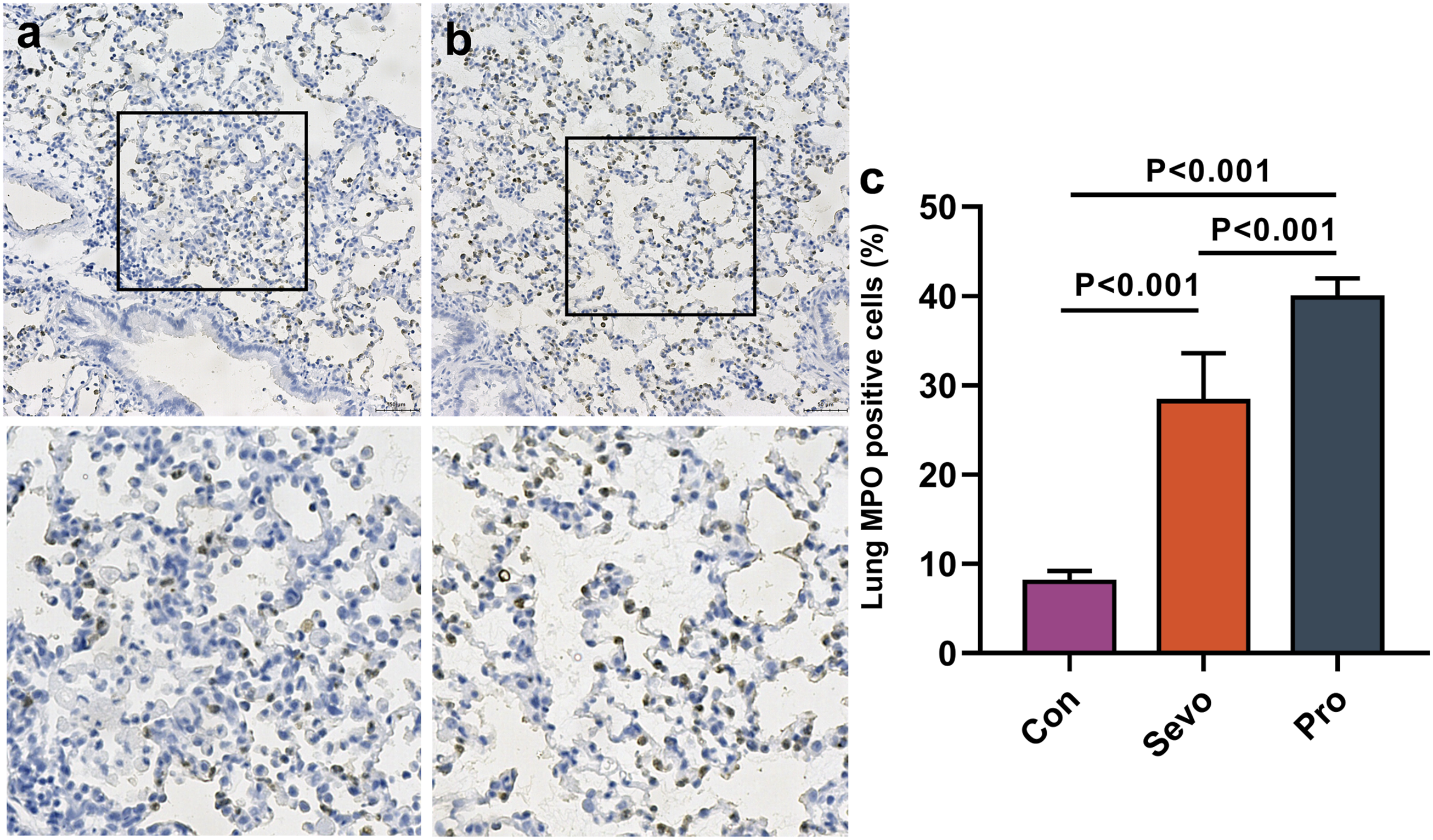
Discussion
This study demonstrated that sevoflurane inhibits lung injury and inflammatory response under VV ECMO conditions. Sevoflurane not only alleviated lung damage but also alleviated pulmonary and systemic inflammation compared with propofol.
In patients with ARDS subject to VV ECMO therapy, sedation is used to improve tolerance of mechanical ventilation, reduce discomfort, and improve patient-ventilator synchrony. 15 Inadequate sedation can cause agitation, accidental extubation or haemodynamic instability. 4 Most sedatives and analgesics, such as propofol, midazolam, fentanyl, etc., are given intravenously. The 2018 Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption in Adult Patients in the ICU (PADIS guideline) also recommended the preference of propofol over benzodiazepines. 16 For the majority of clinical practitioners, these concerns remain theoretical because retrospective and prospective analyses found that use of propofol in patients on ECMO was safe and was not an independent risk factor for oxygenator failure or circuit changes.17-19 It is noteworthy that an in vitro assessment of pharmacokinetic changes in patients on ECMO showed that a significant extent of propofol was lost in the ECMO circuitry and likely related to adsorption to polyvinyl chloride tubing resulting from propofol’s lipophilicity. 20
The ideal sedative would have a rapid onset and offset of action and allow for precise titration to facilitate sedation without accumulation after long-term use. 21 Currently, intravenous sedatives do not cumulatively meet these criteria. As an alternative, volatile anaesthetics (VAs) such as sevoflurane have been introduced as sedatives in the ICU in Europe and Canada, and some countries list them as alternative sedatives in the sedation guidelines. In some studies, sedation with sevoflurane has demonstrated a reduction in post-extubation agitation, delusion, negative feelings and factual memory versus with midazolam or propofol.21, 22 Similarly, a study using the MIRUS™ system demonstrated successful application of inhaled sedation for ECMO therapy in patients with COVID-19-related ARDS. 23
This study found that sevoflurane could improve oxygenation by the following three mechanisms: First, although several studies have investigated the effect of sevoflurane on the intrapulmonary shunt fraction, they have been focused on the potential inhibition of hypoxic pulmonary vasoconstriction by this volatile agent during one-lung ventilation. 24 More importantly, sevoflurane has been predicted to inhibit hypoxic pulmonary vasoconstriction via vascular smooth muscle relaxation and also decrease ventilation-perfusion mismatch by facilitating a more uniformly distributed ventilation. Secondly, sevoflurane inhalation has been shown to protect the lungs from injury by regulating caspase-3 activation and Bcl-xL and Bcl-2 expression, thereby inhibiting excessive epithelial cell apoptosis. 25 Thirdly, in the endotoxin-induced lung injury model, Schläpfer et al. showed that sevoflurane affects formation of lung edema. 26 Overall, during VV ECMO, sevoflurane has been shown to induce notable improvements in the airway and tissue mechanics that is compromised by ARDS.
During the inflammatory process in ARDS, the microvascular barrier of alveolar tissue in the lungs is destroyed, resulting in release of inflammatory mediators by pulmonary endothelial cells, macrophages, and neutrophils. 27 During this process, the microvascular barrier of alveolar tissue is destroyed and inflammatory cells from the blood vessels accumulate in the lung tissue. It is challenging to discern the extent of this inflammatory response that is caused solely by VV ECMO or by critical illness, underlying disease or therapeutic interventions such as complex surgical procedures and mechanical ventilation. An inflammatory response like SIRS might be triggered upon blood exposure to the extracorporeal circuit during VV ECMO. 28 VV ECMO can induce an inflammation associated with morbidity and mortality. Therefore, it can offer therapeutic benefit by limiting determinants of inflammation and thereby facilitate lung support.
Studies show that anaesthetic agents are associated with impaired neutrophil and monocyte function and decreased lymphocyte proliferation; findings related to release of inflammatory biomarkers have been variable. 29 Several studies have confirmed that use of anaesthetics could reduce lung inflammation in patients with ARDS.29-31 Propofol, an intravenous agent, has been shown to inhibit neutrophil chemotaxis, neutrophil oxidative response, macrophage phagocytosis and bacterial clearance. Studies have revealed that the protective effect of propofol on lung tissue is associated with oxidative stress and inflammatory response. 32 On the other hand, sevoflurane, a volatile anaesthetic, affects innate immunity by primarily inhibiting neutrophil function, decreasing lymphocyte proliferation, suppressing cytokine release from peripheral blood mononuclear cells and circulating monocytes, and affecting the function of natural killer and dendritic cells. 33 Sevoflurane is a commonly used inhaled anaesthetic whose anti-inflammatory properties have been demonstrated in lipopolysaccharide-induced lung injury. 34 Sevoflurane showed lower pro-inflammatory mediator levels along with fewer postoperative complications (mostly pulmonary) compared with propofol used in one-lung ventilation surgeries. Sevoflurane exposure attenuated production of proinflammatory mediators in BALF and was associated with reduced lung injury and better oxygenation versus propofol. 35 In a ventilator-induced lung injury model, sevoflurane and isoflurane showed better attenuated neutrophil recruitment and activation versus ketamine and desflurane. 21 In general, our results showed a clear inflammatory response to both propofol and sevoflurane during VV ECMO.
However, this study had the following limitations: (1) We used OA-induced lung damage, which is different from the human ARDS inducement and symptoms. (2) To avoid oestrogen effects, only male rats were used in our study. The results for female rats and their implications are yet to be determined. (3) Finally, VV ECMO support was only used for 3 h to avoid pump-related blood damage.
Conclusions
In conclusion, we found that early use of inhaled sevoflurane was associated with improved oxygenation and a reduction of proinflammatory markers and lung injury in patients with ARDS treated using VV ECMO. These findings may encourage the design of future prospective clinical trials to investigate the impact of inhaled sevoflurane on patient outcome during ARDS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Talent Introduction Plan of the Lanzhou University Second Hospital (No. YJRCKYQDJ-2021-02); the Natural Sciences Foundation of Gansu (No. 20JR10RA760, 20JR10RA745, 20JR10RA733); the Scientific Research Projects of Colleges in Gansu Province (No. 2020B-028, 2020B-049); the Cuiying Scientific and Technological Innovation Program of Lanzhou University Second Hospital (No. CY2019-QN01, CY2019-BJ07); the Scientific Research Projects in Lanzhou (No. 2021-1–107).
