Abstract
Introduction
Lactate levels have been recognized as a reliable tool for monitoring critically ill patients requiring venoarterial extracorporeal membrane oxygenation (VA ECMO) or venovenous extracorporeal membrane oxygenation (VV ECMO) but the reasons behind the overproduction of lactate are different and the influance for survival remains controversial. We analyzed the lactate values and lactate clearance in adult patients in these two forms of extracorporeal support.
Methods
Patient demographics, ECMO duration, 30-day mortality, lactate values and lactate clearance at 24, 48 and 72 h from ECMO initiation of patients supported with VV and VA ECMO at Silesian Centre for Heart Deasese, between January 2011 and April 2020 were retrospectively analyzed. The changes in lactate levels were analyzed using the non-parametric U Mann-Whitney tests and Chi-square test. The ROC curves were draw and the area under the curve was calculated.
Results
The study comprised 91 adult patients, Mortality in the first 30 days from initiation of VV and VA ECMO was 39% and 66%, respectively. Lactate levels were significantly higher in non-survivors that received VV and VA ECMO (
Conclusion
This results emphasized the importance of lactate levels below 2.0 mmol/L at 72 h from both VV and VA ECMO initiation.
Keywords
Introduction
Absolute values of lactate and serum lactate kinetics have been described as prognostic factors in several critical conditions.1–4 Increased lactate concentrations are associated with an increased risk of death in infection, 5 sepsis, 6 trauma, 7 and in surgical patients. 8 Discordance between oxygen supply and demand causes an increase in lactate levels, which reflects the imbalance between production and clearance of lactate in critically ill patients with ARDS 9 and after cardiac surgery.10–12 Venoarterial extracorporeal membrane oxygenation (VA ECMO) is used to provide temporary mechanical circulatory support and venovenous extracorporeal membrane oxygenation (VV ECMO) is used to provide temporary pulmonary support in severe respiratory and circulatory system failure in patients not responding to conventional methods of treatment.13,14
The indications for VV and VA ECMO and the production of lactate in each condition is expected to be quite different.13,14 In patients in circulatory failure, lactate production is due to systemic hypoperfusion. The use of VA ECMO in this population is to restore systemic perfusion, and continuing lactate production would be anticipated to be indicative of inadequate circulatory support and ongoing malperfusion. 10 In the setting of respiratory failure, lactate production is most likely due to systemic malperfusion due to septic shock or, possibly, from profound hypoxia or circulatory failure due to right chambers (RV) dysfunction in the setting of ARDS.9,14
Data about lactate dynamics and its prognostic value in terms of survival in patients supported with VV and VA ECMO is scarce and with results that are inconsistent.15–17 Based on the discrepancies, we attempted to determine the value of lactate levels, as associated with mortality, in the first 72 h after admission of patients with heart failure treated with VA ECMO and in patients with ARDS supported by VV ECMO. In addition, the effects of exogenous catecholamines and vasoconstrictive agents on lactate levels as exponents of circulatory failure in patients treated with VA ECMO, and systemic malperfusion due to septic shock and RV failure induced by ARDS in patients treated with VV ECMO were evaluated.
Material and methods
This study was performed in the Silesian Centre for Heart Diseases in Zabrze, Poland. The study protocol was approved by the Ethics Committee at the Silesian Medical University in Katowice, Poland (KNW/0023/KB1/65/18). The need for informed consent was waived owing to the retrospective study design. We retrospectively analyzed our ICU database in search of all consecutive patients requiring ECMO support between 1 January 2011 and 30 April 2020. Thirty-day mortality, calculated from the time of ECMO initiation, was been chosen as the primary outcome measure. All calculations were performed separately for patients that received VV versus VA ECMO. The primary endpoint of our study was to compare lactate levels during the first 72 h following ECMO initiation among survivors and non-survivors. The secondary endpoint was to assess the prognostic value of lactate kinetics following ECMO initiation in the prediction of ICU death.
Clinical characteristics of patients treated with venovenous extracorporeal membrane oxygenation.
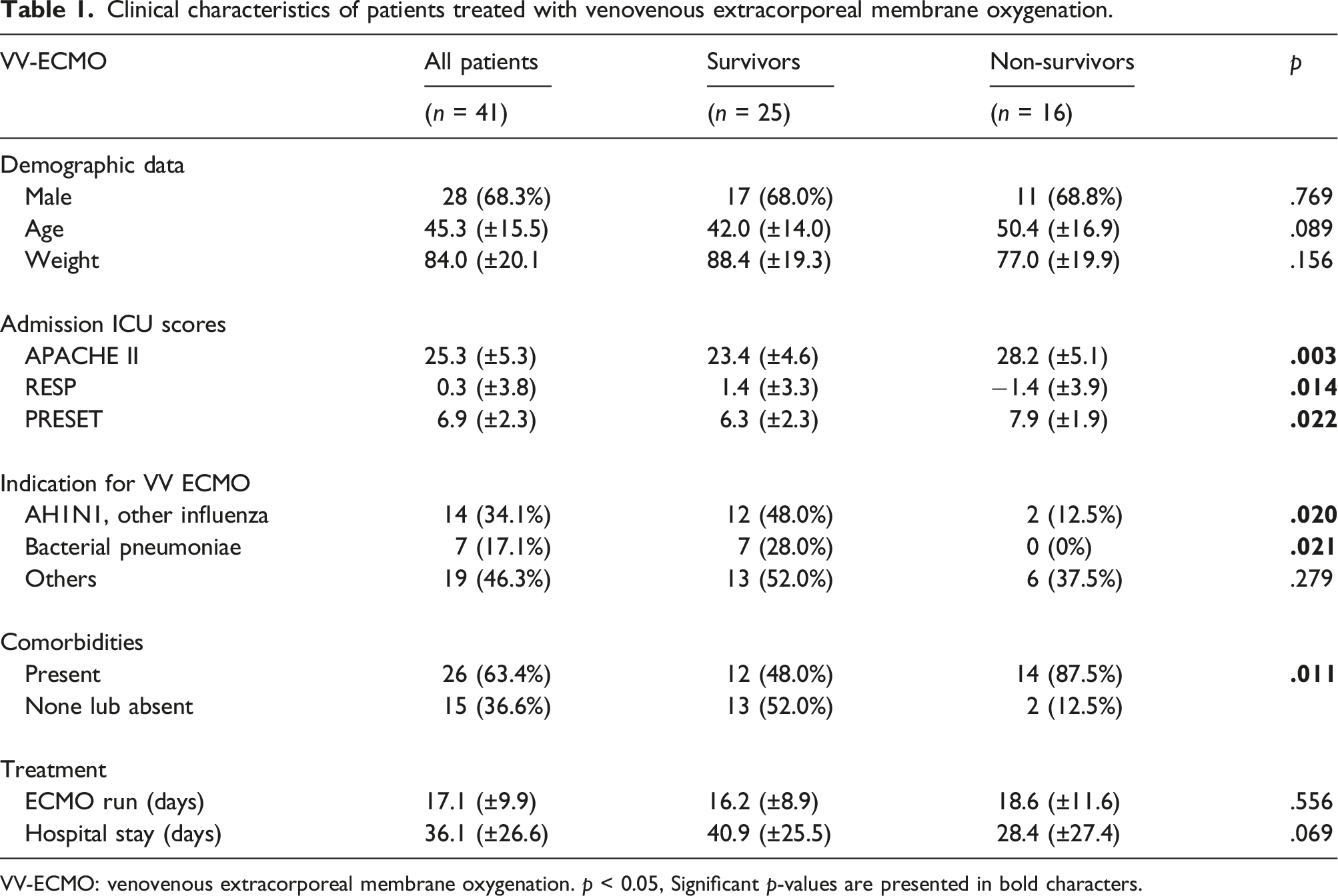
VV-ECMO: venovenous extracorporeal membrane oxygenation.
Clinical characteristics of patients treated with venoarterial extracorporeal membrane oxygenation.
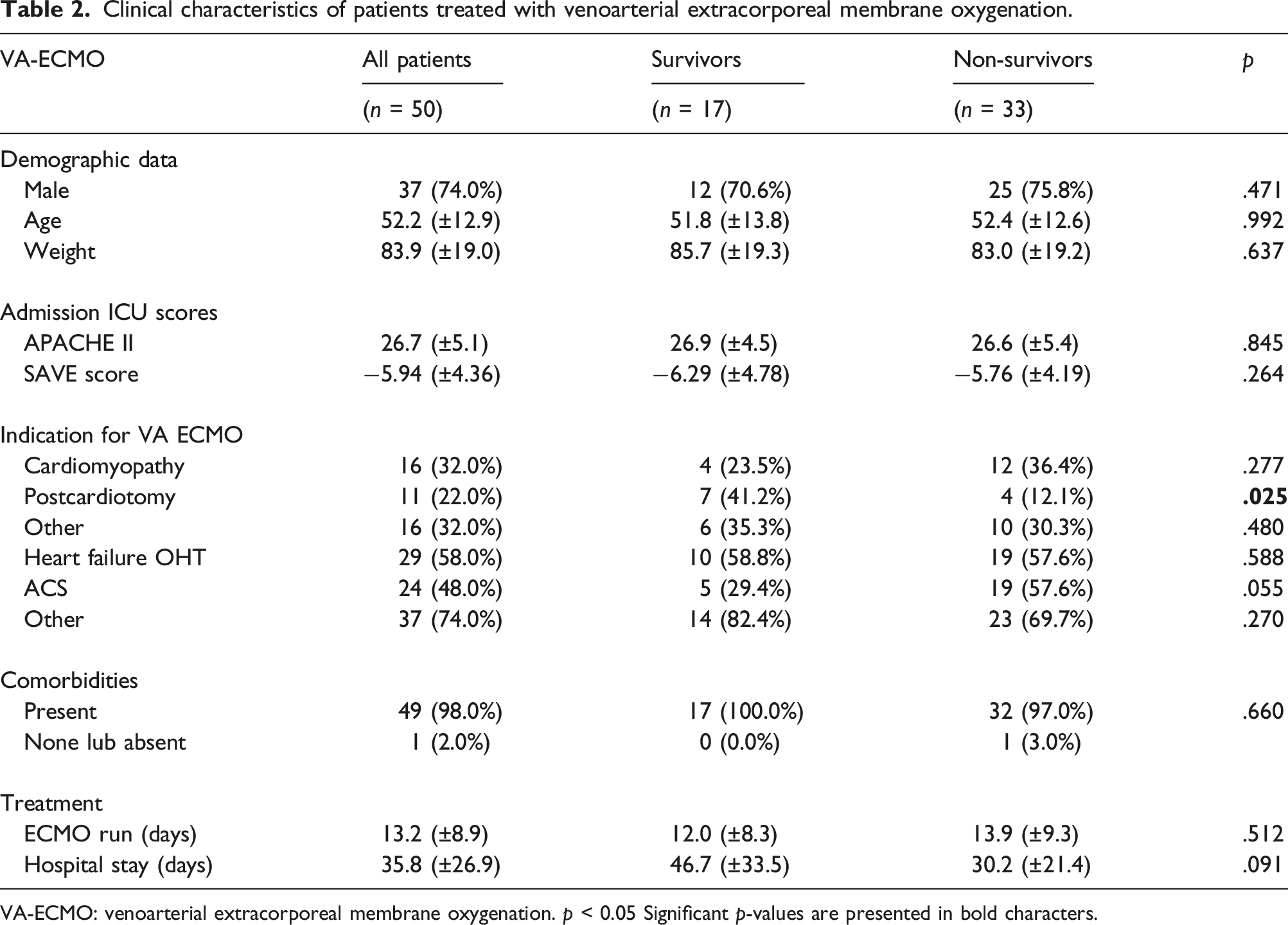
VA-ECMO: venoarterial extracorporeal membrane oxygenation.
Cannulation for VA ECMO was performed either peripherally (via the femoral vein and femoral artery) with distal perfusion cannula (with an additional 5–7 F catheter systematically inserted into the femoral artery to prevent limb ischemia) or centrally (via full or partial sternotomy with venous drainage from right atrium (RA) or right chambers (RV) via aortic cannula placed directly into the aorta. The drainage cannula was placed in the inferior vena cava with the tip at the level of L1–L2, 1–1.5 cms below the right atrium. Demographic data, co-morbidities, indications for ECMO support as well as admission ICU scores, both for general use (APACHE II score 18 and specific for ECMO support: RESP score, 19 and SAVE score, 20 were calculated and analyzed. The dose of catecholamines was calculated for each patient during the first 72 h of ECMO using the vasoactive-inotropic score as proposed by Koponen et al. 21
Within the first 72 h following ECMO initiation, lactate levels and lactate clearance between two time points, T = 24 and T = 72, where T = 24 exhibited the worst lactate levels within the first 24 h after ECMO invitation and T = 72 the worst lactate levels after 72 h of ECMO initiation was (LAC T24-LAC T72)/LAC T24 *100. A negative value means an increase in lactate concentration.
Statistical analysis
The distribution of quantitative variables was tested with the Shapiro-Wilk test. Due to the distributions being significantly different from normal, non-parametric U Mann-Whitney tests were used. Chi-square test with correction for Yates continuity was used for qualitative variables. In order to assess the differentiating properties of lactates, ROC curves were analyzed: sensitivity and specificity for each lactate value were determined and were used to draw a curve and the area under the curve was calculated. The optimal cut-off point was determined using the Youden Index. Differences in survival curves between two independent groups were evaluated using Kaplan-Meier analysis. The results are presented in tables with the mean, mean +/− SD, median and quartile range or counts, and percent value of the tested parameter with SD for quantitative parameters and percentage alone for qualitative parameters. For all of the analyses,
Results
A total of 101 patients were screened. Of these, 10 patients died within 48 h of initiation of ECMO therapy. Overall, 41 patients received VV ECMO and 50 patients received VA ECMO in the analyzed timeframe. Reasons for ECMO implementation varied and included a wide range of clinical indications and conditions: VV ECMO (Table 1) and VA ECMO (Table 2). ICU mortality in the first 30 days of initiation of VV and VA ECMO was 39% and 66%, respectively. Comparison of demographic data, co-morbidities, indications for ECMO support, and admission ICU scores in survivors and non-survivors in ICU at day 30 who underwent VV ECMO and VA ECMO are presented in Tables 1 and 2, respectively. In the VV ECMO group, non-survivors had higher values of APACHE II (survivors: 23.4 ± 4.6, non-survivors: 28.2 ± 5.1,
There was no statistical difference in age, weight, ECMO days, ICU stay, and hospital days between survivors and non-survivors (Table 1). In the VA ECMO group, there were no significant differences between survivors and non-survivors in baseline profiles, in APACHE II and SAVE Score values (Table 2). However, non-survivors with PCS were more frequently compared with survivors (survivors; 7/17, 41.2%, non-survivors 4/33; 12.1%,
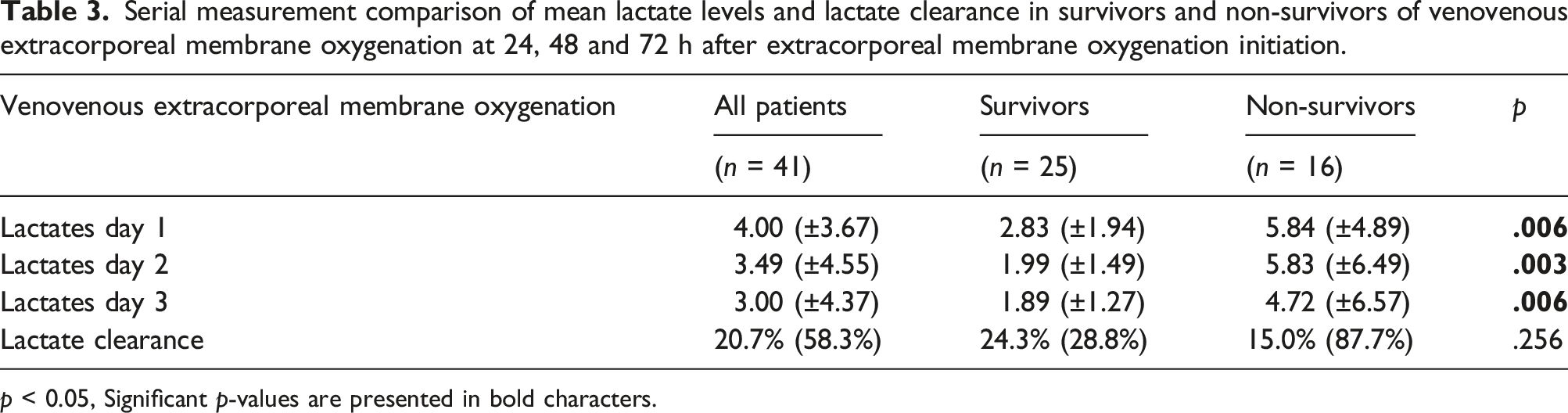
Serial measurement comparison of mean lactate levels and lactate clearance in survivors and non-survivors of venovenous extracorporeal membrane oxygenation at 24, 48 and 72 h after extracorporeal membrane oxygenation initiation.
Serial measurement comparison of mean lactate levels, lactate clearance in survivors and non-survivors of venoarterial extracorporeal membrane oxygenation at 24, 48 and 72 h after extracorporeal membrane oxygenation initiation.
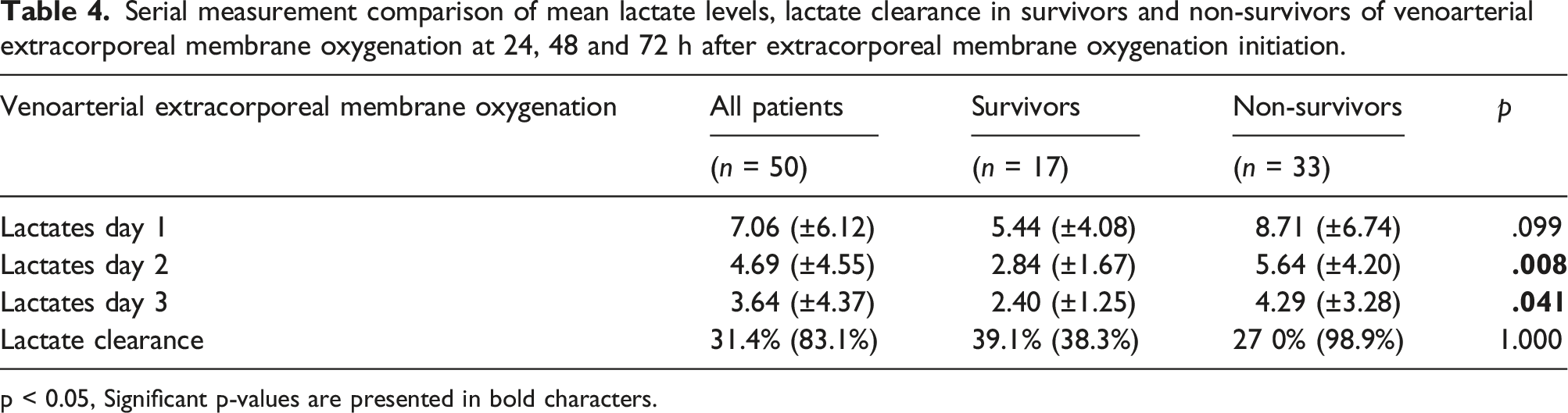
p < 0.05, Significant p-values are presented in bold characters.
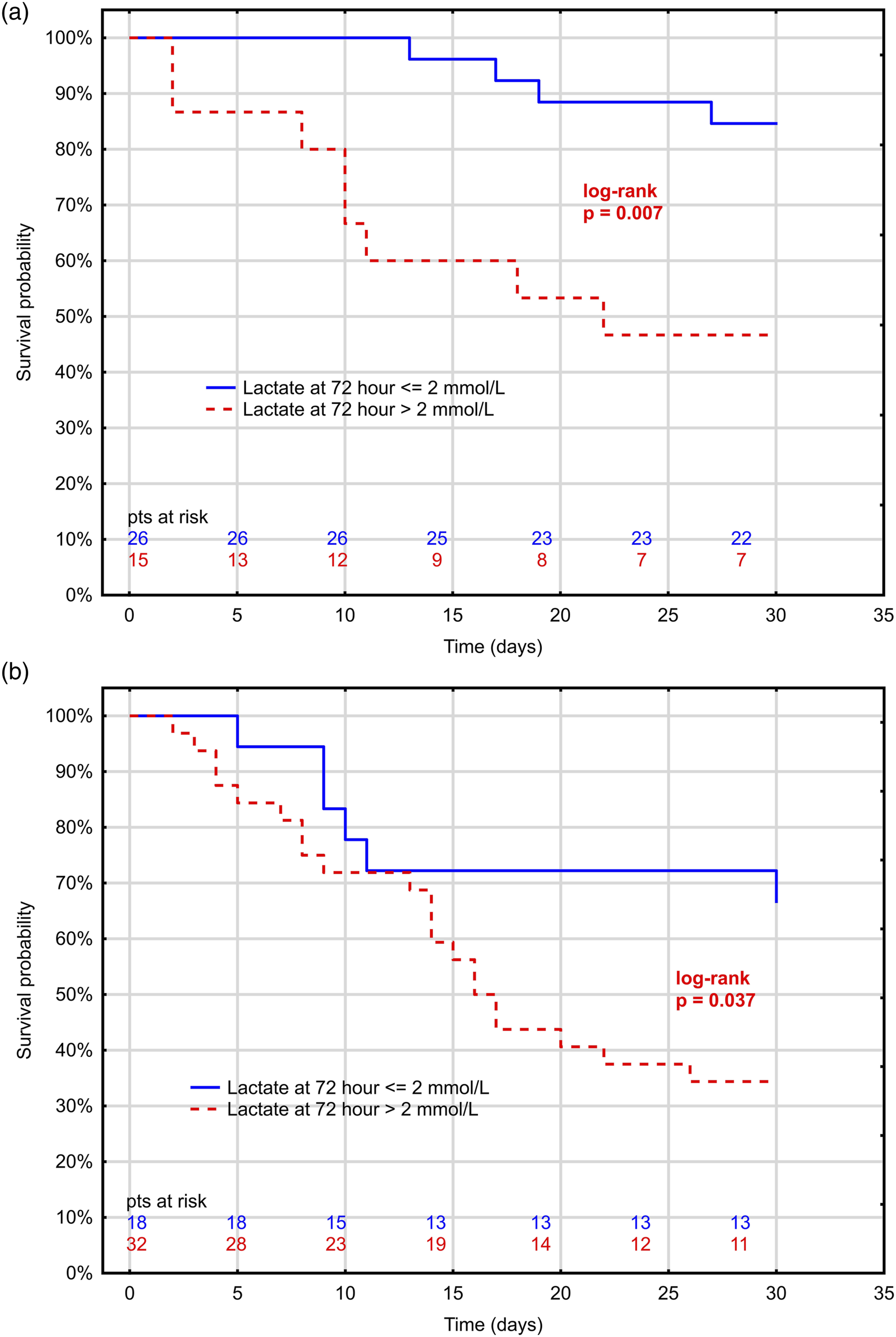
Kaplan–Meier curve with elevated lactate levels, >2 mmol/L, and ≤ 2 mmol/L at 72 h after ECMO initiation. (a) Veno-venous (VV)-ECMO (b) Venous-arterial (VA)-ECMO. ECMO: extracorporeal membrane oxygenation.
Lactate levels at 72 h were found to be predictive of 30-day survival, particularly in patients who underwent VV ECMO (Figure 2(a)). The area under the receiver operating characteristic curve (ROC) was 0.91 for patients who underwent VV ECMO and 0.73 for patients who underwent VA ECMO. In terms of lactate levels at 72 h, the optimal cut-off point indicating 30-day mortality was lactate levels exceeding 2.4 mmol/L for VV ECMO (sensitivity 67%, specificity 86%) and 4.4 mmol/L for VA ECMO (sensitivity 44%, specificity 96%). Lactate levels were substantially better predictors of 30-day survival in the VV ECMO group than in the VA ECMO group as indicated by higher values in the AUC statistics (Figure 2(a)) and much steeper slopes for the relationship between lactate levels and 30-day survival probability. However, unlike absolute value, lactate clearance between 24 and 72 h did not indicate survival for either VV or VA ECMO (ROC curve of 0.415 and 0.554, respectively), as shown in Figure 2(b). (a) ROC curve for lactate levels at 24 and 72 h after the start of ECMO in patients treated with VV-ECMO and VA-ECMO. (b) ROC curve for lactate clearance between 24 and 72 h after the start of extracorporeal membrane oxygenation (ECMO) in VV-ECMO and VA-ECMO. ROC: Receiver operating characteristic; ECMO: extracorporeal membrane oxygenation.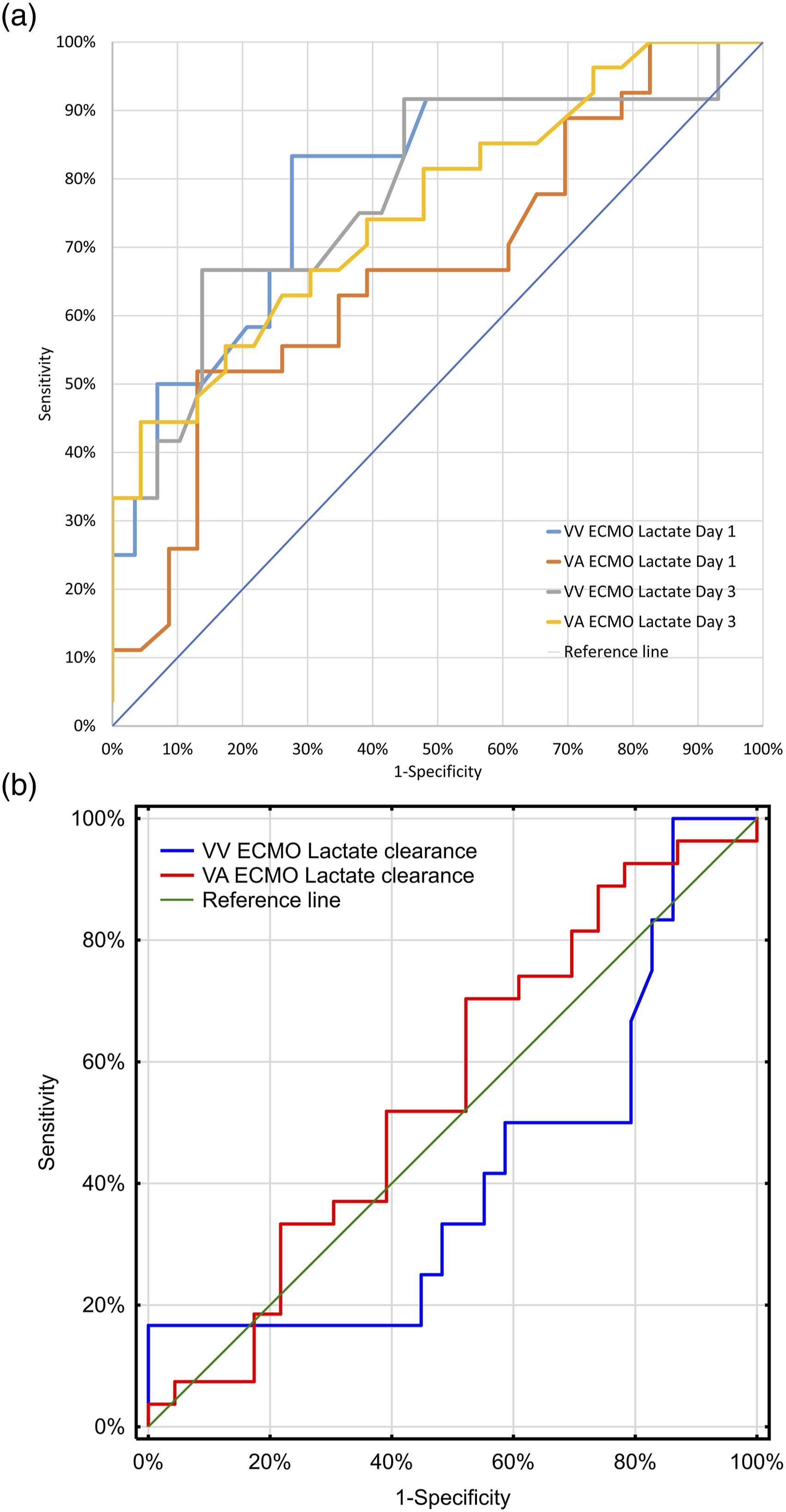
Changes in the absolute values of lactate levels between 24 and 72 h in survivors and non-survivors are presented separately for VV ECMO and VA ECMO (Figure 3(a) and (b), respectively). These curves indicate that although baseline absolute lactate values could widely vary, at 72 h, the range of absolute lactate values tended to be lower and more homogeneous in VV ECMO compared to VA ECMO. Changes in absolute values of lactate levels between 24 and 72 h after ECMO initiation in survivors and non-survivors (a) for VV-ECMO and (b) for VA-ECMO. ECMO: extracorporeal membrane oxygenation.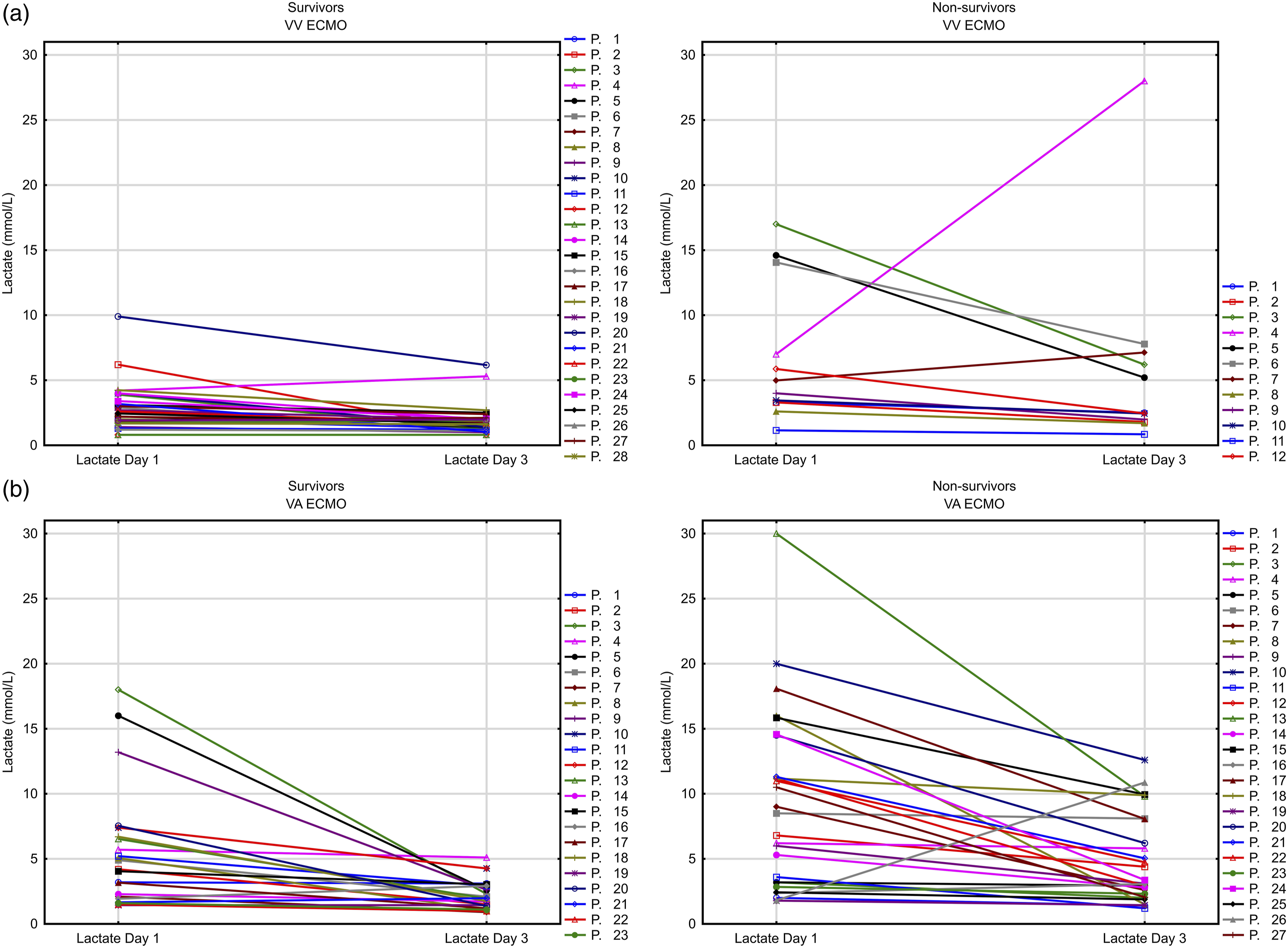
Discussion
Although data on lactate levels in critically ill patients is available, the predictive values of lactate levels in the first 24, 48, and 72 h in 30-day mortality in patients treated with VV and VA ECMO is not well established. In our study, we demonstrated that adult patients with a lactate level above the laboratory normal value, i.e., 2 mmol/L after 72 h of ECMO therapy had a considerably higher risk of death compared to those who did not. This applies to both patients with refractory ARDS supported by VV ECMO and patients with cardiogenic shock after cardiac surgery or with decompensate chronic heart failure supported by VA ECMO.
This is of particular importance to practicing ECMO physicians who treat patients using both types of ECMO. The results obtained in our study suggest that if at 72 h, despite good medical practice and multimodal therapy, a patient’s lactate level is still above normal, i.e., 2 mmol/L, there is a higher risk of death within 30 days compared with a patient whose lactate level at 72 h is below 2 mmol/L.
VV-ECMO
In the setting of respiratory failure, lactate production most likely occurs due to systemic malperfusion due to septic shock or, possibly, from profound hypoxia or circulatory failure due to RV dysfunction in the setting of ARDS. 9 The role of ECMO in reversing lactate production in respiratory failure is likely minimal except for cases of RV failure induced shock that subsequently improves with ECMO support and the ability to modulate ventilatory support to improve RV function. Hypoxemia and hypercarbia before VV ECMO support can lead to an increase in pulmonary vascular resistance, elevated pulmonary arterial pressure, right heart strain, or right ventricular failure. 22 At the same time, hypotension and impaired circuit flow can occur as a result of significant vasoplegia because of a systemic inflammatory response to the exposure to the extracorporeal circuit or hypovolemia related to unrecognized hemorrhage. Volume resuscitation with intravenous crystalloids, colloids, or blood transfusions can lead to fluid overload, exacerbating an already present acute renal failure and right ventricular overload with consequent failure of left ventricular function and impaired tissue perfusion. 22 In most cases, liver failure was also observed as a result of hypoxia and hypovolemic shock. 9 In VV ECMO, the lungs are not commonly considered a source of lactate but have been reported to be a major source of lactate 16 and will provide indirect hemodynamic support through the optimization of pH, PaCO2, and PaO2. 22
However, publications analyzing lactate values between non-survivors and survivors are scarce. In a retrospective analysis, Klinzing et al. evaluated 51 patients supported with VV-ECMO and found no differences in lactate values between non-survivors and those who were alive at 6 months post-ICU. 23 Another retrospective analysis of 12 patients by Beurtheret et al. showed only a trend towards a decrease in mortality associated with lower lactate values after VV ECMO; this was likely due to the small number of patients evaluated. 24 Only a few studies analyzed changes in selected variables, including lactate, associated with outcomes during ECMO support, but rarely did the study trace these trends beyond the early phase of ECMO. In our study, we demonstrated that in VV ECMO patients, non-survivors exhibited significantly higher lactate concentrations than survivors at T = 24, 48, and 72 h of treatment. In addition, we also demonstrated that lactate values above ≥2 mmol/L 72 h after VV ECMO initiation were significantly more often observed in patients with poor outcome. To the best of our knowledge, only a few studies have described higher serum lactate concentrations in VV ECMO non-survivors but they specifically focused on lactate values before or at 24 h after VV ECMO invitation,25,26 which corresponds more to the severity of illness at admission rather than response to therapy.
In addition to the prognostic values of lactate levels, we also tested the prognostic value of lactate clearance between 24 and 72 h from initiation of VV ECMO of therapy for 30-day mortality. A recent study showed that lactate clearance at 72 h after ECMO initiation is an independent predictor of ICU mortality. In particular, patients who showed a lactate clearance of more than 16% had a lower risk of death. 16 In contrast, in our study, lactate clearance between 24 and 72 h did not correlate with survival for either VV or VA ECMO. Similarly, analysis of septic patients treated with VV ECMO6,27 demonstrated that lactate levels had a significantly higher prognostic value than lactate clearance. Our findings add to the existing literature by addressing the kinetics of lactate levels during VV ECMO support. Determining these values allows for the assessment of ECMO efficacy, and potential factors responsible for insufficient perfusion should be immediately identified and corrected.
VA ECMO
In adults treated with VA ECMO, lactate clearance has been studied in only a few reports and similar to VV ECMO patients, the methodology, duration of lactate testing of arterial blood, and the results obtained have not been consistent. Lactate levels in VA ECMO patients were predominately analyzed before ECMO initiation or in the first day after. A retrospective study of 24 patients with refractory cardiac disease showed that lactate values measured upon admission and at 12 and 24 h after ECMO initiation showed no differences between survivors and non-survivors. 28 However, several studies have shown that lactate levels are independent risk factors for mortality in critically ill patients with PCS supported with VA ECMO.12,29–34 In contrast to these results, we found no differences in lactate levels in the first 24 h after VA ECMO initiation between survivors and non-survivors, but the probability of 30-day survival in patients with lactate levels above 2 mmol/L at 72 h from VA ECMO initiation was significantly lower when compared to patients with lactate levels ≤2 mmol/L. The potential mechanism(s) for the higher lactate concentrations observed in non-survivors and why lactate clearance in VV ECMO and VA ECMO from 24 to 72 h was not significant for survival can only be hypothesized. From a pathophysiological point of view, reduction of lactate levels below the upper limit of normal indicates the achievement of hemodynamic goals and restoration of tissue perfusion. As mentioned, the extracorporeal circuit provides indirect hemodynamic support by optimizing the pH, PaCO2, and PaO2, which improves pulmonary arterial pressures, RV dysfunction, coronary oxygenation, and left ventricular function leading to lower lactate levels.
There are only a few studies which have analyzed the cut-off point indicating 30-day mortality for VV and VA ECMO, mostly with similar results.31,35,36
Our study determined that the optimal cut-off point indicating 30-day mortality was a lactate level exceeding 2.4 mmol/L for VV ECMO and 4.4 mmol/L for VA ECMO.
We consider that our findings have several clinical implications. First, normalization of blood lactate during the 72-h period in VV and VA ECMO therapy could be considered a goal of therapy.
Second, dynamic measurement of arterial lactate allows for the assessment of potential factors responsible for insufficient perfusion despite optimal therapy for achieving standard circulatory goals (e.g., mean arterial pressure ≥65 mmHg, cardiac index >2.2 L/minute/m2) while using inotropic and vasoconstrictor agents with lung protection ventilatory strategy, anticoagulation, fluid management, and nutrition during ECMO suport. Clinicians should be particularly careful and immediately identify and address these factors during the first 72 h after VV and VA ECMO implementation.
Limitations
Our study has several limitations. In the VV ECMO group, non-survivors showed higher values of APACHE II, RESP score, PRESET score, and had more comorbidities. Therefore, our analysis may represent the most critically ill VV ECMO patients. In the VA ECMO group, non-survivors were more likely to have pre-existing cardiomyopathy, but survivors were more likely to have PCS, which indicates heterogeneity of the VA ECMO group. Additionally, we only included patients who survived more than 48 h after ECMO implementation, which most likely eliminated the sickest patients. However, our goal was to demonstrate lactate as a predictor of tissue perfusion in response to therapy rather than overall survival. Finally, this retrospective study was limited by factors inherent to the retrospective data analysis and was based on the experience of one institution with a moderate number of patients, thus limiting the generalizability of our results. We focused only on lactate levels and clearance; therefore our results did not reflect the time-course of other physiologic variables.
More prospective research is needed to confirm our findings in a larger group of patients. Further research is required to determine what interventions and therapies are required to achieve the target lactate values in this goal-directed therapy.
Conclusions
Based on our results, lactate levels above 2.0 mmol/L at 72 h from ECMO initiation are prognostic of 30 day-mortality in patients with both VV ECMO for refractory ARDS as well as VA ECMO for patients treated with decompensated chronic heart failure and in patients with postcardiotomy shock.
Footnotes
Acknowledgements
We greatly appreciate the help from Miss Jolanta Cieśla with editorial and for technical advice.
Author contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by ET, PK and DC performed the statistical analyses. The first draft of the manuscript was written by ET. PA, SS, ASS, MT-S, MOZ, KH commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study protocol was approved by the Ethics Committee at the Silesian Medical University in Katowice, Poland.
Informed consent
The need for informed consent was waived owing to the retrospective study design.
