Abstract
Introduction
A basic prerequisite for a good surgical outcome in heart surgery is optimal myocardial protection. However, cardioplegia strategies used in adult cardiac surgery are not directly transferable to infant hearts. Paediatric microplegia, analogous to Calafiore cardioplegia used in adult cardiac surgery, offers the advantage of safe myocardial protection without haemodilution. The use of concentration-dependent paediatric microplegia is new in clinical implementation.
Material and methods
Paediatric microplegia has been in clinical use in our institution since late 2014. It is applied via an 1/8 inch tube of a S5-HLM roller pump (LivaNova, Italy). As cardioplegic additive, a mixture of potassium (K) 20 mL (2 mmol/mL potassium chloride 14.9% Braun) and magnesium (Mg) 10 mL (4 mmol/mL Mg-sulphate Verla® i. v. 50%) is fixed into a syringe-pump (B. Braun, Germany). This additive is mixed with arterial patient blood from the oxygenator in different flow
dependent ratios to form an effective cardioplegia.
Technique
After microplegia application of initially 25 mmol/L K with 11 mmol/L Mg for 2 min, a safe cardioplegic cardiac arrest is achieved, which after release of the coronary circulation, immediately returns to a spontaneous cardiac-rhythm. In the case of prolonged aortic clamping, microplegia is repeated every 20 min with a reduction of the application dose of K by 20% and Mg by 30% (20 mmol/L K; 8.5 mmol/L Mg) and a further reduction down to a maintenance dose (15 mmol/L K; 6 mmol/L Mg) after additional 20 min.
Summary
The microplegia adapted to the needs of paediatric myocardium is convincing due to its simple technical implementation for the perfusionist while avoiding haemodilution. However, the required intraoperative interval of microplegia of approx. 20 min demands adapted intraoperative management from the surgeon.
Introduction
Developments and improvements in Cardiopulmonary Bypass (CPB) have led to the fact that the extracorporeal circulation can nowadays be used routinely even in very small infants. For these small patients, specialised system components such as oxygenators or filters with smaller volumes and reduced surface areas are available. 1 Despite considerable progress, efforts must be made to further reduce morbidity and mortality in paediatric operations involving CPB. To achieve this, the CPB must be designed as physiologically as possible. CPB-systems that are minimised as far as possible reduce the dilution effect and lower the need for foreign blood products.2,3 The type of cardioplegia used for myocardial protection has a significant impact on the CPB-setup and thus a strong influence on the degree of haemodilution. A cardioplegia innovation in infant perfusion could be the use of paediatric microplegia along the lines of Calafiore et al. used in adult cardiac surgery.4,5
When adjusting cardioplegia from a “mature” adult to an “immature” paediatric myocardium, it should be noted that infant hearts in principle have a higher ischaemic tolerance and slightly altered metabolism. 6 However, hearts with congenital defects are more stressed by hypoxia, volume and pressure overload, resulting in lower ischaemic tolerance. Therefore microplegia, tailored towards adult hearts when used in the paediatric cohort provides suboptimal myocardial protection.6,7
O’Brien et al. found higher potassium requirements for inducing asystole and higher magnesium requirements for preventing calcium (Ca) influx into the myocardial cell in neonates and infants. 7 In neonatal hearts, Ca control during the ischaemic and reperfusion phases is of paramount importance to prevent Ca-induced cellular damage. 8
The specific characteristics in these diseased hearts were considered when compiling paediatric microplegia. The basis for paediatric microplegia have been elaborated on, in an animal experimental study by Münch et al.9,10
This Practical Technique article describes the clinical implementation of concentration-dependent paediatric microplegia across the entire patient population.
Material and methods
Paediatric microplegia is based on the mechanism of extracellular electrolyte shift. Here, rapid cardiac arrest is induced by high-dose potassium (K). The additional administration of extracellular magnesium (Mg) inhibits the influx of Ca into the Cell. The extracellular Mg acts as a Ca antagonist with the additional positive effect of membrane stabilisation and reduced postoperative arrhythmias. To counteract possible negative effects of the high extracellular Mg concentration, the plasma Ca concentration is raised to the upper normal limit by selective Ca administration shortly before the end of CPB.
Paediatric microplegia has been used in our institution clinically since the end of 2014. It is applied via an 1/8 inch pump segment in a S5-HLM roller pump (LivaNova, Italy). As a cardioplegic additive, a mixture of potassium (K) 20 mL (2 mmol/mL potassium chloride 14.9% Braun Melsungen, Germany) and magnesium (Mg) 10 mL (4 mmol/mL Mg Verla® i. v. 50%) is prepared in a syringe-pump. This supplement is mixed with arterial patient blood from the oxygenator in adapted ratios and applied as cardioplegia (Figures 1 and 2). Paediatric microplegia: The extracellular ion concentration calculation in the microplegia is assuming the patient’s blood with potassium content of 4.0 mmol/L and magnesium content of 0.9 mmol/L as the carrier medium. Charting of a complete CPB-setup in the application of paediatric microplegia.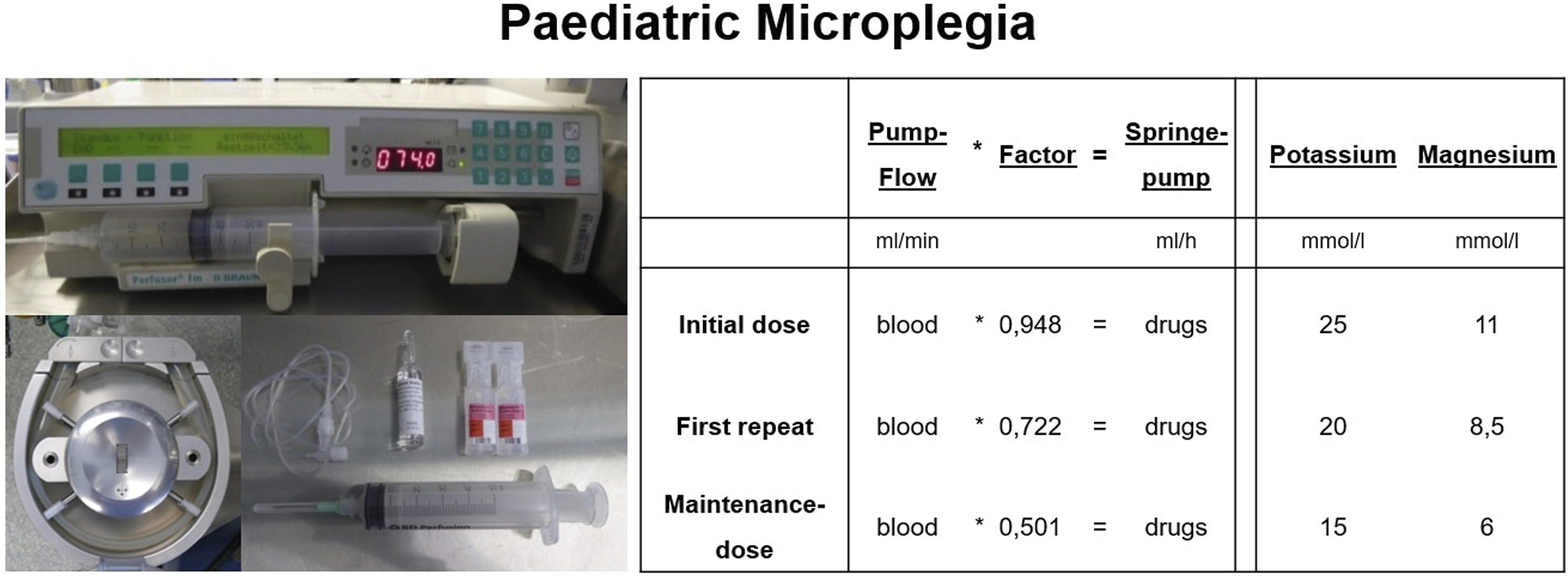
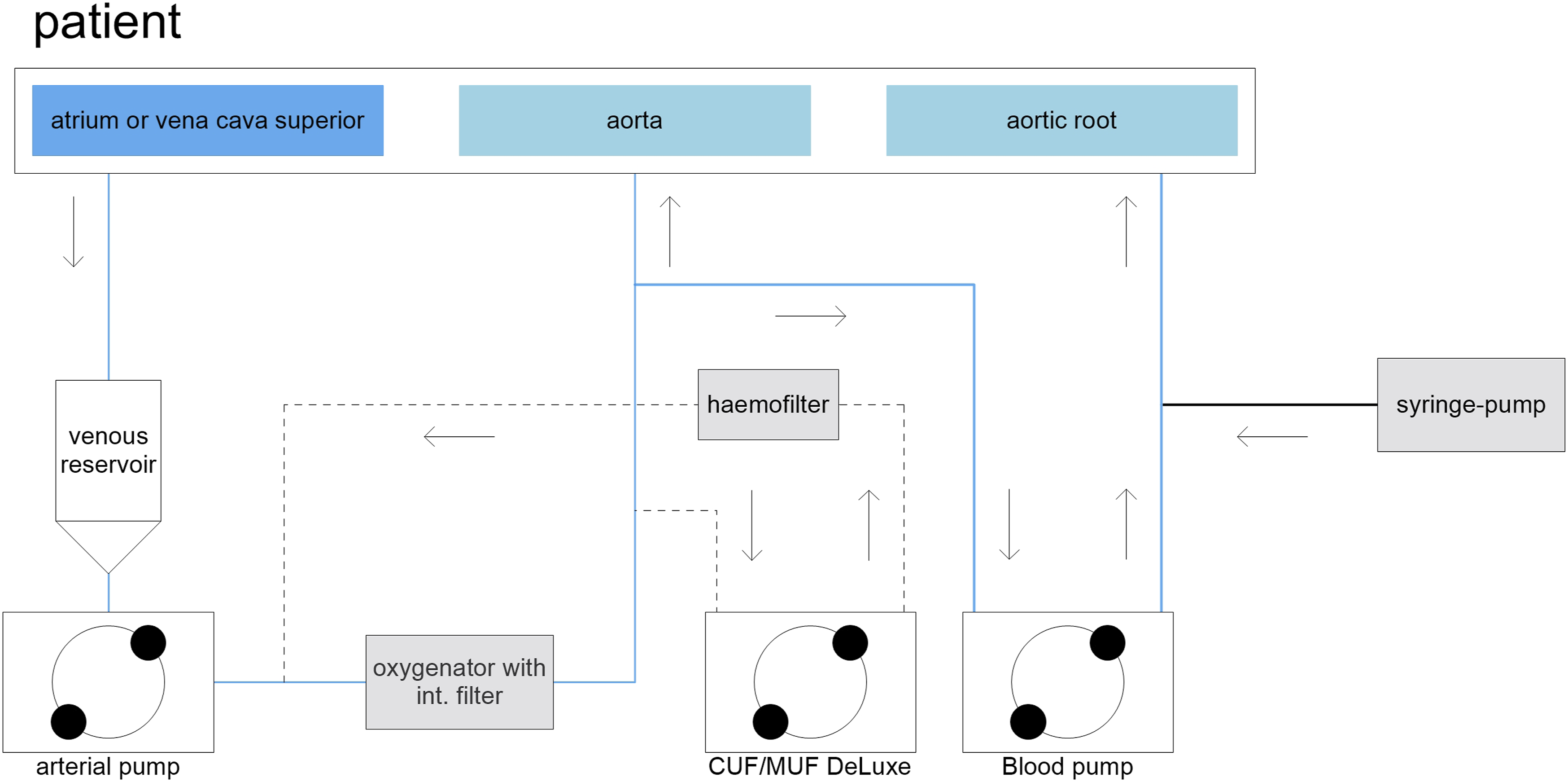
Technique
The goal of the ideal paediatric microplegia was to develop a reproducible and safe method for rapid induction and long-term electromechanical cardiac arrest. Homogeneous distribution is essential in this context. For this purpose, an unloaded heart, which at best is accessed by a vent in the left heart, is ideal and avoids overdistension of the left ventricle. Furthermore, it is important to adjust the application pressure and the intervals between the delivery of the cardioplegia. All together these measures optimize and minimize myocardial oxygen consumption and increase ischemia tolerance. 11
With the initial microplegia application dose over 2 minutes, rapid and safe cardioplegic arrest occurs. Twenty minutes after induction of microplegia, a first repeat-dose is given, then repeated every 20 min. During the first two doses, the application dose of K is reduced by 20% and Mg by 30% to the maintenance dose. The volume application ratio of patient blood to cardioplegic supplement is initially 63/1, first repeat-dose 83/1 and maintenance-dose 120/1. The application requires a certain level of attention from the perfusionist, as intermittent manual adjustment of the syringe-pump speeds to the blood flow of the cardioplegia pump is necessary (Figure 3). Microplegia application increases the magnesium and potassium concentration in the patient’s blood. If the application rates remained constant, the Mg and K concentrations would increase with each cardioplegia re-administration, and the risk of hyperpotassaemia would be unavoidable, especially during prolonged aortic cross clamp time. Adjustment of syringe-pump rate for paediatric microplegia.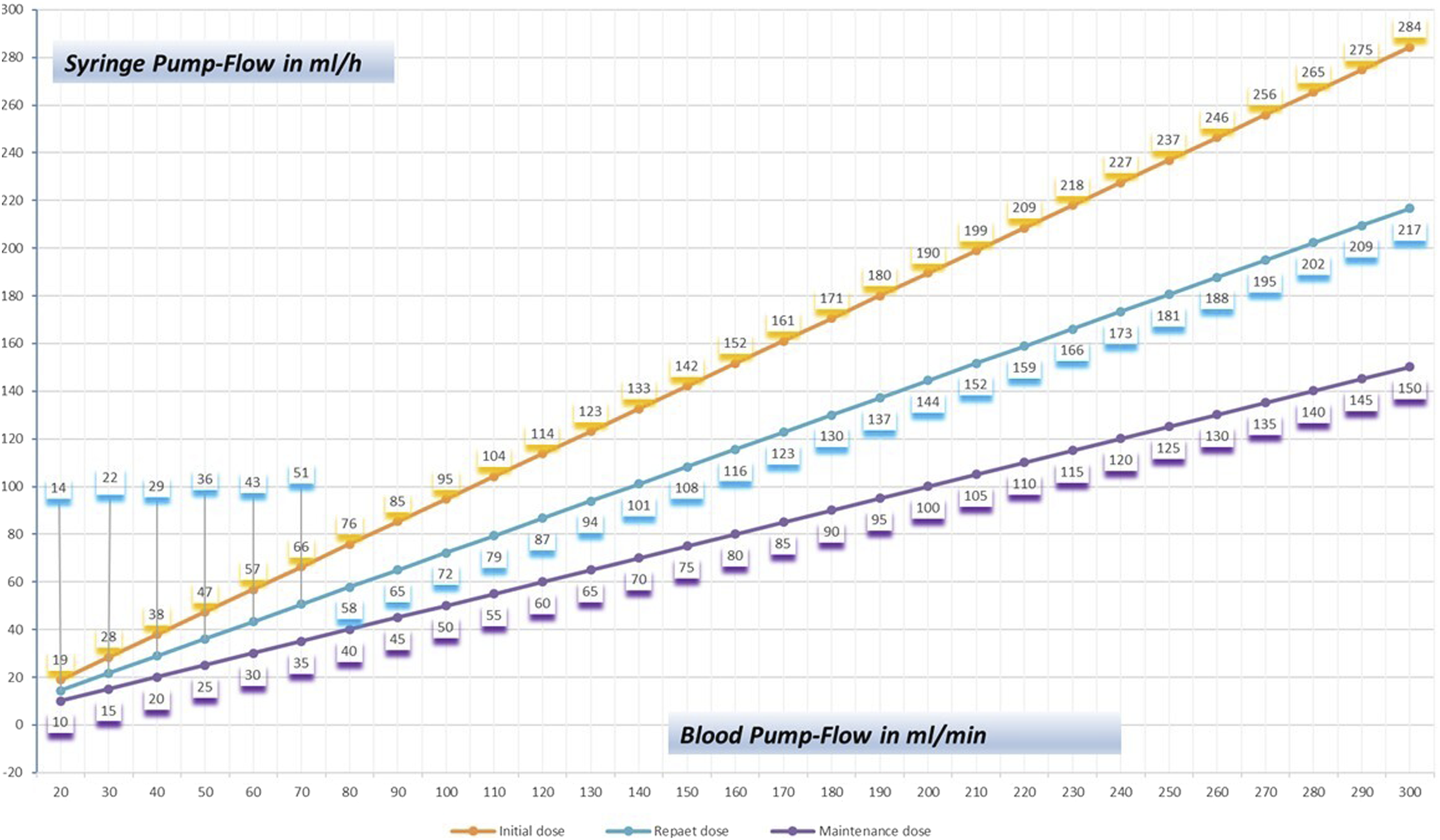
Given a 3 kg neonate undergoing major cardiac surgery and treated with this protective microplegia, it would receive a summed up cardioplegic supplement of 4.9 mL (K = 3.3 mL plus Mg 1.6 mL) when assuming an aortic cross clamp of 120 min.
After opening of the aortic cross clamp and re-establishment of the coronary circulation, the heart usually starts beating spontaneously in its own rhythm.
Discussion
According to Consiglio et al., the ideal cardioplegia provides rapid-onset, reversible cardiac arrest with low haemodilution, little systemic effect, long after-delivery intervals, low cost and ease of use. 11
The described microplegia procedure fulfils most of these criteria. In developing the paediatric microplegia presented here, it was considered that to induce asystole in neonatal and infantile hearts, where a higher K and a higher Mg requirement is necessary. 7 Therefore, the concentration of the initial potassium application dose was increased by 38% and the magnesium dose by 200% compared to Calafiore’s recipe for adult hearts.
The current Calafiore cardioplegia protocol recommends administration of a magnesium bolus at the end of each repeat dose. If there are still waveform movements of the heart, the dose can be increased as needed. In this new microplegia, Calafiore’s blood cardioplegia stood as a model. However, in addition to the Mg concentration, we also adjusted the repeat intervals and dosage. Calafiore et al. also note that other studies have shown that the interval between doses can be extended up to 20–30 min, if necessary, without adverse cardiac effects. Primarily to prevent hyperpotassaemia, especially during long aortic cross-clamping time, the postdosing dose was adjusted, as is also recommended in the current Calafiore protocol. That applied protocols must always be adapted to the surgical techniques and to the patient population, which we have implemented in this approach of paediatric microplegia. (Calafiore et al., 2020)
Systemic wholebody cooling is often indicated, depending on the anatomical features of the congenital heart defects, in order to reduce the oxygen consumption (VO2) of the body. At a 28°C core body temperature, VO2 decreases by 58%, which allows safe handling of patients with reduced CPB flow of 50%. 12
In almost all common cardioplegia procedures, local cooling with a solution temperature down to 4°C is performed to reduce myocardial oxygen consumption (MVO2). 11 However, local cooling of the myocardium seems to be of limited use for myocardial protection, as it increases left ventricular wall stress.9,13,14
On the other hand, Durandy et al. reported on the safe use of normothermic blood cardioplegia in congenital heart disease.15–17 Despite this good experience, normothermic cardioplegia is rarely used in paediatric cardiac surgery. As a compromise between normothermic und deep hypothermic temperatures mild or moderate hypothermia seems to be a good compromise. Several authors reported that intermittent tepid blood cardioplegia causes less metabolic myocardial damage, leading to a better clinical outcome.18–20 Thus, with tepid cardioplegia (28°C), the relationship between increased left ventricular wall tension and reduced MVO2 seems to be ideal. 13
Another advantage of using identical temperatures for systemic perfusion and cardioplegic arrest results in minimising the HLM-setup and reduction of CPB-priming by while omitting an additional cardioplegia heat-exchanger. This results in savings of foreign blood in the treatment of congenital cardiac lesions with improved myocardial function. Another positive side effect is in monetary savings. Thus, it is possible to achieve safe myocardial protection with low overall cardioplegia costs.
Our working group developed the basis for the paediatric microplegia described in this paper in an experimental animal study. In this prospective, randomized trial, improved contractility of the myocardium, less electrolyte shift and reduced haemodilution was observed after myocardial ischaemia induced with microplegia compared to application of cold crystalloid cardioplegia. We were thus able to validate the use of microplegia for the paediatric population.9,10
Ms Klaever’s doctoral dissertation examined the comparison of crystalloid cardioplegia (Custodiol) to paediatric microplegia in simple cardiac defects in a non-inferiority-study. Ms Klaever concluded that lukewarm microplegia provides a safe and effective myocardial protection in infants and young children undergoing cardiac surgery. However, theoretical advantages of microplegia and evidence from other studies that see a significant advantage for cyanotic patients or for patients with longer cross-clamp-times may suggest that this type of blood cardioplegia is preferable for this patient population. 21
Since November 2014, the paediatric microplegia procedure has been included in the university Erlangen hospital’s own Standard Operating Procedure (SOP) and has been continuously applied since then. Due to the very good clinical experience of the cardiac surgeons and the convincing performance, this type of paediatric microplegia has already been adopted by three other paediatric cardiac surgery centres.
Conclusions
The microplegia adapted to the needs of the paediatric myocardium is convincing due to its simple technical implementation for the perfusionist, while avoiding haemodilution. The resulting minimisation of the ECC setup enables a reduction in priming, which is associated with further positive effects for the patient. However, the required intraoperative interval of the microplegia of approx. 20 min requires an adapted operative management from the surgeon.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
