Abstract
Objective
To evaluate a restrictive transfusion policy of red blood cells (RBC) and platelets in pediatric patients submitted to extracorporeal membrane oxygenation (ECMO).
Methods
Retrospective descriptive study of pediatric patients supported with ECMO, from January 2010 to December 2019. Hemoglobin, platelet, lactate and mixed venous oxygen saturation (SvO2) values of each patient while on ECMO, were collected. Transfusion efficiency and tissue oxygenation were statistically evaluated comparing pre-transfusion hemoglobin, lactate and SvO2 with post-transfusion values. Ranges of hemoglobin and platelets were established, and the number of transfusions registered. The bleeding complications and outcome were documented.
Results
Of a total of 1016 hemoglobin values, the mean value before transfusion was 8.6 g/dl. Hemoglobin and SvO2 increased significantly post-transfusion. Red blood cell transfusion varied with hemoglobin values: when hemoglobin value was less than 7 g/dl, 89% (41/46) were transfused but just 23% (181/794) when greater or equal to 7 g/dl. In the presence of active bleeding, the frequency of RBC transfusion increased from 32% to 62%, with hemoglobin between 7 g/dl and 8 g/dl.
The mean value for platelet transfusion was 32 x 109/L. Thirty-eight (43%) platelet values between 20 x 109/L and 30x109/L, and 31 (40%) between 30 x 109/L and 40 x 109/L led to platelet transfusion; between 40 x 109/L and 50 x 109/L, only 7 (9%) prompted platelet transfusion.
Comparing the 2010–2015 to 2016–2019 periods there was a decrease in RBC and platelet transfusion threshold with similar survival (p = .528). Survival to discharge was 68%.
Conclusions
Using a restrictive RBC and platelet transfusion policy was safe and allowed a good outcome in this case series. The presence of active bleeding was an important decision factor when hemoglobin was above 7 g/dl and platelets were above 30 x 109/L.
Keywords
Introduction
Extracorporeal membrane oxygenation (ECMO) is a lifesaving technique used for long-term support of respiratory and/or cardiac function. In the last 30 years, there were significant advances in pediatric ECMO with major changes in indications, cannulation, duration of treatment and outcome. 1 Nevertheless, the transfusion policy, an important aspect of daily patient management on ECMO support, remains in discussion with variable transfusion policies across different ECMO centers.2,3
The Extracorporeal Life Support Organization (ELSO) guidelines defined that the hemoglobin level should be kept within normal limits (12–14 g/dl). 4 The primary goal of red blood cells (RBC) transfusion is to increase the oxygen carrying capacity to allow adequate tissue oxygenation. However, Fiser et al. demonstrated that routine RBC transfusions to maintain a hematocrit above 35% during VA ECMO did not improve venous oxygen saturations (SvO2) or cerebral oxygenation. 5 Furthermore, recent studies showed that higher hemoglobin values could be associated with hemolysis and thrombosis during ECMO support, and consequently a worse prognosis. 6 Other recent reports, designed in the pediatric and adult populations, highlighted that greater RBC transfusion volumes among patients supported with ECMO are independently associated with a significant increase in mortality.2,7–9
Current evidence-based guidelines for critically ill pediatric and adult patients, recommend that transfusions should not be routinely performed for hemoglobin values above 7 g/dl (hematocrit about 21%). 10 Moreover, restrictive transfusion trigger policies (Hg 7 g/dl) were not associated with increased mortality and demonstrated a decrease in the length of stay.11,12 However, that premise was not studied in the pediatric ECMO population. In neonates on ECMO, Keene S. demonstrated that RBC rates are associated with increased mortality, reinforcing the importance of future studies to investigate transfusion practices that minimize the risks. 13
Although the ELSO guidelines admit the possibility of a restrictive approach, considering the lack of clinical evidence on RBC transfusions on neonatal and pediatric ECMO, they still recommend keeping a hematocrit near 40%. 4
Platelet transfusions are common among critically ill children on ECMO. Besides the thrombocytopenia sometimes being a consequence of the underlying pathology, the extracorporeal circuit may induce platelet consumption. 3 The ELSO guidelines consider that platelet transfusion should be performed to keep the platelet count above 80 x 109/L. However, qualitative platelet dysfunction may also occur during ECMO with platelet values above 80 x 109/L. Accordingly, optimal transfusion thresholds are not well established. 14
Considering the current lack of evidence on transfusional policy and highlighting the clinical relevance of those themes, we performed a study to evaluate the safety of a restrictive policy on the transfusion of RBC and platelets in pediatric patients submitted to ECMO. We hypothesized that a more restrictive policy could be safer without compromising the outcome.
Methods
Cohort and characterization
We conducted a retrospective descriptive study of pediatric patients supported with ECMO from 1 January 2010, to 31 December 2019, admitted to the Pediatric Intensive Care Unit (PICU) of a university-affiliated hospital in Lisbon, Portugal. All patients supported with ECMO during the study period were identified for inclusion, regardless of primary diagnosis, type of ECMO and time of ECMO support. Patients who required a major surgery during ECMO support were excluded.
Clinical data to characterize the pediatric cohort were extracted from the electronic medical record system. Information about patients’ demographics, primary diagnosis, venoarterial (VA) or venovenous (VV) ECMO type and time on ECMO was collected. All ECMO patients were anticoagulated with unfractionated heparin to a target activated clotting time (ACT) of 180–220 s. In the presence of active bleeding the target ACT is adjusted and heparin is reduced or even stopped.
Transfusion policy
Transfusion of blood products is determined clinically by the attending physician. Nonetheless, in our unit as a general guideline a cutoff of hemoglobin of 7 g/dL is used for RBC transfusions. In the case of platelet transfusion, the cutoff is 30 x 109/L. This is considered a restrictive policy.
Red blood cells transfusions
To study the RBC transfusion threshold the collected variables were hemoglobin value (g/dl), lactate (mg/dl), mixed SvO2, active bleeding, RBC transfusion number and outcome (survival or non-survival to discharge). Active bleeding was defined as hemorrhage from mouth, nose or pharynx, hematuria, intracranial hemorrhage, gastrointestinal and lung hemorrhage, as well as, hemorrhage from puncture sites of central venous lines, cannulas or any other catheter/drain. All cases of hemorrhage were reported, including mild bleeding.
All hemoglobin values of each patient while on ECMO were collected. For each hemoglobin value, a corresponding lactate value and SvO2, collected at the same time through mixed venous blood gas analysis, were also registered. The presence of active bleeding was also registered. Red blood cell transfusions were analyzed according to the blood test values (hemoglobin, lactate, SvO2) that motivated the transfusion decision. Blood products used for priming the ECMO circuit were not considered. The RBC transfusion decision was made by the attending physician. Mean pre-transfusion values (hemoglobin, lactate and SvO2) were statistically compared with mean post-transfusion values.
To establish a correlation between the transfusion decision and the hemoglobin value, specific intervals were established for hemoglobin values under 12 g/dl. The intervals considered were hemoglobin <6; [6, 7 ]; [7, 8 ]; [8, 9 ]; [9, 10 ]; [10, 11 ]; [11, 12 ](g/dl). Within each interval, the frequency of transfusions was registered. The same study was performed excluding the samples collected during active bleeding.
To study the impact of RBC transfusion threshold on outcome, for each ECMO run, the total RBC transfusion number was evaluated, and it was statistically compared to outcome (survivors vs non-survivors). Death was considered for all patients that died during the ICU stay.
A comparison was made between mean hemoglobin value that prompted transfusion in the first years of ECMO practice (until 2015) and in the last years (2016–2019). The outcome (survival or non-survival to discharge) and the length of run were also compared between these two time periods.
Platelet transfusions
To study the platelet transfusion threshold the relevant variables registered were platelet values, active bleeding, platelet transfusion number and outcome.
All platelet values of each patient while on ECMO were collected. The number of platelet transfusions was registered. The mean platelet value that prompted a platelet transfusion was evaluated. Active bleeding was recorded, including the same hemorrhagic events described above in the RBC transfusion section.
To establish a correlation between the transfusion decision and the platelet value, specific intervals of platelet values were established for values under 100 x 109/L. The intervals considered were: <10 x 109/L; [10 x 109/L, 20 x 109/L ]; [20 x 109/L, 30 x 109/L ]; [30 x 109/L, 40 x 109/L ]; [40 x 109/L, 50 x 109/L ]; [50 x 109/L, 60 x 109/L ]; [60 x 109/L, 70 x 109/L ]; [70 x 109/L, 80 x 109/L ]; [80 x 109/L, 90 x 109/L ]; [90 x 109/L, 100 x 109/L ]. For each interval, the frequency of transfusions was registered. The same study was performed excluding the samples collected during active bleeding.
In order to relate the transfusion practice with outcome, the severe hemorrhagic complications (gastrointestinal, pulmonary and central nervous system (CNS) bleeding) were recorded. The platelet value before each bleeding event was registered.
A comparison was made between mean platelet value that prompted transfusion in the first years of ECMO practice (until 2015) and in the last years (2016–2019). The outcome (severe hemorrhagic complications) was also compared between these two time periods. These periods were chosen to find out if there was a change in transfusion thresholds over the years.
Statistical analysis
Data were analyzed with IBM SPSS Statistics for Macintosh, Version 27.0. Armonk, NY: IBM Corp. Significance was defined as p value of less than 0.05. Means are displayed with standard deviation and median values are displayed with minimum and maximum range values.
Red blood cell transfusion
For variable description, a parametric test (T test) was performed to evaluate hemoglobin and a non-parametric test (Kolmogorov Smirnov test) was applied to assess lactate and SvO2. Comparison between pre and post-transfusion values was assessed using T test for hemoglobin values and Wilcoxon Signed Rank for lactate and SvO2 values. Hemorrhage impact on the transfusion decision was studied using a chi-square test. Correlation between number of RBC transfusion was performed with an Independent Samples Mann-Whitney U test. For comparison of hemoglobin threshold over the 10-year’s experience, T test was used. Chi-square test was used to compare the outcome (survival vs non-survival) between the first 6 years and the last 4 years of the study.
Platelet transfusion
Descriptive tests of normality were applied to a total of 1016 platelet values. Chi-square test was used to study the impact of active bleeding on the transfusion decision. To compare the platelet transfusion threshold between the two time periods considered (2010–2015 and 2016–2019), a T test was used.
Ethical approval
Due to the retrospective and observational nature of the study a formal ethical approval was waived.
Results
Clinical and demographics characteristics
A total of 64 ECMO runs during the years 2010–2019 were included in the analysis. Ten were excluded because these patients were submitted to major surgery during ECMO support. The study was conducted on the remaining 54 ECMO runs, corresponding to 53 patients.
Clinical and demographic characteristics of study population. (Newborn: birth to 28 days of life).
Red blood cells transfusion
A total of 1016 samples were studied. During the ECMO run, the mean value of hemoglobin was 10 ± 1.9 g/dl; mean value of lactate was 18.8 ± 22 mg/dl and the mean value of SvO2 was 81 ± 10.5%. A total of 244 RBC transfusions were performed, with a mean of 4.15 ± 3.36 transfusions/ECMO run.
The mean value of hemoglobin that prompted a transfusion was 8.6 g/dL. There was a significant difference (p < .001) in pre and post-transfusion hemoglobin value, with a 95% confidence interval [−2298; −1841]. The mean hemoglobin value raised from 8.6 ± 1.6 g/dl to 10.7 ± 1.6 g/dl. Venous oxygen saturation showed a significant difference (p < .001) between pre and post-transfusion values, with a mean increase from 75 ± 10% to 79 ± 8%. There was no significant difference for lactate value, with a mean lactate value of 23.6 ± 29.7 mg/dl pretransfusion and 22.9 ± 30.1 mg/dl post-transfusion.
Correlation between hemoglobin value and RBCs transfusion decision
From the total of 840 hemoglobin values below 12 g/dl, 222 (26%) motivated RBC transfusion.
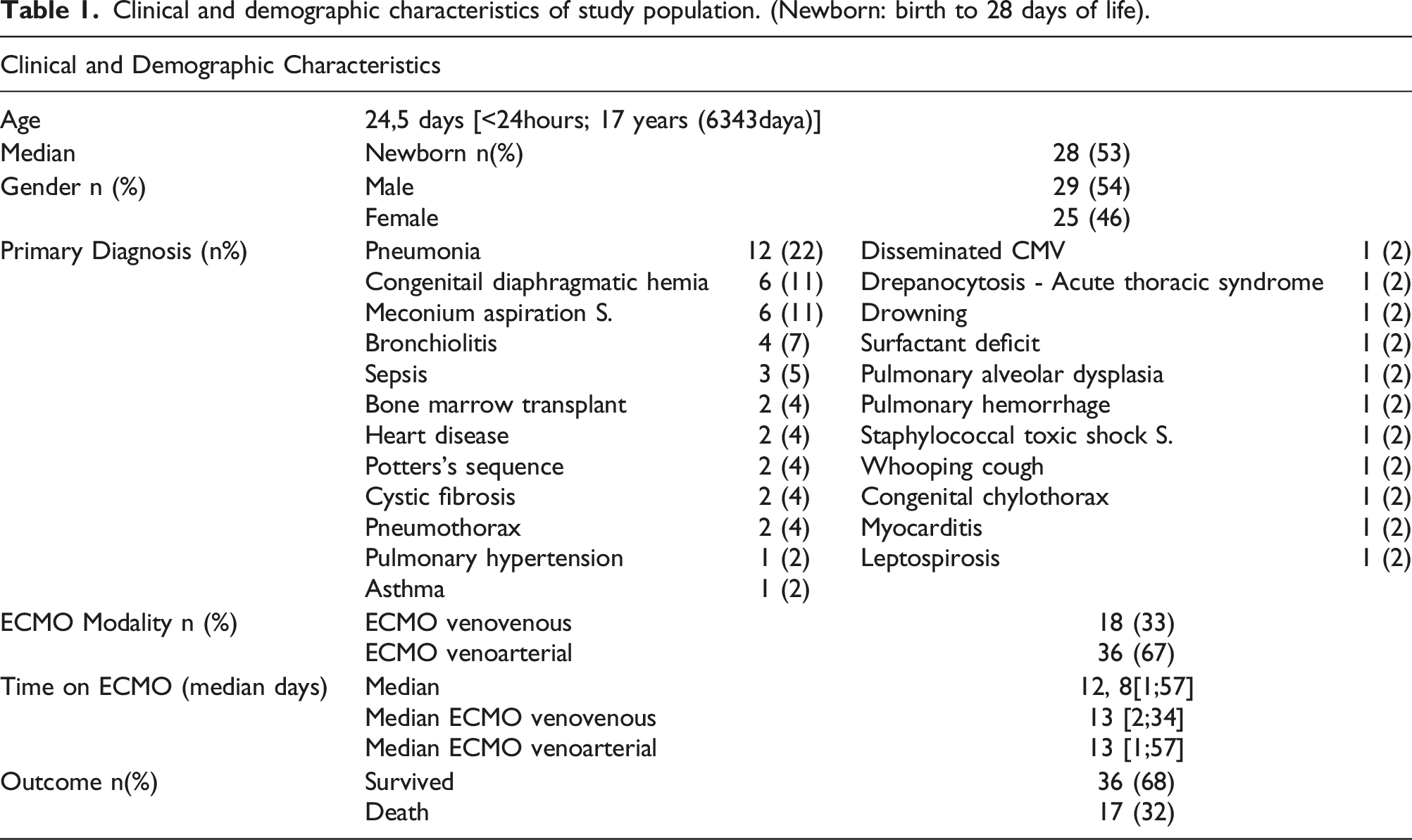
The highest percentage of transfusions occurred in groups with hemoglobin concentration below 6 g/dl and between 6 g/dl and 7 g/dl, with respectively, 3 (100%) and 38 (88.4%) leading to transfusion. When hemoglobin value was less than 7 g/dl, 89% (41/46) were transfused. With hemoglobin value between 7 g/dl and 8 g/dl, RBC transfusions were administered in 43 (40.2%). From the total of 794 laboratory results with hemoglobin value greater than or equal to 7 g/dl, only 181 (23%) were transfused (Figure 1, left graphic) Frequency of red blood cell transfusion for each hemoglobin interval (Left graphic: all samples (n = 1016); Right graphic: samples collected without active bleeding (n = 738)).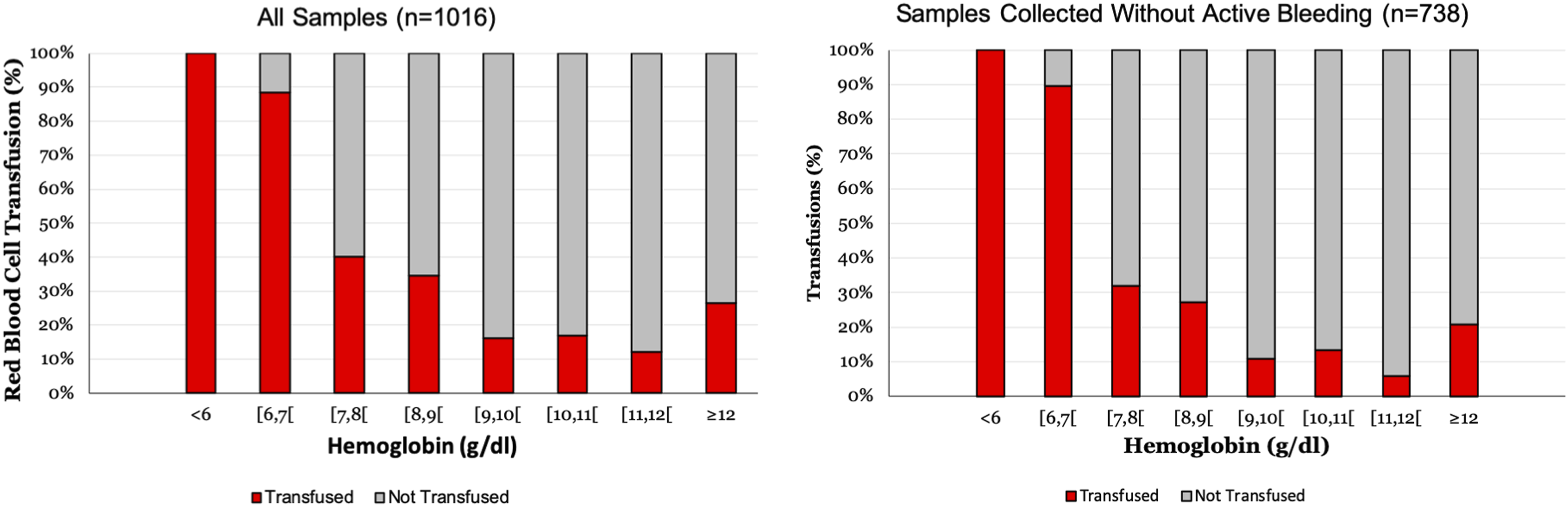
Excluding active bleeding, hemoglobin concentration below 6 g/dl motivated RBC transfusion in all cases and between 6 g/dl and 7 g/dl it was performed in 26 (89.7%). With hemoglobin between 7 g/dl and 8 g/dl, RBC transfusion decreased to 25 cases (32.1%). (Figure 1, right graphic)
Thirty-four (64%) ECMO runs, had at least one episode of active bleeding. Considering hemoglobin values during active bleeding, RBC transfusion was performed in all cases with hemoglobin concentration below 6 g/dl, 12 (85.7%) with hemoglobin between 6 g/dl and 7 g/dl and 18 (62%) with hemoglobin between 7 g/dl and 8 g/dl.
The presence of bleeding significantly increased the transfusion rate with hemoglobin values above 7 g/dl (p < .001). For values below 7 g/L, there is no statistical difference between the groups with and without active bleeding, as shown in Figure 2. Comparison between the frequency of red blood cell transfusion for each hemoglobin interval in case of active bleeding and excluding active bleeding.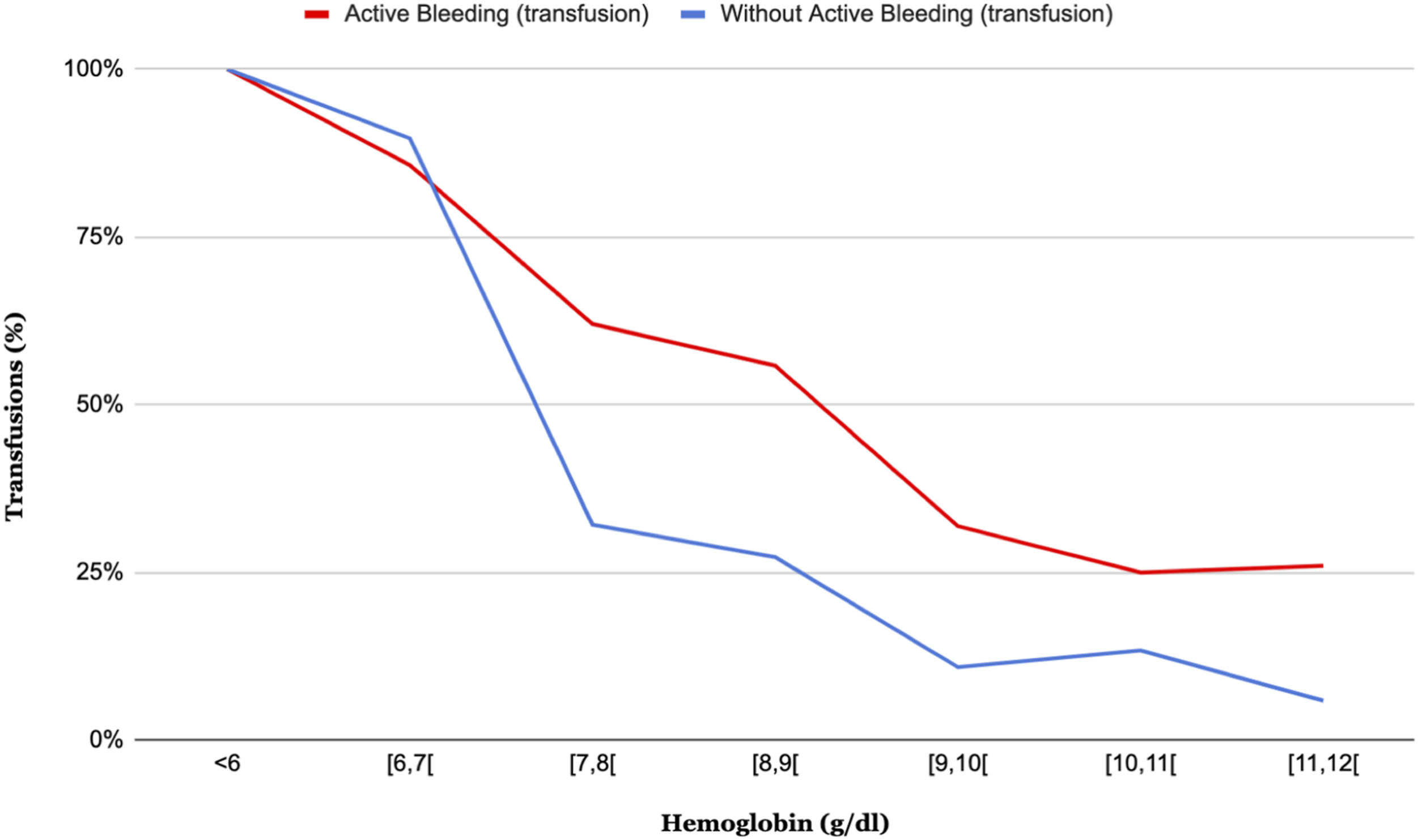
Red blood cells transfusion and outcome
From the total of 53 patients, 17 (32%) died and 36 (68%) recovered. The average number of RBC transfusions in patients who died was 5.6 transfusions compared to 3.5 transfusions in patients who recovered. However, there was no statistically significant difference between the number of transfusions and patient outcome.
Red blood cells transfusions over the years
The average pretransfusion hemoglobin values in the first 6 years of the study (2010–2015) was 9.3 ± 1.5 g/dl, decreasing to 7.98 ± 1.4 g/dl in the last 4 years (2016–2019) (p <0.001).
From 2010 to 2015, 6 out of 22 patients died (27%), compared to the last study period where 11 out of 31 patients died (35%). The outcome was not statistically different (p = .528) between these two periods.
Between 2010 and 2015, the mean duration of ECMO support was 12 days, in comparison with 14 days in the last 4 years.
Platelet transfusion
From the total of 1016 platelet values, the mean was 107 ± 3x109/L. A total of 147 platelet transfusions were performed, on average 2.7 ± 3.7 platelet transfusions for each ECMO run. The mean value for transfusion was 32x109/L.
Correlation between platelet value and platelet transfusion decision
From the total of 609 platelet values below 100 x 109/L, 145 (24%) led to platelet transfusion. When we stratified blood samples by platelet intervals, 44 (62%) samples with platelets below 20 x 109/L motivated transfusion. Thirty-eight (43%) samples with platelet values between 20 x 109/L and 30 x 109/L led to platelet transfusion, as well as 31 (40%) samples between 30 x 109/L and 40 x 109/L. On the other hand, 70 (91%) samples with platelet values between 40 x 109/L and 50 x 109/L were not associated with platelet transfusion. (Figure 3, left graphic) Frequency of platelet transfusion for each platelet interval (Left graphic: all samples (n = 1016); Right graphic: samples collected without active bleeding (n = 738)).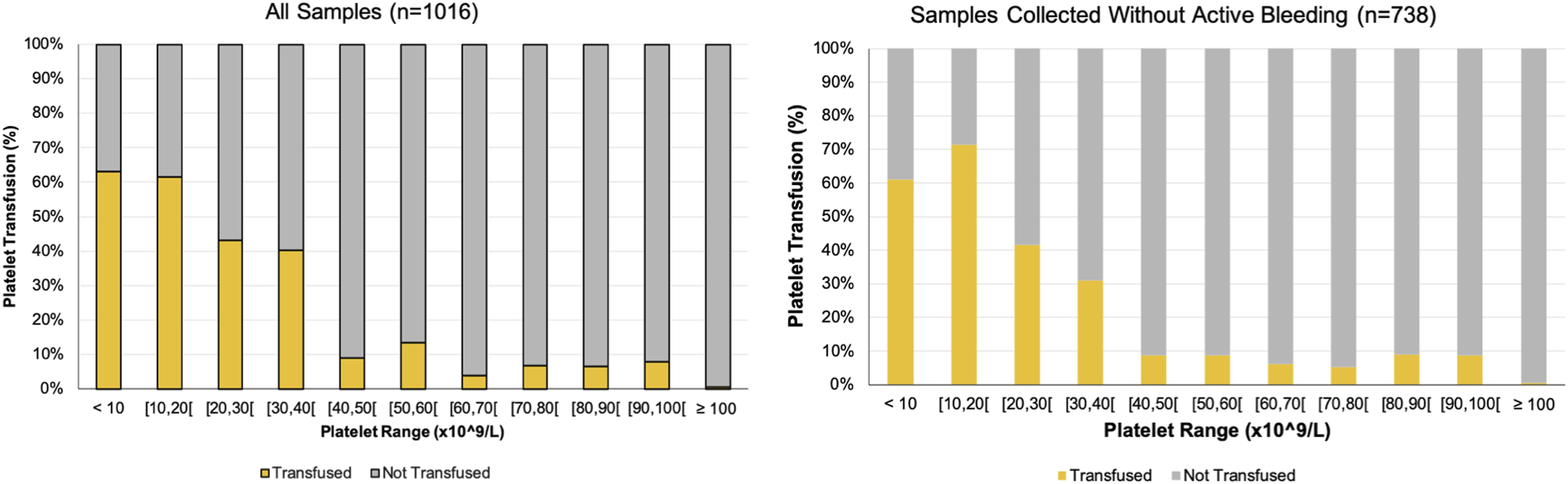
Excluding active bleeding, the total number of platelet transfusions was 92 (22%). Between 10 x 109/L and 20 x 109/L, the number of transfusions increased to 25 (71%). Twenty-two (42%) platelet values between 20 x 109/L and 30 x 109/L led to transfusion; 13 (31%) samples between 30 x 109/L and 40 x 109/L motivated transfusion. In contrast, only 5 (9%) samples with platelet values between 40 x 109/L and 50 x 109/L let to transfusion and the percentage of transfusions decreased further for higher platelet values. (Figure 3, right graphic).
When platelet values were between 30 x 109/L and 40 x 109/L, the number of platelet transfusions rose from 13 (31%) to 18 (51%) in the presence of bleeding, but this difference was not statistically significant (p = .068).
Platelet transfusion and outcome
Severe hemorrhagic complications (gastrointestinal, pulmonary and CNS bleeding) were described in eighteen (33%) patients. Gastrointestinal bleeding occurred in 8 (15%) cases, CNS hemorrhage in 6 (11%) and pulmonary hemorrhage in 4 (7%).
Severe bleeding complications and transfusion decision.
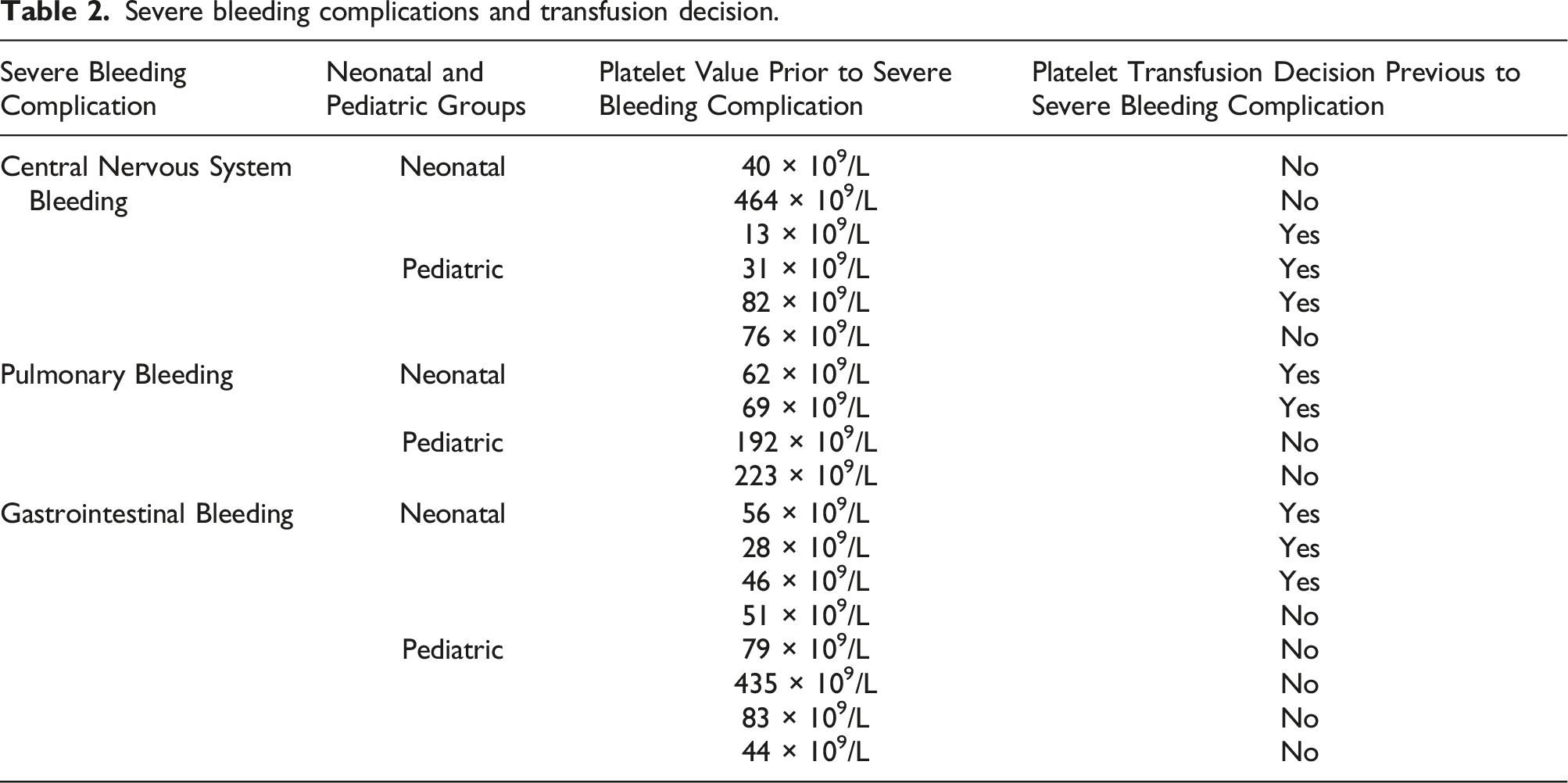
There were four patients with normal platelet values at the time of bleeding.
From the total of 19 cases with platelet counts below 10 x 109/L, none of these values was registered prior to a severe hemorrhagic complication. Between 10 x 109/L and 20 x 109/L, only one case of severe hemorrhagic complication was documented, it was specifically an intracranial hemorrhage in the neonatal period. (Table 2)
Platelet transfusions over the years
The average pretransfusion platelet values in the first 6 years of the study (2010–2015) was 35.05 x 109/L ± 24.77 x 109/L, decreasing to 27.35 x 109/L ± 14.29 x 109/L in the last 4 years (2016–2019) (p = .0369).
Half of the severe hemorrhagic bleeding episodes occurred from 2010 to 2015 (4 cases of CNS hemorrhage, 2 cases of pulmonary bleeding and 3 cases of gastrointestinal bleeding) and half from 2016 to 2019 (2 cases of CNS hemorrhage, 2 cases of pulmonary bleeding and 5 cases of gastrointestinal bleeding).
Discussion
Patients supported with ECMO are frequently exposed to large volumes of blood products, including RBC and platelets. 9 It is unclear whether the benefits of RBC transfusion to support near-normal hematocrit while on ECMO outweigh the risks. Several studies have demonstrated that RBC and platelet transfusion are associated with an increased mortality in critically ill pediatric patients.2,9 However, pediatric patients supported with ECMO are commonly excluded from those studies, so limited evidence is available to guide clinical practice.2,9
Red blood cell transfusion
The threshold for RBC transfusion was evaluated using hemoglobin, lactate and SvO2, important tissue oxygenation markers. Hemoglobin value was used instead of hematocrit for its clinical applicability, in line with recent recommendations. 15
Venous oxygen saturation presented a significant increase post-transfusion, which means an improvement in oxygen delivery to tissues, but the mean value before transfusion was 75% showing that oxygenation was already adequate and not compromised by the restrictive policy used. The hemoglobin threshold below which the majority of patients (89%) was transfused was 7 g/dl. In the hemoglobin range between 7 g/dl and 8 g/dl, the presence of active bleeding was an important factor for transfusion decision. It is of note that most patients in this interval were not transfused if they were not bleeding. When the first 6 years of the study were compared with the last 4 years, the mean pretransfusion hemoglobin value decreased. Over the years, we believe the experience of the ECMO team allowed to reduce the hemoglobin threshold for RBC transfusion, without compromising the outcome.
There is no defined RBC transfusion threshold for pediatric patients supported with ECMO. Extracorporeal Life Support Organization recommends to keep hematocrit above 40% during ECMO to improve oxygen delivery. 16 An international survey, reported lower hematocrit threshold for RBC transfusion for pediatric ECMO, ranging 25%–40%. 17 However, a more recent conference series of international experts in RBC transfusion management of critically ill children, reviewed the existing data and concluded that “there is insufficient evidence to recommend a specific RBC transfusion decision-making strategy” for critically ill children on ECMO. 15
Lacroix J et al., demonstrated that a restrictive transfusion strategy (hemoglobin threshold of 7 g/dl) was as safe as a liberal transfusion strategy (hemoglobin threshold of 9.5 g/dl) in pediatric patients whose condition was stable in PICU. In critically ill adult patients, rates of organ failure and in-hospital deaths were higher with a liberal transfusion strategy. 18 Multiple randomized controlled trials of restrictive versus liberal transfusion strategies in critically ill adults and children have failed to identify benefit to liberal RBC transfusion, however, these studies do not include patients on ECMO support. 19
Red blood cell transfusion could be associated with several risks, such as, immunomodulation, transfusion-related respiratory dysfunction and thrombotic complications.20–24 Several studies have demonstrated that RBC transfusion volume was independently associated with odds of mortality. Two single-center studies reported an increased in adjusted odds of death (9% and 24%) for each additional 10 mL/kg/day of RBC transfusion volume, during pediatric ECMO support.2,7 Our findings are consistent with previous reports and patients who died received a greater number of transfusions, although this difference was not significant. The small number of patients in our cohort could have compromised the statistical evaluation of outcome.
Our Pediatric ECMO center uses a restrictive transfusion policy, without compromising outcome. The mortality rate in our ECMO center is similar to other reference centers in Europe, with 68% of neonatal and pediatric patients surviving to hospital discharge or transfer. 25
Platelet transfusion
Platelet transfusion is not an innocuous procedure and it could be associated with several complications including infections, febrile reactions, thromboembolism and clotting of the ECMO circuit components. A prospective study with 511 children on ECMO, showed that platelet transfusion volume was independently associated with thrombotic events and mortality. 26 Nevertheless, currently transfusion protocols are based on institutional experience with limited evidence to support them.
Quantitative and qualitative platelet dysfunction during ECMO is common and is associated with bleeding and mortality. Several mechanisms could explain platelet dysfunction, including increased destruction, decreased production, sequestration in extravascular sites and platelet aggregation in the circuit tubing. Platelet transfusions are frequently required during pediatric ECMO. However, data available regarding the platelet transfusion threshold remain limited. 27
Despite the platelet transfusion threshold defined by ELSO guidelines of 80 x 109/L, the platelet goals ranged across ECMO centers from 67 x 109/L to 113 x 109/L for VA ECMO and 50 x 109/L to 100 x 109/L for VV ECMO, according with a recent international study. 26
In our study, during the 10-years studied, 147 platelet transfusions were performed. The mean value before transfusion was 32 x 109/L and most transfusions were carried out with platelet values below 20 x 109/L. For platelet values between 30 x 109/L and 50 x 109/L most patients were not transfused. Active bleeding was an important decision factor in this group of patients.
Several studies demonstrated that more liberal platelet transfusion strategies increased complications during ECMO support and mortality, as compared to restrictive platelet transfusion strategies.13,26,28
Our Pediatric ECMO center uses a restrictive transfusion policy, with a mean platelet transfusion threshold of 32 x 109/L, without compromising outcome. Most severe hemorrhagic complications (gastrointestinal, pulmonary and CNS bleeding) occurred with platelet values above 60 x 109/L and some patients even had normal values at the time of bleeding. This supports the idea that lower values are safe and were not associated with severe bleeding complications.
The European Complication Trend Report, October 2021, which included neonatal and pediatric ECMO patients, reported 5% of CNS hemorrhage, 9% of pulmonary bleeding and 3% of gastrointestinal bleeding complications between 2010 and 2019. 29 According to the literature, CNS hemorrhage occurs with rates of 3–11% with higher rates in the neonatal group. In our study, CNS hemorrhage was described in 11%, which is in line in literature. 30 The high percentage of neonatal patients (53%) included in our cohort, could explain the higher number of CNS hemorrhage. The pulmonary bleeding complications reported in our study were similar to the ELSO report and other retrospective studies from Pediatric ECMO centers. 31 Gastrointestinal hemorrhagic complications represented 15% of the total of severe bleeding complications, a higher value than usually reported by ELSO. In our study, all cases of gastrointestinal hemorrhage were reported, including mild bleeding from the nasogastric tube, which could explain the results.
This study is one of the first to investigate the transfusion policies in pediatric patients on ECMO support. Furthermore, this investigation allowed to study the relationship between RBC transfusion and global indices of tissue oxygenation in pediatric patients supported with ECMO.
However, our study is not without limitations. As an observational study, laboratory variables were assessed at the medical records and thus not limited to one specific time window of assessment. The regular blood tests performed in these patients could ameliorate this limitation.
We did not investigate the effect of other clinical variables such as age, underlying pathology and ECMO type on transfusion decision. Age-related physiological changes of the coagulation system could be important in transfusion decision. 32 Moreover, in critically ill children, the same underlying pathology can induce thrombocytopenia and coagulation abnormalities, increasing the risk of bleeding complications. 33 Considering the small series of this study, it was not possible to stratify by age group or pathology without losing statistical power. Randomized, controlled and multicenter trials are needed to evaluate the influence of those variables on transfusion policy for pediatric ECMO.
In this study, for methodologic reasons RBC and platelet transfusions were studied separately. However, the outcome could be influenced by both factors and the interactions between RBC and platelet transfusion could be explored in future studies.
Our cohort did not include post cardiotomy patients, so a restrictive transfusion approach should not be generalized to this group of patients.
Finally, the study was conducted in a single low volume Pediatric ECMO center, so it would be relevant to design a multicenter study to confirm these results. Nonetheless, we consider that our study could support the idea that a restrictive transfusion policy is safe and help in the design of larger prospective studies.
Conclusions
Using a restrictive RBC and platelet transfusion policy was safe, reducing the number of blood products transfusions. In this case series, a restrictive transfusion policy allowed to reach a good outcome, with similar hemorrhagic complications and mortality rate as other European ECMO centers. Prospective studies are needed to complete the investigation about transfusion policies in patients undergoing ECMO, in order to change current guidelines for RBC and platelet transfusions on pediatric ECMO.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
