Abstract
Objectives
To find the imaging mortality predictors in patients with previous myocardial infarction (MI), symptomatic heart failure (HF), and reduced left ventricle (LV) ejection fraction (EF).
Methods
for the study 39 patients were selected prospectively with prior MI, symptomatic HF, and LVEF ≤40%. All patients underwent transthoracic echocardiography (TTE), single-photon emission computed tomography myocardial perfusion imaging (SPECT MPI), 18F-FDG positron emission tomography (FDG PET). 31 patients underwent cardiovascular magnetic resonance (CMR) with late gadolinium enhancement (LGE). Patients were divided into two groups: 1 group – cardiac death; 2 group – no cardiac death. Myocardial scars were assessed on a 5-point-scale. Follow-up data was obtained.
Results
Imaging features disclosed significant difference (p < 0.05) of defect score (CMR and SPECT-PET), LV end-diastolic diameter (EDD) (TTE), LVEDD index (CMR), LV global longitudinal strain (CMR) and LV global circumferential strain (CMR) between the groups. Predictors of cardiac death were: LVEDD index (TTE) and LV global longitudinal strain. The cut-off values to predict cardiac death were: defect score (CMR) 25 (AUC, 79.5%; OR 1.8, 95% CI 1.2–2.7), SPECT-PET defect score 22 (AUC, 73.9%; OR 0.5, 95% CI 0.3–0.7), LVEDD (TTE) 58 mm (AUC, 88.4%; OR 23.6, 95% CI 2.6–217.7), LVEDDi 30 mm/m2 (TTE) (AUC, 73.6%; OR 22.0, 95% CI 1.9–251.5), LVEDDi 33.6 mm/m2 (CMR) (AUC, 73.6%; OR 22.0, 95% CI 1.9–251.5), LV global longitudinal strain −13.4 (AUC, 87.8%; OR 2.1, 95% CI 1.2–3.7) and LV global circumferential strain −16.3 (AUC, 76.1%; OR 1.9, 95% CI 1.2–3.0).
Conclusions
Imaging features, such as defect score (CMR) >25, SPECT-PET defect score >22, LVEDD (TTE) >58 mm, LVEDDi (TTE) >30 mm/m2, LVEDDi (CMR) >33.6 mm/m2, LV global longitudinal strain −13.4 and LV global circumferential strain −16.3, may increase sensitivity and specificity of FDG PET and LGE CMR predicting of late mortality.
Keywords
Introduction
Myocardial viability is a dysfunctional area of the myocardium that has a chance for functional recovery when the blood supply to the myocytes is restored.1–3 Identifying the myocardial viability and the need for coronary revascularization is one of the main targets in patients with ischemic cardiomyopathy when left ventricle (LV) systolic function is impaired.
There are different imaging modalities currently validated for myocardial viability evaluation. Single-photon emission computed tomography (SPECT) is associated with thallium or technetium uptake evaluating the integrity of myocyte membrane and positron emission tomography (PET) shows the uptake of fluorodeoxyglucose as a marker of glucose metabolism in viable hibernating myocardium. Another convenient myocardial viability imaging dobutamine stress echocardiography (DSE) – during inotropic stimulation reveals contractile reserve representing viable myocardium, and cardiac magnetic resonance (CMR) using late gadolinium enhancement (LGE) evaluates the regions with increased extracellular volume as an indication of myocardial scar.
Literature data regarding myocardial viability evaluation imaging method and value on outcome prediction shows conflicting results.4–7 Therefore, we performed the study to make “head-to-head” comparison of different imaging modalities for the most appropriate evaluation of myocardial viability. Furthermore, we made the follow up with the aim to identify the clinical and imaging mortality predictors in patients with previous myocardial infarction (MI), symptomatic heart failure (HF), and reduced LV ejection fraction (EF).
Methods
Study population
39 patients referred to the Cardiology department of Lithuania Health Sciences University Hospital Kaunas Clinics due to ischemic symptomatic HF were selected prospectively to the study. The Inclusion criteria were medical history of previous MI, symptomatic HF with the New York Heart Association (NYHA) functional class II, III or IV, reduced LV EF, ≤40%, and advanced coronary artery atherosclerosis evaluated by invasive coronary angiography when the need for percutaneous intervention was questionable because of suspected myocardial scar tissue.
Clinical data from medical documentation were collected and analyzed: age, gender, comorbidities, medications, symptoms of heart failure or other diseases, risk factors of cardiovascular disease, data of objective investigation, electrocardiographic findings. All patients underwent physical examination, Six Minute Walk Test (6 MWT) evaluation, conventional transthoracic echocardiography (TTE) at rest, rest myocardial perfusion imaging with (SPECT MPI), 18F-fluorodeoxyglucose PET(FDG PET), and cardiovascular magnetic resonance (CMR) with late gadolinium enhancement (LGE) and strain-encoded CMR (SENC). All the subjects gave their informed consent for inclusion before participating in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by Kaunas Regional Biomedical Research Ethics Committee (Project identification code BE 2–29).
Transthoracic echocardiography
Transthoracic echocardiography was performed in all patients. All measurements were obtained according to the valid guidelines (the European Society of Cardiology (ESC), the European Association for Cardio-Thoracic Surgery (EACTS), and American Heart Association (AHA)). 8 The conventional transthoracic 2D echocardiography system (EPIQ 7, Phillips Ultrasound, Inc., Bothell, WA, USA) with 1.5–4.6 MHz transducer was used. We evaluated all morphometric assessments of the chambers using 2D-guided linear and tracing (for volumes evaluation) measurements. In the parasternal long-axis view we performed linear measurements of the left heart: LV end-diastolic diameter (LVEDD) and LV end-systolic diameter (LVESD). Evaluation of the volumes (LV end-diastolic volume (LVEDV), LV end-systolic volume (LVESV)) was achieved by biplane apical tracing. LV exafaraday was calculated using modified Simpson’s biplane method.
Cardiovascular magnetic resonance
CMR: Study protocol
All CMR images were acquired using a 1.5 Tesla scanner (Siemens Magnetom Aera, Siemens AG Healthcare Sector, Erlangen, Germany) with an 18-channel phased array coil in a supine position. The study protocol included an initial survey to define imaging planes.
Cine images were acquired using retrospectively gated balanced steady-state free precession (SSFP) sequence with short periods of breath-holding in three LV long-axis (2-chamber, 3-chamber, and 4-chamber) planes. The ventricular 2-chamber and 4-chamber planes were used to plan a contiguous stack of short-axis slices covering the entire LV. The in-plane resolution of cine images was 0.9 × 0.9 mm, slice thickness of 8 mm with 2 mm interslice gap and 25 phases per cardiac cycle. The following parameters were used: repetition time (TR) = 3.3 ms, echo time (TE) = 1.6 ms, flip angle = 60°, acquisition voxel size = 1.8 × 1.8 × 8.0 mm3, and 25 phases per cardiac cycle.
The late gadolinium enhancement (LGE) images were obtained 10 min after the injection of 0.15 mmol/kg gadobutrol (Gadovist®, Bayer Schering Pharma AG, Berlin, Germany). A Look-Locker sequence was acquired to determine the inversion time to null the signal of the LV myocardium. A 2D inversion recovery fat-saturated spoiled gradient-echo sequence was used to detect scar tissue in three LV long-axis and short-axis orientations.
CMR: Image analysis
All cine images were analyzed offline using Medis Suite, version 3.0 (Leiden, The Netherlands) under a recent consensus document for quantification of LV function and mass using CMR. 9 The end-diastolic and end-systolic cardiac phases were detected visually. After manual contouring of endocardial and epicardial borders, LVEDV, LVESV, LVEF, LVEDM were calculated. Papillary muscles were considered part of the blood pool. LV volumes and myocardial mass were adjusted to body surface area, determined using the Mosteller equation.
The endocardial and epicardial contours drawn on cine images were transferred into LGE images. The presence and extent of LGE were quantified using the signal threshold versus reference mean (STRM) >3 standard deviations (SD) method as it provides the highest accuracy with acceptable reproducibility compared with other signal intensity threshold techniques. 10 The total LGE volume and mass were calculated automatically.
For quantification of myocardial viability, all short-axis images were segmented using a 17-segment model. The LGE extent was assessed and quantified in each short-axis segment by the 5-point scoring system: score 0 = LGE, viable myocardium; score 1 = LGE 0–25% of wall thickness; score 2 = LGE 25–50% of wall thickness; score 3 = LGE 50–75% of wall thickness; score 4 = transmural scar (no viability). The sum of the scores was calculated and named defect score CMR.
Cardiac nuclear medicine imaging
Single-photon emission computed tomography MPI and FDG PET viability analysis were performed by a single-blinded investigator. Myocardial perfusion and viability images were reconstructed and analyzed in the 17-segment model.
SPECT MPI: Study protocol
All patients underwent rest-only ECG gated SPECT myocardial perfusion imaging, after 60 min after the technetium 99 m (99 mTc) labeled sestamibi intravenous injection, adjusted to body mass (280 ± 65 MBq). ECG gated SPECT MPI imaging was performed with a dual-head BrightView XCT (Philips healthcare) gamma camera, using a low-energy, high-resolution collimator, a 20% window at 140 keV, 64 × 64 matrices, an orbit with 120 projections, at 3-degree steps, and a 20 s per step in a supine position with the arms held above the head.
SPECT MPI: Image analysis
Gated and non-gated rest-only SPECT MPI images were reconstructed using OSEM iterative reconstruction, with the dedicated Philips workstation. Analysis was performed using Cedars-Sinai QGS/QPS software package.
Perfusion defects in each LV 17 segments were scored using a 5-point scoring system as follows: 0 - normal perfusion, 1 - minimal perfusion defect, 2 - moderate perfusion defect, 3 - severe perfusion defect, 4 - no perfusion/perfusion defect. And summed rest score (SRS) was obtained in all LV by summing the scores of the corresponding 17 segments.
FDG PET: Study protocol
ECG gated FDG PET myocardial viability imaging was performed in our study. All patients were intravenously injected with an 18F-FDG adjusted to the body mass (4 MBq/kg), after fasting for at least 6 h. The following oral glucose and insulin loading protocol was used in all patients. First, the patient’s glucose level was measured. In nondiabetic patients, 50 g oral glucose was administered if blood glucose level was 8.33 mmol/L and below, and 25 g glucose if blood glucose level was 8.33–13.8 mmol/L. The physician was notified if blood glucose was >11.11 mmol/L. In diabetic patients, 25 g glucose was administered if blood glucose was 8.33 mmol/L and below, and 12.5 g glucose for blood glucose of 8.33–13.8 mmol/L. No glucose was administered if blood glucose was >13.8 mmol/L. 30 min after glucose load blood glucose level was measured, if blood glucose level was below 7.77 mmol/L, repetitive blood glucose level was measured after the 15 min and 18F-FDG was injected intravenously. If the blood glucose level was 7.78 mmol/L and more, adjusted to the blood glucose level insulin injection was made, and the blood glucose level measurement was repeated.
Positron emission tomography images were obtained 60 min after 18F-FDG injection using Discovery XCT PET/CT (Ge Medical Systems, USA). The duration of PET acquisition was 15 min following a low-dose CT scan for attenuation correction.
PET and MPI comparative analysis
Single-photon emission computed tomography MPI and FDG PET viability images were compared visually using short axis, horizontal, and vertical long-axis images as well as polar maps.
Myocardial viability was assessed with visual analysis scoring according to SPECT MPI and FDG PET. Score 4 (absent uptake) was evaluated as non-viable myocardium, score 1–3 (mild, moderate, and severe defect) was described as viable myocardium with a chance of functional recovery after revascularization, and score 0 (normal perfusion) was reported as viable myocardium. The sum of SPECT MPI – FDG PET defect scores was calculated and named SPECT-PET defect score.
Follow-up
We obtained the late follow-up data from the clinical follow-up or telephone interviews made by our investigators-cardiologists.
Statistical analysis
Quantitative variables were expressed as mean (M) ± standard deviation (SD) and categorical variables as frequencies and percentages. The student’s t-test was used for comparisons of continuous variables. The quantitative variable’s normality assumption was verified using the Kolmogorov-Smirnov test.
Univariate logistic regression analysis was performed to determine possible predictors for late mortality. Stepwise logistic regression was performed to identify independent predictors of late mortality. Results were reported as effect sizes [odds ratios (ORs)] with 95% confidence intervals (CIs). Logistic regression was used to reduce the set of patient characteristics into a probability of developing the outcome of interest, late mortality. These model-based probabilities were analyzed using receiver operating characteristic (ROC) curves. ROC curves are a graphic representation of the trade-off between the false-negative and false-positive rates for every possible cut-off probability of death. The area under the curve (AUC) summarizes information about the outcome contained in the ‘‘predictor’’ set, with a value of 1 as maximum.
All reported p values were two-sided, and p values of <0.05 were considered to indicate statistical significance. SPSS software (IBM, SPSS Statistics, Version 22) was used for statistical testing.
Results
Study population
Baseline characteristics.

Continuous variables are expressed as mean (M) ± standard deviation (SD) for symmetric data. Categorical variables expressed as count and percentage of patients.
Abbreviations: BMI, body mass index; CAD, coronary artery disease; STEMI, ST-elevation myocardial infarction; NSTEMI, non-ST elevation myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass grafting; COPD, chronic obstructive pulmonary disease; ICD, implantable cardioverter-defibrillator; NYHA, New York Heart Association; 6 MWT, 6-minute walk test; LAD, left anterior descending artery; CX, circumflex artery; RCA, right coronary artery; ACEI/ARB, angiotensin-converting enzyme inhibitors/angiotensin receptor blockers; CCB, Calcium channel blockers.
Cardiovascular imaging
All patients underwent TTE, MPI SPECT and FDG PET, while CMR was performed in 31 patients (good quality CMR was obtained not for all patients because of arrhythmia, pacemakers, or implantable cardioverter-defibrillators) (Figure 1). According the follow-up results patients were devided into two groups (1 group – cardiac death; 2 group – no cardiac death). The survival rate of whole patients was 74.4% (during the follow-up period died 10 patients, survived 29 patients). The clinical decision on medical treatment alone or combined with revascularization (percutaneous intervention (PCI) or coronary artery bypass graft (CABG) surgery) was more dependent on clinical manifestation of MI induced symptomatic HF, and cardio surgeon ability to perform PCI or CABG for a specific patient in a particular clinical situation. These clinical decisions were not always influenced by the imaging recommendations based on myocardial viability. Nevertheless patient’s wishes, consent to surgery, and comorbidities also affected treatment strategy. Myocardial viability evaluation by nuclear medicine imaging (SPECT and FDG-PET) and cardiovascular magnetic resonance (CMR with LGE and SENC). A- rest myocardial perfusion scintigraphy demonstrates perfusion defect in anterior and apical segments (SPECT defect score 24); B- FDG PET viability imaging represents the viable myocardium in the affected zone (PET defect score 10, PET reversibility score 14); C-D-E- CMR LGE images demonstrate transmural scar in anterior and apical LV segments (CMR defect score 27); F-D- CMR SENC imaging shows changes in myocardial deformation: Fglobal longitudinal strain -13.0, G- global circumferential strain -22.7.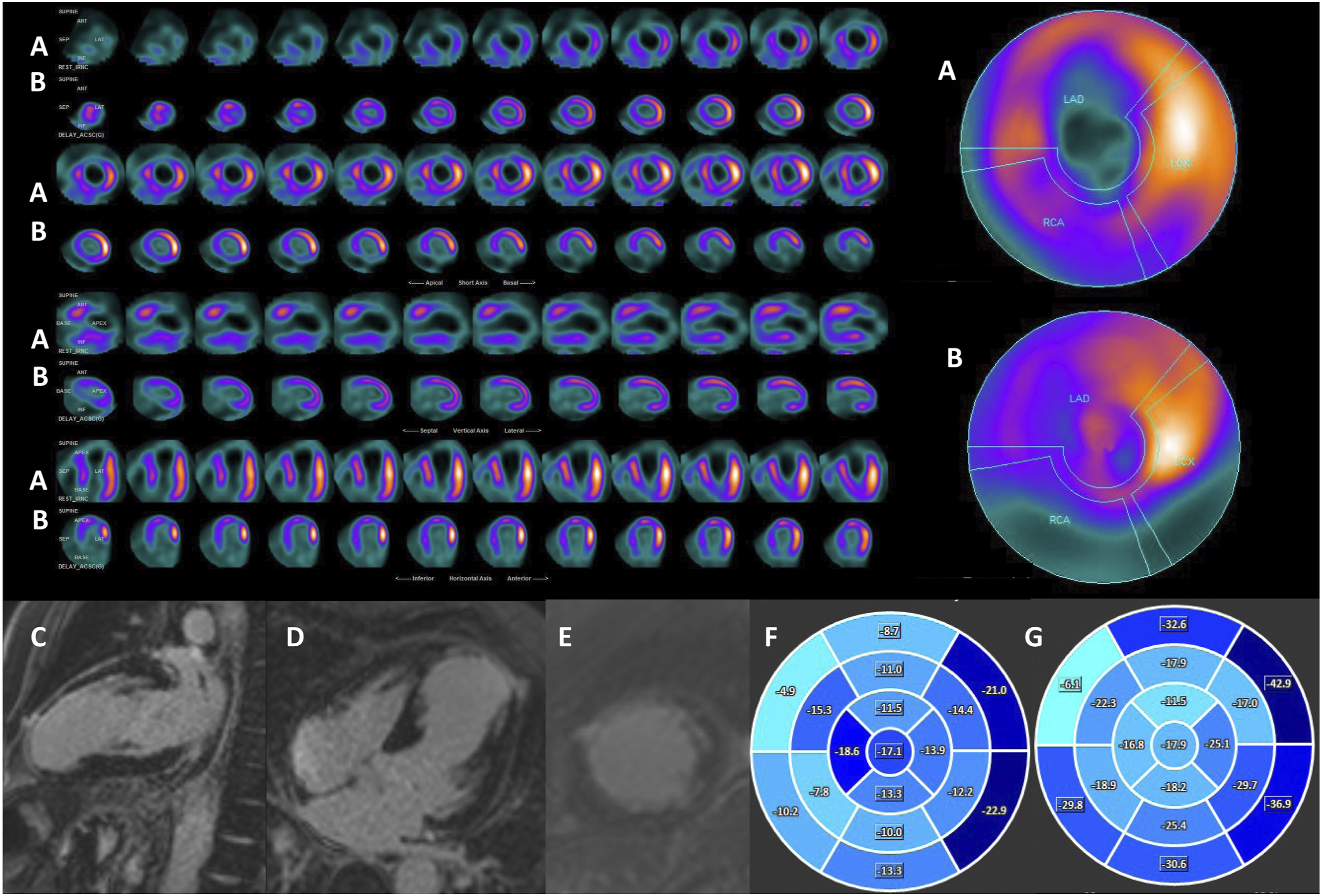
Myocardial viability was evaluated as significant in 21 patients (53.8%), however only 12 patients received recommended treatment (revascularization), others (n = 9) received medical treatment alone. Myocardial viability was absent in 18 patients (46.2%), nevertheless 6 patients received revascularization and 12 patients received medical treatment alone. Treatment strategy according to the viability results was selected in 24 patients (61.5%) and not according to the viability results in 15 patients (38.5%).
Comparison of two groups and univariate analysis for predicting cardiac death (1 group – cardiac death (n = 8 CMR LGE group, n = 10 SPECT-PET group), 2 group – alive patients (n = 23 CMR group, n = 29 SPECT-PET group).
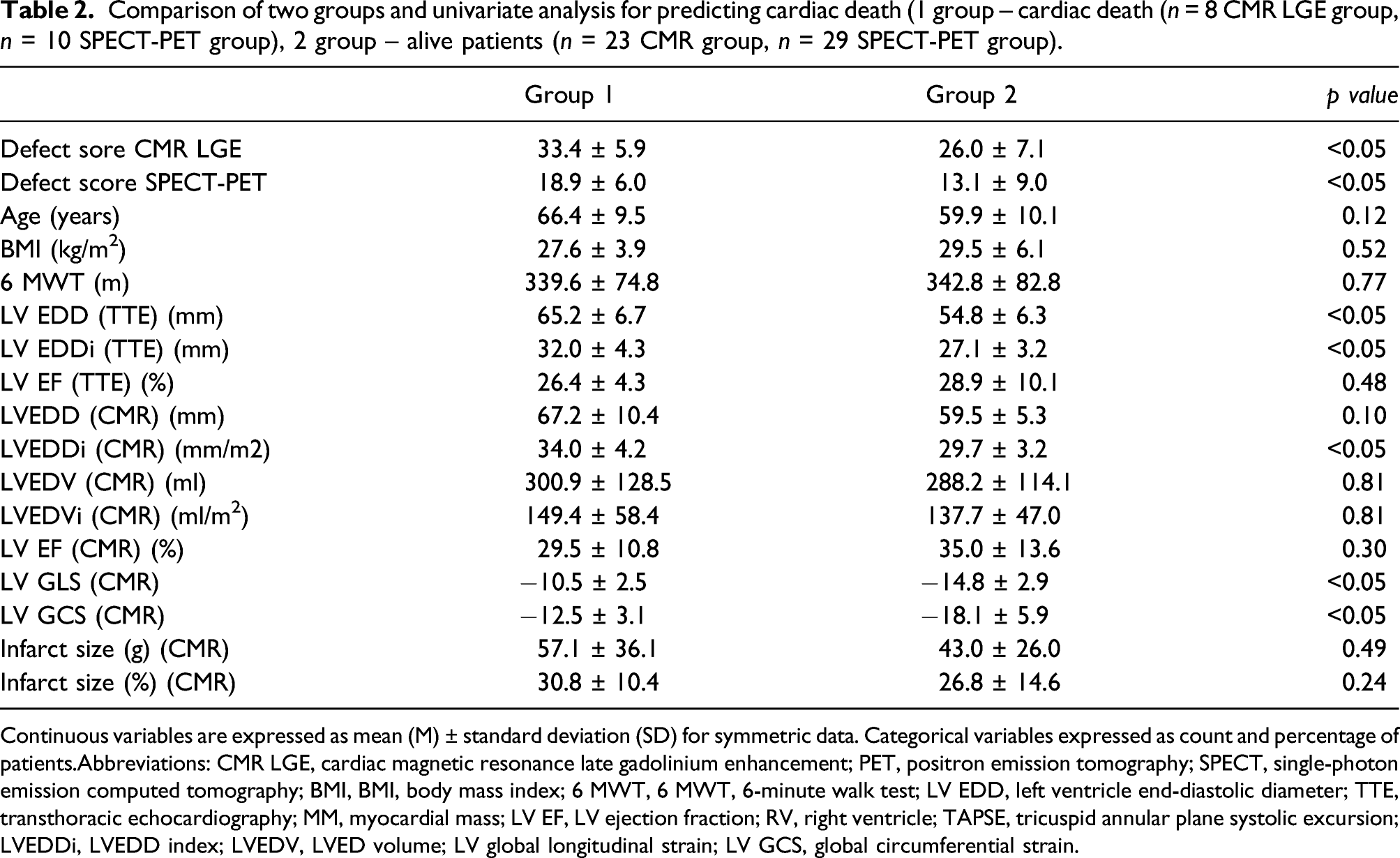
Continuous variables are expressed as mean (M) ± standard deviation (SD) for symmetric data. Categorical variables expressed as count and percentage of patients.
Abbreviations: CMR LGE, cardiac magnetic resonance late gadolinium enhancement; PET, positron emission tomography; SPECT, single-photon emission computed tomography; BMI, BMI, body mass index; 6 MWT, 6 MWT, 6-minute walk test; LV EDD, left ventricle end-diastolic diameter; TTE, transthoracic echocardiography; MM, myocardial mass; LV EF, LV ejection fraction; RV, right ventricle; TAPSE, tricuspid annular plane systolic excursion; LVEDDi, LVEDD index; LVEDV, LVED volume; LV global longitudinal strain; LV GCS, global circumferential strain.
Binary logistic regression analysis model predicting cardiac death (coefficient of determination (R2) is 0.72).

Abbreviations: CMR, cardiac magnetic resonance; CI, confidence interval; LVEDDi, left ventricle end-diastolic diameter index; LV GCS, global circumferential strain.
ROC analysis for predicting mortality (1 group – cardiac death (n = 8 CMR LGE group, n = 10 SPECT-PET group), 2 group – alive patients (n = 23 CMR group, n = 29 SPECT-PET group).
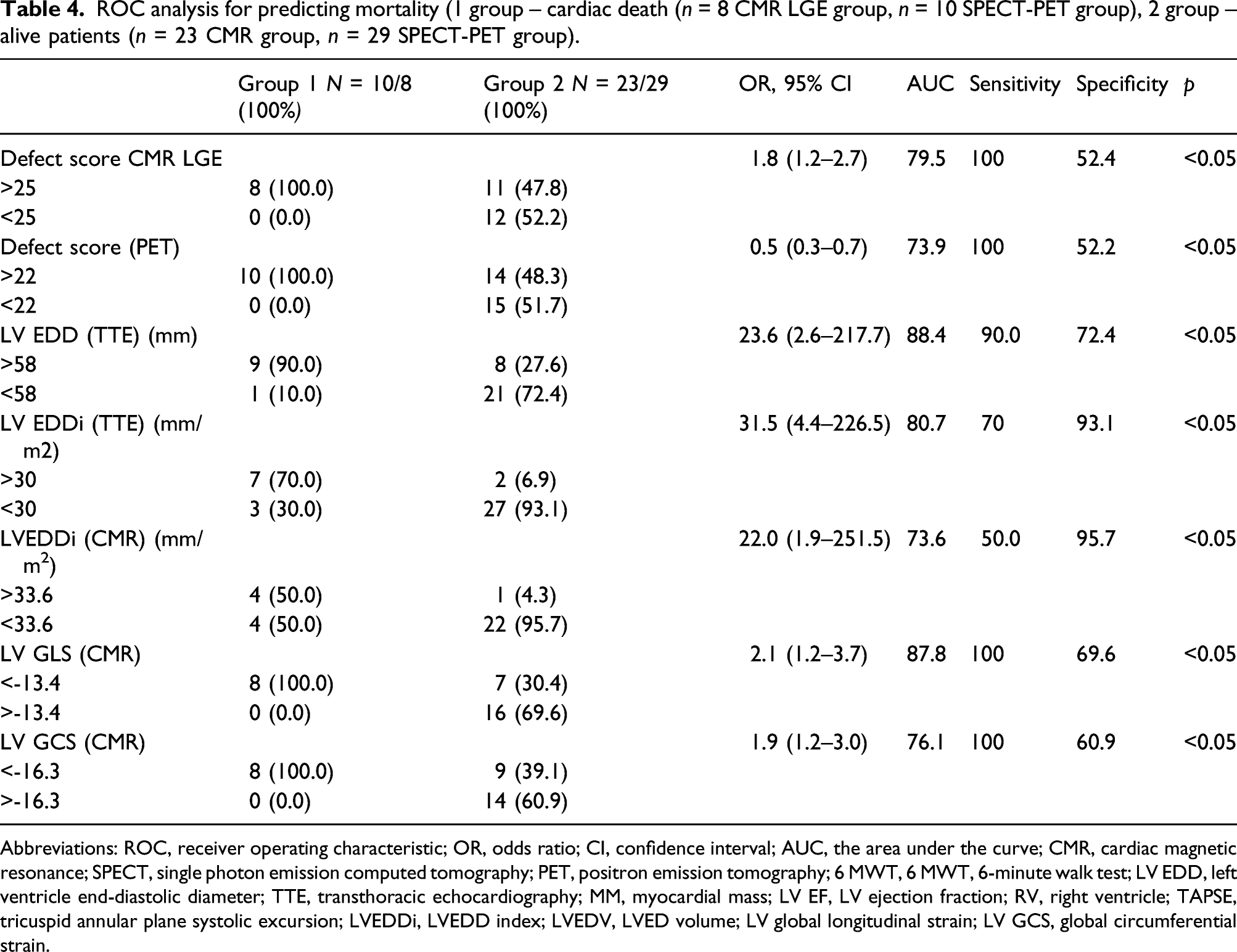
Abbreviations: ROC, receiver operating characteristic; OR, odds ratio; CI, confidence interval; AUC, the area under the curve; CMR, cardiac magnetic resonance; SPECT, single photon emission computed tomography; PET, positron emission tomography; 6 MWT, 6 MWT, 6-minute walk test; LV EDD, left ventricle end-diastolic diameter; TTE, transthoracic echocardiography; MM, myocardial mass; LV EF, LV ejection fraction; RV, right ventricle; TAPSE, tricuspid annular plane systolic excursion; LVEDDi, LVEDD index; LVEDV, LVED volume; LV global longitudinal strain; LV GCS, global circumferential strain.
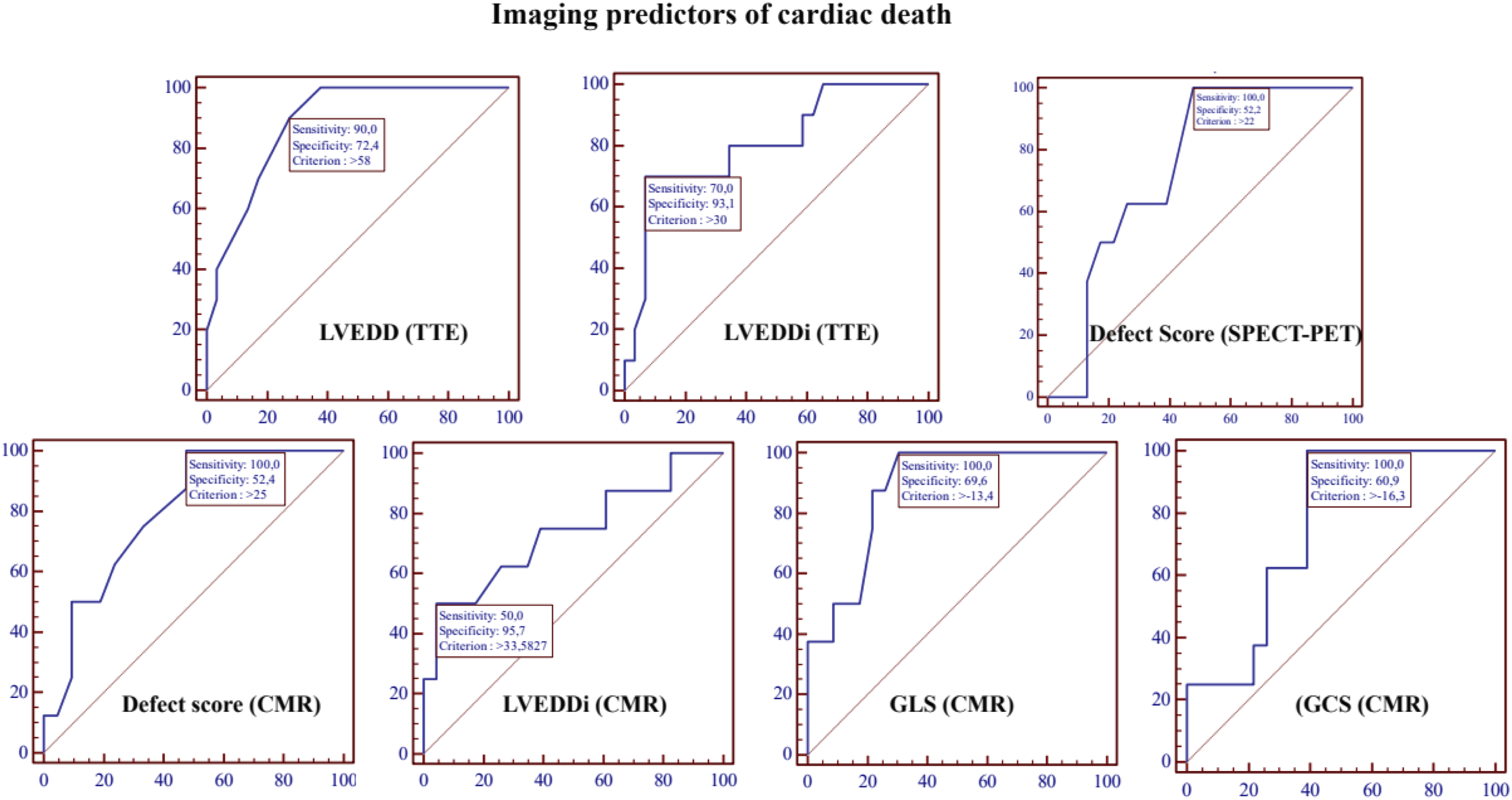
Echocardiographic, nuclear medicine and cardiovascular magnetic resonance imaging predictors of cardiac death.
Discussion
The main finding of our study was that CMR defect score >25 and SPECT-PET defect score >22, LVEDD (TTE) >58 mm, LVEDDi (TTE) >30 mm/m2, LVEDDi (CMR) >33.6 mm/m2, LV GLS (CMR) −13.4 and LV GCS -16.3 imaging features and their cut-off values may increase sensitivity and specificity of SPECT MPI – FDG PET and LGE CMR predicting cardiac death.
Moreover, the present study demonstrates not only a “head-to-head” comparison of SPECT MPI – PET, and CMR imaging for the evaluation of myocardial viability, but also application of CMR SENC as the additional tool to increase sensitivity and specificity of LGE CMR.
During the last decades in clinical cardiology, the benefit of the evaluation of myocardial viability was supported by a wide range of retrospective observational studies.5,11 Myocardial viability in several studies was evaluated as an independent predictor of cardiac death in patients with ischaemic LV dysfunction when revascularisation is not performed,11,12 while other studies demonstrated that the amount of viable myocardium has to be sufficient, therefore the significant extent of viable myocardium (more than 10%) is an independent predictor of survival in patients undergoing revascularisation. 13
However, recent studies suggest that the value of myocardial viability in clinical decision-making remains controversial. Three prospective randomized trials with viability testing on patients with ischaemic LV dysfunction failed to demonstrate benefit from revascularisation compared to optimal medical treatment alone (The viability sub-studies of the Surgical Treatment for Ischaemic Heart Failure (STICH), Heart Failure Revascularisation (HEART), and PET and recovery following revascularization (PARR-2) trials).4,14,15 Nonetheless, despite conflicting results of previously mentioned trials, recent ESC guidelines of myocardial revascularisation 6 suggested that in patients with ischaemic LV dysfunction and coronary artery disease (CAD), non-invasive imaging may be considered for the assessment of myocardial ischemia and viability before the decision making on revascularization or optimal medical treatment.7,11,13
Literature shows conflicting results not only about the impact of viability evaluation but also about the different imaging methods, that might better than others reveal the fact of viable tissue of the myocardium and the significant extent of viability.
Schinkel et al. demonstrated in a meta-analysis of 158 studies the similar impact of evaluating viability by different imaging modalities: PET demonstrates the highest sensitivity and negative predictive value while DSE shows the highest specificity and positive predictive value.16,17 While Romero et al., in a meta-analysis of 24 CMR studies demonstrate that CMR LGE provides the highest sensitivity of 95% in predicting functional recovery after the revascularisation, 18 and provides negative predictive value for predicting improved segmental LV contractile function after revascularization.18–21
However, CMR LGE is an expensive, time-consuming imaging method, associated with several contraindications. Therefore, the strain-encoded CMR (SENC) is widely discussed in recent CMR studies. SENC is a novel CMR tool for evaluation of myocardial deformation, myocardial function, regional myocardial deformation expressed as regional myocardial strain.22–24 Due to this unique characteristic SENC not only increases LGE CMR sensitivity and specificity (100% and 86% respectively) 25 but moreover SENC may distinguish subendocardial and transmural scar. 26 However, despite the relatively large amount of significant scientific evidence, SENC is not yet widely applicable in all CMR centers as quantification analysis is requiring additional time and a dedicated expert.
Therefore, our study was performed to find the imaging mortality predictors in patients with previous MI, symptomatic HF, reduced LV EF, and advanced coronary artery atherosclerosis evaluated by invasive coronary angiography when the need for percutaneous intervention was questionable because of suspected myocardial scar tissue. The ability to use all available non-invasive imaging modalities in difficult patients for the evaluation of myocardial viability might lead to better clinical solution making and might prevent patients from unnecessary invasive procedures and potential risks. Results of our study can add a supplementary understanding of the need for myocardial viability evaluation to prognosticate MI induced symptomatic HF outcomes, and clinical decision making. Imaging modalities without the use of radiation exposure (TTE and CMR) should be the first choice imaging modalities for the viability evaluation of the myocardial. If the CMR results demonstrate a sufficient amount of viable myocardium revascularisation should be recommended. However, according to the results of previous studies, 27 LGE CMR reveals a significantly higher number of scars, therefore FDG PET should be performed in cases when viability is lacking in CMR, as FDG PET appears to be more optimistic in functional recovery prediction. This algorithm of myocardial viability evaluation could be applied in clinical practice, leading to better evaluation of myocardial viability, the cost-effectiveness of selecting exact expensive imaging modalities, but also avoiding unnecessary radiation exposure.
The main limitation of our study was the possibility of anatomical misalignment between different imaging modalities. Evaluation of the 17 segments model of the LV is slightly different comparing nuclear medicine imaging and CMR. Solving this anatomical misalignment problem, the best solution could be a PET/CMR hybrid scan protocol offering a wide evaluation of the heart (anatomical by CMR and functional by PET), however, in our center, it is not technically applicable. Another limitation of our study is the absence of the clinical strategy evaluation (revascularisation vs medical treatment) in the follow-up, which could confirm our prognostic model of parameters for evaluation of the viable/non-viable myocardium and the benefit of possible revascularisation. However, the study population was too small, and clinicians were not always following the recommendations for revascularization versus medical treatment. Therefore, the precise benefit of revascularization in predicting late mortality in our study was not possible.
Conclusions
Taking in to account the imaging features and their cut-off values – defect score (CMR) >25, defect score (SPECT-PET) >22, LVEDD (TTE) >58 mm, LVEDDi (TTE) >30 mm/m2, LVEDDi (CMR) >33.6 mm/m2, LV GLS (CMR) −13.4 and LV GCS -16.3, may increase sensitivity and specificity of FDG PET and LGE CMR for prediction of late mortality. Therefore, identifying a higher risk of cardiac mortality patients, could lead to a more aggressive medical ischemic HF treatment, and the ability for more precise patient selection for the revascularization or heart transplantation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
