Abstract
Background:
Mitochondrial permeability transition pore (mPTP) opening plays a crucial role in cell death during ischemia-reperfusion injury (IRI). Cyclosporine A (CsA) inhibits mPTP opening. This study aimed to investigate the effects of CsA treatment during cardioplegia on the mitochondrial function and cardiac IRI.
Methods:
Landrace pigs (52.9 ± 3.7 kg) were subjected to midline sternotomy, cardiopulmonary bypass at 34°C and 90 minutes of cardiac arrest. They received either a single shot of standard 4°C cold histidine-tryptophan-α-ketoglutarate (HTK)-Bretschneider solution (n = 11) or HTK-Bretschneider plus 1.2 mg/L CsA (histidine-tryptophan-α-ketoglutarate plus cyclosporine A (HTK/CsA); n = 11). During reperfusion global left-ventricular function was assessed and myocardial biopsies were harvested at baseline, during ischemia and 45 minutes following reperfusion. High-resolution respirometry and hydrogen peroxide production were measured. Immunohistochemical stainings for apoptosis-inducing factor and hypoxia-inducible factor-1α as well as a flow cytometry-based JC-1 mitochondrial membrane potential assay were performed.
Results:
Hemodynamic parameters were comparable between both groups. The cytochrome C release (HTK: 930.3 ± 804.4 pg/mg, HTK/CsA: 699.7 ± 394.0 pg/mg, p = 0.457) as well as PGC1α content (HTK: 66.7%, HTK/CsA: 33.3%, p = 0.284) was lower in the HTK/CsA group. Respiratory measurements revealed that the oxygen flux under basal respiration was higher in the HTK/CsA group (8.2 ± 1.3 pmol·O2·s−1·mg−1·ww) than in the HTK group (3.8 ± 1.4 pmol·O2·s−1·mg−1·ww, p = 0.045). There were no significant differences regarding histological surrogates of apoptosis and necrosis.
Conclusions:
Supplementing cardioplegic solutions with CsA enhances the basal mitochondrial respiration thereby exerting a cardioprotective effect and diminishing IRI-induced damage. CsA seems to preserve mitochondrial function via non-ROS related pathways.
Keywords
Introduction
Cardiac surgery requiring cardiac arrest leads to ischemic damage of myocardial tissue driven by hypoxia. Subsequent reperfusion induces an inflammatory response mediated by pro-inflammatory cytokines and enhanced by oxidative stress. 1 The hypoxic effect is enhanced by the resulting microvascular dysfunction in the ischemic tissue.1,2
Ischemia-reperfusion injury (IRI) was described for the first time by Hearse and colleagues. 3 Mitochondria play a key role in cardiac IRI since the loss of mitochondrial function leads to cell death through apoptosis. 4 The process of mitochondrial injury centers around a complex of proteins in the mitochondrial inner membrane that form a non-selective channel, the mitochondrial permeability transition pore (mPTP).5,6 The mPTP is closed during the ischemic period and changes into an open state in the first few minutes of reperfusion. An increased opening time of the mPTP leads to a loss of the gradient across the inner mitochondrial membrane, thus, promoting mitochondrial dysfunction. Opening is modulated by metabolic influences within the cell such as energy availability, mitochondrial Ca2+ overload, production of reactive oxygen species (ROS), or pH status. Cyclosporine A (CsA) can block mPTP opening7,8 and its ability to provide protection against IRI, limit myocardial damage, and preserve cardiac function has been tested experimentally6–9 and in clinical trials.10–13 However, CsA addition to a cardioplegic solution was investigated in isolated hearts only.6,9 Reports on experimental setups investigating the effects of CsA during cardioplegia in animal models are missing. Therefore, the present study was designed to evaluate the cardioprotective effects of CsA as an adjunct to histidine-tryptophan-α-ketogluterate (HTK)-Bretschneider solution (Custodiol®), a crystalloid single-shot cardioplegic solution, in a large animal model.
Methods
Animals and anesthesia
The animal study was approved by the local animal welfare agency (TVV 42/15) and conducted in accordance with 2010/63/EU. Prior to transportation, pigs (50–60 kg) were sedated (midazolam 0.5 mg/kg, atropine 0.02 mg/kg, ketamine 15 mg/kg i.m.). Thereafter, propofol (2 mg/kg i.v.) and sufentanil (0.5 µg/kg i.v.) were administered via the lateral ear vein, and pigs were intubated and mechanically ventilated (Cato, Draeger, Lübeck, Germany) with 50–60 vol% oxygen. Anaesthesia was maintained by propofol (30–35 mg/kg/h i.v.) and sufentanil (0.5 µg/kg i.v.). Propofol was reduced to 25 mg/kg/h during hypothermia. Prior to extracorporeal circulation heparin (300 IU/kg i.v.) was given to prevent clotting of catheters.
Arterial blood pressure was invasively measured via a 5F PiCCO® Catheter (PULSION Medical Systems, Feldkirchen, Germany) in the femoral artery. Blood pressure, electrocardiography, body temperature, O2-saturation, and respiratory parameters were recorded permanently. Blood gases, pH, and electrolyte concentrations in blood samples were measured at ABL 90 flex (Radiometer, Willich, Germany). Heart time and stroke-volume were recorded by PiCCO plus (PULSION Medical Systems).
Surgical technique, extracorporeal circulation, and perfusion
Following midline sternotomy and pericardiotomy, the apex was cannulated with a 7F Maximum™ hemostasis introducer sheath (St. Jude Medical, USA) allowing the introduction of a Millar microtip pressure catheter (Millar MPC-500, Houston, TX, USA) into the left ventricle. Hemodynamic variables were recorded by a 16-channel Ponemah (ACQ-7700; Data Sciences International, St Paul, MN, USA). For extracorporeal circulation (S III, Stöckert, Sorin Group, Milan, Italy and EUROSETS, Medolla, Italy) purse-string sutures were placed in the aortic arch and the right atrial appendage for arterial (25 cm × 22Fr, FemFlex™, Edwards Lifesciences, Irvine, USA) and venous (29 cm × 29F Trim-Flex Dual Stage Venous Drainage Cannula, Edwards Lifesciences) cannulation. A 12F coronary sinus catheter (Edwards Lifesciences) was placed into the coronary sinus for blood gas analysis. A needle vent (12 GA DLP®, Medtronic, Minneapolis, USA) was placed at the ascending aorta.
Before cross-clamping of the aorta and infusion of the cardioplegic solution all animals were equilibrated for at least 40 minutes. During this time baseline variables for hemodynamic measurements and myocardial transmural biopsies from the left ventricle were harvested (Figure 1).
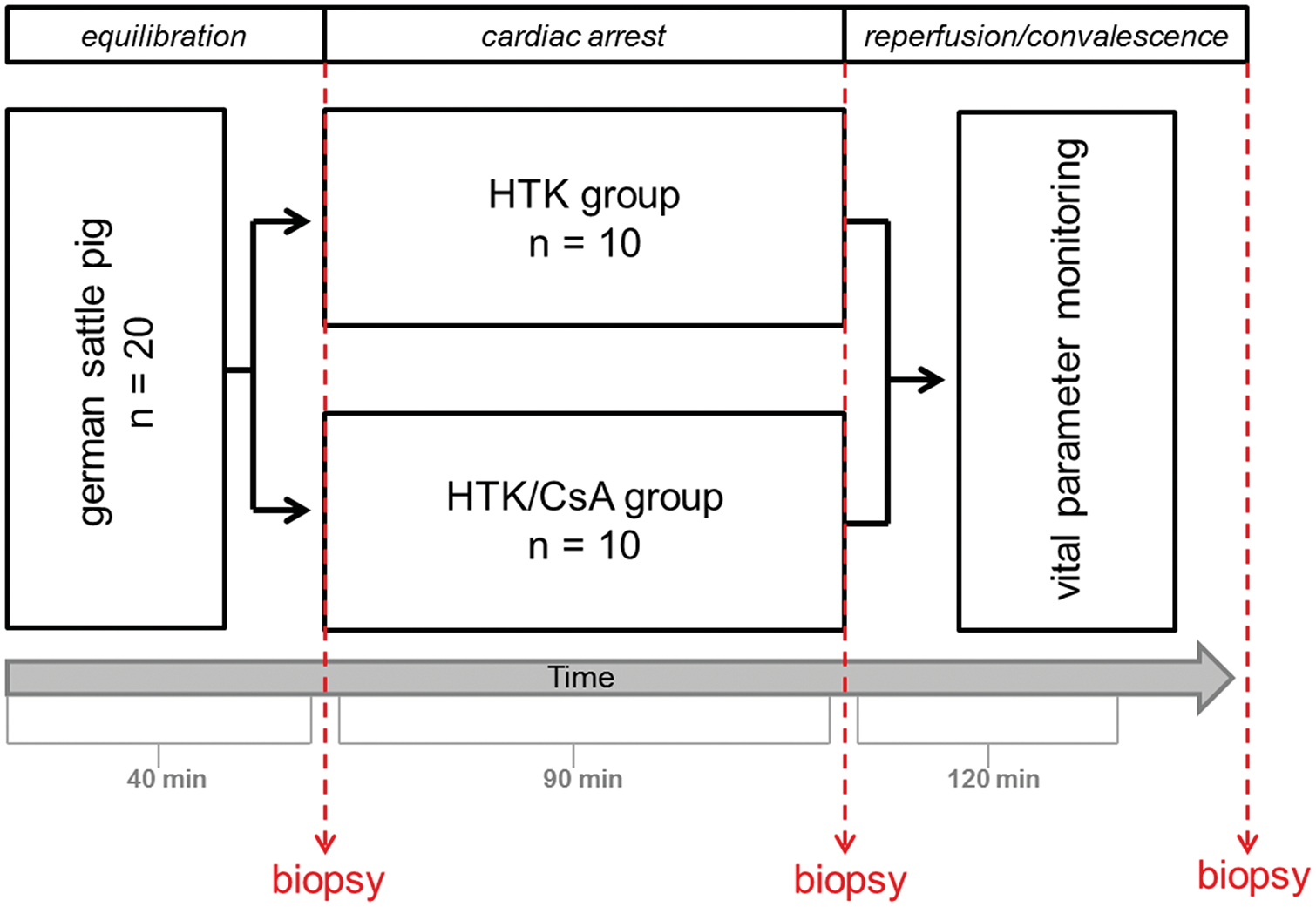
Study design to illustrate the experimental structure, the acquisition of samples including biopsy withdrawal and data collection.
Afterwards animals were randomly assigned (n = 10 per group) to receive either 1 mL/g estimated heart weight/min Custodiol® (HTK-group) (HTK-Bretschneider, Köhler-Chemie, Alsbach-Hähnlein, Germany) or Custodiol plus 1.2 mg/L CsA (HTK/CsA group) (Novartis Pharma GmbH, Nuremberg, Germany) over a time-period of 6–8 minutes. The heart weight was estimated with 0.5% of the body weight. Hearts were arrested for 90 minutes at 34°C body temperature. Second myocardial biopsies were harvested from the left ventricle before reperfusion was started. Subsequently, 30 minutes following reperfusion, the body temperature was restored to physiological conditions. Thereafter, the animals were weaned from extracorporeal circulation and recovery was followed up for 60 minutes. After 120 minutes reperfusion, myocardial biopsies were harvested from the left and right ventricle.
Randomization and blinding
A simple randomization was performed before each operation day. Thus, the generation of an unpre-dictable allocation sequence was guaranteed. Allocation concealment was performed by using sequentially numbered, opaque sealed envelopes (SNOSE).
The surgical team (surgeon, assistants, veterinarian, perfusionists) were blinded and not involved in the randomization process. Preparation of the cardioplegic solutions and transfer of the solution into unlabeled flasks were performed by a third-party person before the cardioplegic solutions were handed over to the perfusionist. Experimenter performing the molecular biological, protein biochemical, histological, and statistical analyses were blinded until statistics were completed.
ROS measurement
Samples for high-resolution respirometry and hydrogen peroxide (H2O2) production were snap frozen in ice-cold BIOPS buffer (2.77 mM CaK2-EGTA (ethylene glycol tetraacetic acid), 7.23 mM K2-EGTA, 5.77 mM adenosine triphosphate, 6.56 mM magnesium chloride, 20 mM taurine, 15 mM Na2-phosphocreatine, 20 mM imidazole, 0.5 mM dithiothreitol, 50 mM morpholino ethanesulfonic acid (MES)), 30% dimethylsulfoxide, and 10 mg/mL bovine serum albumin.14–16 Samples were thawed at 37°C and rinsed twice in the respiration medium, Mir05 (0.5 mM EGTA, 3 mM magnesium chloride, 60 mM lactobionic acid, 20 mM taurine, 10 mM KH2PO4, 20 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 110 mM D-sucrose, and 1 g/L essential fatty acid-free bovine serum albumin). 17 Subsequently, cardiac fibers were dissected and permeabilized for 30 minutes in 1 mL BIOPS buffer containing 50 μg/mL saponin. Fibers were rinsed in Mir05, dry blotted, weighed and transferred to the sealed chambers of the Oxygraph-2K (Oroboros Instruments, Innsbruck, Austria) high-resolution respirometer. The oxygen concentration within the chambers was maintained between 150 and 250 nmol/mL and the derivative of the decline in oxygen concentration over time was calculated as oxygen flux of the permeabilized cardiac fibers corrected for wet weight (ww).
Mitochondrial basal respiration was measured in the presence of L-glutamate, L-malate, and pyruvate, each 5 mM at baseline, end of ischemia, and at the end of the 120 minutes reperfusion period. Measurements of oxidative phosphorylation of complex I, II, and IV or uncoupled respiration was performed by adding 2 mM adenosine diphosphate, 10 mM succinate, 0.5 μM carbonyl cyanide 4-(trifluoromethoxy)-phenylhydrazone, 2 μM rotenone, 2 mM ascorbate, or 500 µM N,N,N′,N′-tetramethyl-p-phenylenediamine. Addition of 2.5 mM antimycin A provided a measure of non-mitochondrial (background) respiration, which was subtracted as a correcting factor.
Mitochondrial H2O2 production was measured simultaneously within the high resolution respirometry using 10 µM Amplex® UltraRed in the presence of 1 U/mL horseradish peroxidase. The slope of ΔF/s was converted to H2O2 production rate by calibrating with three exogenous H2O2 titrations and adjusting the data to account for the decline in fluorescence sensitivity as previously described. 18 The H2O2 production rate was normalized to pmol/s/mg·ww by correcting to sample weight. Exogenous superoxide dismutase with a concentration of 5 U/mL was included to incorporate superoxide released into the cytoplasm.
Immunohistochemical analysis
Myocardial tissue samples were fixed with 4% formaldehyde in phosphate buffered saline (phosphate buffered saline (PBS), pH = 7.4), embedded in paraffin, sectioned (3 µm), and collected on glass slides. Cross-sections were deparaffinized in xylene, dehydrated in ethanol (100%, 96%, 70%), and rehydrated in distilled water. Sections were boiled in 0.01 M sodium citrate for 30 minutes in a microwave at 800 W. After blocking the endogenous peroxidase with 0.1% H2O2 in methanol/tris-buffered saline (TBS; 0.05 M Tris, 0.5 M sodium chloride), unspecific binding sites were blocked with 2% bovine serum albumin in TBS. Primary antibodies against apoptosis-inducing factor (apoptosis-inducing factor (AIF); 1:80, Santa Cruz, Dallas, TX, USA) and hypoxia-inducible factor-1α (HIF-1α; 1:80, Santa Cruz) were incubated overnight at 4°C. After washing with 1x TBS, the sections were incubated with Dako REAL EnVision-HRP labeled secondary antibody (Agilent Technologies, Glostrup, Denmark). The specimens were then stained with 3-amino-9-ethylcarbazole chromogen (Agilent Technologies) and hematoxylin to visualize the nucleus. Further the cross-sections were stained with eosin and hematoxylin to observe histopathological alterations.
Flow cytometric analysis
The JC-1 mitochondrial membrane potential assay (Cayman Chemical, Ann Arbor, Michigan, USA) was performed according to the manufacturer’s instruction. The green/red ratio was calculated to assess mitochondrial integrity. Flow cytometry analysis was implemented on a BD LSR II cytometer with FACS Diva 6.1.3 software (both from BD, New Jersey, USA). Living cells were gated and 10,000 events were analyzed in each sample.
Enzyme-linked immunosorbent assay (ELISA)
Frozen tissue samples were homogenized in lysis buffer (50 mM Tris, 1 mM ethylendiaminetetraacetic acid, 150 mM sodium chloride, 1% nonidet-P40, 0.25% deoxycholic acid sodium salt, 1x protease/phosphatase inhibitor cocktail, 1% phenylmethylsulfonylfluoridin), sonicated by ultrasound and centrifuged at 13,000 × g for 10 minutes at 4°C. Subsequently, protein content was determined by a bicinchoninic acid assay (Thermo Fisher Scientific, Waltham, MA, USA) following manufacturer’s specifications. The peroxisome proliferator-activated receptor γ coactivator α (PGC1α) and cytochrome C were quantified by enzyme-linked immunosorbent assay (ELISA) (Elabscience Biotechnology Co., Wuhan, China and MyBioSource, San Diego, California, USA).
Statistical analysis
Statistical analyses were performed with SPSS Statistics 23 software (IBM, Armonk, New York, USA). Data are represented as mean ± standard deviation, unless stated otherwise. Comparison of the means was performed with Student’s t-test and p values ⩽0.05 were considered as significant.
Results
Hemodynamic parameters
The total amount of cardioplegic solution was applied according to the estimated heart weight (1 mL per g estimated heart weight) and was therefore comparable between the HTK and the HTK/CsA group. Infusion of the modified HTK solution resulted in 1.20 mg/L CsA being provided to the treatment group. There were no significant differences between the groups regarding heart rate (HR), blood pressure, central venous pressure, blood pH, lactate, cardiac output (CO), global end-diastolic volume, dpdtmin and dpdtmax before and after cardioplegic arrest (Table 1). This applies to the vital variables such as heart rate, systolic and diastolic blood pressure as well as the values measured by means of PICCO or Millar catheter (selected parameters are displayed in Table 1). The number of defibrillations or failure of automatically restoration of normal heart rhythm that is sinus-rhythm (HTK: 1.88 ± 1.45 shocks, HTK/CsA: 2.56 ± 2.87 shocks, p = 0.578) was comparable between both groups.
Hemodynamic parameter at baseline, after 40 minutes of equilibration and after 120 minutes reperfusion for the Custodiol and the Custodiol plus CsA group.
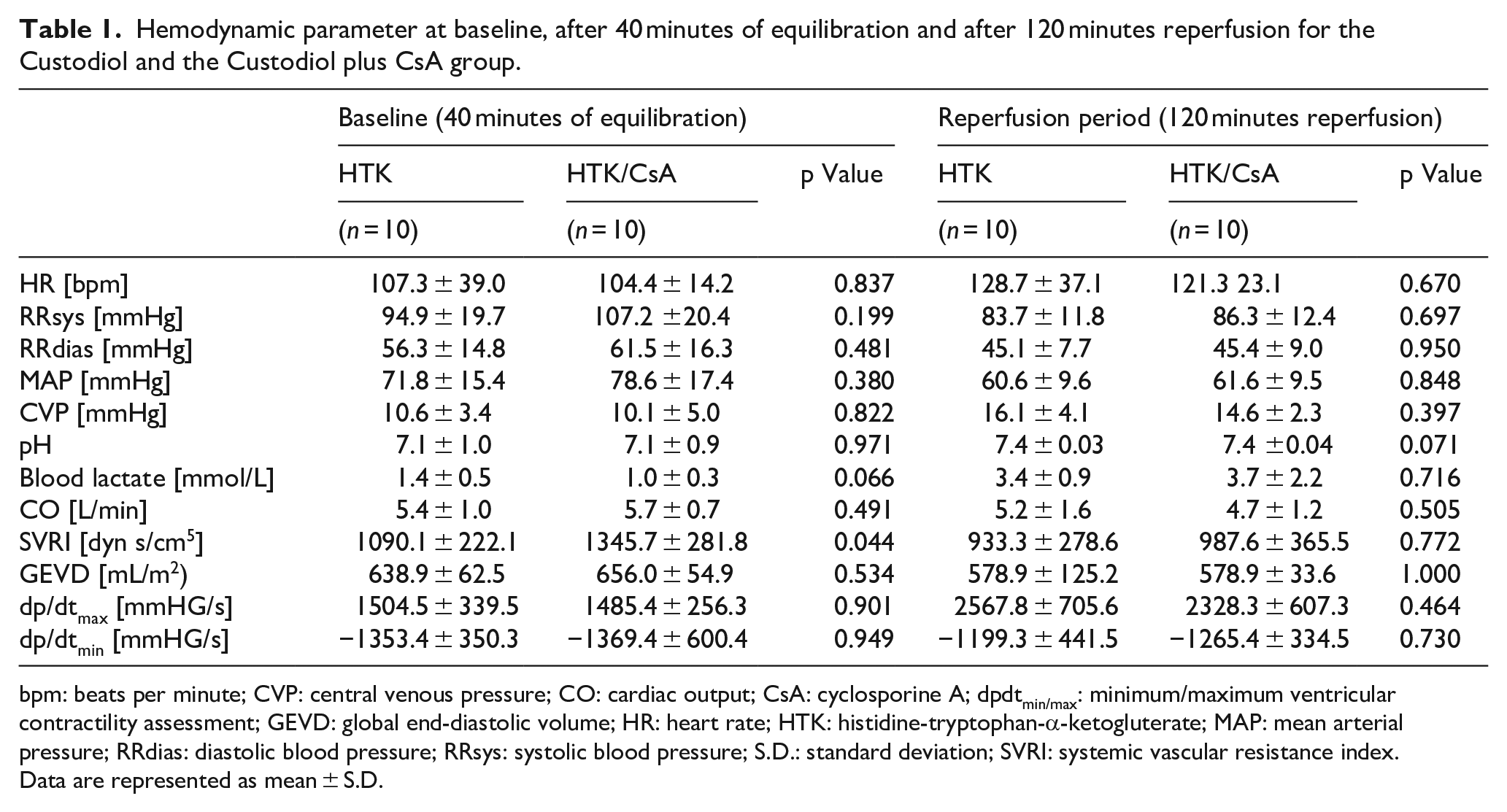
bpm: beats per minute; CVP: central venous pressure; CO: cardiac output; CsA: cyclosporine A; dpdtmin/max: minimum/maximum ventricular contractility assessment; GEVD: global end-diastolic volume; HR: heart rate; HTK: histidine-tryptophan-α-ketogluterate; MAP: mean arterial pressure; RRdias: diastolic blood pressure; RRsys: systolic blood pressure; S.D.: standard deviation; SVRI: systemic vascular resistance index.
Data are represented as mean ± S.D.
Effect on mitochondrial integrity
Mitochondrial integrity was determined by measuring the membrane potential after staining with JC-1. Membrane potential stabilization was detected during cardioplegia in the HTK and the HTK/CsA group which has been evaluated by a decrease of the JC-1 green/red ratio (baseline: HTK 23.0 ± 13.2, HTK/CsA 21.0 ± 14.8, p = 0.689; 90 minutes ischemia: HTK 7.8 ± 4.4, HTK/CsA 10.5 ± 7.0, p = 0.567). The following reperfusion period led to a slight depolarization of the mitochondrial membrane potential according to an increase of the JC-1 green/red ratio (HTK: 15.1 ± 4.3; HTK/CsA 13.4 ± 9.7, p = 0.650) (Figure 2).
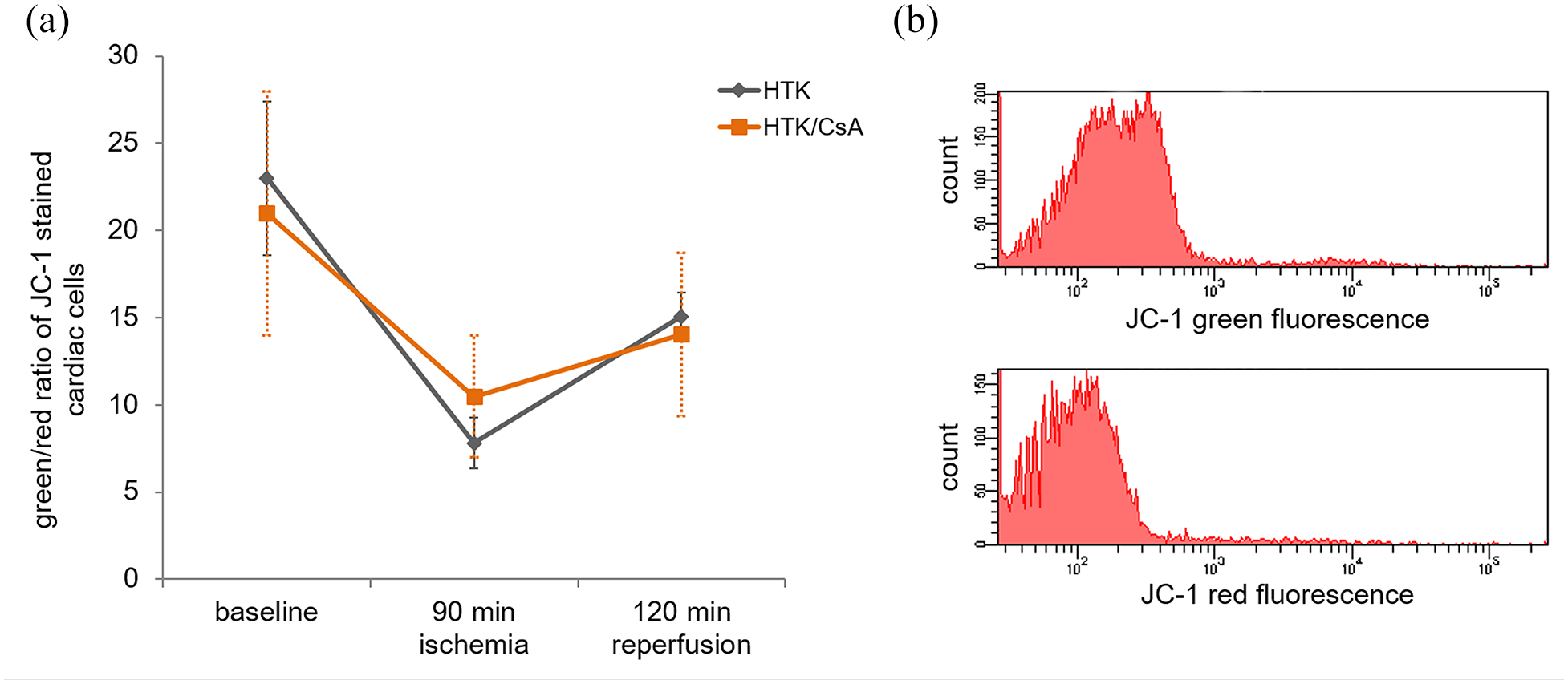
Measurement of mitochondrial integrity. Membrane-permeant JC-1 dye was used to evaluate the mitochondrial membrane potential in the HTK and the HTK/CsA group (a). All values are expressed as mean ± S.E.M. A representative example of the flow cytometric analysis of JC-1 stained cardiac cells was shown in (b).
Furthermore, mitochondrial membrane destabilization during the reperfusion period caused a cytochrome C release with a lower but insignificant mean in the HTK/CsA group (699.7 ± 394.0 pg·cytochrome·C/mg·protein) compared to the HTK group (930.3 ± 804.4 pg·cytochrome·C/mg·protein; p=0.457) (Figure 3).
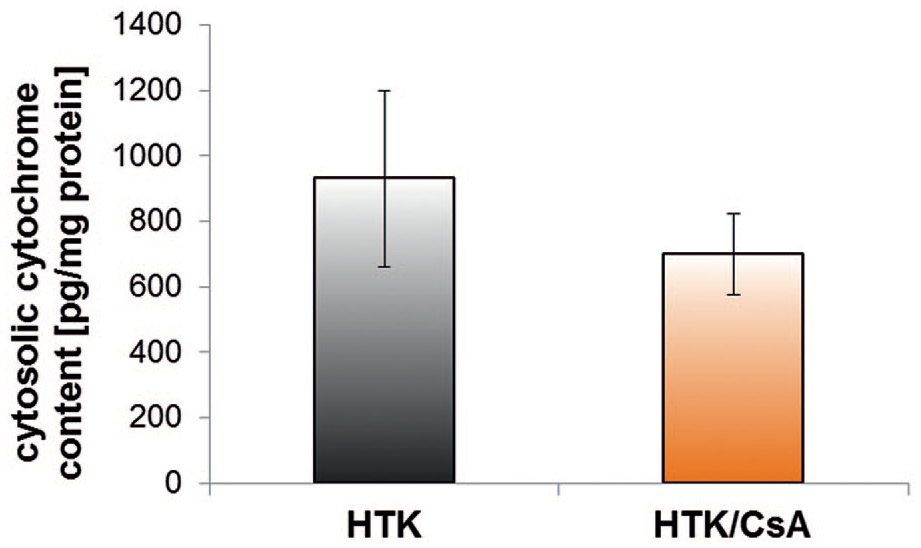
Cytosolic cytochrome C amount in myocardial tissue after cardiac arrest induced by HTK or HTK/CsA cardioplegic solution. All values are expressed as mean ± S.E.M.
Analysis of the transcriptional coactivator PGC1α which regulates genes involved in energy metabolism such as mitochondrial biogenesis revealed that only 33.3% of HTK/CsA-treated individuals had high values of more than 1000 pg/mg PGC1α. In comparison, 66.7% of the HTK-treated pigs showed high values of more than 1000 pg/mg PGC1α (p = 0.284).
Mitochondrial respiration and ROS production
There were no statistically significant differences in the amount of hydrogen peroxide produced in any of the respiratory states. Following reperfusion, we detected a significant increase in the amount of oxygen flux under basal respiration in the HTK/CsA group (8.2 ± 1.3 pmol·O2·s−1·mg−1·ww) compared to the HTK group (3.8 ± 1.4 pmol·O2·s−1·mg−1·ww; p = 0.045) (Figure 4). Uncoupled respiration (HTK: 75.0 ± 9.5 pmol·O2·s−1·mg−1·ww; HTK/CsA: 59.6 ± 8.2 pmol·O2·s−1·mg−1·ww), complex I (HTK: 33.2 ± 3.5 pmol·O2·s−1·mg−1·ww; HTK/CsA: 26.9 ± 2.1 pmol·O2·s−1·mg−1·ww), complex II (HTK: 44.9 ± 7.3 pmol·O2·s−1·mg−1·ww; HTK/CsA: 37.1 ± 4.9 pmol·O2·s−1·mg−1·ww) as well as complex I and II respiration (HTK: 65.1 ± 7.3 pmol·O2·s−1·mg−1·ww; HTK/CsA: 46.0 ± 4.2 pmol·O2·s−1·mg−1·ww) did not differ between both groups after 120 minutes reperfusion.
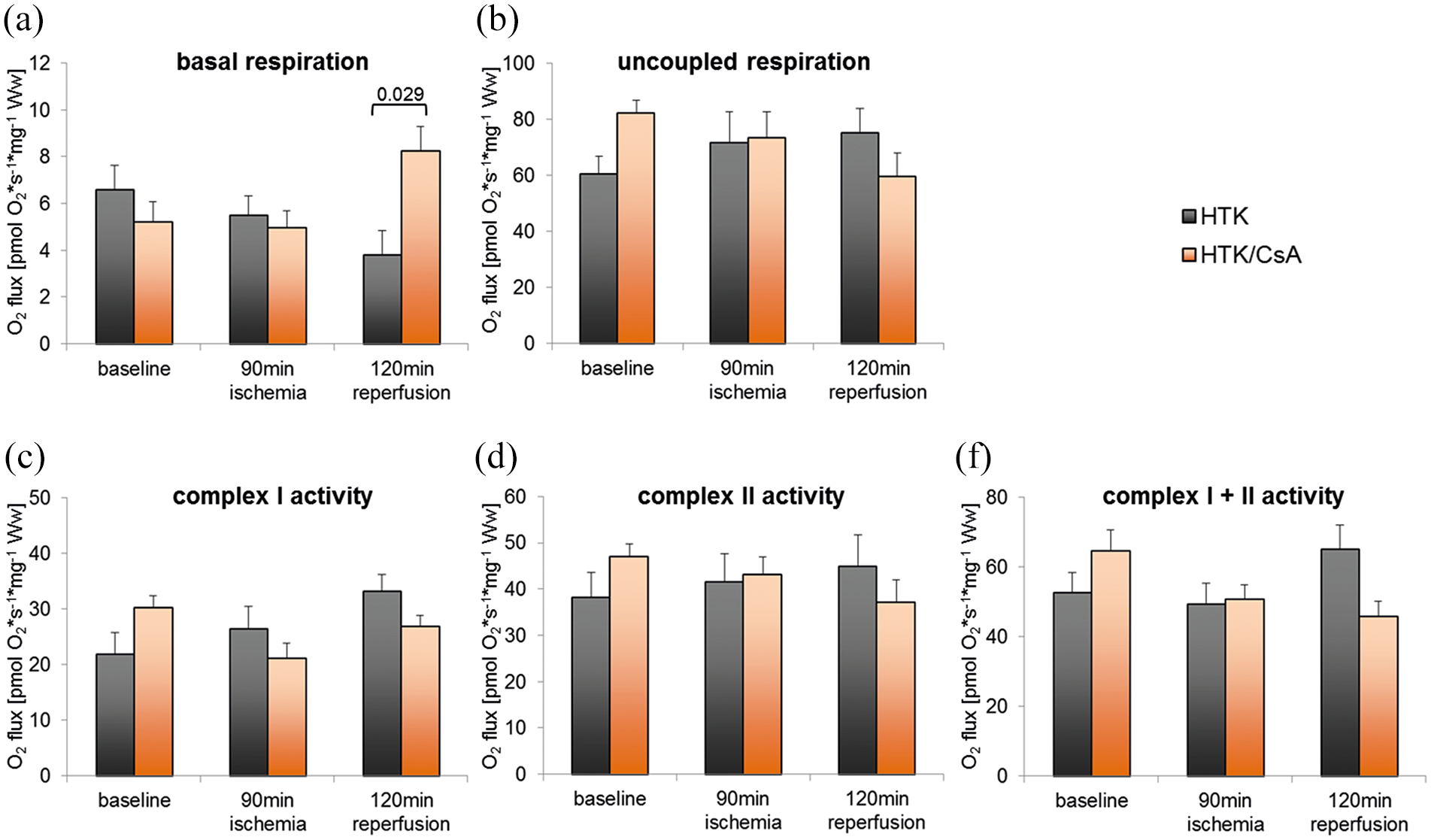
Comparison of mitochondrial respiration in different respiratory states across all time points between the HTK- and HTK/CsA-treated group. Basal (a) and uncoupled respiration (b) as well as complex I (c), complex II (d), and complex I and II (e) respiration were quantified. All values are expressed as mean ± S.E.M. Statistically significant differences (p < 0.05) were shown.
Histology
After 120 minutes reperfusion, no differences regarding the presence of cross striation of cardiomyocytes (p = 0.845), eosinophilia (p = 0.510), edema (p = 0.596), cellular infiltration (p = 0.279), visible bleeding (p = 0.876) as well as the loss of cell boundaries (p = 0.510) were detected between HTK and HTK/CsA-treated animals.
HIF-1α translocation into the nucleus was quantified by immunohistological analysis. The percentage of HIF-1α-positive cells was comparable between HTK and HTK/CsA-treated group at all time points of biopsy withdrawal (baseline: HTK 29.3 ± 9.4, HTK/CsA 30.5 ± 14.7, p = 0.845; after ischemia: HTK 26.1 ± 8.4, HTK/CsA 31.9 ± 13.1, p = 0.302; after reperfusion: HTK 30.7 ± 8.6, HTK/CsA 31.9 ± 6.8, p = 0.776) (Figure 5(a)).
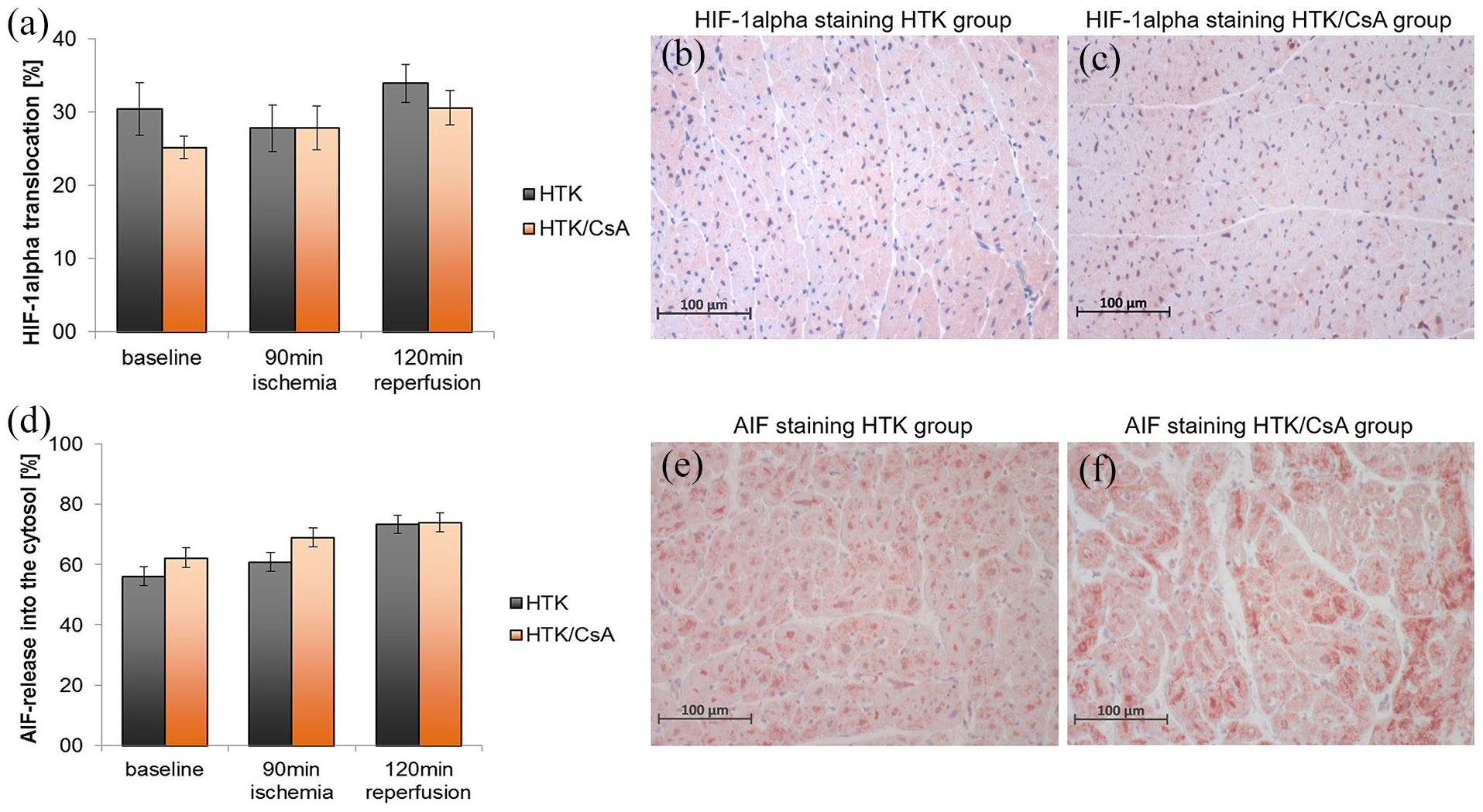
Immunohistochemical analysis of hypoxia inducible factor HIF-1α (a–c) and apoptosis inducing factor AIF (d–f). Exemplary stainings of HIF-1α (b and c) and AIF (e and f) of the HTK and HTK/CsA group were shown.
Analysis of the apoptogenic protein AIF showed that the percentage of AIF-positive cells was similar in the HTK compared to HTK/CsA group at all time points of biopsy withdrawal (baseline: HTK 56.2 ± 13.6, HTK/CsA 62.3 ± 11.9, p = 0.303; after ischemia: HTK 61.0 ± 17.5, HTK/CsA 68.9 ± 9.9, p = 0.226; after reperfusion: HTK 73.4 ± 7.3, HTK/CsA 73.9 ± 3.5, p = 0.838) (Figure 5(b)).
Comment
In the present study, we investigated the cardioprotective role of CsA by adding low-dose CsA to the cardioplegic solution during a surgical intervention including 90 minutes of cardiac arrest. We found lower, but statistically unsignificant levels of cytochrome C release and PGC1α content in the HTK/CsA group. Additionally, a preserved oxygen flux under basal respiration was found after CsA-supplemented cardioplegia.
Over the last 10 years, a number of animal studies have investigated the effect of CsA addition on myocardial damage and cardiac function. The concepts of CsA addition differed enormously in the different pre-clinical studies. Research studies investigating animal models with occlusion of the left anterior descending (LAD) coronary artery added CsA systemic prior to ischemia via the femoral vein8,19 or during post-conditioning. 20 Reports on rabbit or rat isolated hearts added CsA to a perfusion solution before starting cardioplegia, 9 to the cardioplegic solution itself 6 or during post-conditioning. 21 The CsA concentrations used in the LAD occlusion animal models ranged from 1 to 10 mg/kg, while the CsA concentration ranged from 0.2 to 2 µmol/L in the perfused isolated heart studies. The CsA concentration used in this study was low-dosed and set to 1.2 mg/L due to previous results obtained by Pritzwald-Stegmann et al. 6
Our study is the first study which investigated the effect of myocardial protection of CsA addition under surgical conditions in a large animal model. Mitochondrial membrane potential was comparable between the HTK/CsA and the HTK group during the whole surgical intervention including ischemia and reperfusion period. The amount of released cytochrome C was lower in the CsA-treated group. However, this difference was not statistically significant. Previous studies reported a lower cytochrome C release into the cytosol after CsA treatment.9,21 Both research groups investigated the effects of low-dose CsA addition in an experimental setting using Langendorff-perfused rat and rabbit hearts and reported that CsA addition into the crystalloid cardioplegic solution minimized cytochrome C release. Although the difference in cytochrome C content was not significant in our study because of the high biological variance of our pig model, the difference between the groups was visible. Many studies investigated CsA-induced effects on isolated hearts or in inbreeding animal models such as Sprague-Dawley rats. These models show a smaller biological variance than outbred pigs of different litters. However, our model more likely reflects the clinical conditions in human studies. A limitation of our study is the fact that we used healthy pigs with a healthy myocardium for our surgical interventions. Patients who require a cardiac surgery including cardioplegia often show alterations or pathological changes of the myocardial tissue. The mitochondrial effects we measured in healthy pig hearts could be different in pathologically changed human myocardium.
In intact cells, cytochrome C is part of the mitochondrial intermembrane space and functions as an electron shuttle in the respiratory chain. An increased release of cytochrome C indicates a failure of mitochondrial membrane integrity and could mediate activation of apoptotic pathways to induce cell death. Furthermore, it might be that cytochrome C can exit the mitochondria without an important and irreversible mitochondrial outer membrane permeabilization. 22
Beside our group, only one group investigated and reported the mitochondrial oxygen consumption in cardiac cells following IRI. Oka et al. 9 showed that the oxygen flux was higher in the CsA-treated isolated, perfused hearts suggesting that mitochondrial function is preserved and mitochondrial outer membrane integrity is maintained. These results are in accordance with our results which showed that basal respiration was higher in the CsA-treated group.
The histological evaluation did not detect any differences between the groups regarding cellular abnormalities of cardiac cells such as loss of cross striation of cardiomyocytes, loss of cell boundaries and eosinophilia as well as for the occurrence of edema and cellular infiltration. Furthermore, HIF-1α translocation into the nucleus and the release of AIF into the cytosol did not differ between both groups in our study. In response to hypoxia, the cell needs adaptations involved in the regulation of tolerance to oxidative stress. HIF-1α functions as a transcription factor and mediates the expression of genes involved in mitochondrial function, cell survival, and apoptosis. 2 Mitochondria-mediated caspase-independent apoptotic cell death is mediated by activation of AIF. This protein is located within the mitochondrial intermembrane space. During IRI it is cleaved before released into the cytosol. 23 Our results demonstrate no significant difference of HIF-1α activation and AIF positivity in both study groups. However, these findings suggest that there is no adverse effect in using HTK solution supplemented with CsA. Moreover, the period of reperfusion was perhaps too short to detect long-term variations between both groups.
Mitochondria play a central role in energy metabolism and cell survival during stress conditions like oxygen deprivation due to ROS production. IRI within the mitochondria of cardiomyocytes may be initiated by the supraphysiological rise in matrix calcium (Ca2+) during ischemia, which results in the opening of the mPTP upon reperfusion. Consequently, the inner mitochondrial membrane becomes non-selectively permeable to small (<1.5 kDa) solutes of ~3 nm diameter,24,25 resulting in a loss of the mitochondrial membrane potential (∆Ψ), rupture of the outer mitochondrial membrane, and swelling. mPTP opening is promoted under conditions of oxidative stress which may occur in situations of increased ROS production. Interestingly, the current study did not show any evidence of increased ROS production in any respiratory state with the addition of CsA. Nevertheless, we detected that basal respiration was actually enhanced in CsA-treated samples acquired at 120 minutes reperfusion. This suggests that CsA may provide a protective effect upon mitochondrial function via non-ROS related pathways hitherto unknown. Further research is needed to identify the possible mechanisms underlying this protection to elucidate the effects afforded by CsA and to establish if this pathway may be exploited to provide therapeutic protection during cardioplegia. Therefore, we concluded that the reperfusion conditions can be influenced with the help of CsA. A preserved mitochondrial function via non-ROS related pathways was found, and future studies should determine if this preserved mitochondrial function diminishes the IRI-induced damage significantly and the optimal CsA concentration.
Footnotes
Acknowledgements
We thank Prof. Friedrich W Mohr for his great support of the present study. Furthermore, we would like to thank Dr. Susann Ossmann and Johannes Walliser for their veterinary support, Dr. Philipp Kiefer for mentoring, and Carmen Piesker for her assistance with the histological stainings.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
