Abstract
Introduction
There is increasing evidence for extracorporeal cardiopulmonary resuscitation (ECPR) as a rescue therapy for selected patients in refractory cardiac arrest (CA). Besides patient selection, the control of reperfusion parameters is of eminent importance. Especially in out-of-hospital CA, monitoring and individualized, targeted reperfusion remains a great challenge for emergency personnel. The CARL® system is designed to enable an early control of a variety of reperfusion parameters and to pursue a targeted reperfusion strategy in ECPR.
Case presentation
We report the first 10 ECPR applications of the CARL® system in Regensburg, Germany. Early blood gas analysis, oxygen titration and pressure monitoring were feasible and enabled an individualized and targeted reperfusion strategy in all patients. After suffering from refractory CA and prolonged resuscitation attempts, five out of the first 10 patients survived and were successfully discharged from the hospital (CPC one on hospital discharge).
Conclusion
Application of the CARL® system contributed to early monitoring and control of reperfusion parameters. Whether targeted ECPR may have the potential to improve outcomes in refractory OHCA remains the subject of future investigations.
Keywords
Introduction
Across Europe, depending on the region, only 1-18% of all patients with an out-of-hospital cardiac arrest (CA), who receive cardiopulmonary resuscitation (CPR), survive and can be discharged from the hospital alive. 1 A great number of those patients suffer from neurological sequelae. 2 An increasing body of evidence suggests that the use of extracorporeal membrane oxygenation (ECMO) in cardiopulmonary resuscitation - a method called extracorporeal cardiopulmonary resuscitation (ECPR) - may improve survival after CA. 3 Therefore, ECPR is already acknowledged by the current guidelines of the European Resuscitation Council: “We suggest that ECPR may be considered as a rescue therapy for selected patients with cardiac arrest when conventional CPR is failing in settings in which it can be implemented (weak recommendation, very low certainty of evidence”. 4 Besides patient selection, the control of reperfusion parameters is of eminent importance to enable a neurological recovery after CA.5,6
Especially in out-of-hospital CA, monitoring and thus individualized, targeted reperfusion remains a great challenge for emergency personnel. The CARL® system (Resuscitec GmbH, Freiburg, Germany) is the first ECMO system specifically designed for application in resuscitation outside of an intensive care unit or an operation room. The core system consists of the CARL® Controller, which includes a two-pump reperfusion set, and CARL® MOX, a mobile gas blender. The system can be amended by a mobile cooling unit (CARL® Cooler), which can be operated without an external energy source. The combination of CARL® Controller with CARL® MOX enables an immediate control of a variety of reperfusion parameters (i.e. flow, arterial pressure, temperature, venous hemoglobin, oxygen saturation and built-in blood gas analysis) and thereby empowers the ECMO team to pursue a targeted reperfusion strategy in ECPR from the very start7–9 (Figure 1). By mixing ambient air with oxygen (using the blower technology), CARL® MOX can work independently of an external air supply and needs only an oxygen source for oxygen titration in sweep gas. Besides an oxygenator (hilite 7000 LT, Xenios AG, Heilbronn, Germany), the CARL® reperfusion set contains two diagonal pumps (DP3, MEDOS Medizintechnik AG, Stolberg, Germany), which can be used to generate pulsatile flows and therefore higher reperfusion pressures. To improve the transportability of the CARL® system, weight reduction is achieved by using a carbon frame and other low-weight components (weight CARL® Controller: 17.6 kg, CARL® MOX: 8.9 kg). In Regensburg, out-of-hospital ECPR is always performed by at least a team of two: A physician, who performs the cannulation and a cardio technician, who oversees the ECMO system. Inclusion and exclusion criteria are based on the German interdisciplinary consensus statement, which is supported by the German Resuscitation Council.
10
The ECPR team and its strategy have been described previously.11–13 (a) Application of the CARL® system (CARL® controller and CARL® MOX) in a supermarket. (b) Display of CARL® MOX. (c) Display of CARL® controller with online monitoring of reperfusion parameters (i.e. flow, pulsatility, arterial pressure, temperature, venous and arterial blood gas analysis).
Case presentation
We present data of the first 10 consecutive patients who were treated with the CARL® system after out-of-center CA in Regensburg, Germany (Figure 1). Nine patients suffered out-of-hospital CA and one patient suffered CA in an external hospital. The data was collected from the Regensburg ECLS Registry, which was approved by the University of Regensburg Ethics Committee (number 21-2401-104). The need to obtain an informed consent was waived due to the retrospective design.
Patient/case characteristics.
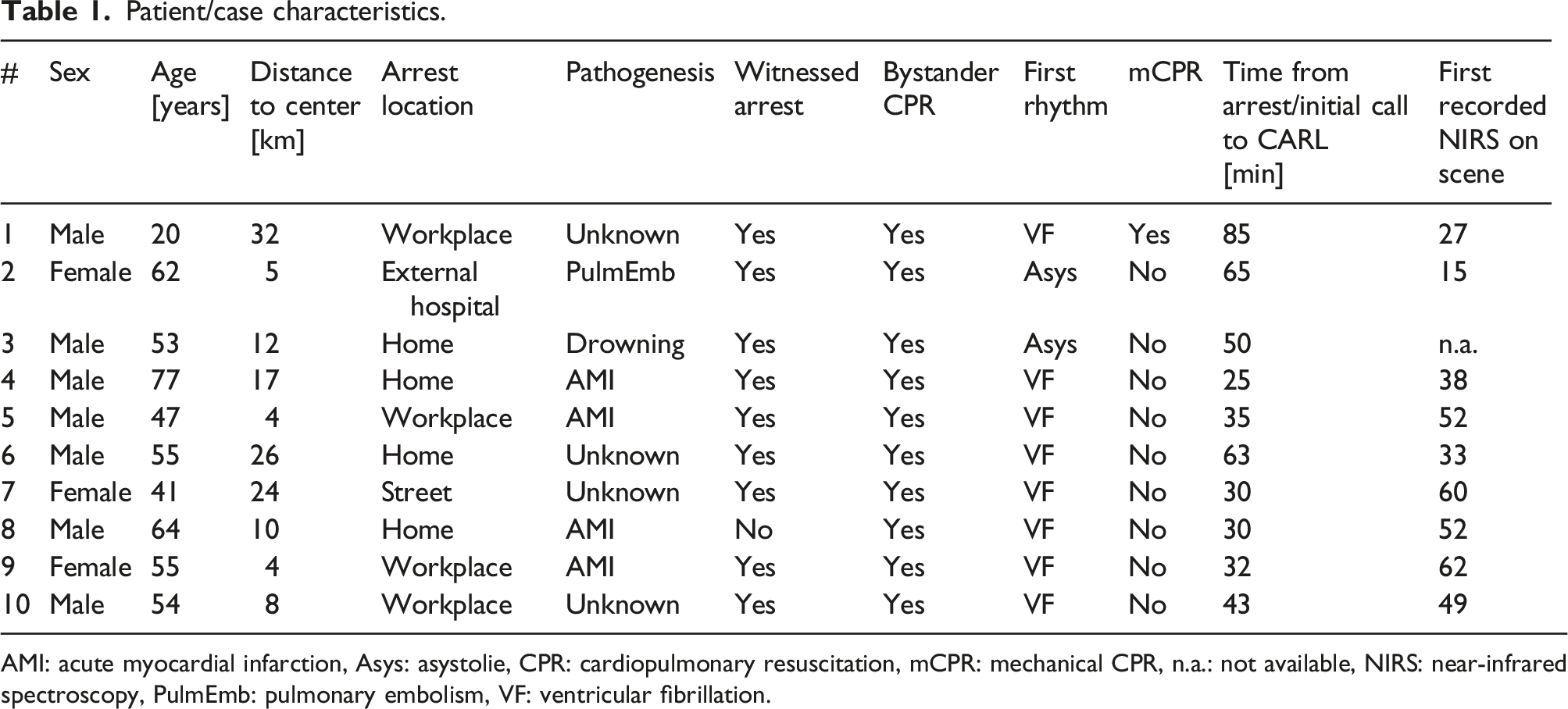
AMI: acute myocardial infarction, Asys: asystolie, CPR: cardiopulmonary resuscitation, mCPR: mechanical CPR, n.a.: not available, NIRS: near-infrared spectroscopy, PulmEmb: pulmonary embolism, VF: ventricular fibrillation.
Parameters at hospital admission/outcome.
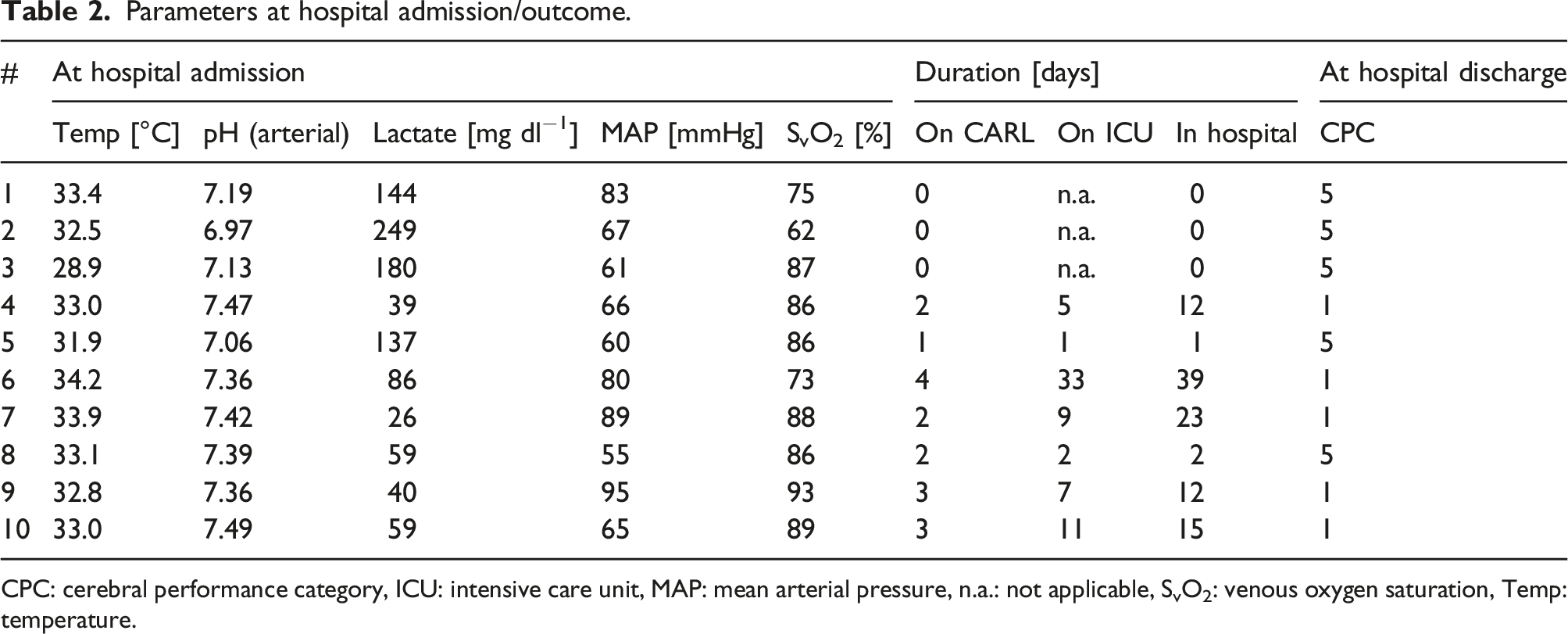
CPC: cerebral performance category, ICU: intensive care unit, MAP: mean arterial pressure, n.a.: not applicable, SvO2: venous oxygen saturation, Temp: temperature.
Conclusion
The application of the CARL® system in ECPR is feasible and enables an extended control of reperfusion parameters (i.e. arterial pressure, oxygen saturation and built-in blood gas analysis) in in- and out-of-hospital settings. Early monitoring of reperfusion parameters allows the ECPR team to pursue an individualized and targeted reperfusion strategy. We believe, that immediate targeted ECPR has the potential to improve outcomes in refractory CA, but this belief has still to be verified by randomized trials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CB is CTO and shareholder of Resuscitec GmbH, a start-up company of the University Medical Center Freiburg. JP is part-time employee of Resuscitec GmbH. The other authors declare that they have no competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
